Abstract
It has been shown previously that increased circulating endothelial cells-derived extracellular vesicles represent an important pathological attribute of pulmonary hypertension. Although it is a well-known fact that inflammatory cells may also release extracellular vesicles, and pulmonary hypertension is a disease associated with abnormal inflammation, there is no profound knowledge with regard to the role of inflammatory cells-derived extracellular vesicles. Therefore, our study demonstrated that circulating levels of extracellular vesicles derived from T-cells are enhanced in various pulmonary hypertension forms and that endothelial cells-derived extracellular vesicles may have distinctive profiles in different clinical subgroups of pulmonary hypertension, which still remains as a poorly treatable and life-threatening disorder.
To the Editor,
Microparticles (MPs) are a type of extracellular vesicles (EVs) and represent shed membrane structures, mostly found in the blood circulation, which originate from different cellular sources during apoptosis or/and activation.1,2 In the previous years, some studies described the altered circulating profiles of endothelial cells-derived and procoagulant MPs in the context of pulmonary hypertension (PH).2–5 Amabile et al. have demonstrated that patients with precapillary PH had significantly higher levels of circulating endothelial cells-derived MPs and some MPs correlated with increased mean pulmonary arterial pressure (mPAP). 2 In addition, EVs may also be active pathological players in pulmonary vascular disease development/progression, considering the fact that they represent carriers for various micro-RNAs. 6 Although there are evidences about the potential involvement of endothelial cells-derived MPs in the PH pathology, there is insufficient knowledge with regard to the inflammatory cells-derived MPs. 2 It is well known that massive accumulation of inflammatory/immune cells is a characteristic of PH, and inflammatory cells-derived MPs were found to play a pathogenic role in some lung disorders.7,8 Finally, the levels of circulating endothelial cells-derived MPs in different clinical forms of PH are still not analyzed in detail.
Therefore, our study aimed to investigate the circulating profiles of different inflammatory (CD3 (T-cells), CD14 (macrophages/monocytes), CD68 (macrophages)) and endothelial (CD62E (E-selectin) and CD144 (VE-cadherin)) cells-derived EVs in various clinical subgroups of PH.
Available clinical data of the patients with different forms of pulmonary hypertension (PH).
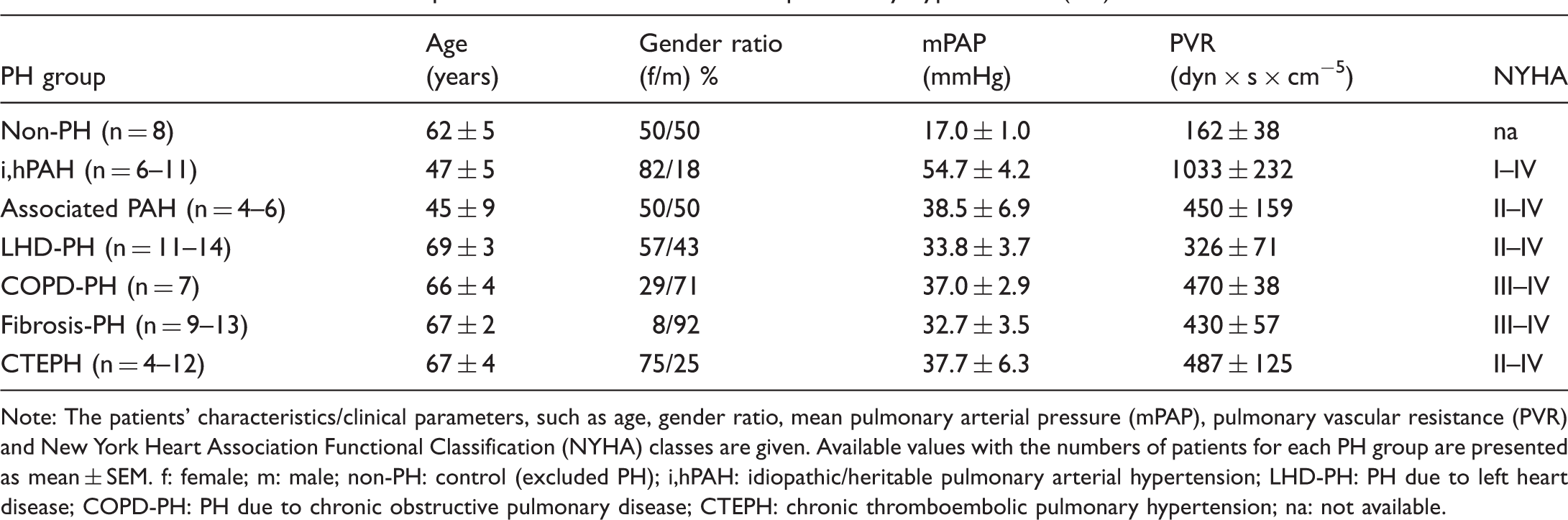
Note: The patients’ characteristics/clinical parameters, such as age, gender ratio, mean pulmonary arterial pressure (mPAP), pulmonary vascular resistance (PVR) and New York Heart Association Functional Classification (NYHA) classes are given. Available values with the numbers of patients for each PH group are presented as mean ± SEM. f: female; m: male; non-PH: control (excluded PH); i,hPAH: idiopathic/heritable pulmonary arterial hypertension; LHD-PH: PH due to left heart disease; COPD-PH: PH due to chronic obstructive pulmonary disease; CTEPH: chronic thromboembolic pulmonary hypertension; na: not available.
Results were expressed as events per microliter of plasma (events/µl) and presented as mean ± SEM in percentage, considering the average values of each non-PH group for all analyzed EVs as 100%. Due to the technical reasons, not all values for all analyzed targets and for all enrolled patients are available. ROUT test was used for identification of outliers. Further, unpaired T-test with Welch’s correction in the case of normally distributed values or Mann–Whitney test when values were not normally distributed were performed to compare non-PH control with respective PH groups. Finally, Spearman test was used for analyses of the correlations.
Profiles of circulating inflammatory and endothelial cells-derived extracellular vesicles (EVs) in different forms of pulmonary hypertension (PH).
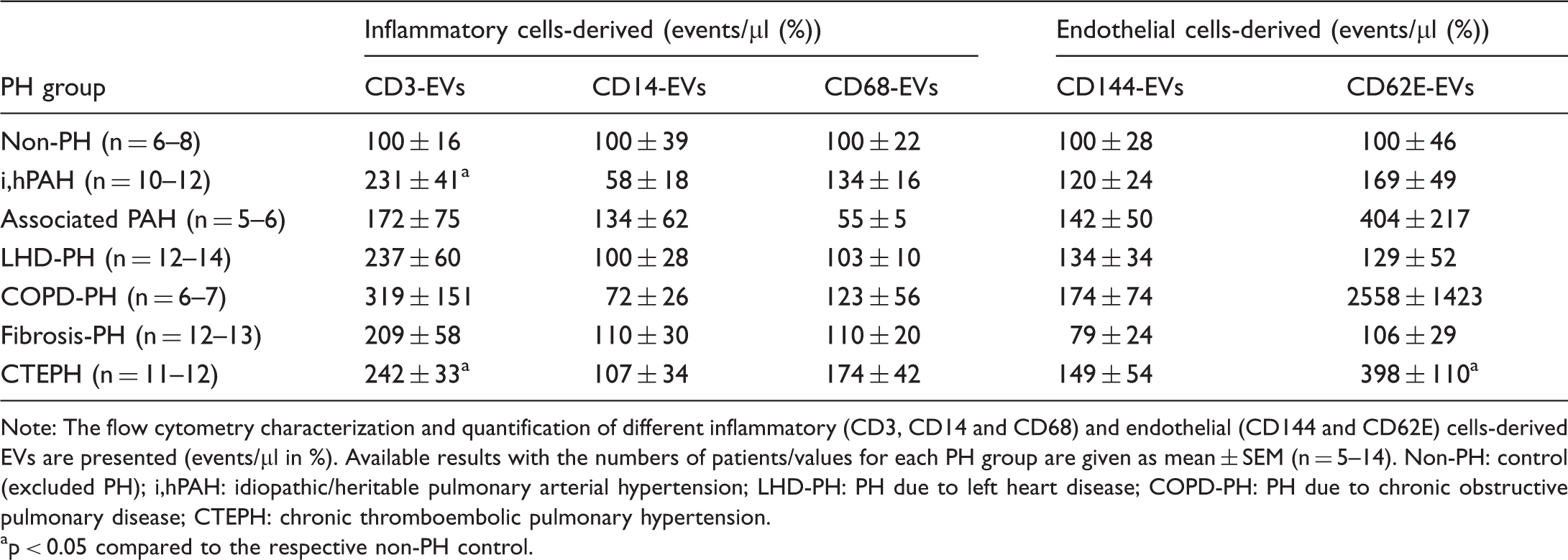
Note: The flow cytometry characterization and quantification of different inflammatory (CD3, CD14 and CD68) and endothelial (CD144 and CD62E) cells-derived EVs are presented (events/µl in %). Available results with the numbers of patients/values for each PH group are given as mean ± SEM (n = 5–14). Non-PH: control (excluded PH); i,hPAH: idiopathic/heritable pulmonary arterial hypertension; LHD-PH: PH due to left heart disease; COPD-PH: PH due to chronic obstructive pulmonary disease; CTEPH: chronic thromboembolic pulmonary hypertension.
p < 0.05 compared to the respective non-PH control.
We have demonstrated for the first time the augmented circulatory levels of EVs derived from T-cells. This finding may fit in the current paradigm of PH as an “inflammatory disorder”, since various inflammatory cells, including T-cells, are accumulated in the remodeled pulmonary vasculature and lungs of idiopathic PAH patients. 8 Interestingly, EVs derived from monocytes/macrophages, inflammatory cells which are also described in the context of this disease, 8 did not show an alteration in the profile. In the past, several studies indicated the involvement of endothelial cells-derived MPs in the pathology of PH.2–5 Finally, the literature suggested the augmentation of circulatory levels of CD62E-MPs in precapillary PH.2,5 We have analyzed the profile of CD62E-EVs in different forms of PH, and found a prominent increase in associated PAH, COPD-PH and CTEPH.
In conclusion, we have found the increased levels of the circulating EVs which originate from T-cells in different clinical forms of PH. Therefore, future studies should focus to identify whether there are promising biomarker properties as well as to reveal a potentially active role of this inflammatory cells-derived EVs in the pathogenesis and progression of PH. Finally, endothelial cells-derived EVs may have distinctive circulating profiles in different clinical PH subgroups.
Some data from this study have been previously reported in the form of abstract during the ATS conference in 2018.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This study was supported by the Universities of Giessen and Marburg Lung Center (UGMLC).
