Abstract
Pulmonary arterial hypertension is a fatal disease, where death is associated with right heart failure and reduced cardiorespiratory reserve. The Sugen 5416, hypoxia and normoxia Fischer rat model mimics human pulmonary arterial hypertension, although the cause(s) of death remains incompletely understood. Here, we hypothesized that these animals develop biventricular diastolic dysfunction that contributes to tissue hypoperfusion coincident with severe pulmonary arterial hypertension. We performed comprehensive echocardiographic and hematologic assessments. Serial echocardiogram at 3–5 weeks was performed followed by blood sampling via aortic or cardiac puncture. Echocardiogram revealed pulmonary arterial hypertension in pulmonary artery Doppler waves, including notched wave envelopes, and decreased pulmonary artery acceleration time/pulmonary artery ejection time ratio and right ventricular outflow tract velocity time integral. Impaired right ventricular systolic function, assessed by decreased tricuspid annular plane systolic excursion and tricuspid tissue Doppler systolic positive wave velocity, was observed in pulmonary arterial hypertension. Tricuspid and mitral pulsed wave and tissue Doppler findings suggested biventricular diastolic dysfunction, with dynamic changes in early and late diastolic filling waves, their fusion patterns, and a decrease in e' velocity. Heart rate and ejection fraction did not change, but cardiac output, stroke volume, and end-diastolic volume were decreased, and inferior vena cava respiratory variation was decreased. Blood electrolyte values were suggestive of intravascular volume expansion early in the disease followed by volume contraction and tissue hypoperfusion in the latter stages of disease. Complete blood count showed thrombocytopenia and non-anemic macrocytosis with reticulocytosis and an increase in red blood cell distribution width. Thus, pulmonary, cardiac, and hematological findings in Fischer animals with pulmonary arterial hypertension are characteristic of humans and provide an insightful experimental platform to resolve mechanisms of disease progression.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a progressive and fatal disease. 1 Due to the nonspecific clinical presentations and heterogeneous patient phenotypes, 2 timely diagnosis is challenging and initiation of therapy is often delayed. 3 Despite rapid advancement in understanding the pathophysiology of PAH, and despite the development of targeted therapies that increase life longevity, 4 the causes of death among PAH patients remain elusive. The direct cause of death in PAH patients was thought to be mainly progression of right heart failure. 5 However, in a cross-sectional study addressing the cause of death of PAH patients, respiratory disease, pulmonary embolism, sepsis, acute renal failure, and other cardiovascular disease accounted for 44% of the deaths. 6 Furthermore, multiple non-PAH specific factors, such as renal failure 7 and thrombocytopenia, 8 independently predict PAH mortality. Recently, unbiased phenome-wide association studies revealed that among 1364 coded medical conditions, red cell distribution width (RDW) and pulmonary hypertension have the strongest association. 9 Although the implications of this finding remain to be determined, they suggest that PAH pathophysiology is not lung-specific, but rather, involves multiple organ systems.
Animal models can help to reveal cause-and-effect relationships of associated diseases when they closely mimic the human condition. A rat PAH model induced by a single injection of Sugen 5416 followed by three weeks of exposure to 10% oxygen and then 3–10 weeks of normoxia, in addition to severe PAH development, uniquely reproduces occlusive vasculopathy of pulmonary arteries, which is not observed in more classic rat PAH models such as the chronic hypoxia and monocrotaline models.10–12 Using the Sugen/hypoxia model, we discovered that PAH progresses to death in Fischer rats, a finding that is atypical among most rodent species. 13 Subsequently, Jiang and colleagues revealed significant differences in disease severity and mortality between rat strains and colonies in this model; in their work, mortality in Fischer rats and selective Sprague-Dawley species was attributed to acute right heart failure. 14 These findings emphasize the contribution of genetic factors to PAH susceptibility, as heterogeneous populations of rats may mirror human PAH populations with variable disease courses. However, examining the Fischer Sugen/hypoxia model with greater emphasis on multiple organ systems may add insight into the complex physiologic milieu that contributes to death in PAH, and in doing so, identify unique subpopulations of patients best represented for translational therapies.
In our previous study, we demonstrated that Sugen, hypoxia, and normoxia exposure (Sugen/hypoxia PAH model) induces severe PAH in Fischer 344 rats, demonstrated by right heart catheterization, echocardiogram, and histopathologic examinations.13,15 Interesting questions left unanswered from that study were why PAH animals are susceptible to sudden death, and whether there is accompanying platelet and red blood cell changes in the setting of hypoxia and occlusive vasculopathy. Here, we hypothesized that PAH rats develop biventricular dysfunction leading to global tissue hypoperfusion, thrombocytopenia, and red blood cell macrocytosis. To test this hypothesis, we performed comprehensive echocardiographic assessments and blood analysis.
Materials and methods
Rat pulmonary hypertension models
All experimental procedures were performed in accordance with current provisions of the U.S. Animal Welfare Act and were approved by the University of South Alabama Institutional Animal Care and Use Committee. Male Fischer rats weighing approximately 200 g were used. PAH was induced by a single subcutaneous injection of 20 mg/kg Sugen 5416 (Semaxanib; MedKoo Biosciences, Chapel Hill, NC) followed by exposure to three weeks of normobaric hypoxia (10% O2) and then re-exposure to normoxia (21% O2) for a total of three to five weeks. 15 Hereafter, this model is referred to as Fischer experimental PAH. Age-matched animals bred in normoxia for three– to five weeks without Sugen 5416 injection or hypoxia exposure were used for control group for all experiments. Exposure to hypoxia for three weeks without Sugen 5416 injection was used as a “hypoxia only” control group for hematologic tests.
Echocardiography
A Vevo 3100 (VisualSonics, Toronto, ON, Canada) with a 30 MHz transducer (MX550D) was used at baseline and three, four, and five weeks after Sugen, hypoxia, and normoxia exposure to evaluate the progression of cardiac dysfunction. Rats were anesthetized but kept spontaneously breathing using isoflurane 1.5% (titrated as needed) in a 1:1 O2–air admixture. Heart rate, electrocardiogram, and respiration were continuously recorded using the sensor-embedded exam pad. To minimize potential cardiovascular stress induced by anesthesia, all measurements were completed within 20 min for each animal. From parasternal long axis view, M-mode was used to obtain left ventricular ejection fraction, stroke volume, and cardiac output. Left ventricular ejection fraction was calculated using modified Quinones method. From the parasternal short axis view at the pulmonary artery level, pulmonary artery acceleration time (PAAT)/pulmonary artery ejection time (PAET) ratio and right ventricular outflow tract (RVOT) velocity time integral (VTI) of pulmonary arterial flow were measured using pulsed wave Doppler. Apical four-chamber view was used to obtain M-mode tricuspid annular plane systolic excursion (TAPSE), mitral and tricuspid pulsed wave Doppler early (E) and late (A) filling waves, and tissue Doppler early diastolic (e'), atrial (a'), and systolic positive (S') wave velocities. In subcostal longitudinal inferior vena cava (IVC) view, minimal and maximum diameters of IVC at the hepatic venous inflow level were obtained. Image acquisition was performed by one operator, and image analysis was performed by two examiners with cardiologist consultation following the American Society of Echocardiography guidelines.16–18
Blood sampling and analysis
Blood was sampled from the left ventricle immediately after the final echocardiogram at three weeks for hypoxia control and five weeks for Sugen/hypoxia animals by aortic or cardiac puncture under isoflurane anesthesia. Complete blood count and serum chemistry analysis were performed at the University of South Alabama Children's and Women's hospital laboratory.
Statistics
Two-way ANOVA, Student's t-tests, and Bonferroni post hoc correction and Chi-square tests were used, as appropriate. Significance was denoted as P < 0.05.
Results
PAH induces characteristic flow pattern changes in the pulmonary artery
To confirm the development of PAH, we performed pulsed wave Doppler on pulmonary arterial flow using the short axis view (Fig. 1a). After Sugen 5416, hypoxia and normoxia exposure, there were characteristic notch pattern changes in the flow velocity envelope (Fig. 1b) and decreases in the PAAT/PAET ratio (Fig. 1c) and RVOT VTI (Fig. 1d). The findings are consistent with those from previous studies in which we also measured concomitantly elevated pulmonary artery pressure by right heart catheterization and echocardiogram.13,19 Thus, severe PAH was confirmed within three weeks of Sugen and hypoxia treatment.
PAH induces characteristic notch pattern changes and decreases PAAT/PAET ratio and RVOT VTI in pulmonary artery flow. (a) Echocardiographic evidence of PAH was assessed on the short axis view at the level of the aortic valves. Pulsed wave Doppler assessment of pulmonary artery flow shows (b) characteristic notch pattern changes (arrow) and (c) decreased PAAT/PAET ratio and (d) RVOT VTI in Fischer rats exposed to Sugen 5416, hypoxia, and normoxia.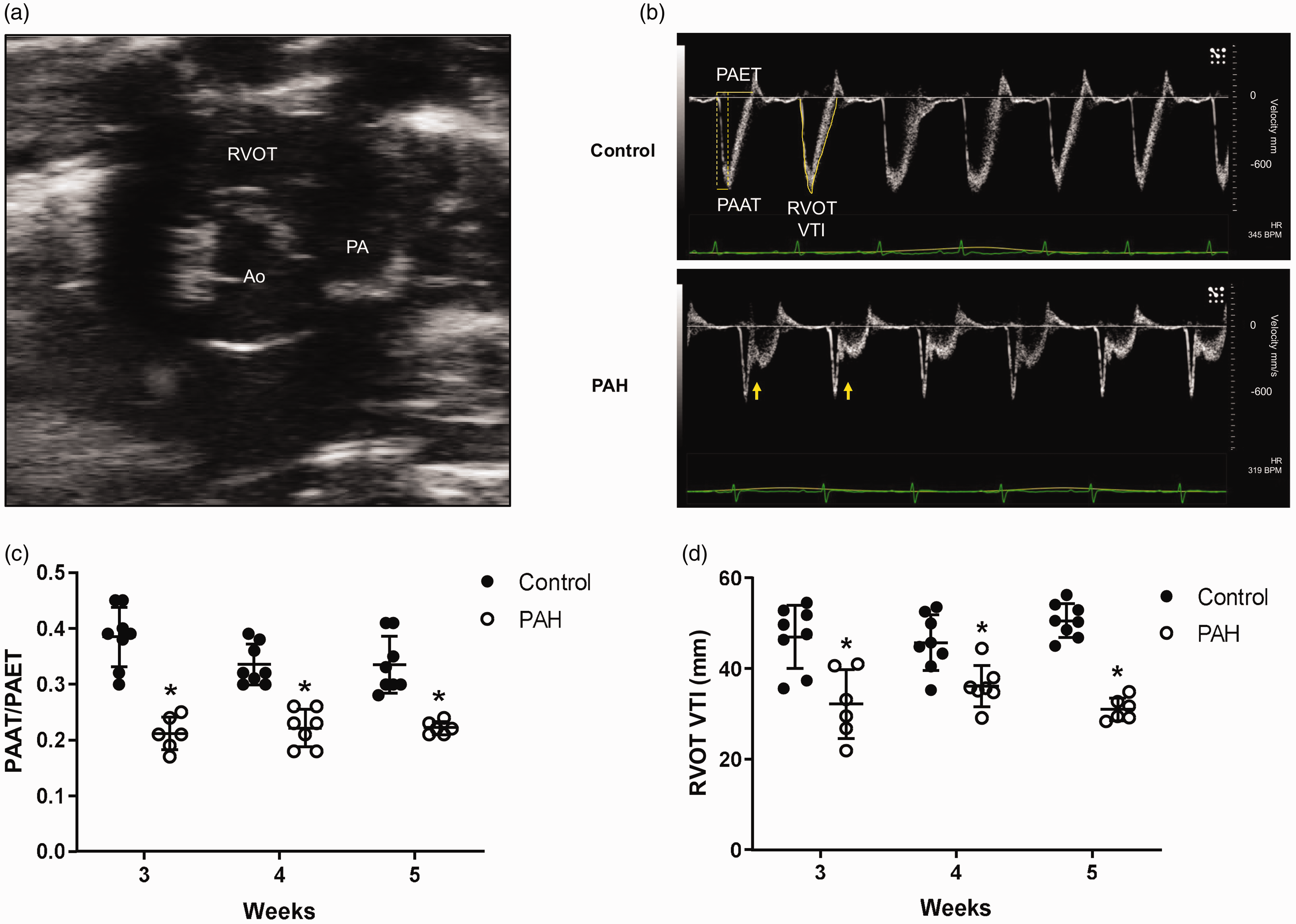
PAH induces right ventricular systolic dysfunction
During PAH development, the right ventricle increases its mass and contractility to overcome increased afterload. As pulmonary arterial pressure further increases, the right ventricle dilates and its systolic function is compromised.
20
In this cohort, PAH animals developed right ventricular dilatation and septal flattening with paradoxical movements (Fig. 2a and b and supplemental movies S1 (https://figshare.com/articles/Supplemental_movie_S1/7949504), i.e. control, and S2 (https://figshare.com/articles/Supplemental_movie_S2/7949528), i.e. PAH). To assess the impact of PAH on the right heart function over time, we assessed TAPSE and S' velocity of the tricuspid annulus, two well-validated parameters of right ventricular systolic function, at three, four, and five weeks. There were significant decreases in both TAPSE (Fig. 2c and d) and tricuspid S' velocity (Fig. 2e and f) in PAH throughout the time course. The finding suggests that maximal development of right ventricular systolic dysfunction is at, or earlier than, three weeks in the Fischer experimental PAH model.
PAH decreases TAPSE and S' velocity. PAH-induced RV dilatation and interventricular septal flattening (arrows) is demonstrated (a) in the short axis view at the level of the mid-ventricular level and (b) in the apical four chamber view. To assess right ventricular systolic function, M-mode of right lateral ventricular wall and tissue Doppler imaging on lateral tricuspid annulus were performed on the apical four chamber view. PAH rats developed right ventricular systolic dysfunction indicated by decreased (c and d) TAPSE and (e and f) lateral tricuspid annulus tissue S' velocity. Please note the change in Y-axis scale among images.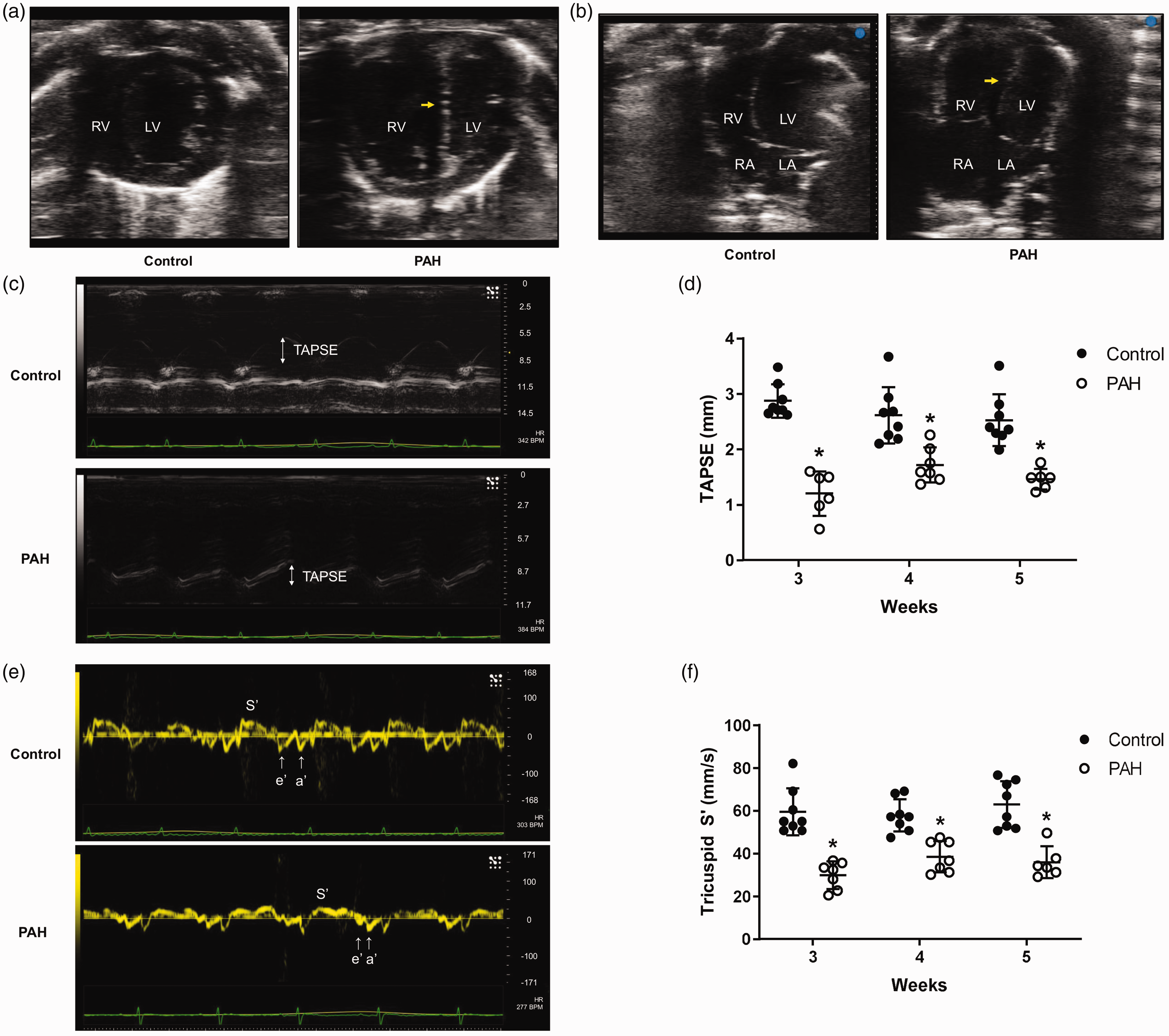
PAH induces evolving right ventricular diastolic dysfunction
Right ventricular diastolic dysfunction complicates clinical management of the PAH patient. When right ventricular relaxation is impaired, a modest degree of tachycardia can trigger sudden circulatory collapse due to insufficient right ventricular filling, subsequently causing a rapid decrease in left ventricular preload and cardiac output.
21
To assess right ventricular diastolic function, we analyzed tricuspid inflow patterns and tissue Doppler imaging of tricuspid lateral annulus over the course of three to five weeks. Compared to control, where E and A waves were clearly distinguishable except in one animal, there was a higher percentage of subjects that developed EA fusion waves, reaching 100% by five weeks, in PAH (Fig. 3a and b). Going with this trend, tricuspid inflow time to cardiac cycle time ratio was lower in PAH, and lower in five weeks compared to three weeks among PAH subjects (Fig. 3a and c). Tricuspid tissue e' velocity was decreased in PAH (Figs. 2d and 3d). There was no statistically significant change in the heart rate throughout the time course (Fig. 3e). The findings suggest evolving right ventricular diastolic dysfunction in PAH.
PAH increases tricuspid E and A wave fusion, decreases tricuspid inflow time to cardiac cycle time ratio, and e' velocity. To assess right ventricular diastolic function, tricuspid pulsed wave Doppler and lateral tricuspid annulus tissue Doppler imaging was performed on the apical four chamber view along with continuous heart rate monitoring. PAH rats developed evolving right ventricular diastolic dysfunction indicated by (a and b) increased frequency of E and A wave fusion (a and c) decreasing tricuspid inflow time to cardiac cycle time ratio over time, and (d) decreased e' velocity (e) without changes in the heart rate.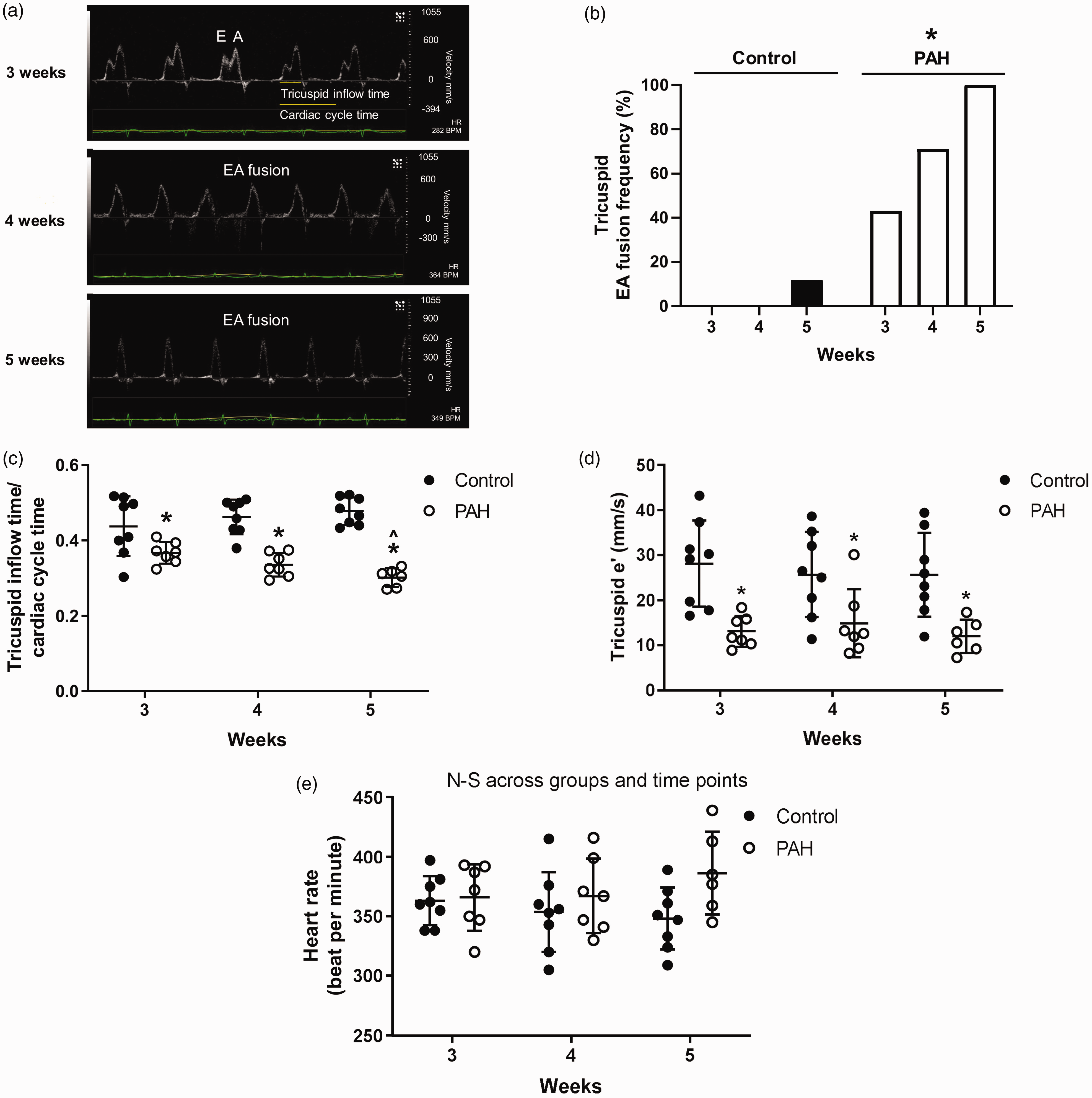
PAH induces left ventricular diastolic dysfunction with preserved ejection fraction
Most PAH studies in animal models mainly focus on assessing right heart function. However, since right and left ventricles do not contract separately, their function should be inter-dependent in nature.
22
Therefore, we sought to determine how left ventricular function is affected by the right heart as PAH and right ventricular dysfunction progress. We first assessed left ventricular ejection fraction. There was no change in left ventricular ejection fraction between control and PAH suggesting preserved left ventricular systolic function (Fig. 4a and b). To assess left ventricular diastolic function, we analyzed mitral inflow patterns and tissue Doppler imaging of the mitral medial annulus over the course of three to five weeks. Similar to tricuspid inflow patterns, there was a higher percentage of E and A wave fusion (Fig. 4c and d) and decreased mitral inflow time to cardiac cycle time in PAH (Fig. 4c and e). However, unlike tricuspid inflow time to cardiac cycle time ratio, which further decreased from three weeks to five weeks, mitral inflow time to cardiac cycle time ratio did not change over time in PAH. Mitral tissue e' velocity was decreased in PAH (Fig. 4f and g). The findings suggest left ventricular diastolic dysfunction with preserved systolic function in PAH.
PAH preserves ejection fraction but increases E and A fusion, decreases mitral inflow time to cardiac cycle time ratio, and decreases e' velocity. To assess left ventricular systolic and diastolic function M-mode of left ventricle, pulsed wave Doppler of mitral inflow and tissue Doppler of medial mitral annulus were performed on the apical four chamber view. (a and b) Left ventricular ejection fraction was preserved in PAH. (c and d) There was a higher percentage of E and A wave fusion and (c and e) decreased mitral inflow time to cardiac cycle time ratio in mitral inflow. (f and g) e' velocity was significantly low in PAH.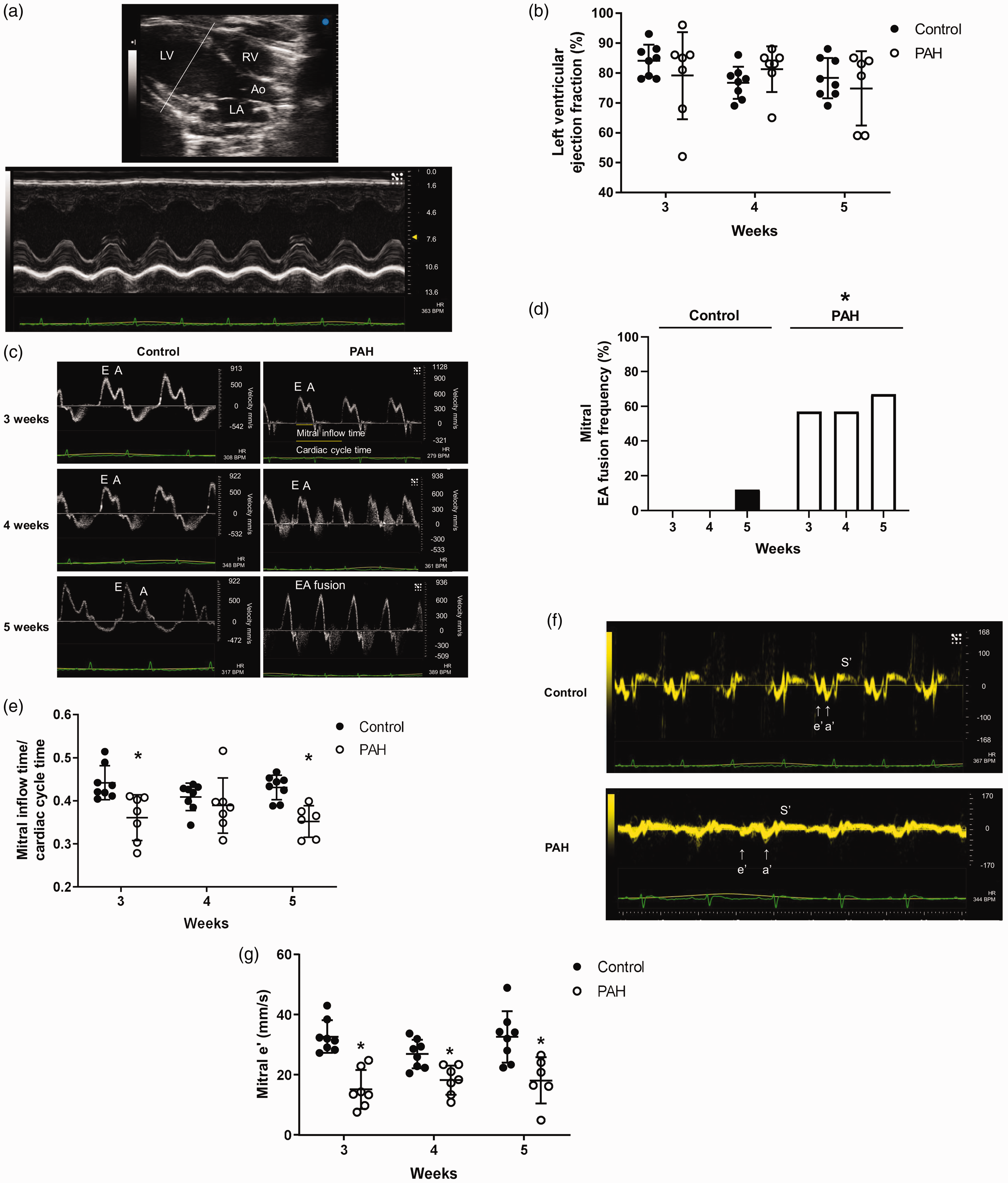
PAH decreases cardiac output and circulating intravascular volume
To assess the impact of biventricular diastolic failure on systemic circulatory status, we estimated cardiac output, stroke volume, and end-diastolic volume and measured IVC respiratory variation. Consistent with our previous report,13,15 cardiac output (Fig. 5a), stroke volume (Fig. 5b), and left ventricular end-diastolic volume (Fig. 5c) were decreased in PAH. Minimal IVC diameter was increased toward four weeks, and then normalized at five weeks (Fig. 5d and e). There was no difference in maximal IVC diameter between groups throughout the time points (data not shown). IVC respiratory variation, a surrogate for right atrial pressure and fluid responsiveness,
23
was decreased at four weeks then normalized at five weeks (Fig. 5f). The findings may indicate early venous congestion that resolves over time as total effective intravascular volume decreases towards five weeks.
PAH decreases cardiac output, stroke volume, and left ventricular end-diastolic volume and temporarily decreases IVC respiratory variation at four weeks. To assess overall intravascular volume status, cardiac output and left ventricular end-diastolic volume were estimated from M-mode measurements of left ventricle on the parasternal long axis view, and IVC diameter and respiratory variation were measured on the subcostal longitudinal IVC view. PAH decreased (a) cardiac output at three to five weeks, (b) stroke volume at three and four weeks, and (c) left ventricular end-diastolic volume at three to five weeks. (d and e) Minimal IVC diameters were increased and (f) IVC respiratory variation was decreased at four weeks indicating relatively increased right atrial pressure.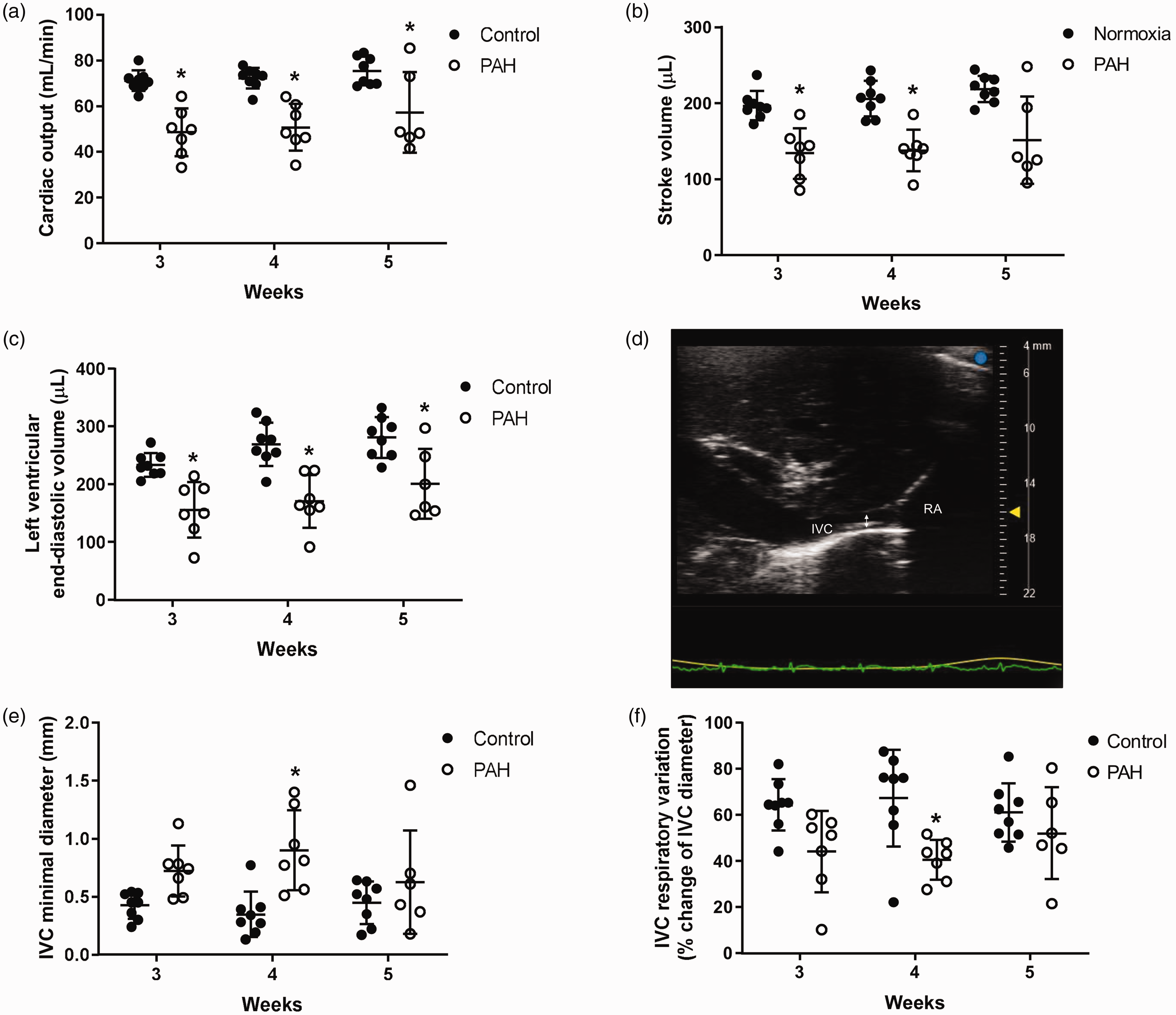
PAH induces biochemical features of volume contraction and tissue hypoperfusion
Comprehensive metabolic panel.
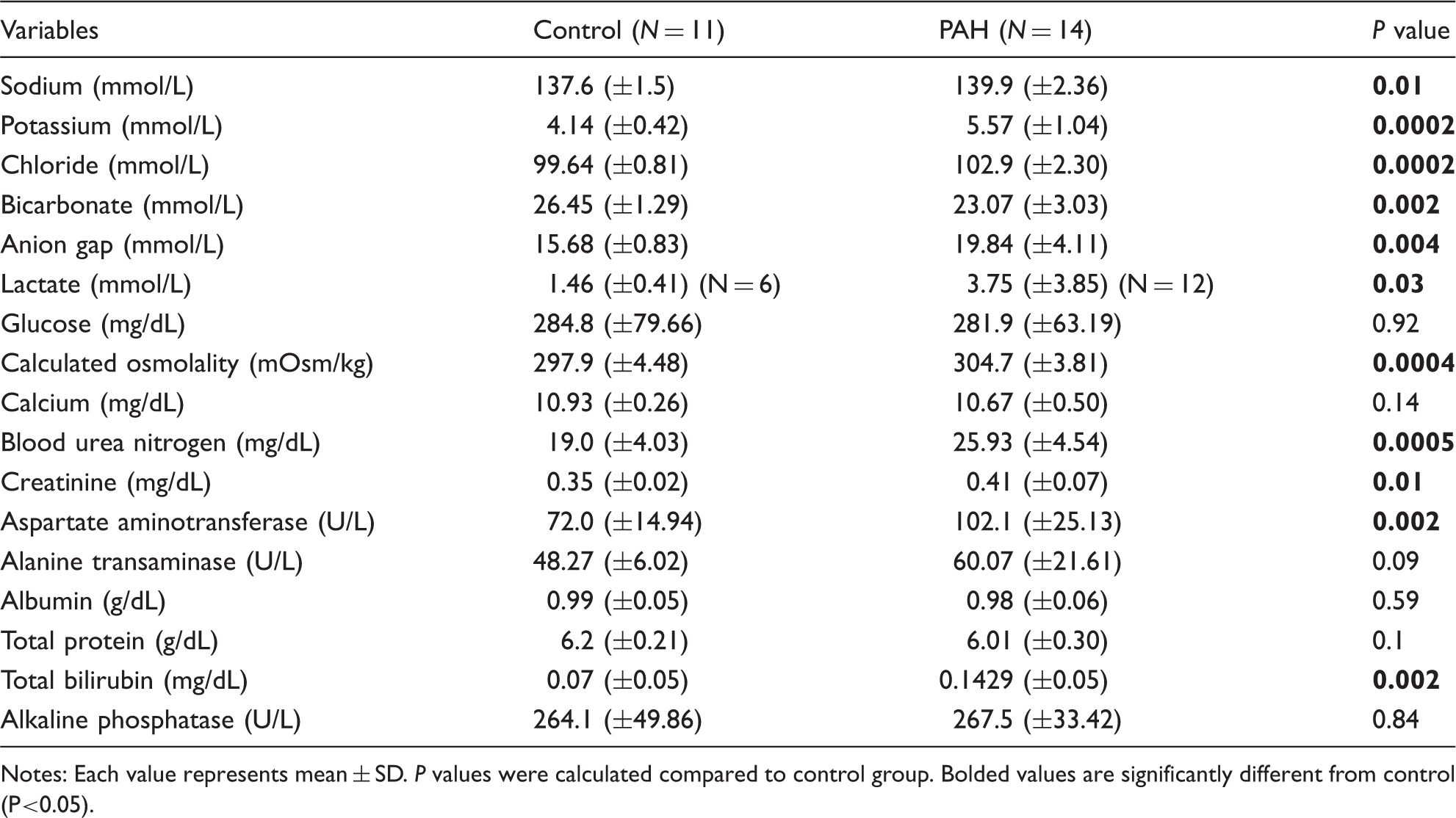
Notes: Each value represents mean ± SD. P values were calculated compared to control group. Bolded values are significantly different from control (P<0.05).
PAH induces thrombocytopenia, red blood cell macrocytosis, and increased red blood cell distribution width
Complete blood count.
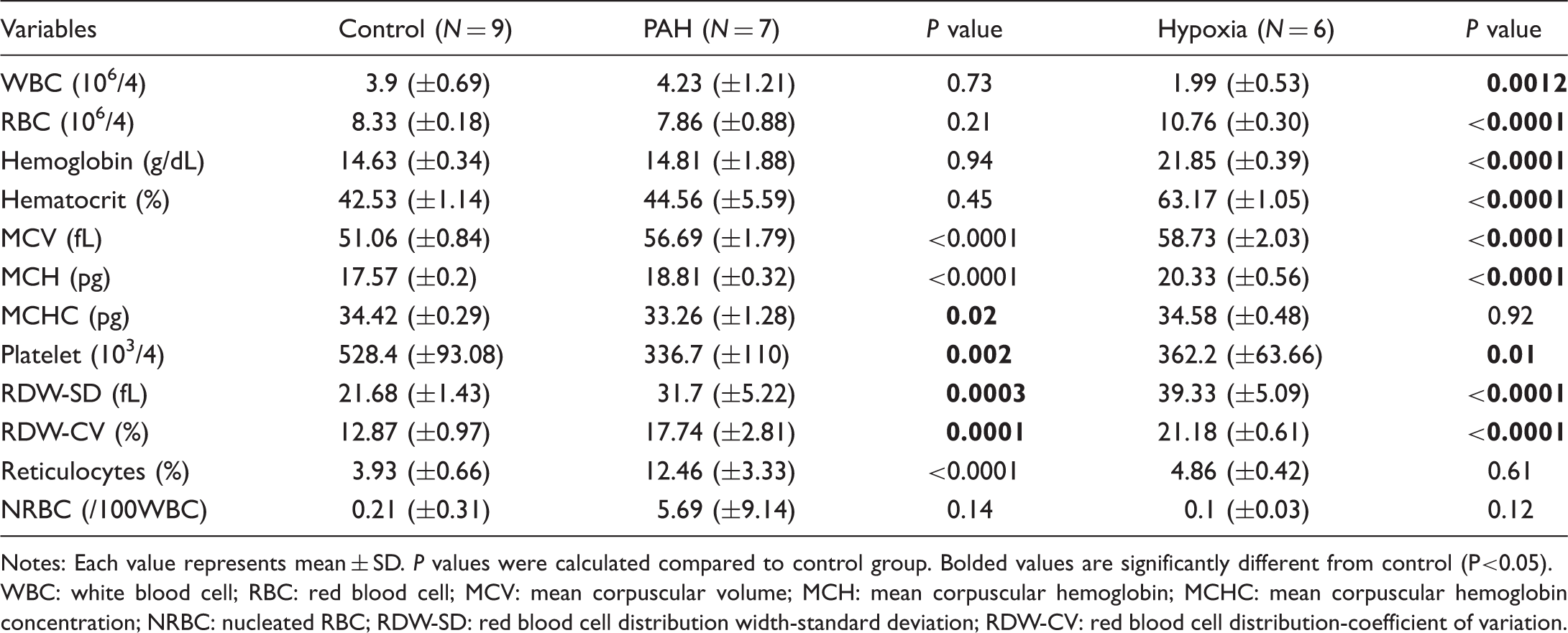
Notes: Each value represents mean ± SD. P values were calculated compared to control group. Bolded values are significantly different from control (P<0.05).
WBC: white blood cell; RBC: red blood cell; MCV: mean corpuscular volume; MCH: mean corpuscular hemoglobin; MCHC: mean corpuscular hemoglobin concentration; NRBC: nucleated RBC; RDW-SD: red blood cell distribution width-standard deviation; RDW-CV: red blood cell distribution-coefficient of variation.
Discussion
In this study, we performed comprehensive echocardiographic and hematologic assessments over three to five weeks on Fischer rats with severe experimental PAH. This time frame represents the typical time points when severe PAH develops but death is yet to occur; a majority of animals die between six and eight weeks.13,14 We report three major findings, that Fischer experimental PAH induces: (1) biventricular diastolic dysfunction; (2) thrombocytopenia; and (3) red blood cell macrocytosis.
Studies on diastolic dysfunction in PAH are relatively limited. When considering the right ventricle, the association between diastolic dysfunction and PAH severity has been described and is thought to be due to myocardial hypertrophy, fibrosis, and sarcomere stiffening.25,26 Neto-Neves et al. has recently shown right ventricular diastolic dysfunction in the Sugen/hypoxia Sprague-Dawley PAH model, using an ex vivo isolated perfused heart preparation (Langendorff preparation). 27 To our knowledge, no study has documented echocardiographic evidence of right diastolic dysfunction in the Sugen/hypoxia PAH model. In our study, decreased e' velocity represented a consistent echocardiographic sign of diastolic dysfunction in the right ventricle. In addition, diastolic parameters such as tricuspid EA fusion frequency and tricuspid inflow time to cardiac cycle time ratio dynamically evolved over time. This finding suggests that right ventricular diastolic dysfunction may help distinguish severity of PAH and can be useful in monitoring the efficacy of therapeutic interventions.
In addition to right ventricular diastolic dysfunction, we found evidence for left ventricular diastolic dysfunction in this model. Left ventricular dysfunction has been documented in both pulmonary hypertension due to left heart failure and in PAH.20,28 In the former example, elevated left atrial pressure elicits remodeling of the pulmonary veins and the microcirculation, leading to elevated pulmonary artery and ultimately right ventricular pressures. This vascular progression culminates in a combined post- and pre-capillary pulmonary hypertension, where the cause of pulmonary hypertension is left ventricular failure that can occur with or without preserved ejection fraction. In contrast to this cause of pulmonary hypertension, PAH with severe right ventricular remodeling leads to ventricular interdependence causing impaired left ventricular filling29,30 and/or coexisting cardiomyopathy. 31 Diastolic dysfunction can occur even when PAH severity is mild. 32 While PAH initiates left ventricular diastolic dysfunction in this case, the resulting elevations in left atrial pressure can subsequently lead to remodeling of the pulmonary veins and microcirculation. 33 We have recently found elevated post-capillary resistance in the Fischer PAH model. 34 Future studies will be required to assess the relevance of left ventricular diastolic dysfunction to remodeling of the pulmonary veins in this model. Nonetheless, it is clear that the cause of left ventricular dysfunction in the Sugen/hypoxia Fischer PAH model is the pre-capillary vasculopathy; this model mimics human pre-capillary PAH that progresses into late-term biventricular failure with post-capillary involvement.
Fluid management in critically ill PAH patients with right heart failure is extremely challenging. 21 Minimal changes in intravascular volume can quickly overwhelm the poorly compliant right heart and result in sudden circulatory collapse. Despite its clinical relevance, fluid status is rarely reported in experimental cardiac disease models. A study with the aorta-to-vena cava fistula heart failure rat model demonstrated water retention and hyponatremia 20 days after surgery, 35 and another study using the coronary artery ligation heart failure rat model showed hyponatremia and heart weight increase three weeks postoperatively, 36 both suggesting “fluid retention” as a consequence of heart failure. As there is no single tool that can absolutely determine intravascular volume status, 37 we combined evidence from echocardiogram and serum chemistry panels. Over the course of three to five weeks, biventricular diastolic failure, respiratory variation of IVC nadiring at four weeks, and upward trending of the heart rate in the setting of serologic evidence of tissue hypoperfusion at five weeks, altogether suggests initial (3–4 weeks) venous congestion followed by later (five weeks) intravascular volume contraction. Although the mechanisms leading to dehydration in our PAH animals is unclear, we speculate that the degree of cardiovascular dysfunction may be severe enough to limit the activity and fluid intake of the PAH animals to ultimately result in failure to thrive. Notably, our late-stage PAH animals are extremely vulnerable to external stimuli, such as cage changes or anesthesia causing sudden death, the phenomenon similar to human PAH patients being at high risk for surgery. The findings emphasize the importance of careful systemic fluid status analysis in interpreting cardiovascular experimental models.
Thrombocytopenia predicts mortality among pulmonary hypertension of all groups, 24 and the same is true when PAH populations are considered separately. 8 Furthermore, there has been a report indicating increased platelet activation in PAH patients when mean platelet volume was used as a marker of platelet activation. 38 However, whether platelets actively drive vascular injury and inflammation or are consumed during PAH disease progression is still debatable, 39 and no randomized controlled trial supports antiplatelet therapies. Endothelial dysfunction in PAH has been well-described, and platelets are mediators of major drug target pathways including nitric oxide, prostacyclin, and endothelin-1. 40 Platelets may contribute to occlusive neointimal lesions in arterioles and our recent study demonstrating hyperpermeable characteristic of the PAH pulmonary vasculature 34 raises the possibility that platelets could be consumed in the leaky perivascular tissue beds, but further studies elucidating pulmonary and extrapulmonary mechanisms of thrombocytopenia are warranted. Studying PAH-specific pathophysiology involving thrombocytopenia in human populations is particularly challenging, due to the high prevalence of multiple confounding factors including comorbidities known to affect platelet counts, such as liver disease, or medications that can potentially decrease platelet counts in the PAH population, such as anticoagulants.41,42 To our knowledge, this is the first report of thrombocytopenia as a phenotype of experimental PAH. Considering the normal lab value range for rats is higher than that of humans, 43 and hemoconcentration due to dehydration may be a confounding effect, the severity of thrombocytopenia in our study is profound. This model may help reveal mechanisms involved in development of thrombocytopenia in selected PAH patient populations.
Thayer et al. recently found that RDW portends poor outcomes for non-anemic PAH patients. 9 The value of RDW as a biomarker for multiple cardiovascular diseases has long been suggested,44,45 but the pathophysiology involved has not been fully elucidated. Interestingly, our PAH rat model developed a non-anemic macrocytic red blood cell phenotype, with increased immature red blood cell production resulting in an increase in RDW. Although hemoglobin was not decreased, it may have been masked by hemoconcentration as discussed above. Since our results indicate actively ongoing concomitant red blood cell loss and production, if we had carried out our study longer, the animals ultimately could have developed iron deficiency.
Anemia and iron deficiency have been reported as poor prognostic markers in pulmonary hypertension. A cohort study of all group pulmonary hypertension demonstrated that hemoglobin levels parallel survival of the patients. 46 Among systemic sclerosis patients, iron deficiency was more prevalent in PAH patients, and among those PAH patients, iron deficiency predicted worse mortality. 47 Our hypoxia-only animal group possessed a hematopoietic response that was characterized by increased red blood cell counts, hemoglobin, hematocrit, MCV, and RDW. Whereas PAH animals showed similarly increased MCV and RDW, their red blood cell counts, hemoglobin, and hematocrit were normal. Furthermore, reticulocyte counts were higher in PAH animals than they were in the control animals, and there was no difference between control and hypoxia-only groups. This normal reticulocyte count in the hypoxia-only group may be because hypoxia-induced polycythemia is prominent, and additional red blood cell production is not necessary. It remains unclear as to whether increased hemolytic activity contributes to normalization of hematocrit in PAH animals. Overall, we speculate that an increase in red blood cell consumption or destruction related to vasculopathy,48,49 and an increase in red blood cell production, in part due to the hypoxic hematopoietic response, 50 resulted in macrocytic red blood cell phenotype with high RDW. Since none of hemolytic parameters are highly sensitive or specific as a standalone test, further comprehensive hemolytic workups would be required to confirm hemolytic process in future mechanistic studies.
In conclusion, experimental PAH in Fischer rats reproduces a number of important phenotypes highly relevant to PAH patients. Prior studies highlighted the severe PAH in this animal model, with medial hypertrophy and adventitial thickening, endothelial hyperpermeability, and neo-intimal occlusive lesions, including grades 3, 4, and 6 lesions. 13 Here, we report the animals display biventricular diastolic failure, thrombocytopenia, and red blood cell macrocytosis with high RDW, ultimately predisposing them to sudden death. Our findings emphasize the importance of understanding PAH as a multi-system disease, and, therefore, its therapeutic approach should not be limited to modifying pulmonary vascular and/or right ventricular remodeling. This animal model will be helpful to test and translate therapeutics applicable to selected PAH subpopulations with relevant phenotypes.
Footnotes
Acknowledgements
The authors thank Dr. Gary Ellis Carnahan and Leigh Ann Wiggins for their assistance with blood test analysis and animal anesthesia, respectively.
Author contributions
Conception and design: J.Y.L. and T.S.; data acquisition: J.Y.L., C.Z., and T.S.; analysis and interpretation: J.Y.L., K.A.F., C.Z., L.B., M.V.C., and T.S.; and drafting the manuscript for important intellectual content: J.Y.L. and T.S.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Ethical approval
All animal studies were approved by University of South Alabama IACUC
Funding
This work was supported by 18CDA34080151 (J.Y.L.), HL66299, and HL60024 (T.S.).
