Abstract
Enhanced vasoconstriction is increasingly identified as an important contributor to the development of pulmonary hypertension. Chronic hypoxia results in enhanced Rho kinase mediated Ca2+ sensitization contributing to pressure-dependent pulmonary arterial tone as well as augmented vasoconstriction to endothelin-1 and depolarizing stimuli. We sought to investigate the interaction between these vasoconstrictor stimuli in isolated, pressurized, pulmonary arteries. We used the K+ ionophore, valinomycin, to clamp membrane potential (Vm) to investigate the role of membrane depolarization in endothelin-1 and pressure-dependent constriction, and endothelin-1 receptor inhibitors to determine whether membrane depolarization or stretch signal through endothelin-1 receptors. Clamping Vm prevented pressure-dependent tone, but not enhanced vasoconstriction to endothelin-1 following chronic hypoxia. Furthermore, endothelin-1 receptor inhibition had no effect on either pressure-dependent tone or vasoconstriction to KCl. As Src kinases contribute to both pressure-dependent tone and enhanced endothelin-1 vasoconstriction following chronic hypoxia, we further investigated their role in depolarization-induced vasoconstriction. Inhibition of Src kinases attenuated enhanced vasoconstriction to KCl. We conclude that membrane depolarization contributes to pressure-dependent tone but not enhanced vasoconstriction to ET-1, and that Src kinases serve as upstream mediators facilitating enhanced Rho kinase-dependent vasoconstriction following chronic hypoxia.
Keywords
Introduction
Pulmonary hypertension (PH) resulting from obstructive pulmonary disease, interstitial lung disease, and sleep apnea is characterized by arterial remodeling and enhanced vasoconstriction leading to increased pulmonary vascular resistance. Increasing evidence demonstrates the importance of vasoconstriction to the pulmonary hypertensive response 1 with a key role for Rho kinase (ROK) mediated Ca2+ sensitization in humans 2 , 3 and animal models. 4 , 5 Vasoconstrictor responses to chronic hypoxia (CH) are multifaceted, including the development of pressure-dependent tone and enhanced vasoconstrictor reactivity to both membrane depolarizing stimuli and many receptor-mediated agonists. Studies from our laboratory have revealed a prominent role for RhoA-mediated myofilament Ca2+ sensitization in each of these vasoconstrictor components. 6 –8 Furthermore, epidermal growth factor receptor (EGFR)- and NADPH oxidase 2-dependent superoxide production are required for enhanced vasoconstriction to endothelin-1 (ET-1) and KCl in addition to the development of pressure-dependent tone following CH. 9 , 10
As membrane depolarization, ET-1, and pressure-dependent tone all signal through a similar pathway following CH, identifying the central upstream mediator of these responses has the potential to lead to the development of novel therapies for the treatment of PH. Increases in intraluminal pressure and ET-1 receptor (ETR) stimulation can result in vascular smooth muscle (VSM) membrane depolarization. 11 –13 Alternatively, both membrane depolarization 14 , 15 and membrane stretch 16 , 17 can be coupled to G-coupled protein receptor (GPCR) activation, potentially suggesting a central role for ETRs to mediate enhanced vasomotor tone in the hypertensive pulmonary circulation. Therefore, we wished to identify whether membrane depolarization or ETRs serve as central mediators for augmented vascular tone in CH-dependent PH. Utilizing isolated, pressurized small pulmonary arteries, we tested the hypothesis that CH increases vasoconstrictor responsiveness by coupling both stretch-induced and ET-1-mediated VSM membrane depolarization to myofilament Ca2+ sensitization. We also investigated the hypothesis the ETRs participate in metabotropic transduction of membrane depolarization and stretch to mediate enhanced vasoreactivity following CH.
Methods
Animals and CH exposure
All protocols and procedures in this study were reviewed and approved by our Institutional Animal Care and Use Committee. Male Sprague–Dawley rats (250–350 g, age three to four months; Harlan Industries, Indianapolis, IN) were used for all experiments. Rats were housed in a hypobaric chamber for four weeks with barometric pressure at ∼380 mmHg; age-matched control rats were housed in similar cages at ambient atmospheric pressure (∼630 mmHg). This CH exposure results in PH, arterial remodeling, and right ventricular hypertrophy. 10 , 18
Isolated pulmonary artery protocols: Vasoreactivity and vessel wall [Ca2+]i
Lungs were removed from rats anesthetized with pentobarbital sodium (200 mg/kg, i.p.) and a dissected pulmonary artery (∼150 µm inner diameter (ID)) was cannulated in a vessel chamber (CH-1; Living Systems, St. Albans, VT, USA). 9 , 10 All isolated arteries were studied at 37°C in physiological saline solution (PSS) containing (in mM) 129.8 NaCl, 5.4 KCl, 0.5 NaH2PO4, 0.83 MgSO4, 19 NaHCO3, 1.8 CaCl2, and 5.5 glucose (all from Sigma–Aldrich; St. Louis, MO, USA) and equilibrated with a 10% O2, 6% CO2, and balance N2 gas mixture (to approximate the normoxic PO2 to which small pulmonary arteries are exposed to in vivo). Arteries were endothelium-disrupted by gently rubbing a single strand of moose mane through the vessel lumen to directly evaluate effects of CH on VSM reactivity to KCl independent of endothelial influences, and disruption of endothelial cells was confirmed by lack of response to acetylcholine (ACh, 1 µM; Sigma) following uridine triphosphate (UTP, 5 µM; Sigma) preconstriction. 7 , 8 For intracellular Ca2+ ([Ca2+]i) measurement, pulmonary arteries were loaded with fura-2 AM for 45 min in darkness followed by a 20 min rinse with PSS. 8 , 10 Background-subtracted fura-2 F340/F380 emission (510 nm) ratios were calculated with IonOptix Ion Wizard software (Milford, MA, USA) and recorded continuously throughout the experiment to measure vessel wall [Ca2+]i, with simultaneous measurement of ID from red wavelength bright-field images. To directly address mechanisms of myofilament Ca2+ sensitization in response to KCl and ET-1, we clamped vessel wall [Ca2+]i by permeabilizing vessels with the Ca2+ ionophore ionomycin (3 µM, Sigma). These vessels were perfused with PSS containing 300 nM Ca2+, a concentration which was chosen to provide optimal vasoreactivity to KCl and ET-1 while having minimal effects on resting tone based on preliminary studies. [Ca2+]i concentration is expressed as fura-2 F340/F380 ratios. Vasoconstrictor responses (% change in inner diameter) to increasing concentrations of KCl (30, 60, and 120 mM) and ET-1 (10−10−10−7 M, Sigma) were assessed in Ca2+-permeabilized arteries from CH and control rats. In some experiments, the role of Src kinases in KCl-dependent constriction was tested with the Src kinase inhibitors SU6656 (10 µM, Cayman Chemical; Ann Arbor, MI, USA) and PP2 (10 µM, Cayman). 19
Pressure–response curves (5–45 mmHg) were performed in non-Ca2+-permeabilized arteries as the development of pressure-dependent tone is not associated with a significant change in vessel wall [Ca2+]i. 7 , 10 We performed pressure steps under both Ca2+-containing and Ca2+-free conditions, and tone was calculated as the percent difference in ID between Ca2+ containing and Ca2+ free conditions at each pressure step. 7 , 10 To evaluate the role of membrane depolarization in this response, experiments were conducted in the presence of the K+ ionophore valinomycin (5 μM, Sigma) 20 and 16 mM KCl to normalize VSM membrane potential (Vm) in CH arteries to that of control arteries and to prevent stretch-induced depolarization. Using the Nernst equation to predict membrane Vm, and assuming an intracellular K+ of approximately 150 mM and that K+ is the only membrane-permeant ion, 16 mM extracellular K+ in combination with valinomycin is predicted to set Vm at ∼ −60 mV (Vm of a control artery under baseline conditions 9 , 11 ). The ability of valinomycin to prevent depolarization was validated using sharp electrode measurement of Vm (see Measurement of membrane potential). We also used valinomycin and 16 mM KCl to eliminate membrane depolarization in response to ET-1 in Ca2+ permeabilized arteries. Assessment of pressure-dependent basal tone was further evaluated in separate sets of arteries from each group treated with the ATP-sensitive potassium channel (KATP) agonist, pinacidil (100 μM, Sigma), to hyperpolarize VSM Vm. We have previously demonstrated that this concentration of pinacidil provides near maximal dilation of the pulmonary circulation in isolated, saline-perfused rat lungs. 21 These experiments with pinacidil were performed in the presence of the L-type Ca2+ inhibitor diltiazem (50 μM, Sigma) 7 to prevent changes in VSM [Ca2+]i due to L-type channels.
As some GPCRs have been implicated in transducing both mechanical 16 , 17 and depolarizing stimuli 14 , 15 to VSM contraction, we hypothesized that ETRs function as a proximal signaling mediator coupling membrane depolarization and vessel wall stretch to myofilament Ca2+ sensitization and vasoconstriction following CH. To test this hypothesis, we assessed pressure-dependent basal tone and vasoconstrictor responses to KCl in pulmonary arteries similar to previous protocols. Experiments were performed in the presence of both the selective ETA receptor antagonist BQ-123 (10 μM, Sigma) and the ETB antagonist BQ-788 (10 μM, Sigma) 8 to determine if membrane depolarization or membrane stretch transduce their signal through ETRs.
Measurement of membrane potential
Small pulmonary arteries prepared as described above were used to measure VSM Vm in arteries from control and CH rats under baseline conditions, in the presence of valinomycin and 16 mM K+, or after pretreatment with pinacidil and diltiazem. Measurements of Vm in arteries treated with ET-1 (10-8 M) were performed in the presence of ionomycin to match vasoreactivity data. Arteries were maintained at 12 mmHg internal pressure unless otherwise stated. VSM cells were impaled from the adventitial surface with microelectrodes (50- to 100-MΩ tip resistance) containing 3 M KCl. Vm was recorded with a Neuroprobe amplifier (model 1600, A-M Systems). Analog output from the amplifier was low-pass filtered at 1 kHz and sent to a Tektronix RM502A oscilloscope and a Dataq data acquisition system. Vm recordings used for analysis contained (1) a sharp negative deflection in potential as the microelectrode was advanced into the cell, (2) a stable Vm for at least 30 s, and (3) an abrupt return to ∼0 mV following retraction of the electrode from the cell. Movement of vessels in response to changes in pressure and ET-1 stimulation prevented the acquisition of continuous recordings from the same cell. Therefore Vm was recorded in several (4–6) cells under both baseline conditions and in response to stimulation once arterial diameter was allowed to stabilize (∼5 min). The mean Vm of all VSM cells recorded from a single artery under each set of conditions was considered a single n for statistical purposes. 9 , 11
Calculations and statistics
Vasoconstrictor responses were calculated as a percent change in diameter from baseline ID. Data are expressed as means ± standard error, and n refers to the number of animals in each group. A t-test, two-way analysis of variance (ANOVA), three-way ANOVA, or repeated measures ANOVA was used to make comparisons when appropriate. If differences were detected by ANOVA, individual groups were compared using the Student–Newman–Keuls test. A probability of P < 0.05 was considered significant for all comparisons.
Results
Membrane depolarization is required for CH-dependent, pressure-induced tone
To test the possibility that depolarization contributes to the development of pressure-dependent pulmonary arterial tone, we permeabilized arteries to K+ with valinomycin in combination with 16 mM extracellular K+ to clamp Vm at ∼−60 mV. In the absence of valinomycin, resting Vm was depolarized in CH arteries compared to controls consistent with our prior findings. 9 , 11 Valinomycin treatment eliminated CH-dependent depolarization at each pressure tested (12 and 35 mmHg), and further blocked pressure-dependent depolarization in both CH and control arteries (Fig. 1(a)). Under vehicle conditions, arteries from CH but not control arteries developed pressure-dependent tone (Fig. 1(b)). Clamping Vm abolished the development of pressure-induced tone in arteries from CH rats without altering vessel wall Ca2+ in arteries from either group (Fig. 1(c)–(e)).
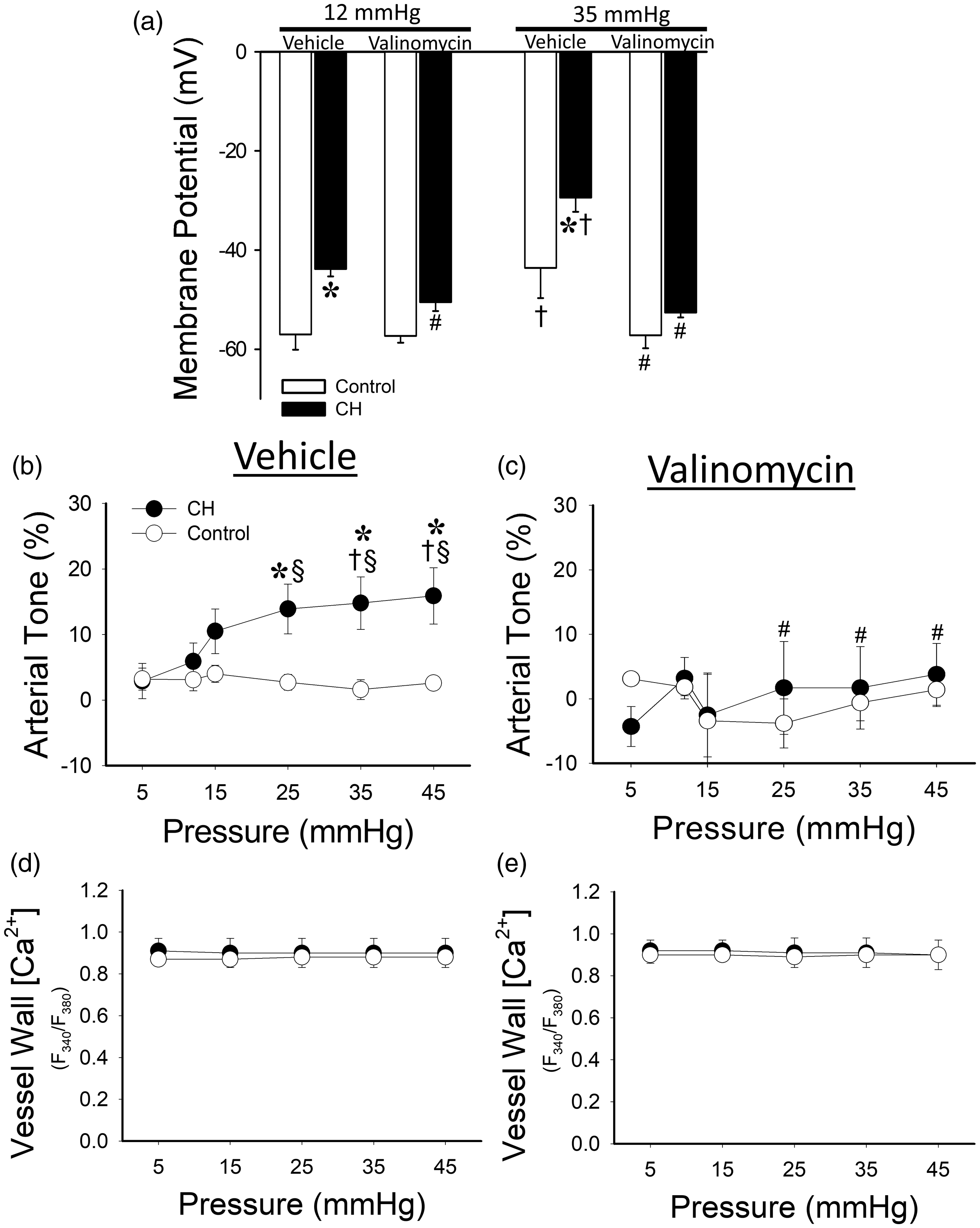
Membrane depolarization is necessary for CH-dependent basal tone. (a) Sharp electrode membrane potential measurements in isolated, endothelium-disrupted pulmonary arteries from CH and control rats under baseline (12 mmHg) and 35 mmHg pressures. Experiments were performed in the presence or absence of valinomycin/16 mM KCl. (b) and (c) Basal tone measurements in arteries from CH and control rats under vehicle (b) and valinomycin/16 mM KCl (c) treatments illustrate loss of pressure-dependent tone in CH arteries when membrane depolarization is prevented. (d) and (e) Fura-2 fluorescence measurements show that vessel wall Ca2+ is not altered by pressure or valinomycin. Values are means ± SE n = 4/group. *P < 0.05 versus control. #P < 0.05 valinomycin versus vehicle. †P < 0.05 versus 12 mmHg. §P < 0.05 versus response at 5 mmHg.
Additional protocols evaluated pressure-dependent responses in non-permeabilized arteries from each group in the presence or absence of the KATP activator pinacidil (100 μM) to hyperpolarize VSM Vm. Experiments were performed in the presence of the L-type Ca2+ channel inhibitor diltiazem to prevent possible effects of pinacidil to reduce VSM Ca2+. Pinacidil caused membrane hyperpolarization in each group, but did not fully prevent depolarizing influences of either CH or pressure (Fig. 2(a)). Consistent with a role for VSM membrane depolarization in pressure-dependent tone following CH, pinacidil reduced the development of tone in arteries from CH rats (Fig. 2(b)) independent of changes in vessel wall Ca2+ (Fig. 2(c)).
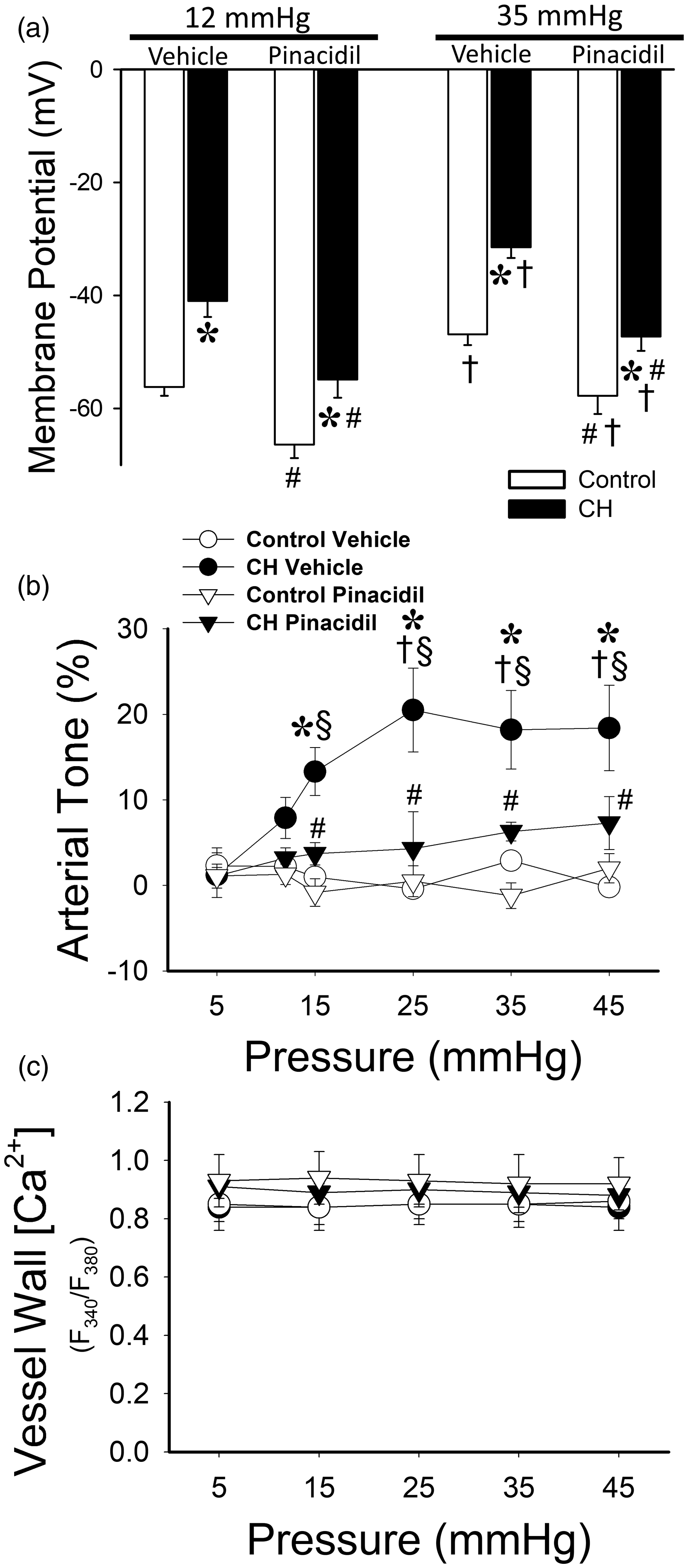
Pressure-dependent tone in CH rats requires depolarization. (a) Sharp electrode measurements of membrane potential at 12 and 35 mmHg in endothelium-disrupted pulmonary arteries from control and CH rats in the presence of pinacidil (100 μM). Both vehicle- and pinacidil-treated arteries were treated with diltiazem (50 μM) to ensure that the effects of pinacidil were not mediated by a reduction in vessel wall Ca2+ influx through L-type Ca2+ channels. Whereas pinacidil led to a more hyperpolarized Vm compared to vehicle conditions, it was not sufficient to prevent pressure-dependent depolarization in CH arteries. (b) Pressure-dependent tone responses indicate that limiting depolarization in CH arteries with pinacidil prevents the development of arterial tone. (c) Vessel wall Ca2+ is not different between groups or treatment conditions. Values are means ± SE n = 4/group. *P < 0.05 versus control. #P < 0.05 pinacidil versus vehicle. †P < 0.05 versus 12 mmHg. §P < 0.05 versus response at 5 mmHg.
Enhanced vasoconstriction to ET-1 following CH does not require membrane depolarization
We utilized valinomycin and 16 mM KCl to set Vm at ∼ −60 mV in Ca2+ permeabilized control and CH arteries to assess the contribution of depolarization to augmented vasoconstriction to ET-1. Surprisingly, while ET-1 caused a slight but significant depolarization in control arteries (Fig. 3(a)), there was no ET-1 dependent depolarization in CH arteries. The combination of valinomycin and 16 mM KCl prevented this depolarizing effect of ET-1 in control vessels and abolished CH-induced depolarization, resulting in similar Vm between groups and treatments. However, in contrast to effects of valinomycin to prevent pressure-dependent tone in CH arteries (Fig. 1), normalizing Vm between groups was without effect on vasoreactivity to ET-1 in vessels from either CH or control arteries (Fig. 3(b) and (c)). Vessel wall Ca2+ remained unchanged by ET-1 or valinomycin verifying [Ca2+]i clamp with ionomycin (Fig. 3(d)–(e)).
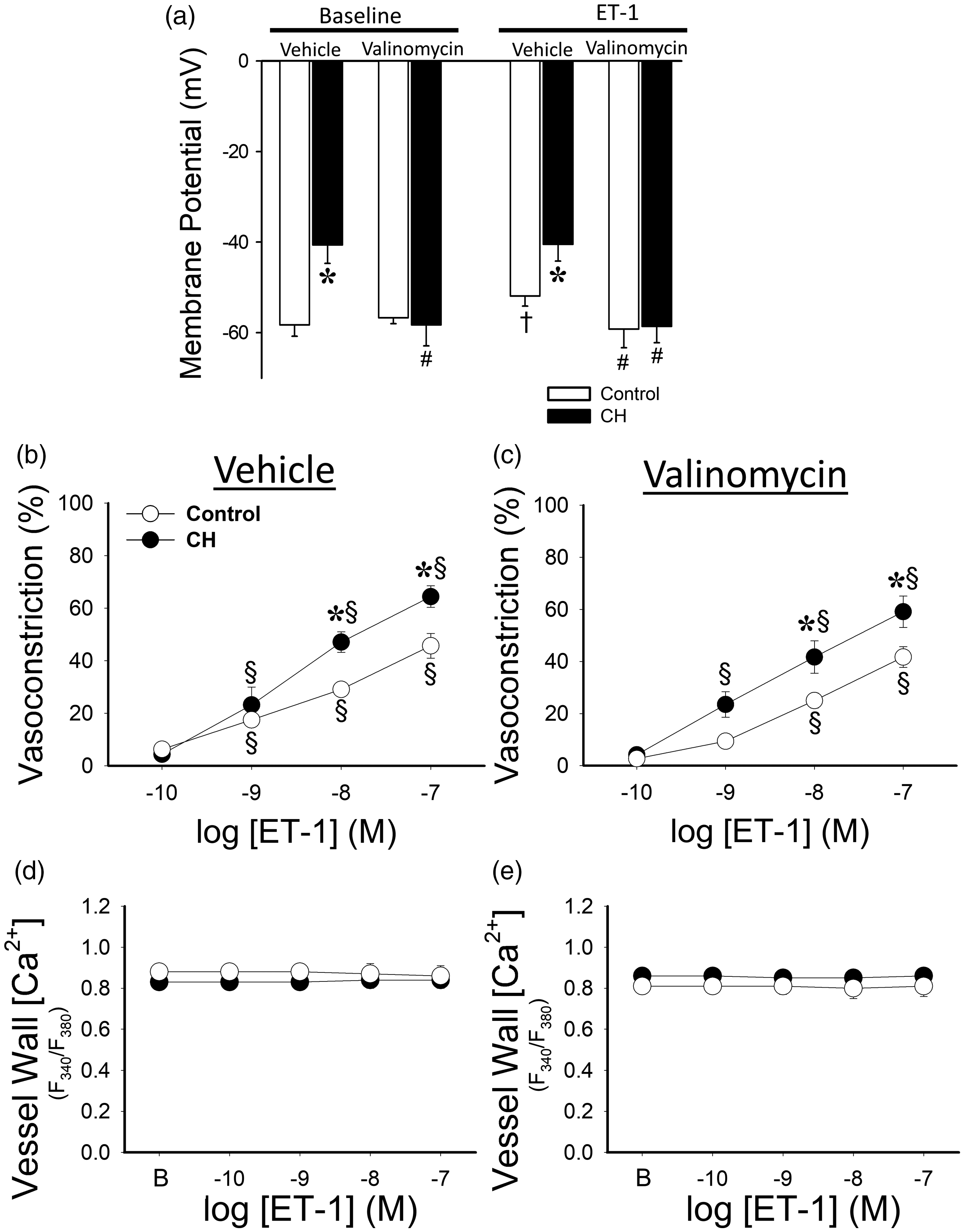
Augmented ET-1 dependent vasoconstrictor reactivity following CH is not dependent on membrane depolarization. (a) Sharp electrode measurements of membrane potential in response to ET-1 (10-8 M) in pressurized, Ca2+ permeabilized, endothelium-disrupted pulmonary arteries from CH and control rats in the combined presence of valinomycin/16 mM KCl and ionomycin or vehicle. (b) and (c) Vasoconstrictor responses to ET-1 arteries from CH and control rats in the presence of vehicle (b) or valinomycin/16 mM KCl (c). (d) and (e) Fura-2 fluorescence measurements verify Ca2+ clamp with ionomycin. B = baseline Ca2+. Values are means ± SE n = 4–5/group *P < 0.05 versus control. #P < 0.05 valinomycin versus vehicle. †P < 0.05 ET-1 versus 12 vehicle. §P < 0.05 versus response to 10−10 M ET-1.
ETRs do not contribute to augmented depolarization-induced vasoconstriction or the development of pressure-dependent tone following CH
Both membrane depolarization 14 , 15 and membrane stretch 16 , 17 can be coupled to GPCR activation. Because ET-1 appears to signal through the same pathway as depolarization and stretch to mediate VSM Ca2+ sensitization, 10 we next tested the hypothesis that ETRs couple membrane depolarization and vessel wall stretch to EGFR-mediated Ca2+ sensitization and vasoconstriction in arteries from CH rats. In agreement with our previous findings, 6 , 9 CH augmented vasoreactivity to KCl in Ca2+-permeabilized arteries (Fig. 4(a)). However, combined inhibition of ETA and ETB receptors was without effect on KCl-dependent vasoconstriction in arteries from CH rats. Furthermore, the development of pressure-dependent tone in CH arteries was also unaltered by combined inhibition of ETA and ETB receptors (Fig. 4(b)). Vessel wall [Ca2+]i remained constant in response to KCl in arteries treated with ionomycin (Fig. 4(c)) and in response to changes in intraluminal pressure (Fig. 4(d)).
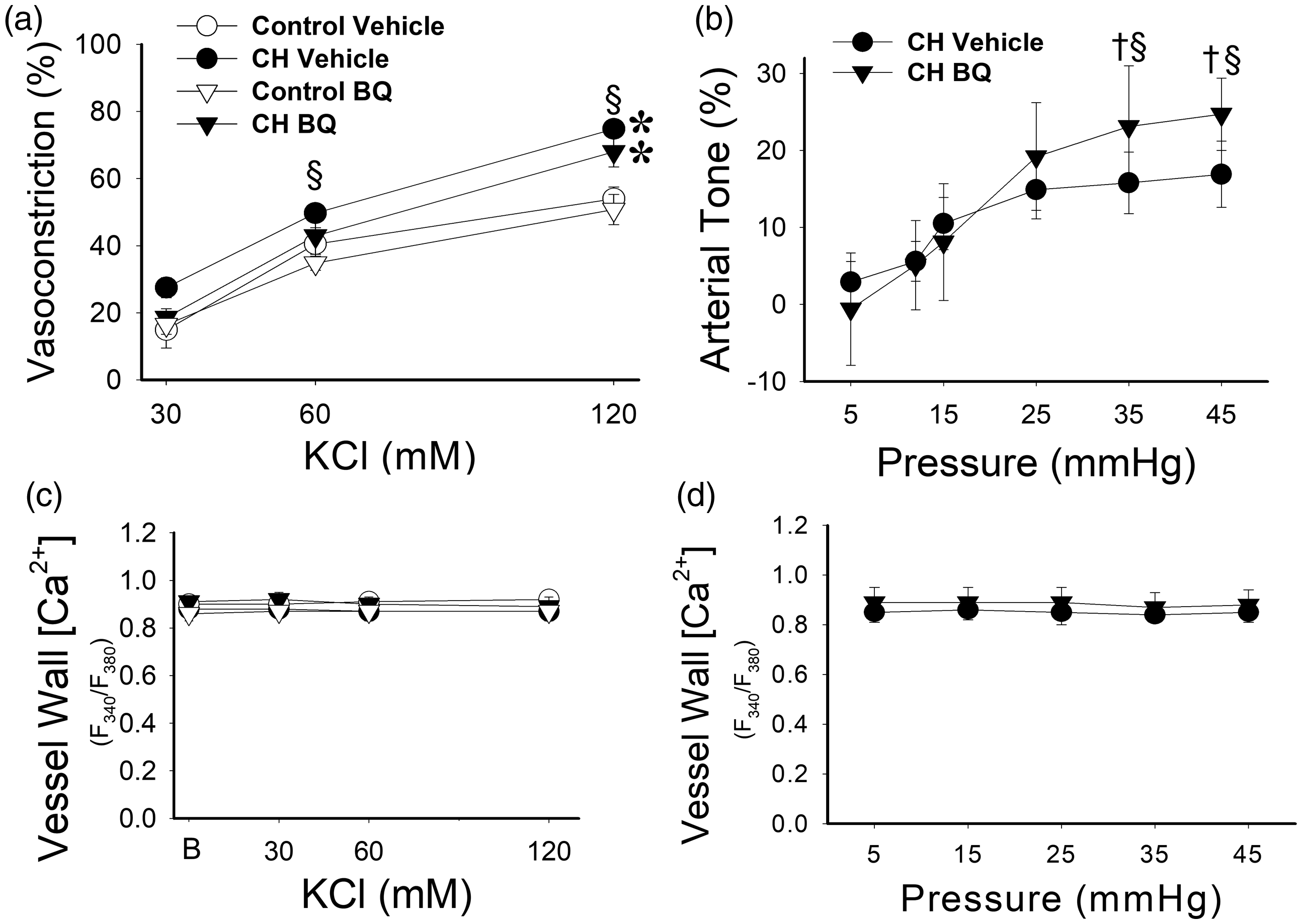
ETRs do not contribute to augmented depolarization-induced vasoconstriction or basal tone following CH. (a) Vasoconstrictor responses to KCl in pressurized, Ca2+ permeabilized, endothelium-disrupted pulmonary arteries from CH and control rats in the combined presence of BQ 123 and BQ 778 (10 μM each) or vehicle. (b) Basal arterial tone in small pulmonary arteries from CH rats in the presence or absence of BQ 123 and BQ 778 (10 μM each). (c) and (d) Fura-2 fluorescence measurements verify that vessel wall Ca2+ is not altered by KCl in Ca2+ clamped arteries (c), or by pressure in nonpermeabilized vessels (d). B = baseline Ca2+. Values are means ± SE n = 4–5/group. *P < 0.05 versus control. §P < 0.05 versus response at 30 mM KCl or 5 mmHg for all treatment groups. †P < 0.05 versus response at 12 mmHg for CH vehicle and CH BQ.
Role of Src kinases in augmented depolarization-induced vasoconstriction following CH
We have previously established that Src kinases contribute to enhanced ET-1 induced vasoconstriction and the development of pressure-dependent tone following CH. 10 Therefore, we sought to test the hypothesis that Src kinases facilitate enhanced depolarization-induced vasoconstriction as well and serve as a central mediator of enhanced vasoconstriction and Ca2+ sensitization in the hypertensive pulmonary circulation of rats. Blocking Src kinases with SU6656 (Fig. 5(a)) and PP2 (Fig. 5(b)) attenuated the effect of CH to augment KCl-induced vasoconstriction, although at the highest concentration of KCl (120 mM), Src inhibition did not fully normalize responsiveness to the level of controls. Vessel wall Ca2+ remained unchanged confirming [Ca2+]i clamp with ionomycin (Fig. 5(c) and (d)). Baseline diameter and tone were not significantly different between control and CH arteries.
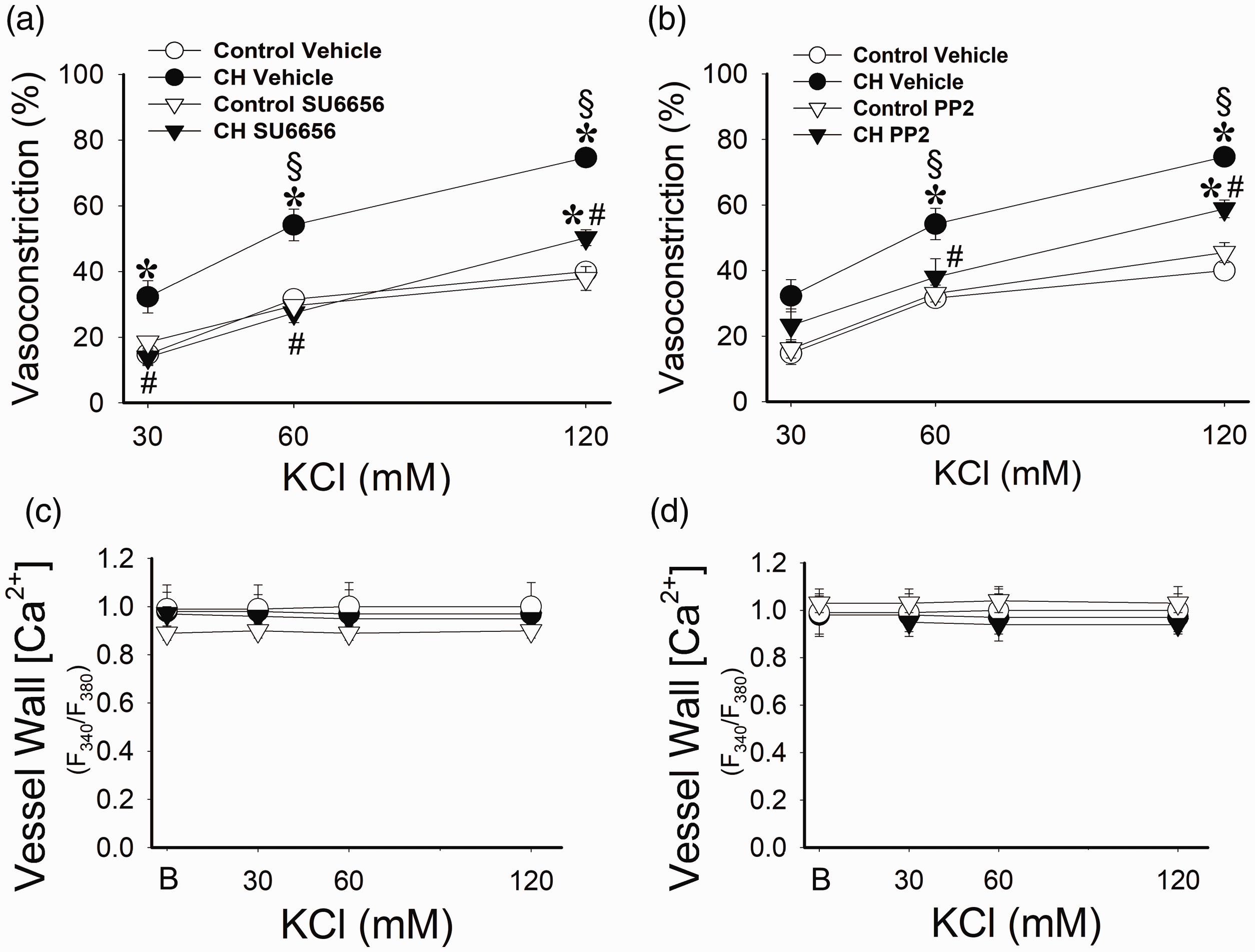
Src kinases are required for enhanced KCl-dependent vasoconstriction and Ca2+ sensitization in arteries from CH rats. (a) and (b) Vasoconstrictor responses to KCl in pressurized (12 mmHg), Ca2+ permeabilized, endothelium-disrupted pulmonary arteries from CH and control rats in the presence of the Src inhibitors SU6656 (a; 10 μM) and PP2 (b; 10 μM). (c) and (d) Fura-2 fluorescence measurements verify that vessel wall Ca2+ is not altered by KCl in Ca2+ permeabilized arteries. B = baseline Ca2+. Values are means ± SE n = 4–5/group; *P < 0.05 versus control. #P < 0.05 versus CH vehicle. §P < 0.05 versus responses at 30 mM KCl for all groups.
Discussion
The objective of the present study was to determine whether various stimuli mediating enhanced pulmonary vasoconstriction following CH signal through the same proximal mediator to augment vasomotor tone via myofilament Ca2+ sensitization. Major findings are that: (1) membrane depolarization contributes to the development of CH-dependent pressure-induced tone but not enhanced vasoconstriction to ET-1; (2) ETRs mediate neither pressure-dependent tone nor enhanced depolarization-induced vasoconstriction; and (3) Src kinases contribute to augmented depolarization-dependent vasoconstriction following CH, similar to that previously observed. 10 Thus, while increases in intraluminal pressure signal through membrane depolarization, Src kinases serve as a common mediator for the various stimuli that mediate augmented vasoconstriction in CH-induced PH.
Exposure to CH increases basal pulmonary arterial tone 7 , 22 and augments vasoconstrictor sensitivity to both receptor mediated agonists 8 , 23 and depolarizing stimuli. 6 , 9 Although mechanisms involving enhanced Ca2+ influx may additionally contribute to greater pulmonary vasoreactivity following CH, 24 , 25 it is clear that myofilament Ca2+ sensitization mediated by ROK provides a major contribution to these responses. 5 –8, 23 ROK inhibition improves pulmonary hemodynamics in patients with PH2, 3 and ROK inhibition acutely diminishes pulmonary arterial pressure in rodents. 4 , 26 We have identified Src, EGFR, NADPH oxidase, and superoxide as intermediaries linking depolarization, ETR stimulation, and increases in intraluminal pressure to ROK activation. 8 –10
Previous reports have demonstrated VSM membrane depolarization associated with increases in intraluminal pressure and ETR stimulation in pulmonary arteries. 11 –13 In addition, exposure to hypoxia reduces K+ currents in pulmonary VSM27 and expression of Kv channels is decreased in PH. 28 Consistent with these findings, we found that VSM cells in CH arteries were depolarized compared to controls, and observed pressure-dependent depolarization in arteries from both groups. Our findings further indicate that increases in intraluminal pressure lead to membrane depolarization and subsequent Ca2+ sensitization and development of tone following CH. Activation of stretch-activated cation channels represents a likely possibility as to how this depolarization occurs and is consistent with the effect of valinomycin to normalize membrane potential between control and CH arteries (Fig. 1). Transient receptor potential channels, for instance, regulate myogenic tone via stretch-dependent depolarization in systemic arteries. 29 However, depolarization alone does not account for the effect of CH, as control arteries depolarized by increasing pressure, or KCl, do not demonstrate Src/EGFR/NADPH oxidase-dependent vasoconstriction. 9 , 10 How this pathway is revealed within the hypertensive pulmonary circulation remains a vital area for future investigation, but may be the result of altered lipid signaling domains following CH. 30
We also examined the role of membrane depolarization in vasoconstrictor responses to ET-1. Consistent with prior studies, 12 we observed no effect of ET-1 to elicit VSM cell depolarization in CH arteries. However, we did detect a modest depolarizing response to ET-1 in control arteries in agreement with previous reports in isolated VSM cells. 12 , 13 In contrast to the development of pressure-dependent tone, depolarization does not appear to contribute to ET-1-dependent Ca2+ sensitization following CH as clamping Vm with valinomycin did not alter vasoreactivity to ET-1 (Fig. 3).
Both membrane depolarization 14 , 15 and mechanical stimuli 16 , 17 can activate GPCRs. Therefore, we examined whether metabotropic signaling of ETRs couple membrane depolarization and mechanical stimuli mediating pressure-dependent tone to Src/EGFR/NADPH oxidase 2-dependent vasoconstriction. However, combined ETAR and ETBR inhibition did not prevent augmented vasoconstriction to KCl in CH arteries, nor did it attenuate pressure-dependent tone. These results suggest that basal tone and enhanced vasoconstriction to depolarizing stimuli occur independent of ETR activation, although they do not preclude a potential role for other GPCRs.
As neither ETRs nor membrane depolarization serve as a central upstream mediator of enhanced Ca2+ sensitization, we sought to examine a potential role for Src kinases in this response. Src family kinases are a large subgroup of non-receptor tyrosine kinases that have similar functions and structures. 31 We have previously identified a role for Src kinases to mediate pressure-dependent tone and augmented vasoconstriction to ET-1 following CH though matrix metalloproteinase and EGFR activation. 10 This response is associated with an increase in phosphorylation of Src kinase at its activation residue, tyrosine 416. 32 Herein, we observe that Src kinases are also required for enhanced depolarization-induced vasoconstriction in rats following CH. The specific Src kinase(s) involved in this response, remains an area for future investigation. However, a likely candidate is c-Src, as it contributes to myogenic tone in systemic arteries 33 and has been linked to RhoA activation in mesangial cells. 19
Although the present study provides insights regarding the signaling mechanisms that link vessel wall stretch, membrane depolarization and ETR, stimulation to enhanced vasoreactivity following CH, further studies are necessary to elucidate how these various stimuli result in Src kinase activation. As GPCRs are capable of Src kinase activation, 34 , 35 enhanced vasoconstriction to ET-1 following CH may result from direct activation of Src kinases via ETRs. Membrane depolarization can also activate Src kinases (Fig. 5 and Ref. 36 ), although whether this occurs from direct activation or via a secondary mediator remains to be identified. Nonetheless, as Src kinases also contribute to the development of monocrotaline-induced vascular remodeling and PH, 37 targeting therapies to Src kinase inhibition represents a potential novel strategy for treating the vasoconstrictor and vascular remodeling components of PH.
Over the last decade, our laboratory has identified a unique signaling pathway in VSM of rats involving Src kinases, EGFR, NADPH oxidase 2, superoxide, and ROK-dependent Ca2+ signaling that mediates enhanced depolarization- and ET-1-dependent vasoconstriction and the development of pressure-induced tone. 6 –10 This signaling pathway is not present within the normotensive pulmonary circulation, and appears to be unmasked only during PH. The present findings demonstrate that increases in intraluminal pressure evoke this pathway through membrane depolarization. Whereas ETRs do not play a role in depolarization-induced vasoconstriction or the development of pressure-dependent tone, Src kinases serve as a central upstream mediator linking various vasoconstrictor stimuli to ROK-dependent Ca2+ sensitization in CH-induced PH.
Footnotes
Acknowledgments
The authors thank Minerva Murphy for technical assistance.
Authors’ contributorship
C.E.N. contributed to study design, performed experiments, analyzed and interpreted data, and drafted and revised the manuscript. B.R.W. and N.L.J. contributed to study design, interpreted data, and revised the manuscript. T.C.R. contributed to study conception and design, supervised study execution, interpreted data, and revised the manuscript.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This work was supported by National Institutes of Health grants R01 HL132883, R01 HL088192, and T32 HL007736 (T.C.R.), R01 HL095640 (B.R.W.), and American Heart Association grant 13PRE14580015 (C.E.N.).
Ethical approval
All protocols and procedures in this study were reviewed and approved by our Institutional Animal Care and Use Committee of the University of New Mexico Health Sciences Center (Albuquerque, NM).
Guarantor
Thomas C. Resta.
