Abstract
Pulmonary Hypertension due to left heart disease is the most common type of Pulmonary Hypertension. Morbidity and mortality significantly increase once Pulmonary Hypertension is present. Treatment is aimed toward optimizing the underlying condition. Targeted therapy has been evaluated in small studies with mixed results. The goal of this systematic review is to identify the possible benefit and safety of Phosphodiesterase 5 inhibitors in Pulmonary Hypertension due to left heart disease with elevated pulmonary vascular resistance, diagnosed by right heart catheterization. Electronic searches using MEDLINE/PREMEDLINE, EMBASE, and The Cochrane Library were searched on 21 October 2018. Randomized clinical trials comparing Phosphodiesterase 5 inhibitors versus placebo in patients with proven Pulmonary Hypertension by right heart catheterization secondary to left heart disease (both heart failure with reduced ejection fraction and with preserved ejection fraction) and reported pulmonary vascular resistance were included. We identified 436 potentially relevant studies. After reviewing the titles and abstracts to exclude irrelevant articles, five randomized clinical trials were considered for the study. Sildenafil was well tolerated among all studies. Sildenafil was found to improve hemodynamics, exercise capacity, and quality of life in patients with elevated pulmonary vascular resistance. Phosphodiesterase 5 inhibitors therapy in patients with proven Pulmonary Hypertension due to left heart disease and elevated pulmonary vascular resistance by right heart catheterization may improve the quality of life, exercise capacity, and pulmonary hemodynamics. Further prospective randomized controlled studies are needed to confirm.
Pulmonary Hypertension due to left heart disease (PH-LHD) is the most common type of Pulmonary Hypertension (PH). PH-LHD results from heart failure (HF), with both reduced and preserved ejection fraction (EF) and valvular heart disease (VHD). 1 The prevalence of PH in patients with heart failure with reduced ejection fraction (HFrEF) in right heart catheterization (RHC) studies has been estimated to range from 40% to 75%. In heart failure with preserved ejection fraction (HFpEF), studies using either echo or RHC indicated a PH prevalence from 36% to 83%. 2 Prevalence was 25% in a recent RHC cohort. 3
Once PH develops in patients with left heart disease, morbidity and mortality increase significantly, with a negative impact on prognosis of the disease. 4 PH has traditionally been divided into post-capillary and combined pre- and post-capillary with definitions varying depending on diastolic pressure gradient (DPG), transpulmonary gradient (TPG), and pulmonary vascular resistance (PVR). The most recent world symposium simplified the definition of combined post-capillary and pre-capillary PH based only on the elevation of PVR > 3 Woods Units (WU). 5 This definition is based on a recent meta-analysis which showed that elevated PVR is associated with worse outcomes and poor prognosis. 6 A recent large cohort confirmed that TPG, DPG, and PVR were predictive of mortality and cardiac hospitalizations. 3 The world symposium strongly urges further study for new therapies in patients with PH-LHD with particular interest in patients with elevated PVR. Experts note there is an urgent need for multicenter trials in patients with Congestive Heart Failure with preserved Ejection Fraction (CHFpEF) who must be hemodynamically well characterized by RHC. 7
Current treatment recommendations for PH-LHD are aimed toward optimizing the underlying condition. Lack of evidence and safety concerns are the reasons why current guidelines do not recommend targeted PH therapy for patients with PH-LHD.5,8 Despite these recommendations, a survey of 30 US PH referral centers found that 77% of the centers prescribed Pulmonary Arterial Hypertension (PAH) therapy for PH-LHD. 9
Targeted therapy for PH-LHD with prostanoids and endothelin receptor antagonists has not shown benefit in patients with HFrEF and HFpEF, and, in fact, has shown an increase in side effects and possibly increased mortality.10–12
Phosphodiesterase 5 inhibitors (PDE5i) increase nitric oxide-mediated vasodilation in patients with congestive HF, and experimental studies have shown improvement in cardiac and pulmonary hemodynamics. 13
Several randomized controlled trials (RCTs) in PH-LHD with PDE5i have shown mixed results. Limitations of these studies include small sample size, single-center studies, and heterogeneous populations. More importantly, very few studies have directed the treatment to specific populations based on hemodynamic PH classification and PVR.
The goal of this systematic review (SR) is to identify the possible benefit and safety of PDE5i in PH-LHD secondary to HF (preserved and reduced ejection fraction) based on PVR > 3 on RHC.
Methods
Literature search strategy
We performed a SR. We followed the recommended guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses. 14
Electronic searches using MEDLINE/PREMEDLINE (EBSCOhost interface), EMBASE (embase.com interface, 1974–present), and The Cochrane Library were searched on 21 October 2018. In the individual database search strategies, each of the four search concepts—(1) PDE5 inhibitors, (2) PH, (3) congestive HF, and (4) randomized controlled trials—was represented by a combination of indexing terms and keywords. Keywords for the drug concept included alternate names for the PDE5 inhibitor class, names for the individual drugs included in the class (obtained from the EMTREE thesaurus), and alternate names for these individual drugs (the English- and foreign-language generic names, any commonly used synonyms for the drug names, investigational names, and U.S. and foreign proprietary names obtained from Martindale: The Complete Drug Reference). Terms for the “randomized controlled trials” concept were based on the validated RCT filter recommended, by the Canadian Agency for Drugs and Technology in Health. The exact strategies used are shown in Online Appendix 1. When possible, searches were limited to articles with English-language full-text and articles indexed as concerning animals that were not also indexed as concerning humans were removed.
Selection criteria
Eligibility criteria were defined a priori. Inclusion criteria for analysis were: (1) RCT comparing a single active agent versus placebo control; (2) active agent belonging to PDE5i class; (3) only proven PH patients with mean pulmonary artery pressure (mPAP) > 25 mm Hg by RHC secondary to HF (HFrEF and HFpEF); (4) reported PVR. Exclusion criteria were: (1) single-dose RCTs using PDE5i; (2) inclusion of patients with VHD as the cause of PH; (3) RCTs using non-invasive measures (ECHO) for diagnosis of PH.
Assessment of methodological quality
Two authors using the Cochrane’s Risk of Bias Assessment Tool evaluated the selected studies independently. High, low, and unknown risk of bias was used for each category. Each study was considered to have a high or unknown risk of bias if they had at least one category with high or unknown risk of bias. 15
Data extraction
One investigator performed title, abstract screening, and data extraction. The senior author reviewed data extraction. The primary outcomes of interest were hemodynamic effect of PDE5i, exercise capacity, quality of life (QoL), and adverse events. Hemodynamics included mPAP, pulmonary capillary wedge pressure (PCWP), and PVR. Exercise capacity was assessed by peak VO2. Different questionnaires for QoL measures were accepted for analysis. When available, 6-min walk testing (6MWT), biomarkers, cardiac output (CO), Cardiac Index (CI) were considered for analyses.
Results
Eligible studies
We identified 436 potentially relevant studies: 263 from EMBASE, 56 from MEDLINE/PREMEDLINE, and 117 articles from the Cochrane Library. After 92 duplicate studies were removed, 344 studies were chosen for further analysis. From these studies, five were excluded due to language other than English. We also searched for additional articles using the bibliographies of each included study and any review articles that we retrieved. After reviewing the titles and abstracts to exclude irrelevant articles, 31 studies were reviewed in detail, 26 of which were rejected (3 SRs and meta-analysis, 12 randomized clinical trials including targeted therapy other than PDE5i, 3 RCTs which addressed only acute changes by PDE5I, 2 retrospective studies, 5 RCTs which did not include hemodynamic measures, and 1 RCT done in patients with PH-LHD associated to valvular disease). Among the five RCTs that met the pre-specified inclusion criteria, two studies were performed in HFrEF16,17 and three studies involved patients with preserved EF (HFpEF).18–20
Despite the known differences in hemodynamics and pathophysiology in patients with HFrEF and HFpEF, the goal of this review was to identify patients with HF and PH with an elevated PVR by RHC and assess their response to treatment with targeted therapy (Fig. 1).
Search flow diagram for RCTs included in the systematic review. RCT: Randomized clinical trials; RHC: right heart catheterization; PDE5i: phosphodiesterase 5 inhibitors.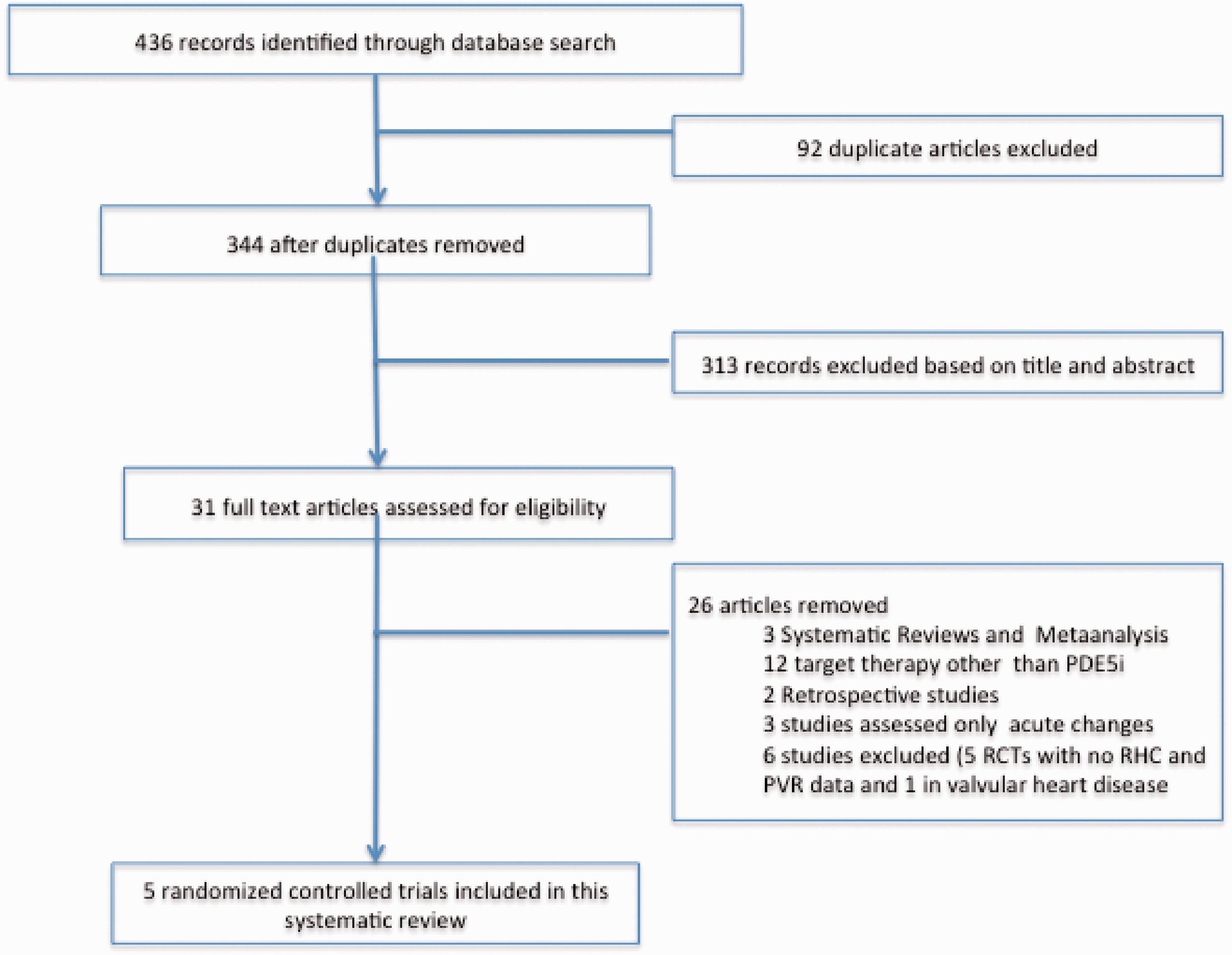
Four studies had a low risk of bias, one had unknown risk of bias (due to allocation concealment and possible conflict of interest), and there were no studies with high risk of bias (Fig. 2) (Online Appendix 2).
Summary of risk of bias analysis.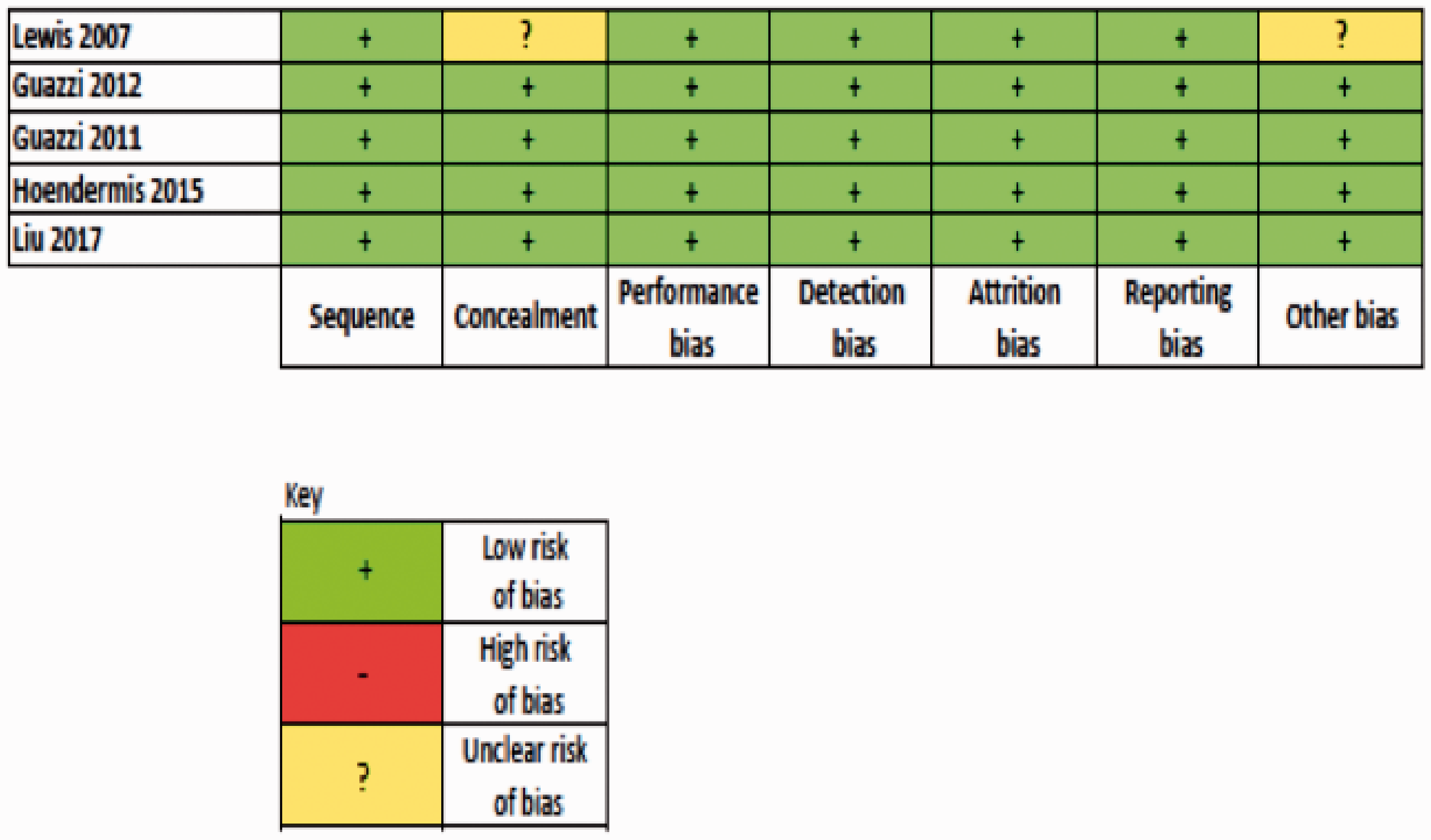
A total of 162 patients were included in this analysis. Eighty-one patients received sildenafil three times a day at different doses, and 81 patients were part of the control group. Patients were followed up for at least 12 weeks and up to one year. All the patients in this review had a diagnosis of HF (either HFrEF or HFpEF), PH based on hemodynamic data, and reported PVR for analysis.
Two studies in HFpEF18,19 had the same patients, but the hemodynamic parameters were reported in a first study, 18 and secondary outcomes including cardiac structure and function, cardiopulmonary exercise testing, health-related QoL measures were reported in a different study. 19
Hemodynamic parameters
All the studies included for this review reported hemodynamic data through invasive right heart cardiac catheterization.16–18,20 PVR, mPAP, and PCWP were reported in all the studies. Only one study reported pulmonary diastolic pressures.
Out of the four studies, mean PVR was elevated at more than 3WU in three studies.16,17,20 The fourth study had a PVR with a mean of 2.25 WU 18 but had a subset of patients with PVR > 3. Within the three articles with an elevated PVR, Sildenafil decreased PVR, PVR/SVR in all three studies. In 34 patients randomized to placebo or sildenafil for 12 weeks, the PVR in the placebo group went from 4.5 WU to 4.25, while the PVR in the sildenafil group decreased significantly from 4.25 WU to 3.5 WU. 16 In 32 patients with CHFrEF, sildenafil reduced PVR from 4.5 WU to 3.32 WU while there was no change in the PVR of the placebo group. 17 In the study with 44 patients with CHFpEF, the PVR decreased from 3.88 to 1.00 in the sildenafil group and increased from 3.27 to 3.96 in the placebo group. 20 Decreases in mPAP and PCWP were reported in two studies.17,20 Only one study showed an increase in CI. 20 None of the three studies showed that sildenafil had an impact on Mean Arterial Pressure or Heart Rate (HR). The one study with PVR less than 3 WU 18 found that sildenafil had no change in PVR, PAP, CI, or CO and, in fact, PCWP decreased in the placebo group. This study had a subgroup with patients who had elevated PVR, which constituted 35% of the study population (eight patients in the treatment arm). Separate results for this very small group were reported: sildenafil did not reduce PAP after 12 weeks of treatment.
All studies reported a TPG of 12 to 14 in these patients with Congestive Heart Failure (CHF) and PVR > 3WU. Only one study reported diastolic pulmonary pressures, and when DPG was calculated in this study, it was significantly reduced with treatment with sildenafil. 20
Exercise testing and 6 MWT
Two studies used upright cycle ergometry,16,17 one study used a treadmill for assessment of exercise capacity, 19 and one study did not report exercise capacity results. 20
Sildenafil increased peak VO2 in the two studies where PVR was elevated at baseline.16,17 One of these studies also showed an increase in CO and 6MWT distance. 16 The other study showed a decrease in VE/VCO2 slope. 17
The only study where patients had a PVR less than 3 WU, sildenafil did not change peak VO2 or VE/VCO2 slope. There was a significant decrease in peak HR and in systolic and diastolic blood pressure in patients who received sildenafil. 19
Quality of life
Unfortunately, all the studies evaluated QoL with different questionnaires and tools.
In the three studies of patients with PH and elevated PVR, sildenafil compared to placebo improved QoL measures. Two studies used Chronic Heart Failure Questionnaire (CHQ) with focus on emotional function, breathlessness, and fatigue. Sildenafil improved all of these symptoms.17,20 One study used the Minnesota Living With Heart Failure questionnaire, which showed the score decreased in patients taking sildenafil compared to placebo. This study also showed that New York Heart Association functional class improved in patients taking sildenafil. 16
The only study with patients with PH and PVR < 3WU assessed QoL based on the Kansas City cardiomyopathy questionnaire. The study found improvement in both groups but no difference between sildenafil and placebo. 19
Safety
Overall, sildenafil was well tolerated in all the studies. Withdrawal from the studies did not differ between sildenafil and placebo.
Known adverse effects of sildenafil (headache, flushing, orthostatic hypotension, increased erection, and dyspepsia) were more common in the sildenafil group.16–18
One study showed that compared to placebo, the sildenafil group had fewer HF exacerbation hospitalizations. 16 In two studies, patients in the placebo group were more likely to require up titration of diuretics. In the sildenafil group, down titration of diuretics was more common.17,20 These findings were reported in the three studies in patients with PVR > 3 WU.
Discussion
The present SR, which evaluated patients with group 2 PH due to HF with elevated PVR by RHC, showed that treatment with sildenafil improves exercise capacity, QoL, and hemodynamics with no difference in side effects compared to placebo. The study by Hoendermis et al. had a subgroup of patients with elevated PVR where no difference was found in hemodynamics. This subgroup only had eight patients who received sildenafil, which makes the sample too small to draw conclusions or find the benefit that was seen in other larger studies.
Pathophysiology in PH-LHD due to HF involves two pathways: first there is a passive increase in left atrial pressure with backward transmission of filling pressures resulting in venous congestion in the pulmonary circulation causing an isolated post-capillary PH. Then, vascular remodeling may occur with hypertrophy and fibrotic changes in the pulmonary vasculature, which is characterized by an increase in PVR > 3 WU. A reduction of endothelium-derived Nitric Oxide and elevated levels of endothelin-1 is involved in the development of endothelial dysfunction and vascular remodeling that leads to intimal fibrosis and medial hypertrophy.2,21 These pathophysiological changes in the pulmonary circulation could explain the benefits of sildenafil seen in patients with HF who have developed Pulmonary Vascular Disease with a PVR > 3 with no improvement seen in patients with PVR < 3.
In these studies, the population with elevated PVR was more likely to be older and have HFrEF, hypertension, and not a particularly high body mass index (BMI). In other studies of pulmonary venous hypertension, the phenotype has been older females with diabetes, diastolic dysfunction, and obesity. While it is possible that this particular phenotype could play a role in the response to sildenafil, it is PVR that has been recognized as a marker of prognosis and with worse outcomes in this population. 6 Therefore, from these studies, treatment with sildenafil in patients with elevated PVR may be beneficial despite how different their phenotype may be.
Several non-invasive studies have assessed the effect of PDE5i in patients with HF (either reduced or preserved EF) and PH13,22–26 with conflicting results. The common weakness in all of these papers is that ECHO was used to evaluate for PH. This elucidates the uncertainty of objective PH and elevated PVR without a RHC in these patients. There is clear evidence on how poorly echo measures correlate to RHC data.27,28 This weakness likely explains the conflicting results and elucidates the difficulty of trying to find the ideal group of patients who could benefit from PDE5i therapy.
Charles-Antoine Guay et al. performed a SR and meta-analysis to assess the effect of PH-targeted therapies on exercise capacity in patients with group 2 PH. They included 22 studies, with only 10 PDE5i studies. From these 10, only 5 had hemodynamic measures and 1 included patients with VHD. The authors reported significant improvement in exercise capacity; this finding was driven by the subgroup of patients receiving PDE5i. A decrease in pulmonary pressures was also noticed in patients treated with PDE5i. An exploratory analysis suggested PDE5i may be associated with a decrease in cardiac hospitalizations in some patients with HF but not in patients with valvular disease. 29 These findings are consistent with our study despite differences in patient population (echo diagnosis of PH and inclusion of VHD with no mention of PVR in their SR). The authors also reported a significant increase in treatment discontinuation, with no mention in the subgroup population taking PDE5i. This finding is different than our study and is likely related to the other therapies included in this trial.
Jacob Y Cao et al., in their SR and meta-analysis, assessed the effects of all types of advanced therapy in group 2 PH, 30 including one study in patients with VHD. They also included two non-invasive studies, one of them using Udenafil. This study reported no difference in all cause and cardiovascular mortality but stated that numerically these outcomes were higher in the advanced therapy group. At the same time, the study reported that HF exacerbation was not statistically different, but in a subgroup analysis the numbers favored patients treated with PDE5i which is consistent with our findings. Right heart hemodynamics and functional capacity improved in the treated group without achieving significance. The difference in outcomes in this study compared to ours may be related to the inclusion of VHD patients and therapies other than PDE5i.
Our findings correlate with several hemodynamically well-characterized cohorts of HF patients. In a cohort of 226 patients with RHC confirmed CHFpEF with mean PVR 7 WU who were treated with targeted PH therapy (mostly PDE5i), an improvement in 6MWT, functional class, and natriuretic peptides was found but they had a higher medication discontinuation rate of 18%. 31 In another retrospective cohort of 40 patients with CHFpEF who were optimally diuresed and tolerated sildenafil for a year, there was again an improvement in 6MWT, N-terminal prohormone of Brain Natriuretic Peptide (NTproBNP), and World Health Organization Functional Class (WHO-FC). 32
The main limitation of the current SR includes the small sample of patients included in the studies. All the RCTs included for the review shared the common flaw of small samples, heterogeneity of patient demographics, duration, and dosage of medication.
The phenotype of HF with PH warrants further evaluation. Interestingly, in these RCTs, where PVR was elevated, the patients were mainly hypertensive males, a Caucasian population, and were overall less sick when compared to the two RCTs where the majority of the population had a PVR less than 3 WU, which included females, higher BMI, patients with atrial fibrillation, diabetes, and hypercholesterolemia. All these differences may influence the results of the study.
It is also important to mention that follow up for a year was done in only two studies, making safety and adverse events difficult to evaluate in the review given that three studies only followed up patients for 12 weeks. Sildenafil dose was different in all the RCTs included, and in all trials the dose was higher than the usual prescribed dose for patients with PAH.
Characteristics of included studies.
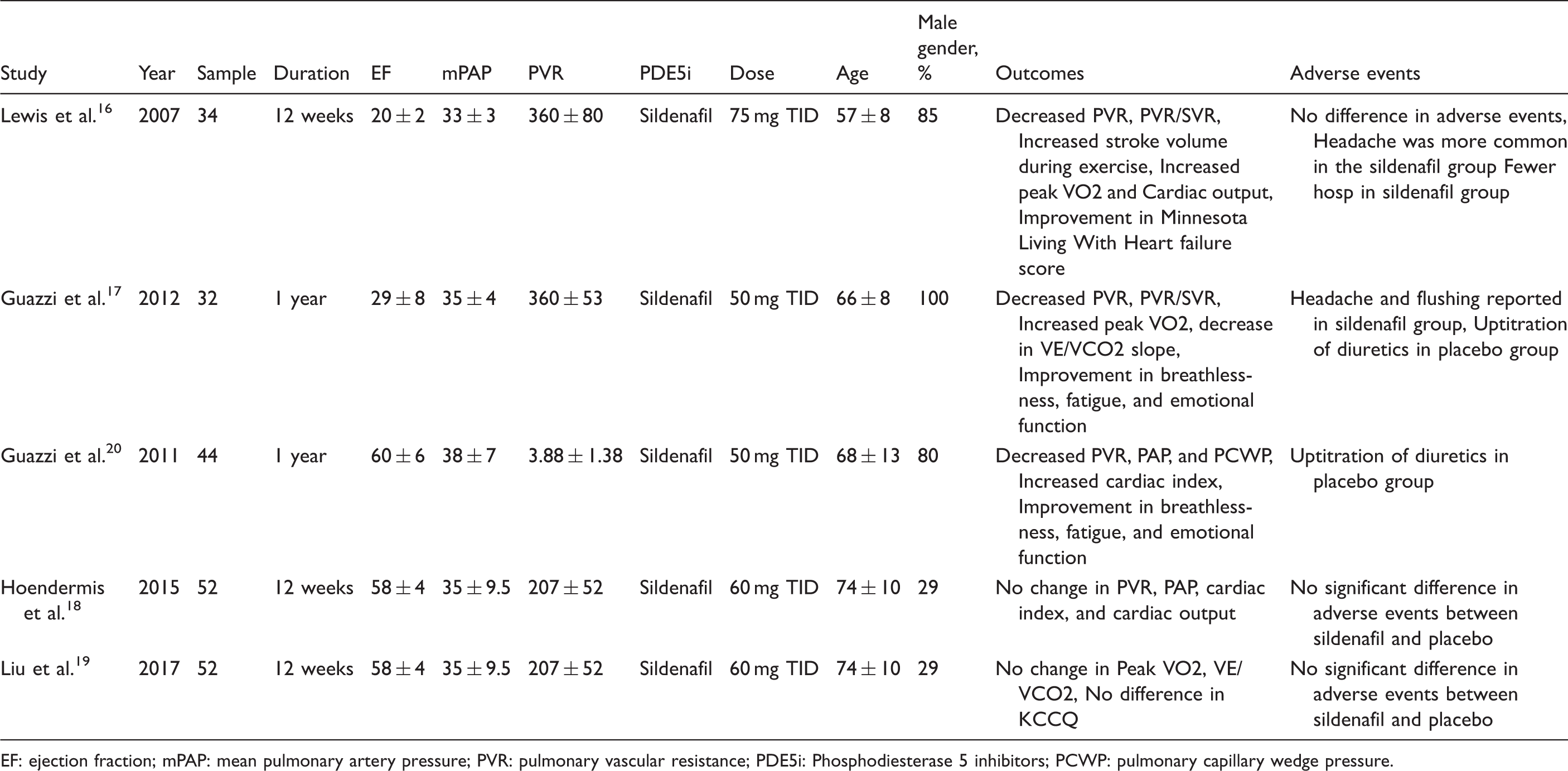
EF: ejection fraction; mPAP: mean pulmonary artery pressure; PVR: pulmonary vascular resistance; PDE5i: Phosphodiesterase 5 inhibitors; PCWP: pulmonary capillary wedge pressure.
Supplemental Material
sj-xlsx-1-pul-10.1177_2045894020948780 - Supplemental material for Targeted therapy with phosphodiesterase 5 inhibitors in patients with pulmonary hypertension due to heart failure and elevated pulmonary vascular resistance: a systematic review
Supplemental material, sj-xlsx-1-pul-10.1177_2045894020948780 for Targeted therapy with phosphodiesterase 5 inhibitors in patients with pulmonary hypertension due to heart failure and elevated pulmonary vascular resistance: a systematic review by Galo M. Sanchez Palacios, Cindy Schmidt and Tammy Wichman in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Guarantor
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
