Abstract
Pulmonary hypertension in interstitial lung diseases is associated with increased mortality and hospitalizations and reduced exercise capacity. Interstitial pneumonia with autoimmune features (IPAF) is a recently described interstitial lung disease. The characteristics of pulmonary hypertension in IPAF patients are unknown. We sought to characterize patients with IPAF based on their echocardiographic probability of pulmonary hypertension and compare patients with and without pulmonary hypertension identified by right heart catheterization. We conducted a retrospective study of patients seen in the interstitial lung disease clinic from 2015 to 2018. Forty-seven patients with IPAF were identified. Patients were classified into low, intermediate and high echocardiographic pulmonary hypertension probabilities. A sub-group analysis of patients with pulmonary hypertension and without pulmonary hypertension (IPAF-PH vs. IPAF-no PH) identified by right heart catheterization was also performed. Linear regression analysis was performed to study the association between 6-min-walk-distance (6MWD) and pulmonary vascular resistance (PVR) while adjusting for age and body mass index. Right ventricular hypertrophy (>5 mm), right ventricular enlargement (>41 mm) and right ventricular systolic dysfunction defined as fractional area change% ≤35 was present in 76%, 24%, and 39% of patients, respectively. Pulmonary hypertension was identified in 12.7% of patients. IPAF-PH patients had higher mean pulmonary artery pressure and lower cardiac output compared to the IPAF-no PH group (34 mmHg vs. 19 mmHg, p = 0.002 and 4.0 vs. 5.7 L/min, p = 0.023, respectively). Lower 6MWD was associated with higher PVR on regression analysis (p = 0.002). Pulmonologists should be aware that a significant number of IPAF patients may develop pulmonary hypertension. Reduced 6MWD may suggest the presence of pulmonary hypertension in IPAF patients.
Keywords
Introduction
The prevalence of pulmonary hypertension (PH) in interstitial lung diseases (ILDs) is approximately 14%. 1 Interstitial pneumonia with autoimmune features (IPAF) is a recent denomination for undifferentiated forms of connective tissue disease (CTD) with associated ILD endorsed by the American Thoracic Society and European Respiratory Society. 2 While the risk of developing PH in well-defined ILDs such as idiopathic pulmonary fibrosis (IPF) or CTD-ILDs has been well described, the risk of PH in patients with IPAF remains uncertain.
Patients with ILDs who develop PH have lower 6-min walk distances (6MWD), more rapid decline in right ventricular systolic function, and higher mortality compared to their ILD counterparts without PH.1–5 Even mild elevation in mean pulmonary artery pressures (mPAP) ≥21 mmHg in IPF patients is associated with lower survival. 3 Since little is known about the features of PH in patients with IPAF, we sought to characterize patients with IPAF who also have PH and identify factors associated with the presence of PH. Our preliminary findings were presented as an abstract at American Thoracic Society International Conference 2020 (https://www.atsjournals.org/doi/pdf/10.1164/ajrccm-conference.2020.201.1_MeetingAbstracts.A3391).
Methods
Study subjects
The study was approved by The University of Florida’s Institutional Review Board (protocol no. 201802309). We conducted a retrospective review of IPAF patients evaluated at The University of Florida’s Interstitial Lung Disease Clinic between January 2015 and December 2018. To identify patients with IPAF, we queried our institution’s integrated data repository database and compiled a list of all patients with the following International Classification of Diseases (ICD-10) diagnoses: interstitial pulmonary disease (J84.9), other specified interstitial pulmonary diseases (J84.89), other systemic involvement of connective tissue (M35.8), and pulmonary hypertension due to lung diseases and hypoxia (I27.23). The diagnosis of IPAF was made per the American Thoracic Society’s and European Respiratory Society’s consensus statement. 2 Namely, patients had to have the following (1) evidence of interstitial pneumonia on computed tomography (CT) scan of the chest and/or lung biopsy, (2) exclusion of other causes, (3) does not meet criteria for a known connective tissue disease and at least one point from at least two of three domains: clinical domain (specific extra thoracic manifestations), serologic domain (specific serum antibodies), and morphologic domain (suggestive radiologic or histopathologic patterns). 2 Two pulmonologists with expertise in ILDs (D.G.M and D.P) independently verified all patients’ diagnosis of IPAF. A patient was not included in the final cohort if there was disagreement between the reviewers regarding the diagnosis.
Classification based on echocardiographic PH probability
We classified IPAF patients into low, intermediate, and high PH probability based on Doppler echocardiographic measurement of tricuspid regurgitation velocity (TRV) and right ventricle to left ventricle (RV/LV) ratio measured at the base: TRV ≤2.8 meters/second (m.s−1) or not measurable as low, TRV 2.9–3.4 m.s−1 or TRV ≤2.8 m.s−1 with RV/LV >1 as intermediate, TRV >3.4 m.s−1 or TRV 2.9–3.4 m.s−1 with RV/LV >1 as high probability. 6
IPAF patients with PH vs. IPAF patients without PH
We then classified IPAF patients with right heart catheterization (RHC) data into IPAF patients with and without PH (IPAF-PH vs. IPAF-no PH) and performed a subgroup analysis. IPAF-PH patients were defined using the 2018 world symposium on PH (WSPH) consensus to define PH in patients with chronic lung diseases: IPAF-PH as those with mean pulmonary artery pressure (mPAP) 21–24 mmHg with pulmonary vascular resistance (PVR) ≥3 Wood units (WU) or mPAP ≥25 mmHg. 7 The IPAF-no PH group was defined as patients with mPAP <21 mmHg or mPAP 21–24 mmHg with PVR <3 WU. 7 Pre-capillary pulmonary hypertension was defined according to the 2018 WSPH consensus as mPAP ≥21 mmHg, pulmonary arterial wedge pressure (PAWP) ≤15 mmHg, and PVR ≥3 Wood units by RHC. 8
Clinical data
We collected pulmonary function tests (PFTs), 6MWD, Borg dyspnea scores at rest and exertion, oxygen requirement at rest and exertion, number of hospitalizations, main pulmonary artery size on CT scan of the chest, transthoracic echocardiography (TTE) parameters and RHC data for the entire IPAF cohort. All tests were performed within 12 months of diagnosis of IPAF. If multiple tests were performed within 12 months of the IPAF diagnosis, data closest to the time of diagnosis was used. The mean time difference between obtaining PFTs and IPAF diagnosis, TTE and IPAF diagnosis, and TTE and RHC was 104 ± 141 days, 159 ± 189 days and 212 ± 209 days, respectively. We compared the clinical data between the three echocardiographic PH classes and then between IPAF-PH versus IPAF-no PH groups.
Pulmonary function tests
We obtained spirometry, plethysmography, and diffusion capacity for carbon monoxide (DLCO) data (Zan 500 Body, nSpire Health Inc., Louisville, Colorado, USA). Post bronchodilator values were used when available. Tests were performed according to ATS guidelines and predicted values according to the Third National Health and Nutrition Examination Survey were used. 9 PFT values were not available in 1 (2%) patient.
6MWDs and Borg dyspnea scores
The 6MWDs were performed according to ATS guidelines on a hard, flat 30 m track. 10 Borg dyspnea scores and oxygen requirements at rest and exertion were obtained before and after the 6MWD.
Pulmonary artery diameter
We measured the widest diameter perpendicular to the long axis of the main pulmonary artery at the level of the bifurcation of the pulmonary artery as noted on CT scan of the chest. 11
Echocardiography
Standard transthoracic echocardiography was performed utilizing a Philips EPIQ 7 system (Philips Healthcare, Andover, MA, USA). Two cardiologists with expertise in echocardiography and PH (R.H and T.L) reviewed the exams and measured standard right ventricle (RV) measurements including basal and mid RV diameter and right atrial (RA) area and volume from the apical four chamber view. The presence of RV hypertrophy was assessed by measuring the RV free wall thickness in the subcostal view. RV function was assessed using multiple methods. Fractional area change% (FAC%) was calculated by subtracting the end-systolic area from the end-diastolic area and then dividing the resulting value by the end-diastolic area, all measured in an apical four chamber view. The RV index of myocardial performance (RIMP) was calculated by measuring the pulmonic valve ejection time (ET) using pulse-wave Doppler in the right ventricular outflow tract (RVOT) and tricuspid closure to opening time using continuous-wave Doppler on the tricuspid regurgitation (TR) jet as described in the American Society of Echocardiography right heart guidelines. 12 The tricuspid annular plane systolic excursion (TAPSE) was measured using M-mode tracing of the tricuspid annulus obtained in the apical four chamber view. Right ventricular systolic pressure (RVSP) was estimated by adding the peak TR jet gradient to the estimated RA pressure obtained using standards defined by the inferior vena cava diameter and inspiratory collapsibility. The presence of mid-systolic notching of the RVOT Doppler profile was noted if present. 13 Assessment of left ventricular filling pressures was performed by measuring the ratio of the peak mitral inflow E-wave velocity obtained from the apical four chamber view to the averaged medial and lateral mitral annular tissue Doppler-derived e' velocity.
Right heart catheterization
RHC was performed in patients undergoing a lung transplant evaluation or if they had high echocardiographic probability of PH. RHC was performed via the internal jugular vein or femoral vein approach using a 7-French balloon tipped catheter. All variables were measured at end-expiration. The variables included right atrial pressure, right ventricular systolic and diastolic pressures, pulmonary artery systolic and diastolic pressures, mPAP, PAWP, cardiac output (CO) and mixed venous oxygenation of a blood sample obtained from the main pulmonary artery. CO was determined either by thermodilution or the indirect Fick method described by LaFarge et al. 14 PVR was calculated as the difference between mPAP and PAWP divided by CO and expressed in WU.
Statistics
We summarized the distribution of baseline characteristics as percentages for categorical variables and medians with interquartile range (IQR) for continuous variables. Kruskal–Wallis H or Mann–Whitney U tests were used as indicated to compare continuous variables between low, intermediate, and high echocardiographic PH probability groups and between IPAF-PH and IPAF-no PH groups, respectively. Fisher’s exact test was used to compare categorical variables between different groups. To identify non-invasive parameters that could identify the presence of PH, we examined whether there was any association between DLCO, 6MWD, TAPSE, RIMP and FAC% and PVR using linear regression analysis. A recent study found TAPSE and FAC% were associated with PVR in chronic lung diseases. 15 We adjusted for age, and BMI as these could impact 6MWD independently. 16 A two-sided p-value <0.05 was considered statistically significant. All analyses were conducted using IBM SPSS Statistics for Windows, Version 23.0 (released 2015, IBM Cor, Armonk, NY).
Results
A total of 561 patients were identified using the ICD codes listed. Forty-seven (8%) patients met the IPAF criteria based on ATS-ERS consensus statement. Other diagnoses were as follows: undifferentiated ILD (18%), CTD-ILD (34%), IPF (12%), sarcoidosis (7%), chronic hypersensitivity pneumonitis (7%), and other ILDs (14%). Other ILDs included radiation pneumonitis, organizing pneumonia, drug-related ILD, ILD due to aspiration, and combined pulmonary fibrosis and emphysema.
Clinical characteristics are summarized in Supplementary Table 1. In our cohort, morphologic criteria, serologic criteria, and clinical criteria for IPAF were present in 94%, 89% and 32%, respectively. Sixty-six percent of patients had nonspecific interstitial pneumonia (NSIP) pattern on CT chest. The most common serologic antibody was anti-nuclear antibody which was present in 31/47 (66%) of patients. Thirty-nine (83%) had an echocardiogram and 16 (34%) had RHC (one patient had RHC performed with no echocardiogram data available). The mean 6MWD in the entire cohort was 335 ± 132 m. Twelve patients (25.5%) had at least one hospital admission for ILD exacerbation during the study period and five (10.6%) required intensive care unit stay.
Echocardiographic PH probability groups
Patients were classified into low, intermediate, and high echocardiographic probability of PH based on TRV and right ventricle to left ventricle (RV/LV) ratio measured at the base: TRV ≤2.8 m.s−1 or not measurable as low, TRV 2.9–3.4 m.s−1 as intermediate, TRV >3.4 m.s−1 or TRV 2.9–3.4 m.s−1 with RV/LV > 1 as high probability.
Kruskal–Wallis test was used to compare continuous variables and Chi-square test was used for categorical variables.
Percent predicted valves are reported for FEV1, FVC, TLC, and DLCO.
Pulmonary hypertension per the WSPH definition of pulmonary hypertension in patients with chronic lung disease (chronic lung disease with PH (mPAP 21–24 mmHg with PVR ≥3 WU, or mPAP ≥25 mmHg).
CI: cardiac index; CO: cardiac output; CT: computed tomography; DLCO: diffusion capacity of carbon monoxide; FAC: fractional area change; FEV1: forced expiratory volume 1 s; FVC: forced vital capacity; IPAF: interstitial pneumonia with autoimmune features; LVEF: left ventricular ejection fraction; mmHg: millimeters of mercury; m/s: meters/second; mPAP: mean pulmonary artery pressure; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; RAP: right atrial pressure; RIMP: right ventricular index of myocardial performance (measured by pulsed Doppler method, normal ≤0.43); RV: right ventricle; RVOT FVE: right ventricular outflow tract Doppler flow velocity envelope; RVSP: right ventricular systolic pressure; SV: stroke volume; SVO2: mixed venous oxygenation; TAPSE: tricuspid annular plane systolic excursion; TLC: total lung capacity; TR: tricuspid regurgitation; TRV: tricuspid regurgitant velocity.
Patients in the high probability group required higher oxygen flow with exertion (6 L/min) as compared to the intermediate (5 L/min) and low (3 L/min) probability groups (p = 0.036). No differences in BNP, 6MWD, O2 saturation at rest and with exertion, Borg dyspnea score, pulmonary artery diameter or PFT parameters were noted between the three groups. The high probability group had higher TRV and RVSP when compared to the low and intermediate groups (3.6 m.s-1 and 54 mmHg vs. 2.1 m.s-1 and 21 mmHg vs. 3 m.s-1 and 40 mmHg respectively, p = 0.001). An increment was also noted from the low probability to the high probability groups in RV basal diameter (p = 0.049), RV mid-diameter (p = 0.029) and RV free wall thickness (p = 0.034). Out of seven patients who underwent RHC evaluation in the low probability group, two patients (28.5%) were diagnosed with PH as compared to 0 out of 4 in the intermediate group and 3 (75%) out of 4 in the high probability group (p = 0.074). The high probability group had higher PVR (p = 0.044) and tended to have lower CI (p = 0.059) when compared to the other two groups. There were no differences noted in mPAP and the other hemodynamic parameters (Table 1).
IPAF-PH vs. IPAF-no PH groups
Patients with available right heart catheterization data were classified into PH and no PH groups based on the WSPH definition of pulmonary hypertension in patients with chronic lung disease (chronic lung disease with PH (mPAP 21–24 mmHg with PVR ≥3 WU, or mPAP ≥25 mmHg).
Mann–Whitney U test was used to compare continuous variables and Chi-Square test for categorical variables.
Percent predicted valves are reported for FEV1, FVC, TLC, and DLCO.
One patient from the IPAF-PH group did not have an echocardiographic examination available.
Only five patients in the pulmonary hypertension group had echocardiography data.
CI: cardiac index; CO: cardiac output; CT: computed tomography; DLCO: diffusion capacity of carbon monoxide; FAC: fractional area change; FEV1: forced expiratory volume 1 s; FVC: forced vital capacity; IPAF: interstitial pneumonia with autoimmune features; LVEF: left ventricular ejection fraction; mmHg: millimeters of mercury; m/s: meters/second; mPAP: mean pulmonary artery pressure; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; RAP: right atrial pressure; RIMP: right ventricular index of myocardial performance; RV: right ventricle; RVOT FVE: right ventricular outflow tract Doppler flow velocity envelope; RVSP: right ventricular systolic pressure; SV: stroke volume; SVO2: mixed venous oxygenation; TAPSE: tricuspid annular plane systolic excursion; TLC: total lung capacity; TRV: tricuspid regurgitant velocity.
Demographic and clinical summary of IPAF patients with precapillary pulmonary hypertension.
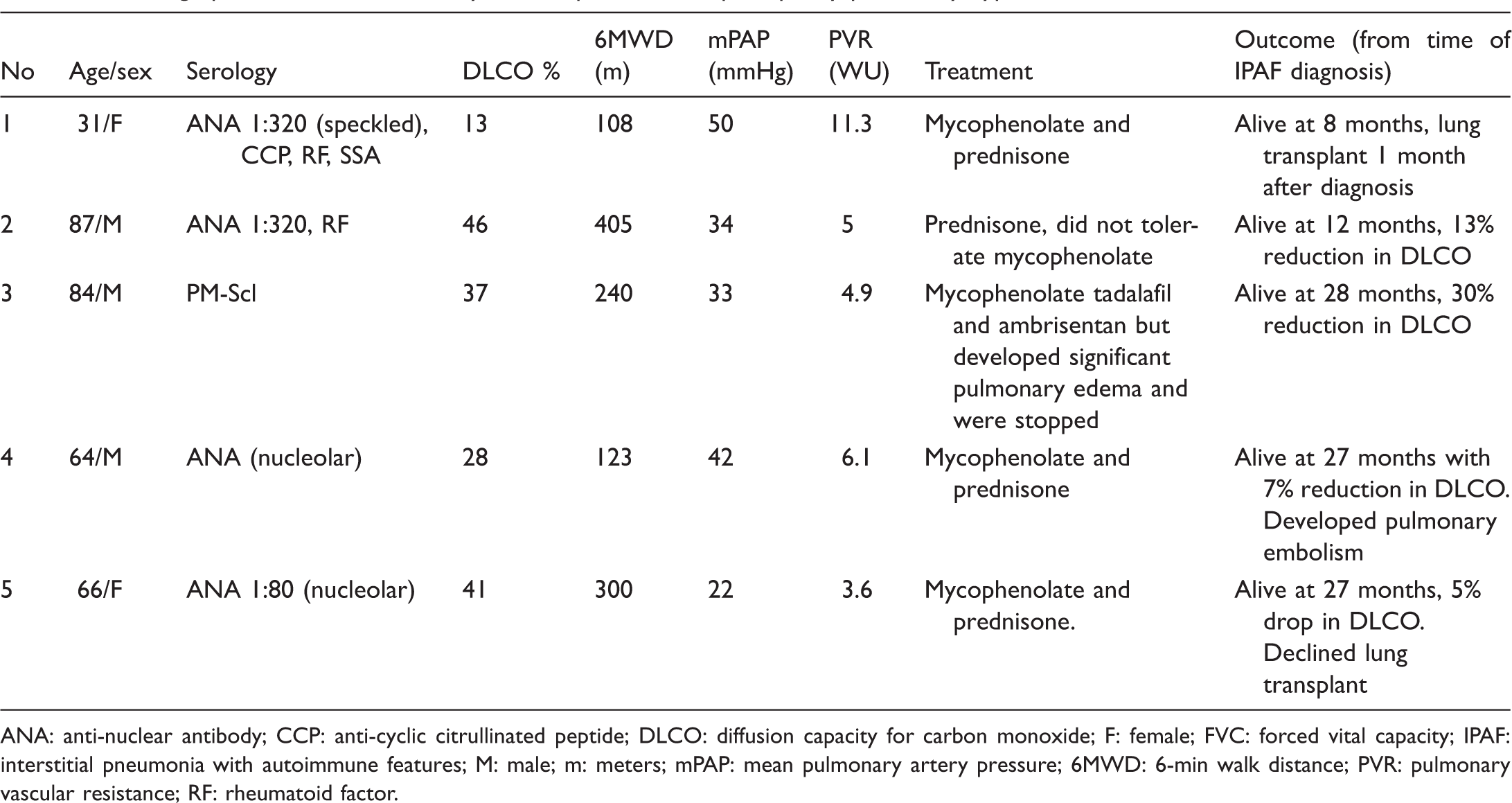
ANA: anti-nuclear antibody; CCP: anti-cyclic citrullinated peptide; DLCO: diffusion capacity for carbon monoxide; F: female; FVC: forced vital capacity; IPAF: interstitial pneumonia with autoimmune features; M: male; m: meters; mPAP: mean pulmonary artery pressure; 6MWD: 6-min walk distance; PVR: pulmonary vascular resistance; RF: rheumatoid factor.
Regression analysis
To identify non-invasive parameters that could suggest the presence of PH in IPAF patients, we tested whether 6MWD, DLCO, TAPSE, FAC%, and RIMP were associated with PVR. Only 16 patients were with RHC data were used in this analysis. We found that 6MWD was inversely associated with PVR (p = 0.002, Fig. 1) while controlling for age and BMI as they could affect 6MWD independently. We found no significant association between the other variables and PVR (data not shown).
Linear regression (with line of best fit) showing the association between log transformed data of pulmonary vascular resistance (PVR) and 6-min-walk-distance (6MWD) (p = 0.0024).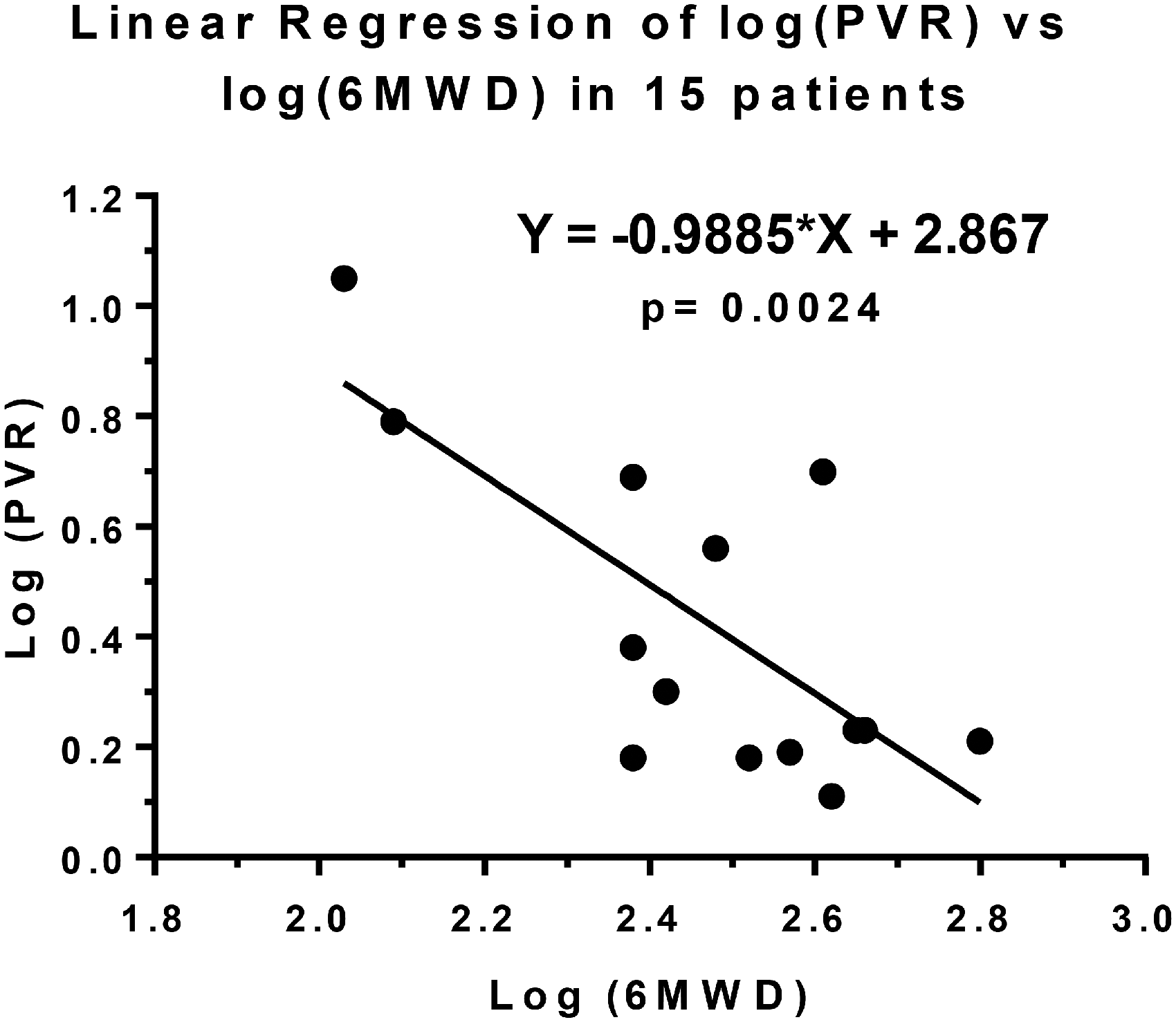
Discussion
To our knowledge, this is the first study in published literature to characterize PH in the IPAF patient population. The major findings of our study are as follows: (1) PH was identified in 12.7% of patients with IPAF, (2) RV systolic dysfunction and RV remodeling (hypertrophy and enlargement) were present in a significant number of patients, and (3) 6MWD was inversely associated with PVR.
The reported prevalence of PH in ILDs ranges from 14% to 31%.1,2,16–18 This wide variation is likely due to the type of ILD studied and the variation in methodology to describe PH. In our cohort of 47 patients, 6 (12.7%) met the definition of chronic lung disease with PH; however, RHC was only performed in one-third of the cohort. We used the recommended 2018 WSPH consensus definition using hemodynamic measurements to define PH. 7 While only 16 patients in our cohort had RHC data, the percentage of patients with PH in this group are similar to other studies. For example, in the DETECT study, 19.1% and 6% of systemic sclerosis patients were classified as group 1 PH and group 3 PH, respectively. 18 Another study evaluating IPF patients reported PH (mPAP ≥35 mmHg) in 14% of subjects. 19
A significant number of patients in our study cohort had abnormal RV function and remodeling. The median RV wall thickness in our PH group was 6 mm. Similar findings were noted in another study of 16 patients with IPF and PH who had an average RV wall thickness of 5.1 ± 1.6 mm. 20 While RV abnormalities were more common in patients with high echocardiographic PH and intermediate echocardiographic PH probability groups, they were also present in patients in the low echocardiographic PH probability group. For example, two patients in the IPAF-PH group had low echocardiographic PH probability despite having elevated mPAP. Based on previous literature, PH may be present despite a low echocardiographic PH probability. A study by Amsallem et al. 21 found PH in 16.7% of patients despite having a low echocardiographic probability. As such, echocardiography may not identify all patients with chronic lung disease patients with PH. In the same study, amongst those with high probability echocardiography PH, 96% of them were found to have PH. In our cohort, three out of four patients who underwent RHC in the high echocardiographic probabilty group actually had PH. Another important finding in our echocardiography group comparison was the lower TAPSE/PASP ratio in the high echocardiography probability PH group compared to the other groups suggesting poor right ventricular-pulmonary arterial coupling. In a recent study by Tello et al. 22 TAPSE/PASP ratio <0.26 in patients with chronic lung disease with PH was associated with worse five-year survival. In our sub-group analysis of IPAF-PH versus IPAF-no PH, we found reduced FAC% in the IPAF-PH group. The remainder of the echocardiographic variables in our sub-group analysis was non-significant which is likely due to a small sample size. While the significance of RV remodeling in IPAF patients remained to be determined, RV systolic dysfunction has been associated with increased risk of hospitalization and death in patients with ILDs. 23
The pathogenesis of PH in IPAF remains unknown. However, there may be overlapping mechanisms in CTD-associated PH and IPAF-associated PH as both entities have an autoimmune component. Both conditions may be the result of a dysregulated immune system. For example, patients with systemic sclerosis have increased endothelial cell apoptosis, CD8+ T cell recruitment, production of profibrotic factors such as transforming growth factor beta, excess extracellular matrix accumulation, and reduced capillary density.24–26Other contributory mechanisms could include elevated levels of angiogenic factors, increased generation of reactive oxidative species, and reduction in endothelial progenitor cells.27–29 Together, this may lead to pulmonary vascular remodeling and ultimately, the development of PH.
We found no significant differences in the PFT values and 6MWD between the IPAH-PH and IPAF-no PH group. While other studies have reported DLCO <60% as a marker of PH in chronic lung diseases and reduced 6MWD in those with chronic lung disease-associated PH, we found no differences between the two groups.16,30 This is likely due to the small sample size. We found higher supplemental oxygen flow rates during exertion in the high echocardiographic PH probability group compared to the other groups. In our sub-group analysis, the IPAF-PH group required higher supplemental oxygen flow rates compared to the IPAF-no PH group both at rest and during exertion but was not statistically significant. Similar findings were reported by Lettieri et al. 30 in a group of IPF patients (n = 25) where they found that oxygen use was more common in the IPF-PH group and these patients also had lower oxygen saturation nadir during the 6MWD test compared to those without PH. Another significant finding in our study was that the 6MWD was inversely associated with PVR. While we were able to control for age and BMI which could impact the 6MWD independently, we were unable to assess whether patients had arthritis which could have limited their walk distance.
To our knowledge this is the first study describing PH in IPAF patients. We have described TTE characteristics in this cohort and identified clinical factors such as reduced 6MWD associated with the presence of PH. Our study has limitations. First, it is a retrospective study from a single institution and limited by a small sample size. Second, our study was conducted at a tertiary center where patients tend to be sicker, limiting our study’s generalizability to the general population of ILD patients. Third, only 16 patients (34%) had right heart catheterization data. However, the percentage of patients with PH in our cohort was consistent with other studies of patients with PH in ILDs, also examined at tertiary centers.16–19 Lastly, the median time difference between obtaining TTE and RHC was 212 days which could have caused a discrepancy in pulmonary pressures between the two diagnostic tools.
In summary, our study identified PH in 12.5% of patients with IPAF and lower 6MWD was associated with higher PVR. While echocardiographic is a useful screening tool for PH in the general population, it may not identify all patients with PH in IPAF. PH is an important complication of fibrotic lung diseases which may increase the risk of hospitalization, impair exercise capacity, and increase mortality. Prospective studies investigating a larger group of patients are needed.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020944117 - Supplemental material for Pulmonary hypertension in patients with interstitial pneumonia with autoimmune features
Supplemental material, sj-pdf-1-pul-10.1177_2045894020944117 for Pulmonary hypertension in patients with interstitial pneumonia with autoimmune features by Bashar N. Alzghoul, Robert Hamburger, Thomas Lewandowski, Brandon Janssen, Daniel Grey, Wei Xue, Ali Ataya, Hassan Alnuaimat, Diana C. Gomez-Manjarres, Divya Patel and Raju Reddy in Pulmonary Circulation
Footnotes
Acknowledgements
We would like to thank Dr. Borna Mehrad (University of Florida) and Dr. Jess Mandel (University of California San Diego) for their support in helping with study design and editing the manuscript.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Ethical approval
The study was approved by The University of Florida’s Institutional Review Board (201802309).
Guarantor
The members of the writing committee (Bashar Alzghoul, Robert Hamburger, Raju Reddy) assume responsibility for the overall content and integrity of this article.
Contributorship
Bashar Alzghoul – Data curation, Conceptualization, Methodology, Investigation, Project administration, Supervision, Writing – original draft, Writing – reviewing and editing.
Robert Hamburger – Data curation, Conceptualization, Methodology, Investigation, Project administration, Supervision, Writing – original draft, Writing – reviewing and editing.
Thomas Lewandowski – Data curation, Investigation, Project administration, Supervision, Writing – reviewing and editing.
Brandon Janssen – Data curation, Investigation, Project administration, Writing – reviewing and editing.
Daniel Grey – Data curation, Investigation, Project administration, Writing – reviewing and editing.
Wei Xue – Formal analysis, Project administration, Writing – original draft Writing – reviewing and editing.
Ali Ataya – Investigation, Project administration, Writing – reviewing and editing.
Hassan Alnuaimat – Investigation, Project administration, Writing – reviewing and editing.
Diana Gomez-Manjarrez – Data curation, Investigation, Project administration, Writing – reviewing and editing.
Divya Patel – Data curation, Investigation, Project administration, Writing – reviewing and editing.
Raju Reddy – Data curation, Conceptualization, Investigation, Methodology, Project administration, Supervision, Writing – original draft, Writing – reviewing and editing.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


