Abstract
Diagnostic World Health Organization (WHO) Group 1 pulmonary arterial hypertension (PAH) and Diagnostic Group 1' pulmonary veno-occlusive disease (PVOD) and/or pulmonary capillary hemangiomatosis (PCH) are progressive and fatal disorders. Past registries provided important insights into these disorders, but did not include hormonal exposures or genomic data. The United States Pulmonary Hypertension Scientific Registry (USPHSR) will provide demographic, physiologic, anorexigen and hormone exposure, genomic, and survival data in the current therapeutic era for 499 patients diagnosed with PAH, PVOD, or PCH. The USPHSR also will explore the relationship between pharmacologic, non-pharmacologic, and dietary hormonal exposures and the increased risk for women to develop idiopathic or heritable PAH.
Background
Pulmonary arterial hypertension (PAH), a pulmonary vascular condition which includes the historic diagnosis previously described as primary pulmonary hypertension (PPH), was once a progressive and fatal disorder for which there was no effective treatment.1,2 Over the past four decades, investigators have made basic discoveries3–5 and completed pivotal clinical trials of 12 medications which have received regulatory approval for the treatment of PAH. 6
As treatment evolved, national patient registries provided important data to inform the pulmonary hypertension (PH) community. In the United States (US), National Institutes of Health (NIH) Registry investigators enrolled 187 patients diagnosed with PPH during 1983–1987, an era before specific treatment for this disorder. 7 NIH Registry investigators reported that women were more likely than men to be diagnosed with PPH; 7 they used hemodynamic measurements from the NIH Registry population to derive a prediction equation for survival. 8 Twenty years later, Registry to Evaluate Early and Long-term PAH Disease Management (REVEAL) investigators enrolled 3515 patients diagnosed with World Health Organization (WHO) Group 1 PAH. 9 REVEAL patients also included disproportionately more women than men and more diverse phenotypic patterns than the NIH Registry cohort. 10 Furthermore, in an era of PAH targeted therapies, REVEAL investigators observed improved survival of patients with baseline characteristics matched to NIH Registry patients. 11 REVEAL investigators also derived and validated a new method to predict survival for PAH patients treated in the REVEAL era.12,13
Four medications received regulatory approval for the treatment of PAH after REVEAL. Riociguat, 14 macitentan, 15 and an oral formulation of treprostinil 16 received FDA approval in 2013; selexipag 17 received FDA approval in 2016. These new medications altered the therapeutic landscape for PAH.
Advances in genomic understanding of PAH paralleled advances in treatment of PAH. The discovery that heterozygous BMPR2 mutations cause familial PPH18,19 and recognition that BMPR2 mutations were often the unrecognized cause of sporadic PPH20,21 led to an updated clinical classification of PH which included heritable PAH (HPAH). 22 Subsequent discoveries of mutations in ALK1, 23 ENG, 24 SMAD8, 25 CAV1, 26 KCNK3, 27 and TBX4 28 expanded the known causes of HPAH. In 2014, investigators reported that homozygous or compound heterozygous mutations in EIF2AK4 caused heritable pulmonary capillary hemangiomatosis (PCH) and pulmonary veno-occlusive disease (PVOD).29,30 A recent study of > 1000 patients diagnosed with idiopathic PAH (IPAH) identified mutations in ATP13A3, SOX17, AQP1, and GDF2 and suggested additional genes that will require further validation. 31 SOX17 variants were also identified in patients with PAH associated with congenital heart disease. 32 These discoveries permitted researchers to organize the first United States registry to incorporate genetic characteristics of PAH patients.
The United States Pulmonary Hypertension Scientific Registry (USPHSR) is a contemporary PAH registry which will provide data for PAH patients diagnosed between 2012 and 2018. Patients in the USPHSR will be characterized using targeted sequence and copy number analysis, genome-wide single nucleotide polymorphism (SNP) data, and whole exome sequencing. USPHSR investigators will use reproductive histories and hormonal exposure data to examine these potential contributors to the disproportionate numbers of women diagnosed with PAH.
USPHSR study overview
USPHSR is a multicenter, longitudinal, observational, and hypothesis-driven study of 499 patients diagnosed with WHO Group 1 PAH or PVOD/PCH. The study protocol was approved by the Institutional Review Boards at each participating center. USPHSR differs from the NIH Registry and REVEAL in three important ways: (1) USPHSR patients were diagnosed, characterized and treated in the current genomic, imaging, and therapeutic era; (2) USPHSR includes both observational data and pre-specified hypothesis driven research; and (3) USPHSR links with the NHLBI sponsored National Biological Sample and Data Repository for PAH (PAH Biobank, NHLBI HL105333).
Study objectives
Primary objectives of USPHSR.

Additional objectives of USPHSR.

Patient population
A total of 499 consecutively screened consenting adult (age > 18 years) patients diagnosed with WHO Group 1 PAH or Group 1' PVOD/PCH 33 at participating study sites (Supplementary Appendix 1) enrolled in USPHSR after they were enrolled in the PAH Biobank (https://www.pahbiobank.org, PI William Nichols, PhD). Only patients who enrolled in the PAH Biobank within five years of PAH diagnosis were eligible for enrollment in USPHSR. Consonant with REVEAL, the date of diagnosis was defined as the date of the diagnostic pulmonary artery catheterization.
Inclusion criteria
Before enrollment in the study, candidates met the following inclusion criteria:
diagnosis of PAH, PVOD, or PCH; documentation of the following hemodynamic parameters by pulmonary artery catheterization
∘ mean pulmonary arterial pressure (PAPm) ≥25 mmHg at rest; ∘ pulmonary artery wedge pressure (PAWP) or left ventricular end-diastolic pressure (LVEDP) ≤15 mmHg in the absence of mitral stenosis; ∘ pulmonary vascular resistance (PVR) >3 Wood Units (WU).
Exclusion criteria
Patients not eligible for enrollment included:
patients unwilling or unable to provide written consent for USPHSR; patients with an underlying medical disorder (e.g. metastatic cancer) with an anticipated life expectancy of <2 years; patients with complex adult congenital heart disease associated with PAH.
Informed consent procedure
Patients eligible for enrollment were asked to provide informed consent for participation in USPHSR. To maintain patient confidentiality according to HIPAA, each patient was assigned a unique patient-identifier. In addition, patients were asked to consent to data sharing for collaborative research among investigators conducting research deemed by the USPHSR steering committee to be of value to the PH community.
Data collection
Baseline data.

6MWD, 6-min walking distance.
Environmental and hormone exposure data were collected by a structured interview using a questionnaire. Study interviewers administered an expanded version of the previously validated and extensively published Nurses' Health Study (NHS) I and II questionnaire (http://www.channing.harvard.edu/nhs/index.html).34,35 NHS questionnaires address a person's exposure to hormones via a large array of questions related to contraceptive therapy (CT) and hormone replacement therapy (HRT), menstruation, menopause, pregnancy, abortions, and parity. In addition, basic data elements concerning exposure to tobacco smoke, alcohol use, illicit substance use, and anorexigen exposure were collected.
Each patient's vital status will be determined at one-year intervals after enrollment without altering clinical practice. Patients will be followed for the duration of the study with plans for minimum follow-up of one year for the last patient enrolled in USPHSR and with permission to contact the patient or a designated contact in the future.
USPHSR Investigators will access biologic data according to protocols established by the PAH Biobank Steering Committee and the Steering Committee of the USPHSR. Biologic specimens include DNA, RNA, plasma, serum, and EBV immortalized cell lines. Genetic data generated by the PAH Biobank include Illumina OMNI5 SNP data (∼4.5 million SNPS), coding sequence data for BMPR2, ACVRL1, ENG, CAV1, SMAD9, KCNK3, EIF2AK4, ABCC8, GDF2, KCNA5, SMAD4, and TBX4, and dosage data for BMPR2, ACVRL1, and ENG by MLPA. As of 30 October 2018, sequencing analyses were completed for 2688 PAH patients of 2853 patients enrolled in the PAH Biobank. In addition, whole exome sequencing data generated by the Regeneron Genetic Center are available on 2517 patients in the PAH Biobank.
Sample size
A total of 499 patients diagnosed with WHO group 1 or 1' PAH were enrolled over a period of three years. These patients were a subset of the 2853 patients enrolled in the PAH Biobank. The sample size was selected based upon budgetary constraints.
Data analysis
The USPHSR will support two approaches to data analysis. The first approach is for USPHSR demographics and the second approach is for individual studies which test specific hypotheses. Individual studies will have a specific statistical analysis plan to address specific research questions.
The statistical plan will incorporate standards for reporting observational research 36 and the checklist provided by the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 37
For the observed characteristics of the study cohort, descriptive statistics will be employed. In brief, we will describe quantitative variables using mean, median, standard deviation, and minimum to maximum intervals. To compare quantitative data of two groups (e.g. men compared to women), we will use the Student's t test or the Wilcoxon rank-sum test as appropriate, depending on the normality of the data. In the event of multiple group comparisons, quantitative data will be analyzed using analysis of variance or Kruskal–Wallis non-parametric tests, as appropriate. For categorical variables, description will be made using typical measures with analysis via χ 2 tests or Fisher's exact test. The level for determination of statistical significance will be set at P < 0.05 unless otherwise noted.
We will construct Kaplan–Meier survival curves for time to event endpoints and use the Cox proportional hazards regression model to analyze survival in the current era of diagnosis and treatment. Multivariate regression analysis will be employed to adjust for non-random treatment assignments. Multivariate Cox proportional hazards regression will be employed to identify predictors of outcomes, including time to death or lung transplant.38,39 As done previously in REVEAL, methods that account for survivor bias will be used due to the variable time from diagnosis to enrollment. 40 This will allow us to account for variable length of follow-up.
A major goal of the USPHR is to update clinical prediction tools with genomic data. We will determine the accuracy of clinical prediction tools, with and without genomic data, to predict survival of PAH patients diagnosed and treated after REVEAL. Updated algorithms will be evaluated for calibration and discrimination. Model calibration will be determined by comparing one-year survival predicted by an updated algorithm with observed one-year survival of the USPHSR cohort. Calibration plots will be employed to demonstrate agreement between predicted and observed survival.
Evaluating hormone exposures
USPHSR investigators will test the hypothesis that PAH patients have a higher exposure to exogenous sources of female sex hormones than controls. We are currently planning to recruit control individuals from Research.Match.org. These controls will be matched to newly diagnosed PAH women according to age, self-reported race, ethnicity, and body mass index (BMI).
Analyses will be stratified by sex. For women, the primary exposure endpoint will be > 1 year of CT and/or HRT use. For cases, only data before diagnosis will be collected. For controls, the entire lifetime will be evaluated. For male cases, the primary exposure endpoint will be BMI (kg/m2) > 24.9 at diagnosis. For the primary exposure, conditional logistic regression will be used to generate odds ratios (OR) and 95% confidence intervals (CI) for the association between CT/HRT exposure >1 year and PAH in women, as well as the association between BMI >24.9 kg/m2 and PAH in men. We will also analyze hormone exposure and BMI as continuous variables, comparing cases and controls, using Student's t test for normally distributed data, or the non-parametric Mann–Whitney U test for data that are not normally distributed. We will determine the univariate association of the other exposures determined, such as tobacco smoke exposure, and PAH/control status, and assess for effect modification by biologically relevant genes as appropriate. In addition, multivariable conditional logistic regression models will be used to calculate adjusted ORs and 95% CIs for each sex. The models will include covariates and sources of effect modification. Analysis for continuous outcome variables, such as age at diagnosis among cases, will be similar but utilize techniques appropriate to a continuous variable, such as Student's t test and linear or Cox regression analyses. All distributions will be examined for deviations from normality (and transformed as necessary, if indicated, or non-parametric analysis methods may be applied). To avoid multiple comparisons and over-fitting the separate model for each sex, a limited number of covariates will be included in each. Those covariates will be identified a priori and will include those relevant biologically and by univariable analysis. The logistic models will be assessed for adequacy of the fit for the matched case-control data. We will employ both graphical and numerical methods according to previously published approaches. These techniques allow for examination of the functional form of each covariate, as well as the overall adequacy of the models.41–43
Evolving research needs
To adapt to the evolving science and research needs of the PAH community, separate analysis plans will be developed for new objectives. Minor changes (if approved by the Steering Committee) in the electronic case report form (eCRF) may occur in response to new diagnostic approaches and treatments. Ongoing assessment of enrollment and data captured will also be performed by the Steering Committee to be certain the goals of the Registry are being met.
General issues
Except for the exposure questionnaire, clinical practice should not be changed in any way to accommodate collection of additional data. The exposure questionnaire was completed in person at the time of enrollment, by telephone interview, or by mail. Participants may have follow-up studies performed at institutions different from the enrolling site. Investigators/coordinators were encouraged to request copies of these results and enter data into the eCRF.
Discussion
Past United States Registries of patients diagnosed with PAH.
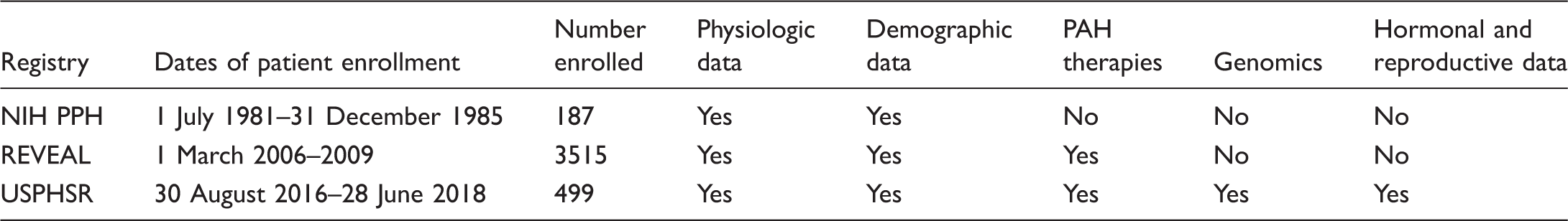
First, USPHSR is designed to describe the genomic characteristics of patients diagnosed with PAH. Prior US PAH registries provided extensive demographic, clinical, and physiologic data, but genomic data were not available.7,45 The USPHSR study design assures detailed genomic data through its link with the National Biological Sample and Data Repository for PAH (PAH Biobank). By design, all patients enrolled in USPHSR are also enrolled in the PAH Biobank. The PAH Biobank research team will provide genome wide SNP genotypes, sequencing for established PAH genes, multiplex ligation probe dosage data, and whole exome sequencing on samples from patients enrolled in USPHSR.
A second design feature that distinguishes USPHSR from previous US PAH registries is the prospective collection of hormonal and environmental exposures. Past registries provided limited data with respect to hormonal and environmental exposures, while they demonstrated that IPAH and HPAH affect women more often than men.7,45,46 REVEAL investigators reported that PAH patients were older and more often women than previously reported, an important observation with implications for an interplay between hormonal, genetic, and environmental exposures.45,47 In addition, exposure to anorexigens was more common among women than men with PAH. 47 Clinician investigators have suggested that exogenous estrogens (e.g. oral contraceptives) accelerate pulmonary vascular disease and PH, both in patients with predispositions (e.g. congenital heart disease, familial PAH, or in combination with anorexigens known to cause PAH) and in patients without known predispositions.48–50
A third key design feature of USPHSR is a planned report of survival for a cohort of PAH patients diagnosed and treated in the contemporary diagnostic and therapeutic era. NIH registry investigators provided the first description of survival of patients diagnosed with PPH in an era (1981–1991) when there was no specific therapy. 8 The median survival from diagnosis was 2.8 years. REVEAL investigators documented improved survival of PAH patients in an era (2006–2012) when treatment was available with prostacyclins, endothelin receptor antagonists, and phosphodiesterase inhibitors.11,45 Four medications received regulatory approval for the treatment of PAH after completion of REVEAL.14–17 USPHSR patients were diagnosed and treated in an era when these medications were available and in an era when clinicians often provided initial therapy with a combination of PAH-specific therapies.
A fourth key design feature is a planned evaluation of the contribution of genomic data to prediction of survival. Prior US Registries lacked access to genomic data. The NIH PPH registry investigators derived an equation to predict survival based upon hemodynamic data alone 8 and REVEAL investigators derived and validated a risk assessment tool (REVEAL Risk Calculator) based upon multiple clinical variables.12,13 Familial PAH, a surrogate for PAH caused by pathogenic gene mutations, was an important predictor of survival in the REVEAL Risk Calculator, but a family history of PAH is either incomplete or non-existent for many IPAH patients who carry genetic mutations which cause PAH.51,52 Genomic data may improve survival predictions because mutations in BMPR2, ALK1, and EIF2AK4 identify patients with more severe disease and reduced transplant free survival.52–57 Furthermore, the list of pathogenic mutations continues to expand, with data suggesting that many IPAH patients have pathogenic mutations in other genes (e.g. TBX4) responsible for more severe PAH with reduced survival. 31 Correct identification of the influence of a broad spectrum of pathogenic mutations on mortality underlies the USPHSR plan to evaluate the contribution of genomic data to survival prediction.
Finally, a rich source of data which includes sex-hormone-related exposures and reproductive histories, exposures, and genomics will also allow USPHSR investigators to explore other relationships. For example, limited evidence suggests that combinations of a genetic predisposition such as a pathogenic BMPR2 mutation and exposure to an environmental stimulus such as an anorexigen increases the risk of developing severe PAH. 58 The USPHSR was designed to provide data for additional genes and exposures which may increase an individual's risk to develop severe PAH. Enhanced understanding of these risks may allow individuals with genetic predispositions to avoid exposures that increase their risk to develop PAH.
In conclusion, the USPHSR represents the first US Registry of PAH patients to provide demographic, physiologic, and genomic data along with hormonal and reproductive data and environmental exposures. The USPHSR will provide this data to the PH community in the current era of PAH diagnosis and treatment.
Footnotes
Acknowledgments
Samples and/or data from the National Biological Sample and Data Repository for PAH, which receives government support under an investigator-initiated grant (R24 HL105333) awarded by the National Heart Lung and Blood Institute (NHLBI), were used in support of this study. We thank contributors, including the Pulmonary Hypertension Centers who enrolled patients, completed questionnaires, and collected samples used in this study, as well as patients and their families, whose help and participation made this work possible.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This work was supported by an unrestricted grant from Actelion Pharmaceuticals US, Inc. and donations from BikePHifty. WCN, MWP, and KAL were funded by HL105333.
