Abstract
This study investigated whether dilated bronchial arteries are associated with reperfusion pulmonary edema in patients with chronic thromboembolic pulmonary hypertension. Results showed that the extent of enlarged bronchial arteries was not associated with the development of reperfusion pulmonary edema, whereas the residual pulmonary hypertension had a significant association.
Introduction
Chronic thromboembolic pulmonary hypertension (CTEPH) is a type of pulmonary hypertension characterized by occlusion of the pulmonary arteries with chronic thrombi. 1 Pulmonary endarterectomy (PEA) is a surgical technique for removing organized thrombi from proximal pulmonary arteries. 2 Reperfusion pulmonary edema (RPE) is a common complication after PEA, and the occurrence is approximately 10–40%. 3 RPE is characterized by hypoxemia and infiltration that appears in reperfused lung areas after PEA3,4 and it can be a risk factor for prolonged mechanical ventilation and hospitalization in intensive care units (ICUs). 3
The bronchial circulation has anastomoses to the pulmonary circulation and plays a role in providing nutrients and maintaining the airway and vessels. 5 Enlarged bronchial arteries (BAs) are commonly observed in CTEPH patients,6,7 and are speculated to be associated with the development of RPE. 4 A transcatheter occlusion of enlarged BAs before PEA can suppress the incidence of RPE, 8 but the detailed measurement data of the BAs was not shown in the paper. Conversely, several reports have demonstrated that enlargement of BAs is related to hemodynamic improvement and a good survival after PEA.9–11 However, data on RPE were not shown in those reports. Thus, the association between enlarged BAs and RPE development remains controversial.
In this study, maximal diameters and cross-sectional areas of BAs were measured using enhanced computed tomography scans. The relationship of BA enlargement to the development of RPE in CTEPH patients has been investigated.
Methods
Details of the methods are described in the Supplemental Methods. Sixty-five patients were diagnosed with CTEPH and underwent PEA between December 2011 and June 2017 at the Chiba University Hospital. Hemodynamic assessment by right heart catheterization was conducted at diagnosis and one to two months (mean: 41.9 ± 15.4 days) after PEA. Eight patients with airway bleeding during PEA were excluded as per the exclusion criteria of RPE. Thus, the clinical data of 57 patients were retrospectively analyzed. RPE was diagnosed according to previously described research criteria. 3 The severity of RPE was assigned according to the required respiratory managements 12 : grade 0 (no RPE); grade 1 (respiratory management with minimal care); grade 2 (non-invasive positive-pressure ventilation and/or nasal high flow); grade 3 (prolonged artificial ventilation or extracorporeal membrane oxygenation); and grade 4 (related to perioperative death). Computed tomography scans at diagnosis with a thickness of 0.5 mm were used for BA evaluation. Two parameters were used to evaluate each patient: (1) the maximal diameter of the BA (BAmax) and (2) the total area of the BAs (TABAs), which was the sum of the cross-sectional areas of the BAs. 6
Results
The characteristics of the 57 patients are shown in Supplemental Table 1. Thirty-four of the 57 patients (59.6%) developed RPE. Thirty-one of the 34 patients (91.2%) developed RPE on the day of PEA, and the remaining three patients (8.8%) developed it on postoperative day 1. The numbers of patients with each grade of RPE were as follows: grade 0: 23 (40.4%); grade 1: 3 (5.3%); grade 2: 20 (35.1%); grade 3: 10 (17.5%); and grade 4: 1 (1.8%). A positive correlation between the RPE grade and ICU stay (
One-hundred twenty-one BAs were identified in the 57 patients (2.1 vessels per patient), and the average diameter of the BAs was 2.2 ± 0.7 mm (range, 1.1–4.9 mm). No differences were found in the BAmax and TABAs between patients with and without RPE (BAmax: Severity of reperfusion pulmonary edema and indicators of bronchial arteries. (a) The maximal bronchial artery diameter (BAmax) and the severity of reperfusion pulmonary edema (RPE). (b) The total area of the BAs (TABAs) and the RPE grade. (c) The BAmax of male and female patients (male, 3.9 ± 0.7; female, 3.2 ± 0.4 mm; 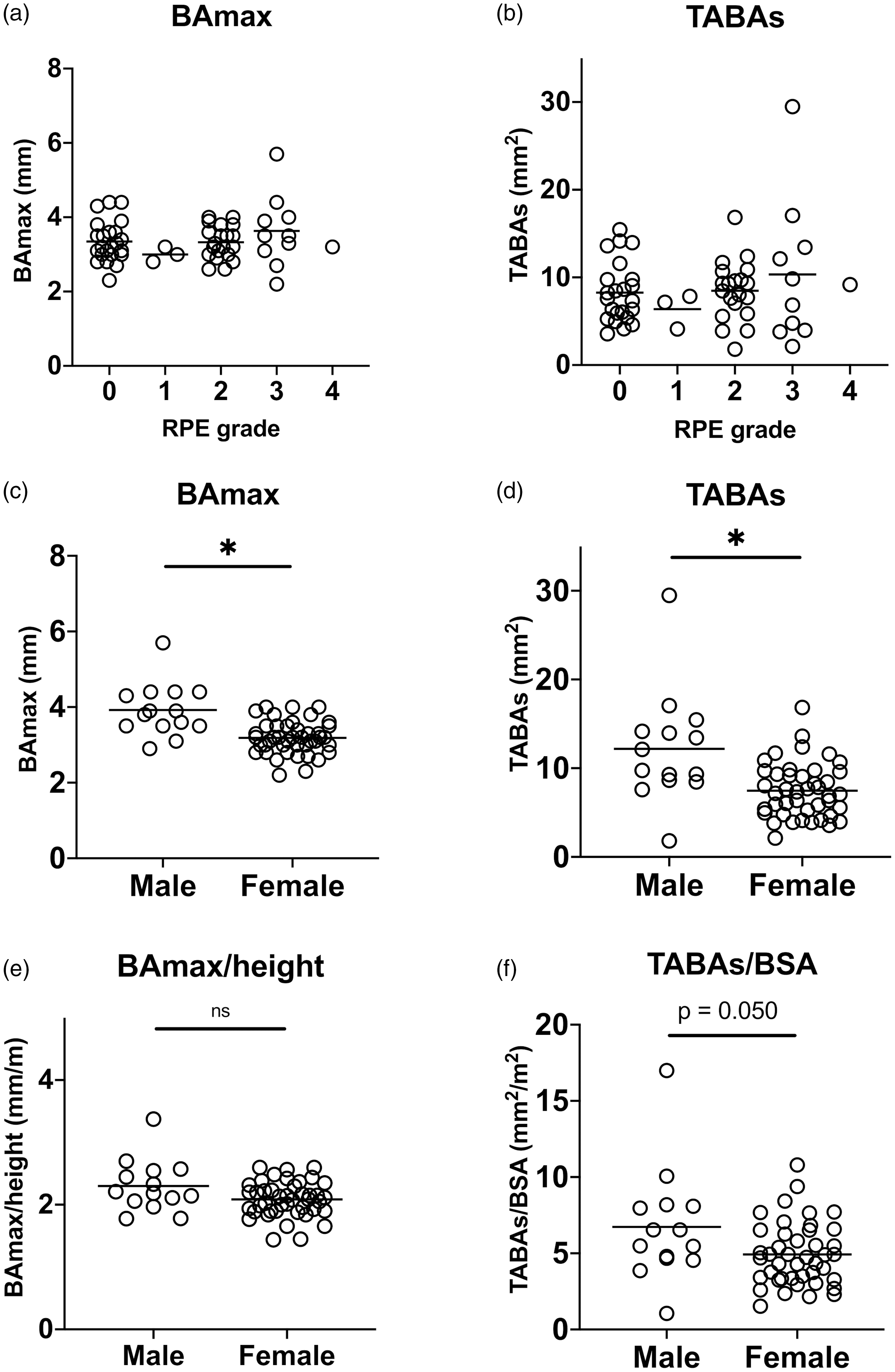
The preoperative mean pulmonary arterial pressure (mPAP) was significantly correlated with the RPE grade (
Logistic regression analyses demonstrated that elevated postoperative PVR was significantly associated with the development of RPE (odds ratio: 4.69, 95% confidential interval: 1.29–17.10,
Discussion
In this study, enlarged BAs were observed in most CTEPH patients; however, the extent of enlarged BAs was not correlated with the development of RPE. Logistic regression analysis confirmed that the association of enlarged BAs with the development of RPE is limited.
No clear association between the development and severity of RPE and enlarged BAs was found in this study. Maintenance of blood supply via bronchial circulation can avoid ischemia of the lung, and alleviate the increased vascular permeability and subsequent RPE. 13 Blood flow through BAs is negatively correlated with the severity of RPE in rabbits that received pulmonary arterial ligation. 14 In CTEPH patients, the blood supply via BAs increases up to 36% of cardiac output, whereas the percentage is <1% in healthy subjects. 15 In this study, even patients with RPE grades 0 or 1 had enlarged BAs, which might contribute to suppression of RPE. Otherwise, grade 3–4 RPE occurred in patients with enlarged BAs, suggesting that maintenance of blood flow via enlargement of BAs could not completely prevent RPE and other factors were involved in the development of RPE.
Our result was inconsistent with that of the study by Gan et al., which suggested that preoperative occlusion of BAs could suppress the development of RPE. 8 This discrepancy could be explained by three factors. First, the criteria for RPE in their report did not exclude other causes of infiltration including pulmonary hemorrhage. It may be that some of their patients had been misdiagnosed with RPE. Infiltration due to pulmonary hemorrhage can be suppressed by BA embolization, because the embolization may decrease the risk of airway bleeding in CTEPH patients. 16 Second, their report did not show detailed BA measurement data. Thus, the impact of enlarged BAs on RPE may have differed between their study and this study. Third, in terms of the indications for embolization of BAs, their study enrolled patients with a history of airway bleeding or active hemoptysis. 8 This may have resulted in potential selection bias and affected the incidence of infiltration. The occlusion of BAs before PEA is a matter of debate; therefore, further investigation is needed.
It is known that residual pulmonary hypertension after PEA is associated with the development of RPE,3,17 which supports our results. Our previous study reported that severe obstruction of muscular pulmonary arteries is associated with residual pulmonary hypertension and the development of RPE. 12 Residual pulmonary hypertension can increase sheer stress in the reperfused lung area, which may induce the development of RPE. 12 Conversely, the relationship between the preoperative hemodynamics and RPE was limited in this study. Some reports have suggested that elevated preoperative mPAP and PVR levels are correlated with the occurrence of RPE,17,18 although the definitions of RPE are different between those reports and the current study. The observed discrepancy may have therefore resulted from the difference of the RPE definition.
Gender-related differences of BAmax and TABAs remain controversial. Esparza-Hernández et al. reported that male patients had a larger BA diameter than female patients 19 ; however, Battal et al. found that the difference was not significant. 20 Our results suggested that the body size could be responsible for the gender-related difference.
This study had several limitations. First, this was a single-center retrospective study. Second, the sample size was small. Third, the number of PEAs performed in our institution was relatively small. Fourth, the decision regarding respiratory management depended on the clinicians in the ICU, which may have affected the severity classification of RPE. Finally, the definition of RPE has not been standardized to date, and these results may differ if a different definition was applied.
In conclusion, the extent of enlarged BAs was not associated with the development of RPE. Characteristics of BAs in CTEPH patients, including those based on gender-related differences, remain limited; therefore, further investigation is needed.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020968677 - Supplemental material for The extent of enlarged bronchial arteries is not correlated with the development of reperfusion pulmonary edema after pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension
Supplemental material, sj-pdf-1-pul-10.1177_2045894020968677 for The extent of enlarged bronchial arteries is not correlated with the development of reperfusion pulmonary edema after pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension by Yumiko Ikubo, Takayuki J. Sanada, Nobuhiro Tanabe, Akira Naito, Hiroki Shoji, Jun Nagata, Ayaka Kuriyama, Asako Yanagisawa, Takayuki Kobayashi, Keiko Yamamoto, Hajime Kasai, Rika Suda, Ayumi Sekine, Toshihiko Sugiura, Ayako Shigeta, Keiichi Ishida, Seiichiro Sakao, Masahisa Masuda and Koichiro Tatsumi in Pulmonary Circulation
Supplemental Material
sj-pdf-2-pul-10.1177_2045894020968677 - Supplemental material for The extent of enlarged bronchial arteries is not correlated with the development of reperfusion pulmonary edema after pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension
Supplemental material, sj-pdf-2-pul-10.1177_2045894020968677 for The extent of enlarged bronchial arteries is not correlated with the development of reperfusion pulmonary edema after pulmonary endarterectomy in patients with chronic thromboembolic pulmonary hypertension by Yumiko Ikubo, Takayuki J. Sanada, Nobuhiro Tanabe, Akira Naito, Hiroki Shoji, Jun Nagata, Ayaka Kuriyama, Asako Yanagisawa, Takayuki Kobayashi, Keiko Yamamoto, Hajime Kasai, Rika Suda, Ayumi Sekine, Toshihiko Sugiura, Ayako Shigeta, Keiichi Ishida, Seiichiro Sakao, Masahisa Masuda and Koichiro Tatsumi in Pulmonary Circulation
Footnotes
Acknowledgements
The authors would like to thank Editage for their English editing.
Ethical approval
This study was approved by the Ethics Committee of Chiba University (approval number 826).
Guarantor
Takayuki J. Sanada is the guarantor of the data shown in this paper.
Contributorship
T.J.S. designed this research studies. Y.I., N.T., A.N., H.S., J.N., A.K., A.Y., T.K., K.Y., H.K., R.S., A.S., T.S., A.S., K.I., S.S., M.M., and K.T. contributed to the data acquisition, analysis, and interpretation of the data. T.J.S. and Y.I. wrote the draft and the other authors contributed to revising the manuscript. All authors approved the final version.
Conflict of interest
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: T.J. was a member of an endowed department sponsored by Actelion Pharmaceuticals Ltd. and received a scholarship from MSD Life Science Foundation. N.T. was a member of an endowed department sponsored by Actelion Pharmaceuticals Ltd. and received personal fees from Actelion Pharmaceuticals Ltd., Bayer AG, the Daiichi-Sankyo Company, and Nippon Shinyaku Co., Ltd. S.S. received lecture honoraria from Nippon Shinyaku Co., Ltd, Bayer AG, Actelion Pharmaceuticals, and Pfizer. A.S. was a member of an endowed department sponsored by Actelion Pharmaceuticals Ltd. and received a scholarship from the Uehara Memorial Foundation. K.T. received lecture honoraria from Actelion Pharmaceuticals and Nippon Boehringer Ingelheim. The other authors had no potential conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was supported by the Japanese Ministry of Health, Labor and Welfare research grants specifically designated to the Respiratory Failure Research Group and Cardiovascular Diseases and the Pulmonary Hypertension Research Group from the Japan Agency for Medical Research and Development (No. 16ek0109127h0002). The funder had no role in the study’s design, collection of data or their analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
