Abstract
Increased risk and severity of idiopathic pulmonary arterial hypertension (iPAH) is associated with elevated estradiol in men and postmenopausal women. Pulmonary arteries synthesise estradiol via aromatase and metabolise it via CYP1B1 to mitogenic metabolites; SNPs in aromatase and CYP1B1 have been associated with PAH. This suggests that estradiol metabolism could be altered in iPAH. This proof-of-concept study profiles estradiol and several metabolites of estradiol simultaneously in serum from iPAH patients and controls. We show that the estradiol and metabolite profile is altered in iPAH and that 16-hydroxyestrone and 16-hydroxyestradiol accumulate in iPAH patients with 16-hydroxyestrone levels relating to disease severity.
Introduction
Increased risk and severity of idiopathic pulmonary arterial hypertension (iPAH) is associated with elevated plasma estradiol in both men and postmenopausal women,1,2 suggesting that estradiol is not ovarian-derived. Aromatase is responsible for the synthesis of estradiol. Remodeled pulmonary arteries, lesions, and human pulmonary artery smooth muscle cells (hPASMCs) from pulmonary arterial hypertension patients have high levels of aromatase.3 Cytochrome P450 Family 1 Subfamily B Member 1 (CYP1B1) can convert estradiol to mitogenic 16-hydroxyestrogens and this enzyme is over-expressed in diseased pulmonary arteries from pulmonary arterial hypertension (PAH) patients.4 Existing assays have not been sensitive or specific enough to detect levels of many estradiol metabolites in PAH patient blood. Using a newly developed liquid chromatography tandem mass spectrometry (LC-MS/MS) assay,5 here we investigate the profile of several estradiol metabolites in iPAH patients.
Methods
Serum from 22 subjects with iPAH (12 males, 10 females) and 34 healthy control subjects (17 males, 17 females) were analyzed by LC-MS/MS for estrone (E1), 17α-estradiol (17αE2), 17β-estradiol (E2), 16-hydroxyestrone (16OHE1), 16-hydroxyestradiol (16OHE2), 2-methoxyestrone (2MeOE1), 4-methoxyestrone (4MeOE1), 2-methoxyestradiol (2MeOE2), and 4-methoxyestradiol (4MeOE2). 5 Data were compared by a Mann–Whitney U test with associations assessed using Pearson’s correlations. Values below the confirmed limit of quantification (LOQ) were imputed as the LOQ (2 pg/mL E1, E2, 16OHE2, 2MeOE1, and 6 pg/mL 16OHE1).
To determine if 16OHE2 was present at bioactive concentrations, proliferation was studied in hPASMCs and cell migration was studied in human blood outgrowth endothelial cells (BOECs). hPASMCs were isolated from distal pulmonary arteries (0.3–1 mm diameter) from macroscopically normal controls or patients undergoing lung transplantation. BOECs were isolated from PAH patients as described previously 6 with cells cultured as described previously. 7 The patients were male and female (heritable PAH/iPAH). Cells were stimulated with 16OHE2 following preliminary concentration-response analysis (10 nM hPASMCs and 1 nM BOECs) for 48 h in phenol red-free media. Cell counts and cell migration (Corning® Transwell inserts) were assessed using a Countess II FL cell counter (Life Technologies, UK). A student’s t-test was applied for comparison of 16OHE2 vs vehicle-treated cells for each group.
Results
Clinical characteristics and estrogen concentrations for controls vs iPAH patients.
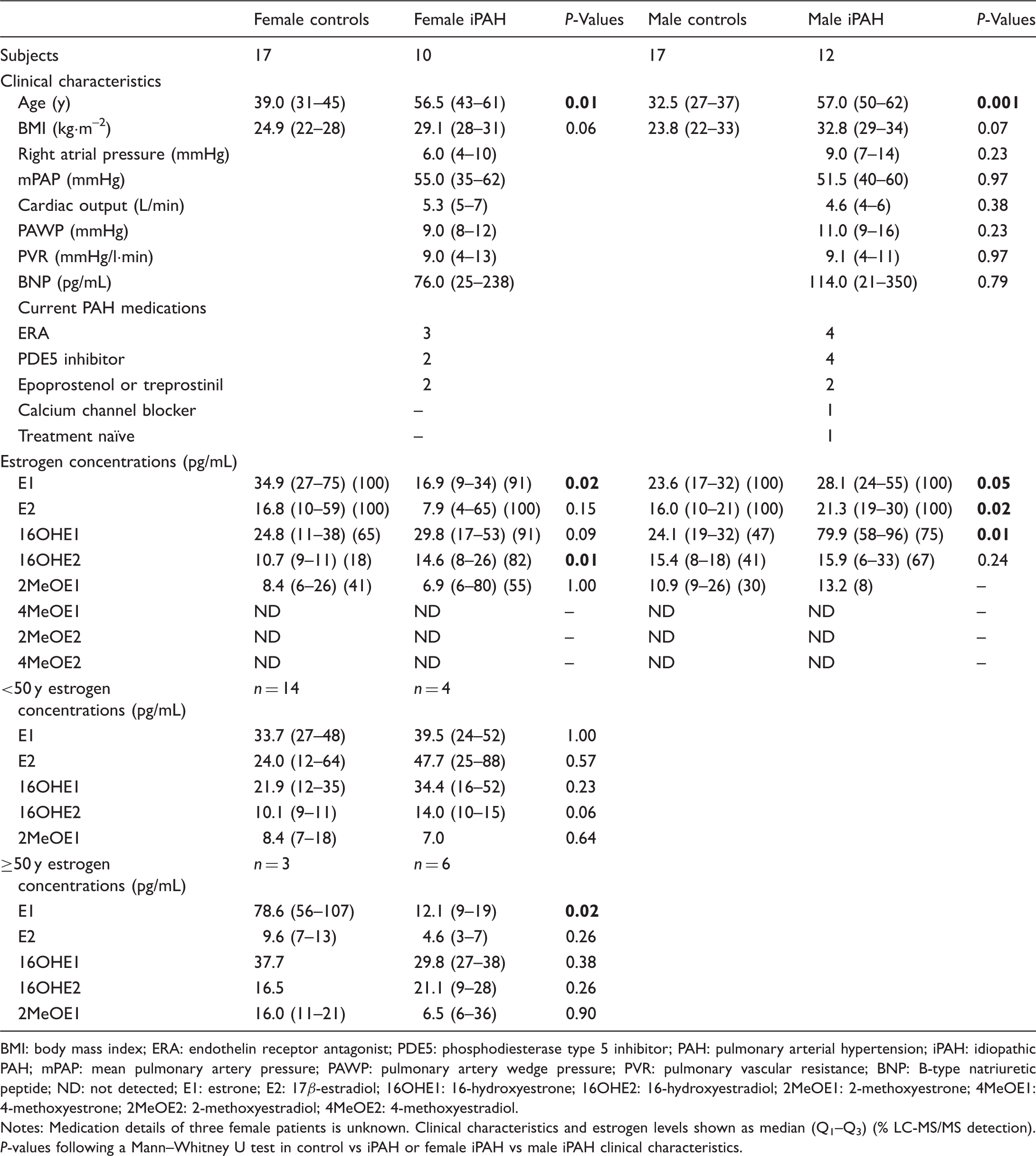
BMI: body mass index; ERA: endothelin receptor antagonist; PDE5: phosphodiesterase type 5 inhibitor; PAH: pulmonary arterial hypertension; iPAH: idiopathic PAH; mPAP: mean pulmonary artery pressure; PAWP: pulmonary artery wedge pressure; PVR: pulmonary vascular resistance; BNP: B-type natriuretic peptide; ND: not detected; E1: estrone; E2: 17β-estradiol; 16OHE1: 16-hydroxyestrone; 16OHE2: 16-hydroxyestradiol; 2MeOE1: 2-methoxyestrone; 4MeOE1: 4-methoxyestrone; 2MeOE2: 2-methoxyestradiol; 4MeOE2: 4-methoxyestradiol.
Notes: Medication details of three female patients is unknown. Clinical characteristics and estrogen levels shown as median (Q1–Q3) (% LC-MS/MS detection). P-values following a Mann–Whitney U test in control vs iPAH or female iPAH vs male iPAH clinical characteristics.
In female iPAH patients, increased E1 was associated with increasing pulmonary artery wedge pressure (PAWP) (R2 = 0.5, β = 0.7, p = 0.03), decreased E2 was associated with increasing age (R2 = 0.7, β = –0.8, p = 0.02), and increasing E2 with increasing PAWP (R2 = 0.6, β = 0.6, p = 0.05) while increased 16OHE1 correlated with a reduction of cardiac output (R2 = 0.7, β = –0.6, p = 0.01). In male iPAH, increased E2 was associated with increased PAWP (R2 = 0.5, β = 0.6, p = 0.02) while increased E1 and E2 associated with increasing BNP levels (R2 = 0.6, β = 0.8, p = 0.01 and R2 = 0.4, β = 0.6, p = 0.04).
16OHE2 caused proliferation of hPASMCs from female PAH patients (by 63%) but not controls (male or female) (Fig. 1a–c). 16OHE2 caused increased migration of BOECs from PAH patients, both in males (by 55%) and females (by 67%) (Fig. 1d).
16OHE2 stimulation increases proliferation and migration of human cells. 10 nM 16OHE2 increases proliferation in female PAH (a) with no effect on female control (b) and male control (c) human PASMCs. 1 nM 16OHE2 increased migration of male and female BOECs derived from PAH patients (d).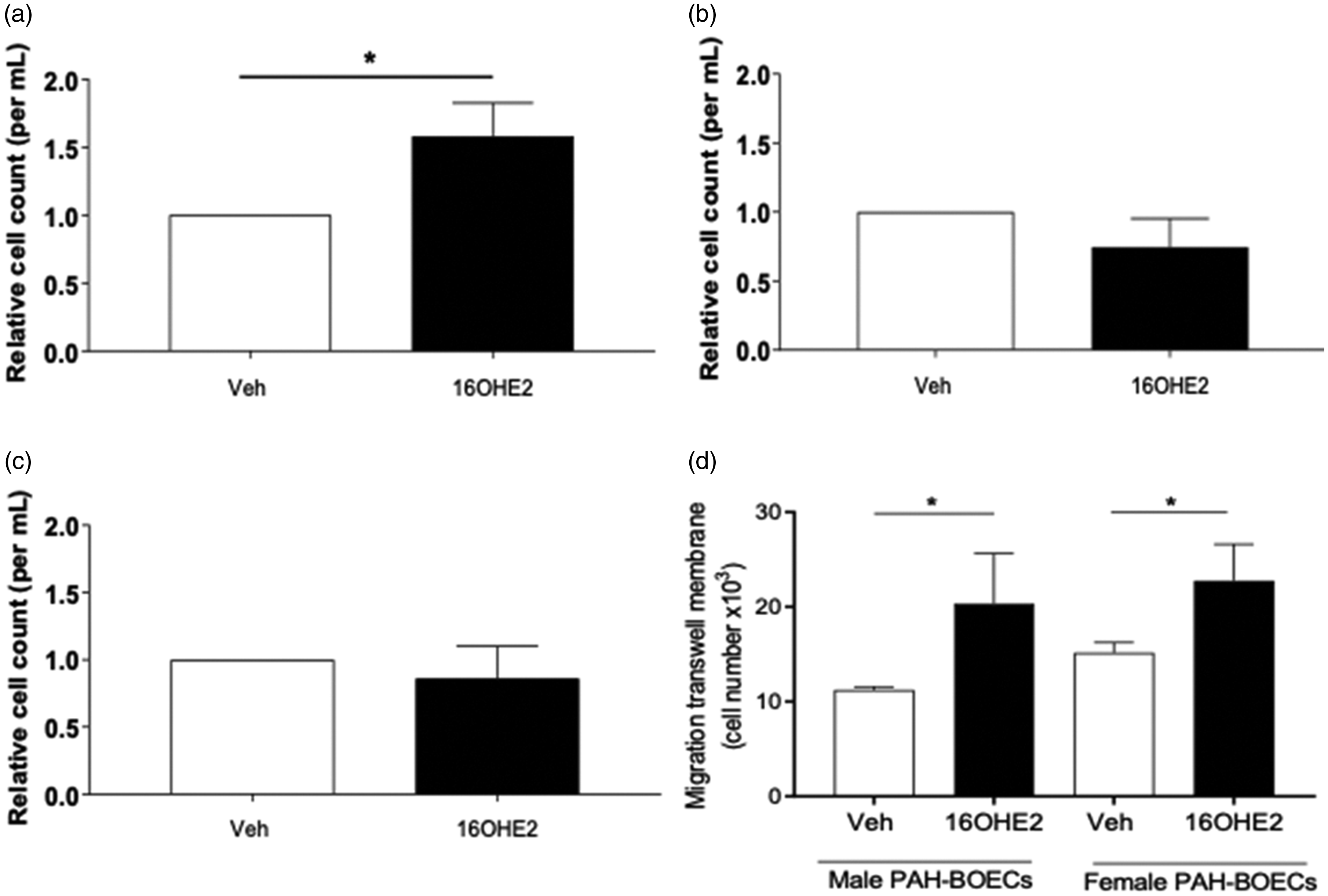
Discussion
This study is the first to simultaneously quantify several estrogens in controls and PAH patients as our methodological paper measured levels in pooled plasma samples. 5 LC-MS/MS allowed discrimination between isomers of E2 and its bioactive metabolites such as 16OHE1, 16OHE2, and methoxyestrogens. Increased 16-hydroxylation of E1/E2 promotes formation of proliferative metabolites and we identified accumulation of 16OHE2 in female iPAH patient serum for the first time. We confirmed increased levels of 16OHE1 in patients which had been suggested by previous studies.8,9 In female iPAH, 16OHE2 was also increased. There was increased E1 and E2 levels in male iPAH patients, consistent with a previous study using immunoassay detection. 1 The E2 concentrations we determined here were lower compared with previous studies determined using immunoassays. This is consistent with immunoassays being limited by cross-reactivity and over-estimation of E2 levels compared with LC-MS/MS. 10
Aromatase and CYP1B1 are increased in PAH patient pulmonary arteries and pulmonary vascular lesions.3,4 Increased aromatase activity may account for increased plasma E2 in men and post-menopausal women with iPAH.1,2 In female iPAH, there was a decrease in E1 with no change in E2. The change in E1 may reflect an increased conversion to 16OHEs via CYP enzymes as reflected by increased 16OHE1 and 16OHE2 in these patients. Where there is a decrease in E1 with no change in E2, this could also be due to the increased metabolism of E2. Baird et al. recently demonstrated that E2 levels are elevated in post-menopausal iPAH female patients compared with controls; however, E2 levels from controls in that study were 5–7 pg/mL, commensurate with post-menopause levels. 2 The majority of females in this current study were pre-menopausal, with sampling not routinely timed to consider menstrual cycle, and mean E2 concentrations in controls and patients were ∼34 pg/mL. Hence, it is perhaps unlikely that a further increase would be expected in this patient cohort. Future clinical studies could consider blood sampling from pre-menopausal women at the same stage of the menstrual cycle. 3
As 16OHE1 can be derived directly from E1 and male patients have higher E1, this may also contribute to the elevated 16OHE1 in these patients. Consistent with increased 16OHEs observed in iPAH patients, polymorphisms in CYP1B1 are observed in PAH patients associated with functional differences in 16-hydroxylation, cancer risk, and E2-mediated carcinogenicity.11,12 Conversion of E2 to 16OHE1 by CYP1B1 may contribute to the pathogenesis of experimental pulmonary hypertension and cause proliferation of hPASMCs via increased oxidative stress.7,8
We have already demonstrated that 16OHE1 can cause proliferation of both male and female hPASMCs and male mouse PASMCs at concentrations observed in plasma.4,7,13 16OHE1 acts through the ERα receptor in hPASMCs7,14 and indeed we have shown that ERα receptors are more highly expressed in female PAH hPASMCs. 15 This is consistent with female hPASMCs being more proliferative to E2, also due to decreased basal bone morphogenetic protein receptor II (BMPRII) signaling in female hPASMCs. 16 16OHE2 is a novel metabolite with unknown effects that was increased in the serum from female iPAH patients compared with controls. This could also be due to altered CYP1B1-mediated 16-hydroxlation.4,13 16OHE2 is an E2-receptor agonist, capable of cellular proliferation associated with cancer risk and E2-mediated carcinogenicity. 12 Consistent with this, we now show that, at a pathophysiological concentration, 16OHE2 can also induce proliferation in female human PAH PASMCs with no effect in female or male control PASMCs where ERα expression is not so high. However, we show that 16OHE2 increases cell migration in both male and female PAH BOECs. As 16OHE1 has been shown to act as a “second hit” to uncover a pulmonary hypertensive phenotype in BMPRII mutant male and female mice, 9 this suggests that both 16OHE1 and 16OHE2 may also facilitate pulmonary vascular remodeling in males and females by acting as a “second hit” to BMPRII mutations. 16OHE2 is also produced in abundance during pregnancy and it is interesting to speculate that it may contribute to pregnancy being a risk factor for PAH. 17
It should be noted that all of the medications that the patients were prescribed are metabolized in the liver by CYP enzymes such as CYP3A4 and CYP2C9/8 and have no known effects on lung or liver CYP1B1 or CYP1A1 activity. One limitation of this study is that the patients were older than the controls. However E2 and 16OHE1 levels correlated with indicators of disease severity within the patient groups and decreased E2 was associated with increasing age in iPAH patients. This suggests that increased E2/metabolite levels in PAH patients are related to disease status rather than difference in age.
The results suggest that estrogen metabolism is altered in PAH and both 16OHE1 and 16OHE2 accumulate, with 16OHE1 levels relating to disease severity. This study is limited by the small size but important as it warrants further multi-center fully powered studies to confirm and correlate metabolite levels with severity and sub-groups of PAH. Future studies on CYP enzymes and CYP1B1 inhibitors in patients with PAH may also be valuable.
Footnotes
Ethical approval
Yes.
Guarantor
Margaret R. MacLean.
Author contributions
Nina Denver: all LC-MS/MS, data analysis, hPASMC experiments, and preparation of manuscript; Natalie Homer and Ruth Andrew: analysis of LC-MS/MS; Katie Y. Harvey: BOEC experiments; Nicholas Morrell: isolation of BOECs; Eric D. Austin: blood sampling from all patients and controls; and Margaret R. MacLean: PI, grant holder, preparation of manuscript, and data analysis.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
The project described was supported by the British Heart Foundation, through grants RE/13/5/30177 and RG/16/2/32153, the Wellcome Trust (108468/z/15/z), the National Institutes of Health (R01HL134802 and P01HL108800), and a BBSRC iCASE PhD award (BB/N503691/1).
