Abstract
Treprostinil is a prostacyclin analogue approved for the treatment of pulmonary arterial hypertension. Apart from the inhaled formulation, there is neither a target dose nor a ceiling dose to guide clinicians using treprostinil; doses are individualized for each patient based upon tolerability and clinical improvement. Using combined data from the pivotal subcutaneous and oral treprostinil studies, we evaluated the effect of treprostinil dose on hospitalization and exercise capacity to better define the treprostinil dose–response relationship. Data from the pivotal subcutaneous and oral treprostinil studies were combined by converting oral doses to weight-based continuous doses (ng/kg/min) accounting for patient weight and bioavailability. Patients were divided into dose tertiles (lowest, middle, highest 33%) and retrospectively analyzed. Analysis 1 assessed the effect of dose on pulmonary arterial hypertension-related and all-cause hospitalizations. Analysis 2 evaluated the effects of dose on six-minute walk distance, Borg dyspnea score, and World Health Organization functional class. Results showed that, in Analysis 1, higher doses of treprostinil were associated with significantly longer times to first pulmonary arterial hypertension-related and all-cause hospitalization. In Analysis 2, there was a trend toward improvements in six-minute walk distance with higher doses. In patients with pulmonary arterial hypertension on systemic treprostinil therapy, higher doses were associated with significantly longer time to first pulmonary arterial hypertension-related and all-cause hospitalization. There was a trend toward improvements in six-minute walk distance. Collectively, these results underscore the importance of managing prostacyclin adverse events in order to achieve appropriate dose titration. Further studies are required to confirm these findings and to better characterize the dose–response relationship of treprostinil.
Introduction
Pulmonary arterial hypertension (PAH) is a progressive and severely disabling disorder characterized by luminal narrowing in the small- and medium-sized pulmonary arteries, which leads to an increase in pulmonary vascular resistance and may culminate in right ventricular failure and premature death. 1 There are currently four therapeutic drug classes targeting three distinct molecular pathways approved for the treatment of PAH: endothelin receptor antagonists (ERAs), phosphodiesterase type 5 (PDE-5) inhibitors and soluble guanylate cyclase (sGC) stimulators, and prostacyclin analogues/receptor agonists. 2
Evidence suggests that in patients with PAH, there are abnormalities in prostacyclin metabolic pathways within the pulmonary vasculature which lead to vasoconstriction of pulmonary arteries and endothelial cell proliferation. 3 Treprostinil is a prostacyclin analogue that reduces pulmonary arterial pressure through direct vasodilation of the pulmonary and systemic arterial vascular beds. Additional effects include inhibition of platelet aggregation and in vitro reversal of pulmonary artery remodeling via reduction in smooth muscle cell proliferation.4–7 Emerging pre-clinical data suggest anti-fibrotic properties as well.8–10 These mechanisms lead to improvements in pulmonary gas exchange, systemic oxygen transport, and cardiac output with minimal alteration to heart rate. 11
As established by United States Food and Drug Administration and International Conference on Harmonization (ICH) guidelines on studying dose–response relationships, a well-controlled dose–response study would ideally evaluate patients randomized to different doses. Associations can then be drawn between dose and measures of efficacy or safety.12,13 With systemically administered treprostinil, this ideal trial design is challenging because there is no known dose target and treprostinil doses are individualized based on tolerability and clinical improvement. Therefore, patients cannot be randomized to different drug exposures without considering the ethical implications of prolonged use of a possibly sub-therapeutic treprostinil dose for a life-threatening condition such as PAH. Low doses of prostacyclin therapy have been shown to acutely improve hemodynamics, 14 but in clinical practice, patients are not maintained at a low dose. Instead, they are gradually titrated to the highest dose that balances clinical metrics, patient symptomatology, and manageable adverse events. Therefore, it would also not be clinically appropriate to randomize a patient to a treprostinil dose which may be supratherapeutic and harmful for the purposes of a dose–response study. Finally, treprostinil is a very potent vasodilator with potential for significant adverse events and inherent route-specific complications; achieving adequate doses to derive dose–response relationships cannot be safely or ethically administered to healthy volunteers.
As these obstacles preclude the formal study of treprostinil dose–response in a prospective, randomized manner, physicians have derived a large portion of their understanding from clinical experience. Traditional PAH clinical trials have utilized the six-minute walk distance (6MWD) as the primary endpoint and more recent trials have employed composite endpoints evaluating time to clinical worsening, with results largely driven by hospitalization and disease progression.15–18 Both of these endpoints are clinically relevant and warrant evaluation. In this paper, we present two novel analyses that combine data from patients enrolled in the subcutaneous (SC) treprostinil registration study with data from patients from the FREEDOM-M registration study for oral treprostinil.19–22 Both pivotal studies were 12 weeks long, had the same primary endpoint of 6MWD, studied systemically administered treprostinil monotherapy, and titrated treprostinil to the maximum tolerated dose.19,20 Using these combined data, we evaluate the effects of higher treprostinil doses on both hospitalization rates and functional capacity in patients with PAH.
Methods
Analysis 1: effect of dose on hospitalization
Patients were included in the analysis if they completed the randomized, pivotal oral or SC treprostinil studies, and entered the open-label studies.21,22 Inhaled treprostinil was not included in the analysis because unlike parenteral and oral treprostinil, the manufacturer recommends a maximum target dose. 23 The manufacturer's global drug safety database was retrospectively analyzed for hospitalizations reported per severe adverse events associated with the open-label extension studies. Hospitalizations were adjudicated for relatedness to PAH by physicians employed by the drug manufacturer who are familiar with treprostinil's safety profile. Hospitalizations were considered unrelated to PAH if the patient was treated for another underlying etiology or experienced decompensation due to acute medical problems. To combine the dosing data, oral doses were converted to weight-based continuous doses (ng/kg/min), accounting for patient weight and bioavailability using the formula in the manufacturer package insert. Patients were grouped into tertiles based on last known oral or SC treprostinil dose (i.e. lowest 33% “low”, middle 33% “medium”, highest 33% “high”) in the open-label extension studies. Pairwise comparisons were made between dose groups to evaluate difference in time to first PAH-related and all-cause hospitalizations. Cox proportional-hazards model was used to compare risk of PAH-related and all-cause hospitalizations between dose groups. Kaplan–Meier curves and log-rank tests were used to compare dose groups. An adjusted analysis using a Cox proportional model was repeated to control for confounding differences in baseline characteristics known to impact hospitalizations in PAH. 24 All statistical calculations were completed using SAS® version 9.2 (SAS Institute, Inc. Cary, NC).
Analysis 2: effect of dose on 6MWD
Data from patients in the active arm (i.e. not placebo) in the SC and oral treprostinil pivotal studies were combined.19,20 Dosing data were combined as described in the methods for Analysis 1. Patients were grouped into tertiles based on dose at Week 12 of the clinical studies (i.e. lowest 33% “low”, middle 33% “medium”, highest 33% “high”). Last observation carried forward was used to impute last observations of 6MWD, Borg dyspnea score (BDS), World Health Organization functional class (WHO FC), and dose. One-way analysis of variance and Jonckheere–Terpstra tests were used to assess differences and linear trends in 6MWD, BDS, and WHO FC based on treprostinil dose received. An adjusted analysis using multiple linear regression was repeated to control for confounding differences in baseline characteristics known to impact 6MWD in PAH. 25 All statistical calculations were completed using SAS® version 9.2 (SAS Institute, Inc. Cary, NC).
Results
Analysis 1: effect of dose on hospitalization
Analysis 1—baseline patient characteristics.
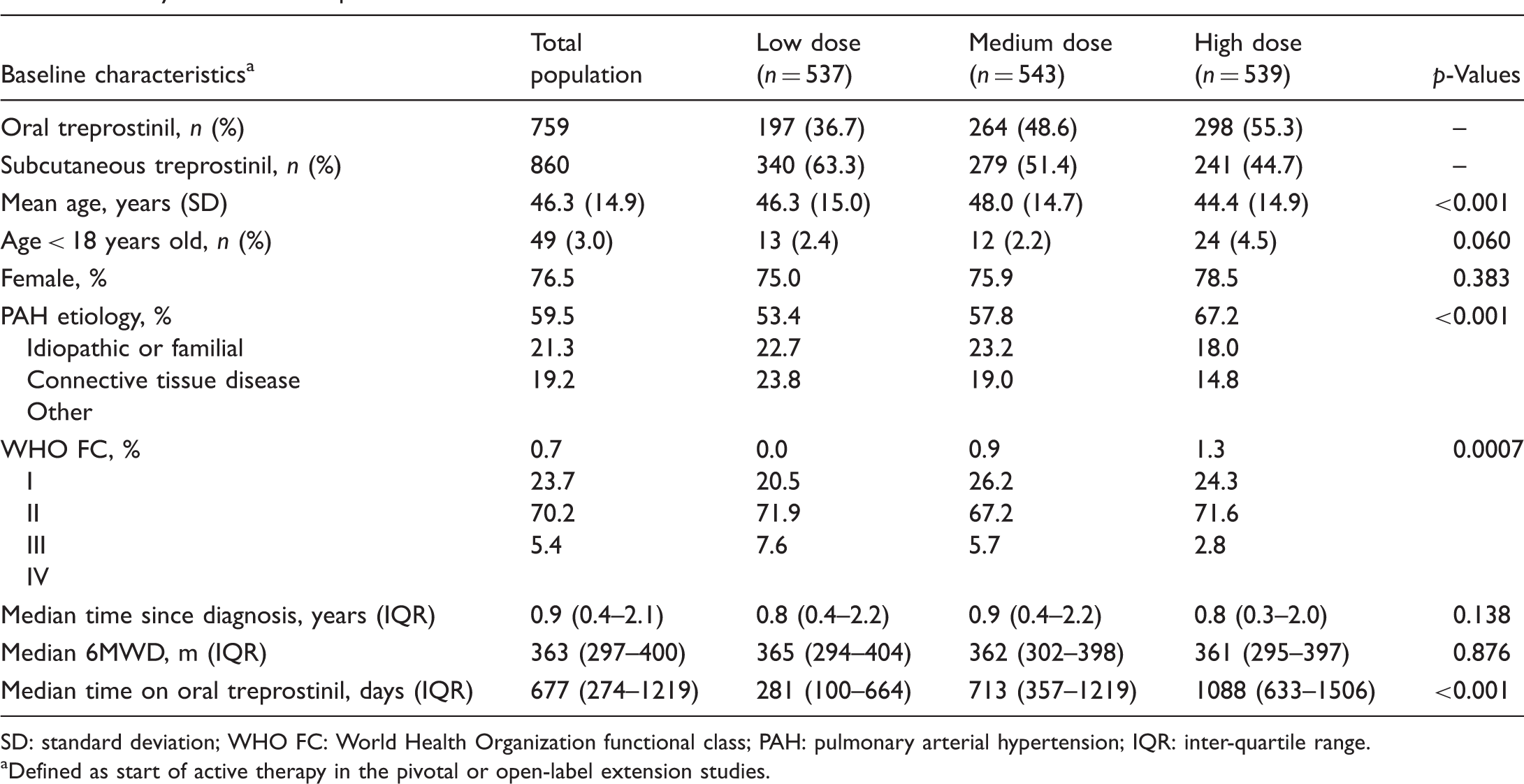
SD: standard deviation; WHO FC: World Health Organization functional class; PAH: pulmonary arterial hypertension; IQR: inter-quartile range.
Defined as start of active therapy in the pivotal or open-label extension studies.
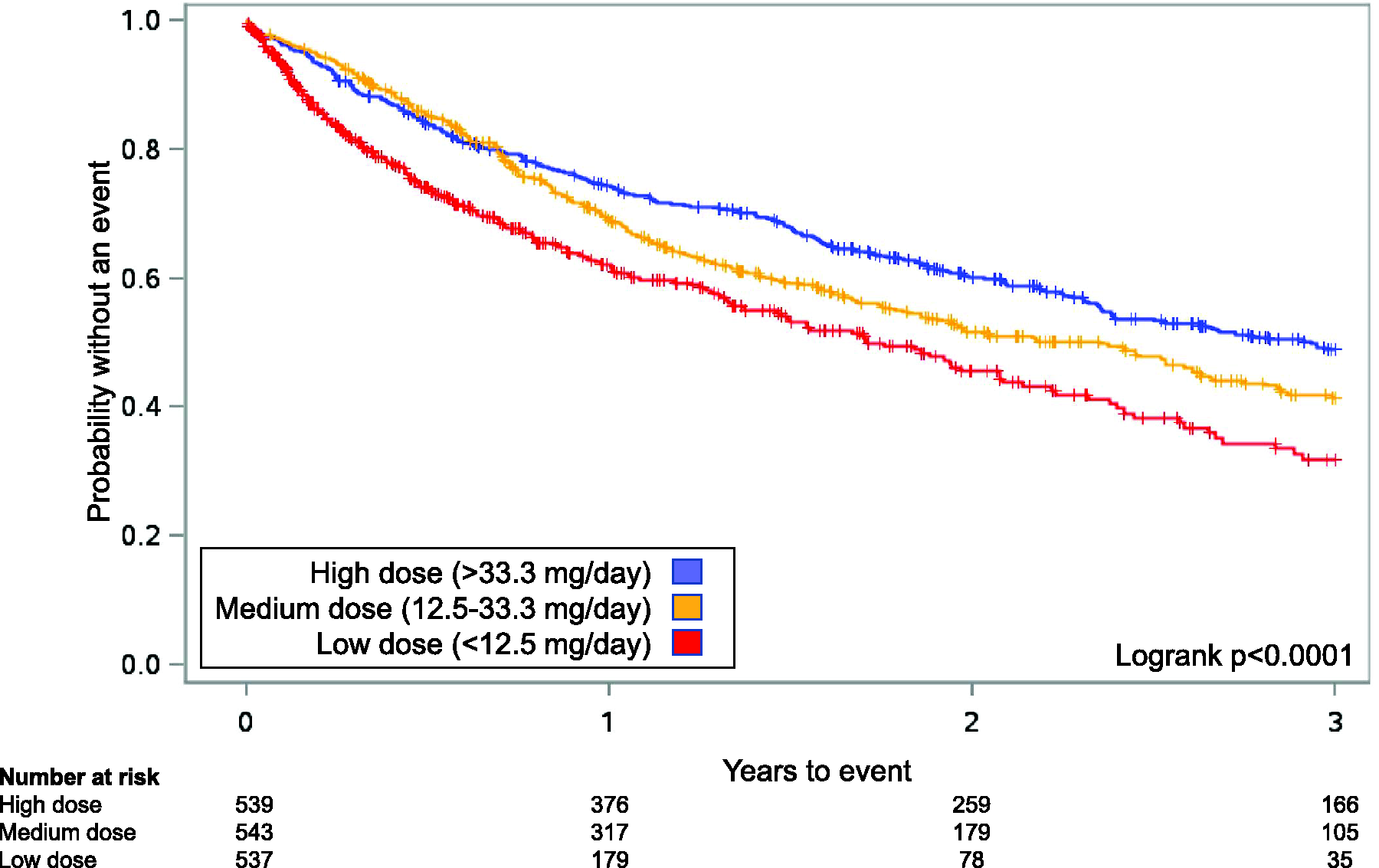
Of the 1619 patients, 730 patients (45.1%) experienced a hospitalization, including 396 (24.4%) due to PAH. Median time to first PAH-related hospitalization for low, medium, and high dose groups was 209, 378, and 529 days ( Kaplan–Meier curves for Analysis 1 for time to first pulmonary arterial hypertension (PAH)-related hospitalization for patients receiving oral treprostinil and subcutaneous (SC) treprostinil in the open-label extension studies after the randomized registration studies.21,22 Patients were grouped into tertiles based on the last known dose (i.e. lowest 33% “low”, middle 33% “medium”, highest 33% “high”). In the combined analysis for oral and SC treprostinil, a significant treatment effect in favor of higher doses was observed (logrank Kaplan–Meier curves for Analysis 1 for time to first all-cause hospitalizations for patients receiving oral treprostinil and subcutaneous (SC) treprostinil in the open-label extension studies after the randomized registration studies.21,22 Patients were grouped into tertiles based on the last known dose (i.e. lowest 33% “low”, middle 33% “medium”, highest 33% “high”). In the combined analysis for oral and SC treprostinil, a significant treatment effect in favor of higher doses was observed (logrank Analysis 1—hazard ratios for PAH-related and all-cause hospitalizations between dose groups. PAH: pulmonary arterial hypertension; HR: hazard ratio; CI: confidence interval; WHO FC: World Health Organization functional class.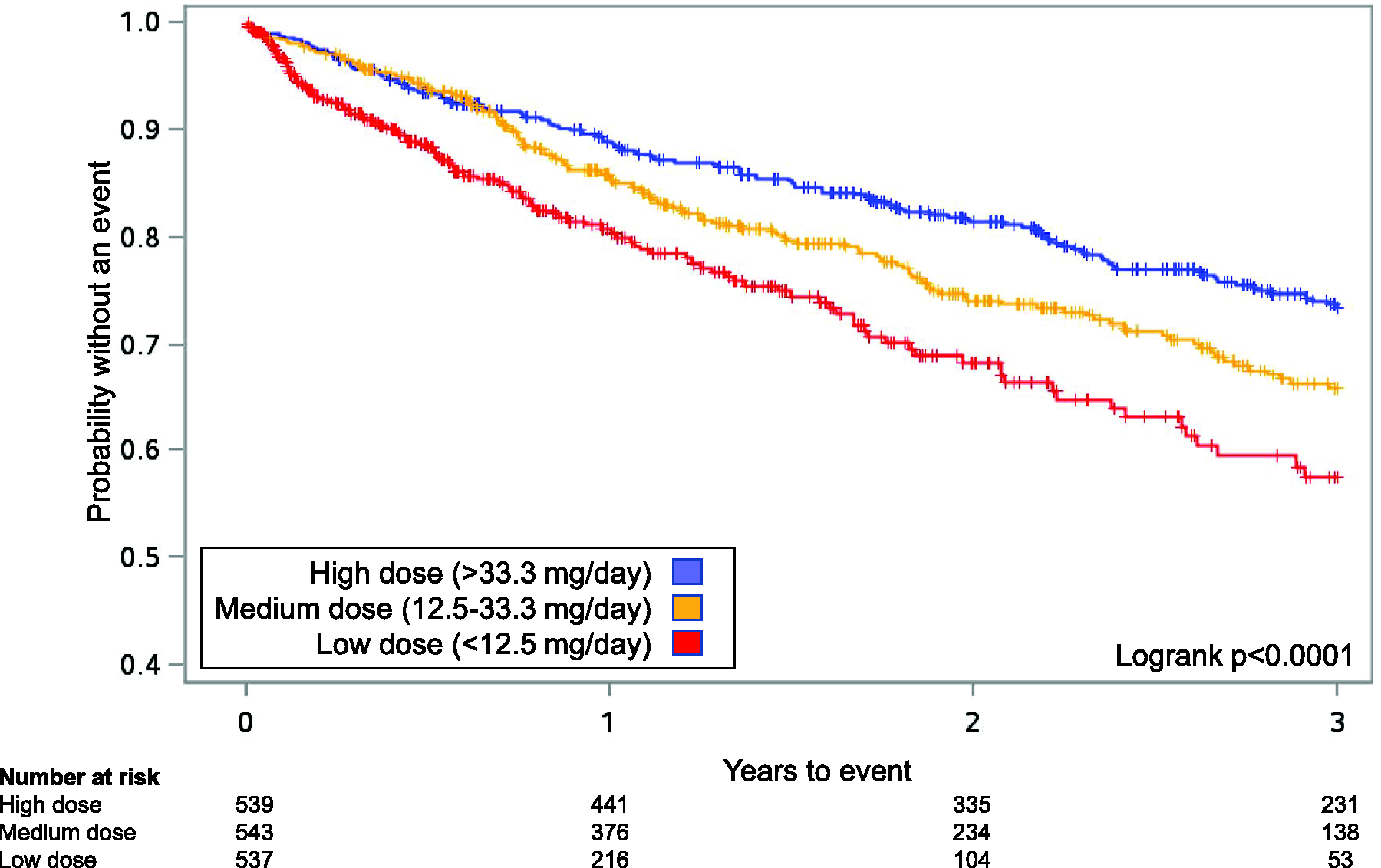
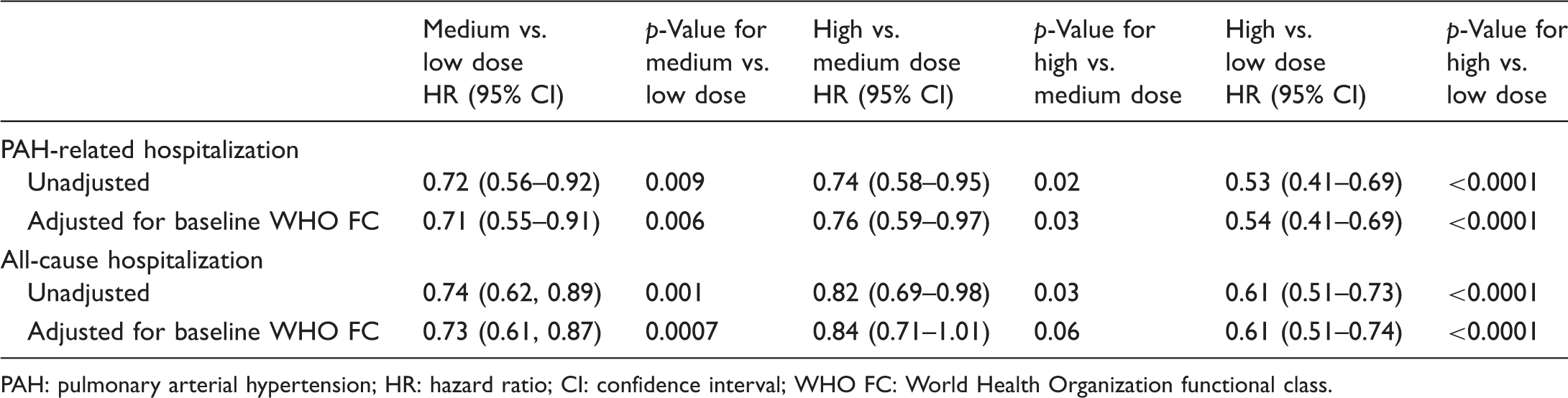
Noting that WHO FC was significantly different between dose groups at baseline, we performed an adjusted analysis based on published literature suggesting disease severity may influence hospitalization.
24
Disease severity reflected by 6MWD, age <18 years old, and comorbidities were also considered as confounding variables. Baseline 6MWD and age <18 years were balanced between dose groups. Baseline comorbidities could not be corrected for because these data were not available from the parent studies used in Analysis 1. In the adjusted analysis controlling for WHO FC, higher doses remained associated with significant decreases in risk of PAH-related hospitalization (
To evaluate whether results differed by treprostinil route of administration, SC and oral dose data were evaluated separately. Patients on low, medium, and high doses of oral treprostinil had a median time to first PAH-related hospitalization of 339, 412, and 658 days, respectively (
Analysis 2: effect of dose on 6MWD
Analysis 2—baseline patient characteristics.
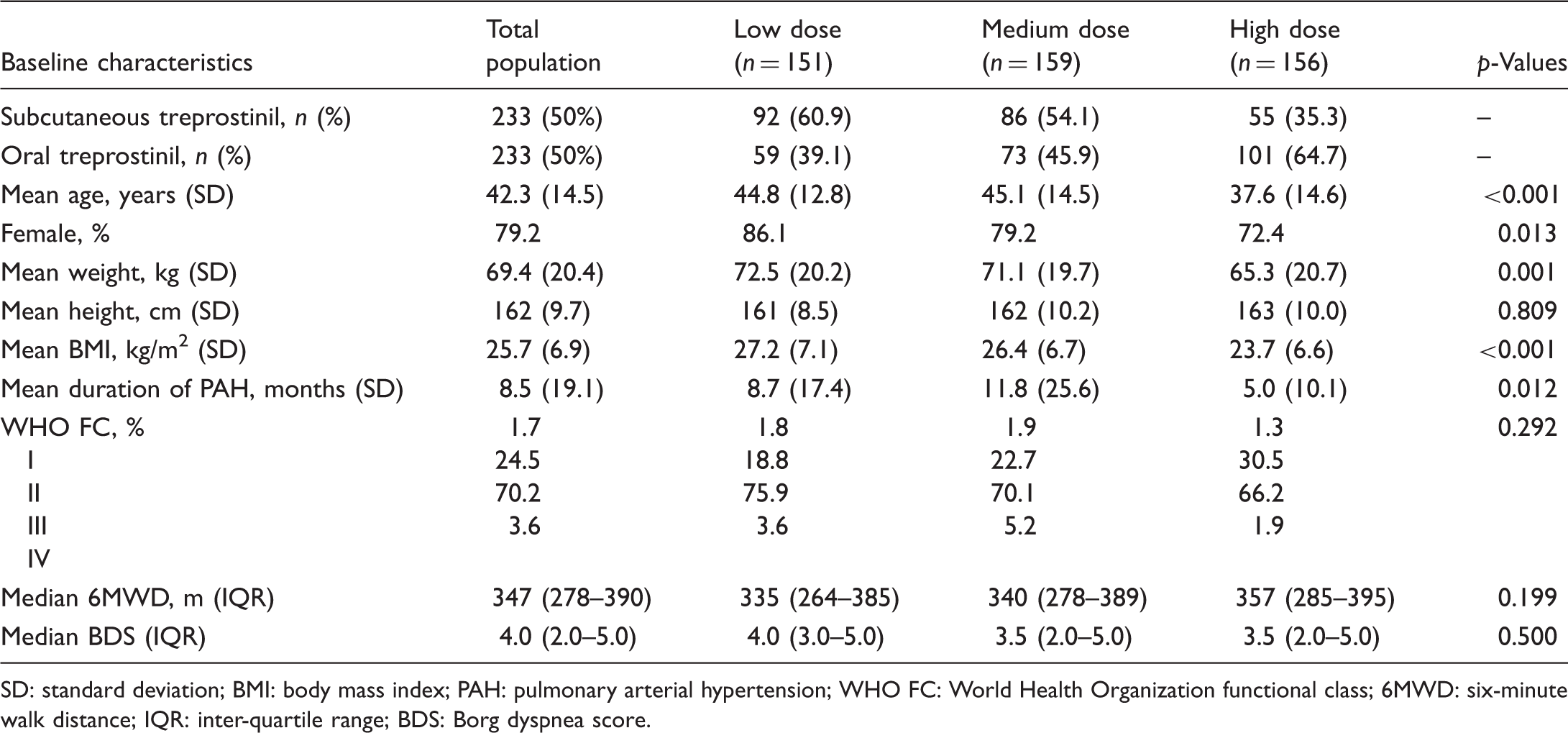
SD: standard deviation; BMI: body mass index; PAH: pulmonary arterial hypertension; WHO FC: World Health Organization functional class; 6MWD: six-minute walk distance; IQR: inter-quartile range; BDS: Borg dyspnea score.
Analysis 2—change in 6MWD, BDS, and WHO FC by dose group.
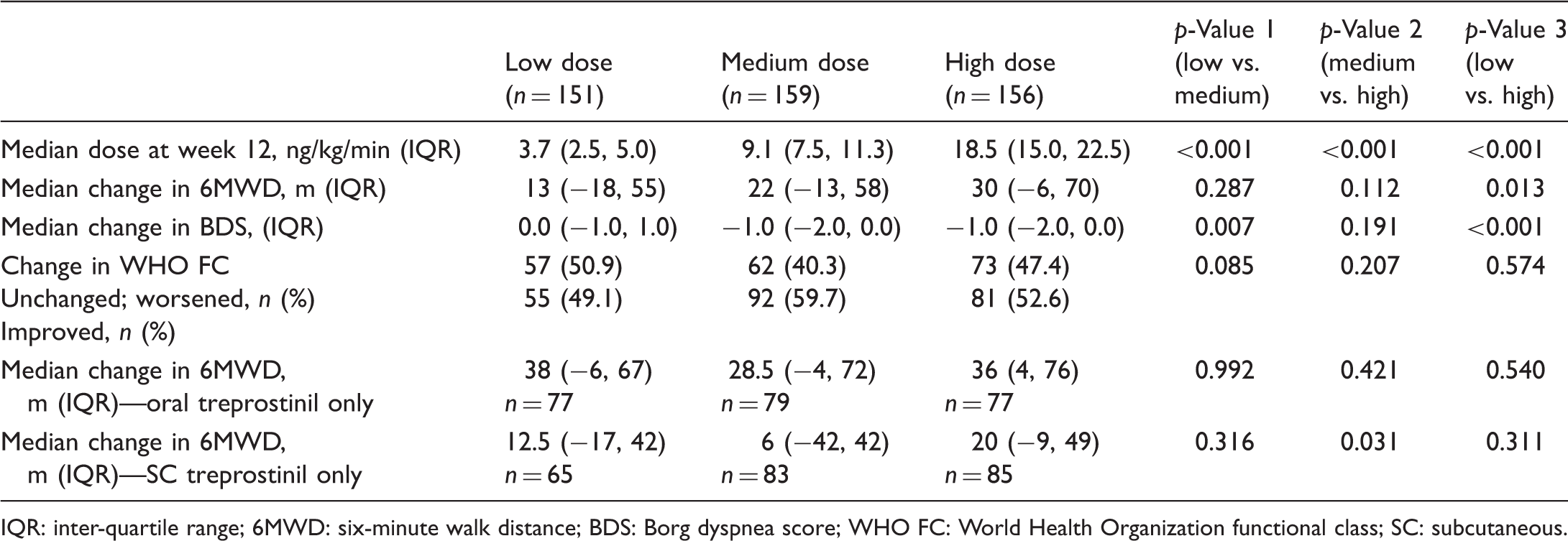
IQR: inter-quartile range; 6MWD: six-minute walk distance; BDS: Borg dyspnea score; WHO FC: World Health Organization functional class; SC: subcutaneous.
The low, medium, and high dose groups had respective median 6MWD improvements of 13, 22, and 30 m (Fig. 3). There was a statistically significant difference in 6MWD improvement between low/high dose groups ( Boxplot showing change in exercise capacity by treprostinil dose tertile for Analysis 2 (unadjusted). Numbers within the colored boxes represent median and the interquartile range (IQR) 6MWD change from baseline with last observation carried forward (LOCF) for each dose tertile. Horizontal lines within the boxes represent the median, with maximum and minimum values appearing above the boxes. 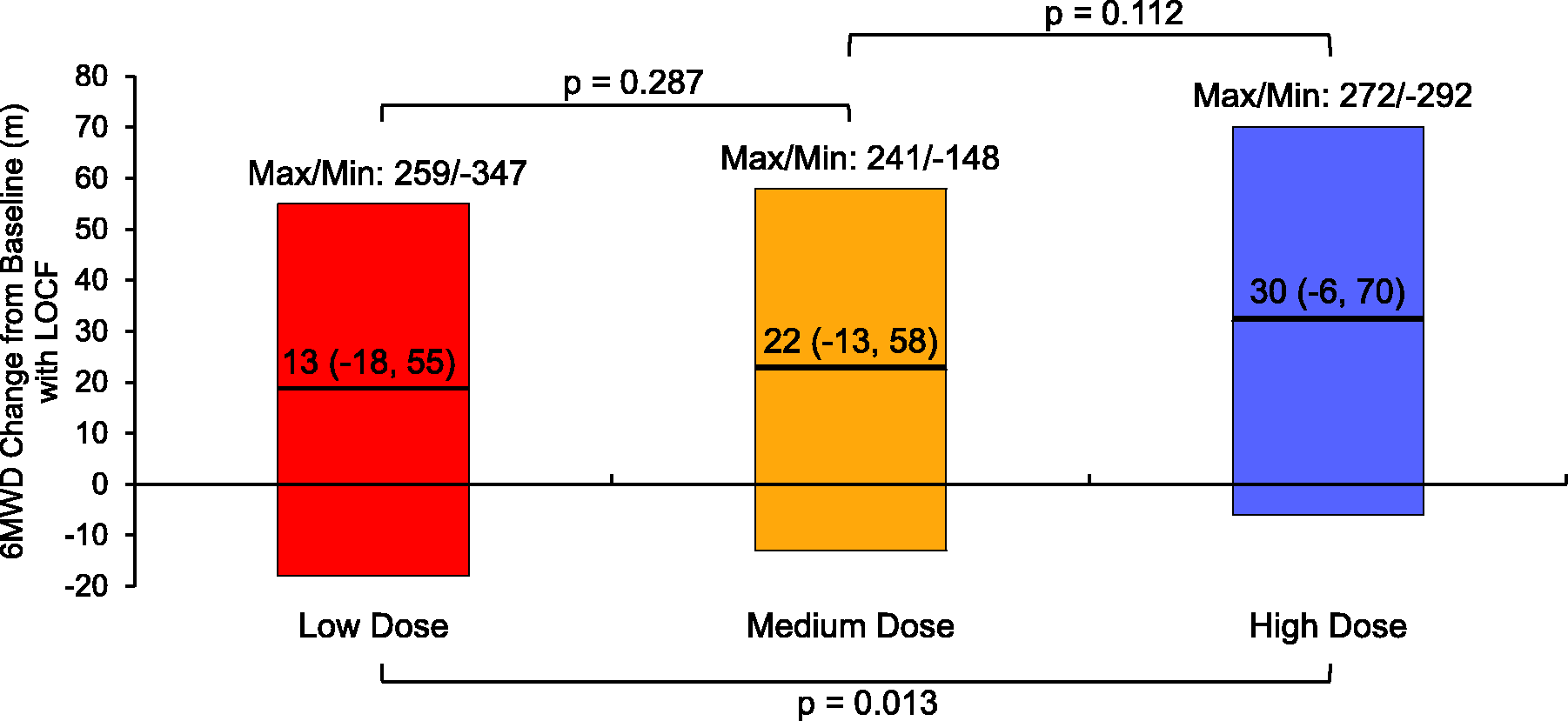
Noting that age, gender, and BMI were significantly different between dose groups at baseline, we performed an adjusted analysis based on published literature suggesting this may influence 6MWD.
25
PAH etiology was considered as well but was balanced between the dose groups at baseline. When controlling for age, sex, and BMI, we did not observe any significant differences in 6MWD between dose groups (
To evaluate whether 6MWD results differed by treprostinil route of administration, SC and oral dose data were separated and the analysis repeated. For oral treprostinil patients, the low, medium, and high dose groups had respective median 6MWD improvements of 38, 28.5, and 36 m (
Discussion
Until recently, 6MWD has been used as the primary endpoint in nearly all pivotal Phase 3 PAH clinical trials. Much about what is currently understood about treprostinil dose–response is derived from increased exercise capacity associated with escalating doses. Although the validity of the 6MWD as a surrogate endpoint is often questioned, findings from European PAH registries have confirmed that 6MWD at both baseline and at follow-up is a strong clinical prognosticator.26,27 Recent clinical trials have emphasized the importance of preventing clinical worsening and PAH-related hospitalizations.15–18 In the contemporary era, both endpoints are highly relevant. In these novel analyses, we have summarized how patients achieving higher treprostinil doses demonstrate lower risk of hospitalization and a trend toward improved exercise tolerance.
In Analysis 1, the high and medium dose Kaplan–Meier curves for PAH-related and all-cause hospitalization separate from the low dose curve early on, and there was not much separation between the high and medium dose groups until month 9. This could be due to the dose cutoffs used in this study which were based on unbiased tertiles; it is possible that early on in the course of therapy, certain dose thresholds are needed to achieve noticeable benefits and our cutoffs do not adequately reflect these thresholds. In our adjusted analysis controlling for baseline differences in WHO FC, the high dose group still demonstrated significant reductions in risk when compared to the low dose group. Results from evaluating SC and oral treprostinil patients separately suggest that both routes of administration independently demonstrate a dose–response relationship with regard to hospitalization.
The improvements in 6MWD observed in Analysis 2 are relatively modest but it is important to note that the treprostinil doses analyzed are low compared to those routinely used in contemporary clinical practice. Furthermore, many factors can impact 6MWD and when controlling for differences in baseline demographics between dose groups, we did not observe a significant difference in the Week 12 6MWD associated with dose. It is possible that 6MWD may not be a sensitive marker of response to treprostinil in the way that hospitalization may be, or that doses utilized in this study were too low to demonstrate a statistically significant improvement.
Summary of dose–response findings from studies of SC/IV and oral treprostinil.
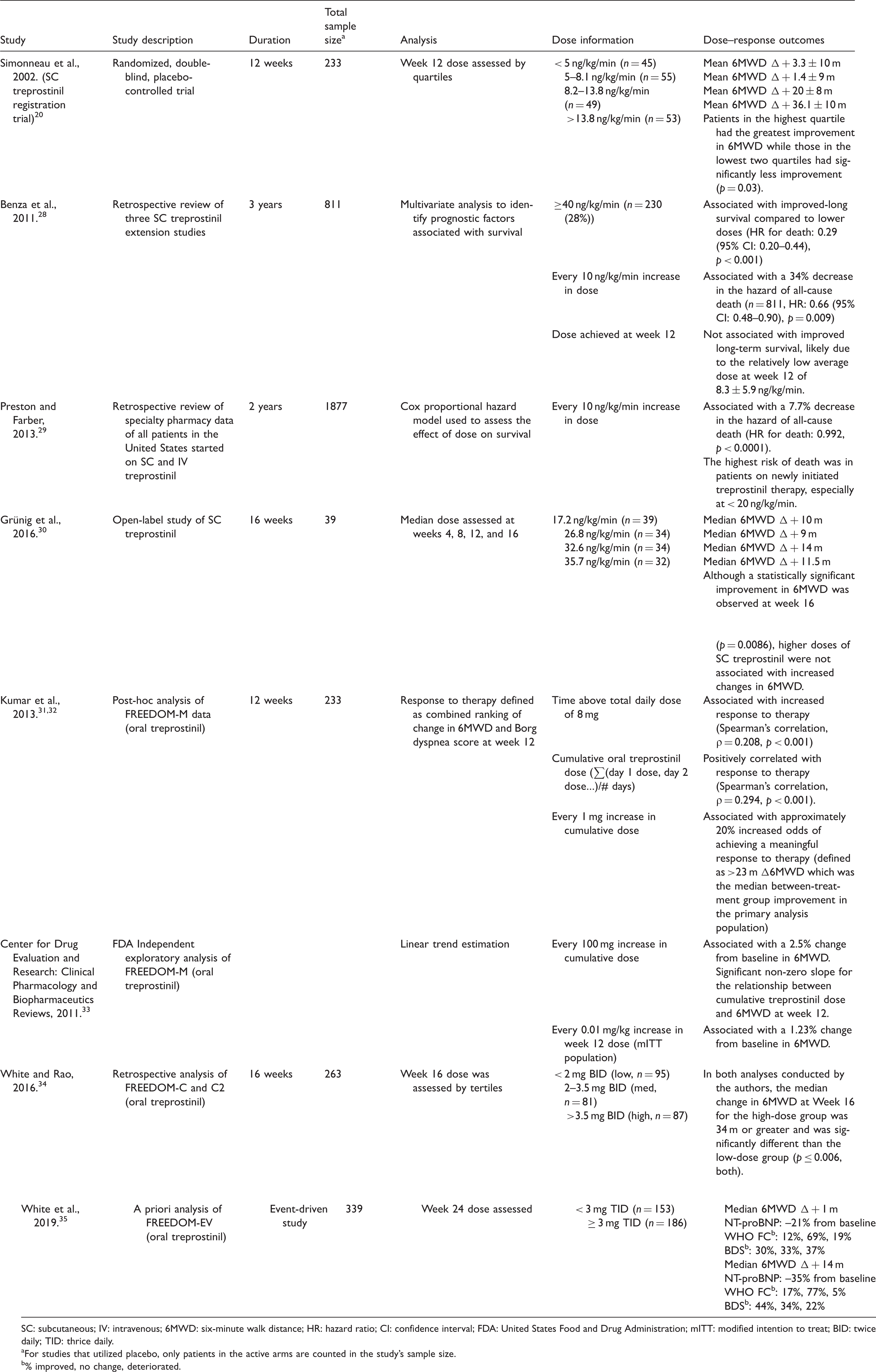
SC: subcutaneous; IV: intravenous; 6MWD: six-minute walk distance; HR: hazard ratio; CI: confidence interval; FDA: United States Food and Drug Administration; mITT: modified intention to treat; BID: twice daily; TID: thrice daily.
For studies that utilized placebo, only patients in the active arms are counted in the study's sample size.
% improved, no change, deteriorated.
Similar findings have been observed for oral treprostinil (Table 5). Several post-hoc analyses suggest a dose–response relationship of oral treprostinil monotherapy in which higher doses correspond to greater improvements in functional capacity.31–33 In another analysis of oral treprostinil used as combination therapy, White and Rao found a significant difference in 6MWD change from baseline between the low and high dose groups at week 16. 34 More recently, longer term dose–response findings in the FREEDOM-EV study were published showing that patients achieving ≥ 3 mg thrice daily (TID) at week 24 had greater improvements in 6MWD, N-terminal pro-brain natriuretic peptide (NT PRO BNP), WHO FC, and BDS compared to the <3 mg TID group while experiencing a similar rate of adverse events. 35
There are limitations to these present analyses. Although combining SC and oral data is a novel concept supported by the systemic nature of drug exposure for both treprostinil formulations, there is evidence that parenteral and oral routes are different. 36 Analysis 1 was retrospective in nature, and hospitalizations were adjudicated by physicians employed by the drug manufacturer rather than treating clinician. Results may be confounded by changes or addition of background PAH therapy, which were unavailable for assessment. Dose data were extrapolated from last known dose in the open-label study and the actual dose for patients may be different. Analysis 2 was retrospective in nature and dose groups varied with respect to age, sex, weight, and duration of PAH at baseline. The doses used in Analysis 2 were relatively low which may limit the applicability of the findings; for instance, the median dose in the high dose group was 18.5 ng/kg/min, which corresponds to an oral treprostinil regimen of approximately 3 mg TID in a 70 kg patient. There are also limitations to using 6MWD as an endpoint, as improvements in this clinical measure may be influenced by numerous parameters other than dose. 25 Finally, there were no significant 6MWD findings when SC and oral treprostinil doses were evaluated separately, likely due to the small sizes of each group.
Prostacyclin dose–response relationships are also confounded by the time it takes to titrate patients to clinically appropriate doses. This is especially apparent when considering the mean dose of 26 ng/kg/min at one year in the long-term SC treprostinil study used in Analysis 1, which is low compared to the mean dose of 55 ng/kg/min achieved at one year in contemporary clinical practice.21,37,38 It is possible that longer exposure time rather than increased dose is responsible for the improved clinical benefit over time; in a post-hoc study of patients on oral treprostinil, Kumar and colleagues found that the time a patient spent above a total daily dose of 8 mg was associated with an increased response to therapy.31,32 This suggests that there may be a temporal component to the dose–response relationship of treprostinil.
Along with epoprostenol, parenteral and oral treprostinil therapies are unique in PAH as they are the only drugs without a ceiling dose. In patients who achieve clinical improvement and stability, incremental prostacyclin dose increases are often required over time. Whether the need for dose titration arises from disease progression or drug tolerance is uncertain. The benefit of the dose–response relationship for systemically administered prostacyclins is that drug is titratable with no ceiling dose, allowing this drug class to remain a viable long-term treatment option. In effect, dose titration can outpace disease progression. In contrast, ERAs, PDE-5 inhibitors, sGC stimulators, and prostacyclin receptor agonists are employed with dose limits and when a patient experiences disease progression on these therapies, additional treatment with drugs from other therapeutic classes or conversion to prostacyclins is required.
There are potential risks associated with progressively higher doses of prostacyclins. These have been best characterized with parenteral epoprostenol. A high cardiac output state has been described with higher doses of epoprostenol. 39 High output heart failure should be considered in previously stable patients at high doses who develop worsening flushing, gastrointestinal symptoms, and palpitations. Right heart catheterization often reveals a markedly elevated cardiac output and symptoms subside with dose reduction without worsening of PAH symptoms. High output heart failure has not been well described with oral treprostinil. Increased incidence of adverse events such as headache, nausea, flushing, and jaw pain may also be associated with higher doses of prostacyclins, so proper follow-up remains important as patients uptitrate on dose.
These two novel post-hoc analyses demonstrate that increased treprostinil dose is associated with improvements in 6MWD and significantly longer time to hospitalization. Despite the differences between the oral and parenteral formulations of treprostinil and the heterogeneity of the doses achieved in clinical trials, the significantly longer time to first PAH-related and all-cause hospitalization associated with higher, clinically relevant doses of treprostinil supports the importance of aggressive dose titration. The exact nature of the dose–response relationship (e.g. linear vs. non-linear) remains difficult to characterize owing to the background progression of disease, and whether this relationship continues to exist at very high doses is unknown. It is also unclear whether the dose–response relationship exists for certain PAH phenotypes that are more aggressive in nature and less responsive to pharmacotherapy. In addition, these results should be interpreted cautiously as the use of higher treprostinil doses is dependent on a patient's PAH symptomatology as well as drug tolerability. Nonetheless, these unique properties of treprostinil distinguish it from other non-prostacyclin therapies. Further research into the dose–response relationship may focus on the relationship between side effects and therapeutic benefits, and better understanding the physiologic and biochemical mechanisms that lead to clinical improvement with higher treprostinil doses.
Footnotes
Acknowledgements
We would like to thank Erick Borg, PharmD for his work on Analysis 1 and his guidance with data presentation. Preliminary data were presented at ATS 2018 International Conference, 18–21 May 2018, San Diego, CA; and at the CHEST Annual Meeting 2018, 6–10 October 2018, San Antonio, TX.
Guarantor
Dr Gautam Ramani
Contributorship
GR and SC collected data, drafted manuscript, directed statistical analyses, and incorporated author feedback for the final submission. ES, MB, and AW assembled data tables, drafted methods section, and responded to author queries for data. MB and AN collected data and offered critical feedback. QS aggregated data and performed statistical analyses as directed by authors. GR, SC, ES, and MB guarantee the integrity of the work as a whole.
Conflict of interest
The University of Maryland contracts with United Therapeutics Corporation and Actelion Pharmaceuticals Ltd. for GR to perform research. SC and AW do not have any conflicts of interest to declare and does not have affiliations with institutions, organizations, or companies mentioned in the manuscript or whose products or services are discussed. ES, MB, QS, and AN are employees or contractors of United Therapeutics Corporation.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
