Abstract
Pulmonary arterial hypertension (PAH) is characterized by progressive limitations in physical activity and health-related quality of life (HRQoL). HRQoL deficits may extend beyond the traditional domains of physical activity, psychological health, and emotional wellbeing to sexual health and function. Sexual HRQoL has not been studied in PAH, nor has the impact of PAH therapies themselves on sexual health and intimacy. In this initial investigation, we sought to explore HRQoL among women diagnosed with PAH and to determine if PAH treatment type (intravenous or subcutaneous prostanoids versus oral medications) was associated with levels of self-reported HRQoL assessed by validated measures for PAH-specific, general, and sexual HRQoL. We administered the emPHasis-10, Short Form (SF)-36, Female Sexual Dysfunction Scale-Revised (FSDS-R), and the Arizona Sexual Experience Scale (ASEX) to 35 women with self-reported World Health Organization Group 1 PAH at the 2016 Pulmonary Hypertension Association International Conference and Scientific Sessions. HRQoL instruments demonstrated excellent internal reliability. Women with PAH had high levels of sexual distress captured with the FSDS-R scale. The FSDS-R (but not ASEX) was significantly correlated to emPHasis-10 (r = 0.64, p < 0.01) and most SF-36 domains (r = − 0.36 to − 0.64, p < 0.05). Participants treated with intravenous or subcutaneous prostanoids had higher (worse) FSDS-R scores than those on oral therapies while ASEX, emPHasis-10, and SF-36 scores were similar across treatment types. Sexual HRQoL may impact overall quality of life in PAH and specific assessment of sexual health and functioning within intimate relationships may detect deficits in wellbeing not addressed by established HRQoL metrics. Further study to address all aspects of HRQoL in PAH is required.
Introduction
Pulmonary arterial hypertension (PAH) is a non-curable pulmonary vasculopathy characterized by progressive dyspnea, fatigue, and limitations in physical exertion that disproportionately affects women.1,2 PAH adversely impacts everyday life and requires daily treatment ranging from oral medications to continuous parenteral (i.e., intravenous or subcutaneous) infusions depending on the progression of the disease.3,4 Numerous therapies are now available that delay clinical worsening and prolong survival,5–9 but how treatments themselves impact various aspects of health-related quality of life (HRQoL) has not been well studied. Sexual health and function is an important component of HRQoL,10,11 and while several tools to assess overall HRQoL have been developed and used in PAH,12–17 sexual HRQoL (SHRQoL) in PAH has not been studied and, in fact, has been understudied in many chronically ill populations. 18
Sexual health and function may be uniquely affected in PAH for several reasons. First, cardiopulmonary symptoms in PAH worsen with exertion. Prior studies suggest that patients with other chronic pulmonary diseases fear experiencing dyspnea during sex and have high rates of sexual dysfunction.19–23 Second, PAH therapies themselves may directly impact SHRQoL. Pumps, catheters, and skin sites required for continuous infusion prostacyclin analogue therapies may pose challenges to engaging in sexual behaviors and a commonly used class of oral PAH medications (phosphodiesterase type-5 inhibitors (PDE5i)), also approved for erectile dysfunction, may directly affect sexual function. Third, since as many as 80% of prevalent patients with PAH are women, 1 fears of pregnancy may impact SHRQoL in women of childbearing age and their partners because pregnancy is strongly discouraged in PAH and because certain PAH medications are teratogenic.24–26 Finally, anxiety and depression is common in PAH and it is conceivable that this may be driven in part by as yet unquantified effects on sexual relationships and feelings of intimacy.27–29
Many patients and clinicians consider sexual health a particularly sensitive topic. Most patients never discuss sexual health with clinicians, and rarely discuss these matters with their partner(s). 30 There are consensus guidelines for sexual activity in left heart failure patients that outline the importance of psychosexual counseling but also note the limitations in clinician skills and training in this area. 30 There are currently no validated clinical measures for evaluating patients’ SHRQoL and no uniformly adopted recommendations for sexual activity in PAH. Further, there are no published data identifying this as a knowledge gap in the field.
In this formative study, we sought to establish whether SHRQoL is affected in women with PAH as compared to SHRQoL in both the general population and among those with reported sexual dysfunction. We assessed for correlations in scale scores between validated SHRQoL questionnaires, the Female Sexual Distress Scale-Revised (FSDS-R) and the Arizona Sexual Experience Scale (ASEX),31,32 and HRQoL metrics established in PAH, the emPHasis-10 and the Short Form [SF]-36 Health Survey.17,33 We also assessed whether SHRQoL tracked with disease indicators including time since diagnosis, functional class, and type of PAH treatment. We sought to determine whether: (1) women with PAH in our sample would demonstrate impairments in SHRQoL as compared to population norms and with diagnosed sexual dysfunction, (2) there would be a correlation between SHRQoL measures and HRQoL measures, and (3) patients with PAH on parenteral therapies (i.e., intravenous or subcutaneous medications) and those with more PAH-related impairment (i.e., higher functional class) would have greater deficits in SHRQoL as compared to patients on oral therapies only and less physical limitations (i.e., lower functional class).
Methods
We conducted a cross-sectional study of PAH patients at the 2016 Pulmonary Hypertension Association (PHA) International Pulmonary Hypertension Conference and Scientific Sessions in Dallas, Texas. A dedicated Research Room for investigators was available for use as part of the PHA Conference contingent on selection and approval of a research application by the PHA Conference’s Research Room Leadership Committee, as well as applicable Institutional Review Board (IRB) approval. This study was approved by the Rhode Island Hospital IRB (Board reference number 408516).
Participants
Participants were a convenience sample recruited from the Research Room with a self-reported history of World Health Organization (WHO) Group 1 PAH. The data presented here is part of a larger study that included surveys with men and women as well as sexual partners of PAH patients. For this substudy, only five men with PAH (13% of the study sample) enrolled, consistent with the known sex distribution of PAH from prevalence registries. 1 Among the four men who provided complete self-reported medical data, all were functional class II. It was therefore not statistically feasible (or sound) to conduct analyses on men. We did not combine men and women for this study given marked physiologic and psychologic differences in sexual function by sex. Participants were also given the opportunity to participate in a qualitative interview about their sexual health and quality of life (data analysis ongoing for both men and women).
Study procedures
Participants approached study personnel in the Research Room at the PHA conference. After receiving a brief description of what participation would entail, written informed consent was obtained from interested individuals. Participants were then assigned a research identification number to preserve confidentiality, and were given a short tutorial (if needed) on use of an electronic tablet for survey completion. Participants were provided a quiet, private space to fill out the four self-report surveys using a computer-assisted self-interview format on a web-based tablet. Common demographic and medical data, made available to approved study personnel, were collected by PHA either online (self-report) prior to attending the conference or by PHA staff if patients enrolled in Research Room activities on-site. Detailed information on disease characteristics for individual participants, such as hemodynamics or other biomarker data, was not available.
Measures
Participants’ demographic and medical history data included age, sex, date of PAH diagnosis, WHO functional class, and a list of prescribed PAH medications. Self-report surveys included the emPHasis-10, 17 the SF-36, 33 the FSDS-R, 31 and ASEX. 32
General HRQoL surveys
The emPHasis-10 is a PAH-specific HRQoL tool that is unidimensional and pragmatic (10 items). 17 It has been translated into multiple languages with cultural and linguistic validation. 34 The emPHasis-10 assesses breathlessness, energy, feelings of dependency, and perceived control that pulmonary hypertension (PH) has over life, among other relevant constructs. emPHasis-10 items are rated on a 0–5 Likert scale, with potential sum scores ranging from 0 to 50 and higher scores indicating more disruption to life due to PH symptoms. 17
The SF-36 comprises eight subscales which assesses various general aspects of quality of life. 33 It is validated in other chronic heart–lung diseases, correlates with common PAH outcomes (e.g., functional class), has been used as a secondary endpoint in randomized clinical trials of approved PAH therapies, and tracks with survival.13,14,16,35–37 Higher scores represent higher quality of life in the specific domain.
SHRQoL surveys
The FSDS-R is a validated survey which captures anxiety and distress related to impaired sexual health and function (13 items). 31 The revised scale is a unidimensional psychometric tool which detects distress related to sexual function in women with hypoactive sexual desire and serves as a screen for women with high or low sexual function with strong internal consistency and reliability across different languages and ages of women.38–40 It has been used in conditions associated with PAH, such as systemic sclerosis and liver disease,41,42 and in female cancer survivors. 43 FSDS-R items are rated on a 0–4 Likert scale, with potential sum scores ranging from 0 to 52 and higher scores indicating higher levels of sexual distress. 31
The ASEX is a five-item survey that assesses sexual drive, arousal, penile erection/vaginal lubrication, ability to reach orgasm, and satisfaction from orgasm. The ASEX is appropriate for patients of any sex and sexual orientation, with and without partners. Responses are scored on a 1–6 Likert scale with a potential range of 5–30; higher scores indicate worse sexual function, 32 and absolute scores ≥19 indicate sexual dysfunction. It has been shown to have excellent reproducibility. 32 It has been validated in populations with chronic conditions, 44 and also used to assess sexual function in cardiovascular disease. 45
There are no published data regarding the utility of the FSDS-R or ASEX (or any SHRQoL survey) in patients with PAH. Nor has it been studied to date whether the FSDS-R and/or ASEX could serve to characterize SHRQoL in patients with PAH.
Statistical analysis
For the current study, we report descriptive statistics and Pearson’s r correlations between all scales and subscales. We conducted correlational analyses between age, time since diagnosis (in years), and each of the scales and subscales. We also conducted analyses of variance to determine whether patient treatment type and functional class were related to scale scores.
Results
Participant demographics
Thirty-five female conference attendees with PAH were enrolled. Of those who reported age (n = 34), the average was 49 years (SD = 13, range 31–72). Of those who reported ethnicity (n = 29), four (14%) were Latina/Hispanic, and of the 32 who reported race, 21 (66%) were white, four (13%) black, four (13%) Asian, one white and Native American (3%), and two (6%) unknown/other.
Participant characteristics.
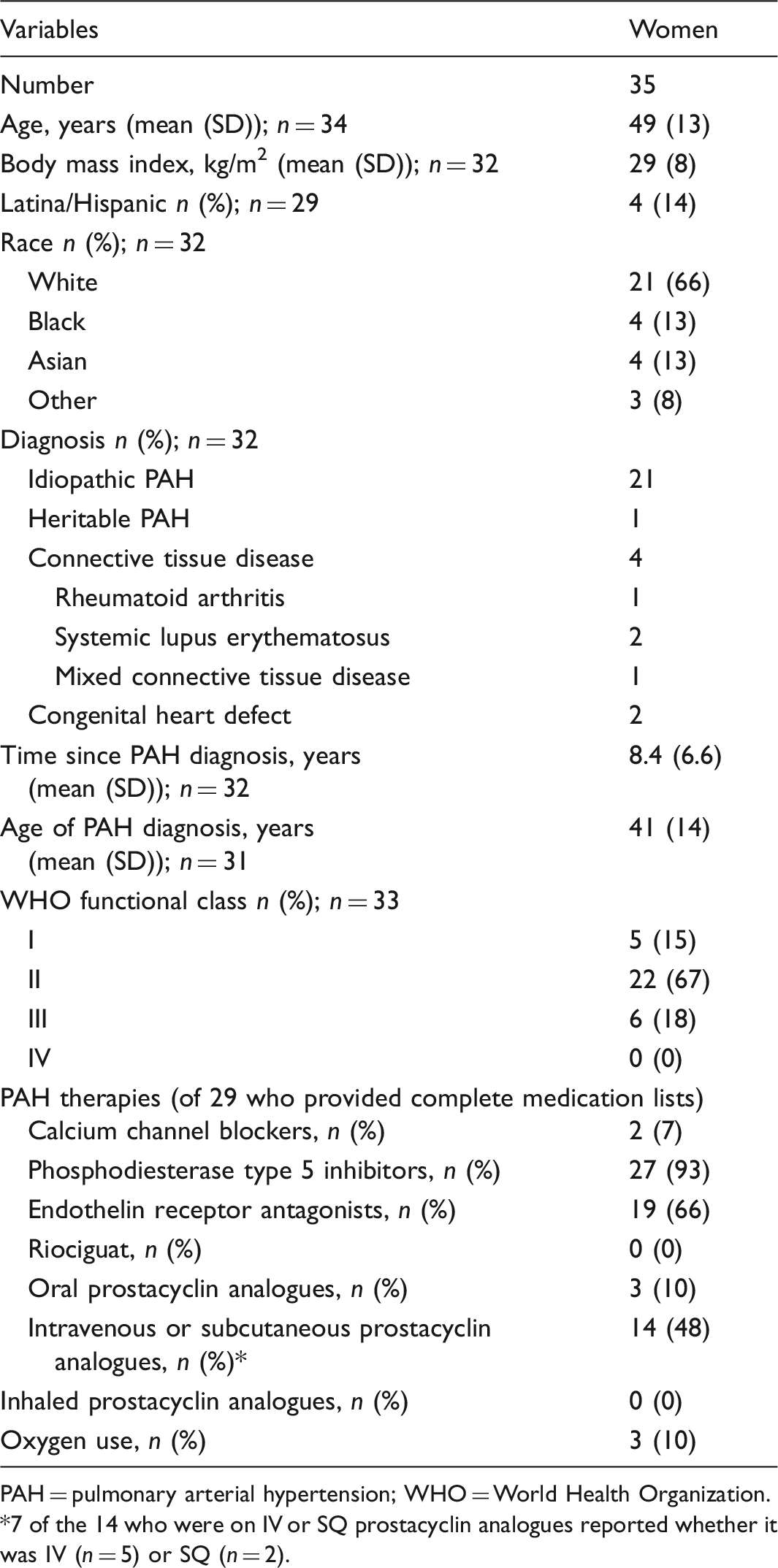
PAH = pulmonary arterial hypertension; WHO = World Health Organization.
7 of the 14 who were on IV or SQ prostacyclin analogues reported whether it was IV (n = 5) or SQ (n = 2).
Performance characteristics of scales
The HRQoL and SHRQoL measures performed well in this sample. The emPHasis-10 demonstrated excellent internal consistency (Cronbach’s α = 0.94). Each of the SF-36 subscales demonstrated very good or excellent internal consistency: physical functioning (10 items, Cronbach’s α = 0.89), role limitations due to physical health (4 items, Cronbach’s α = 0.97), role limitations due to emotional problems (3 items, Cronbach’s α = 0.93), energy (4 items, Cronbach's α = 0.81), emotional wellbeing (5 items, Cronbach’s α = 0.89), social functioning (2 items, Cronbach’s α = 0.93), pain (2 items, Cronbach’s α = 0.90), and general health (5 items, Cronbach’s α = 0.79). The FSDS-R demonstrated excellent internal consistency (Cronbach’s α = 0.97) and ASEX exhibited strong internal consistency in this sample (Cronbach's α = 0.89).
Sample comparisons in quality of life measures
Overall means, standard deviations, and ranges of scores for all quality of life measures.
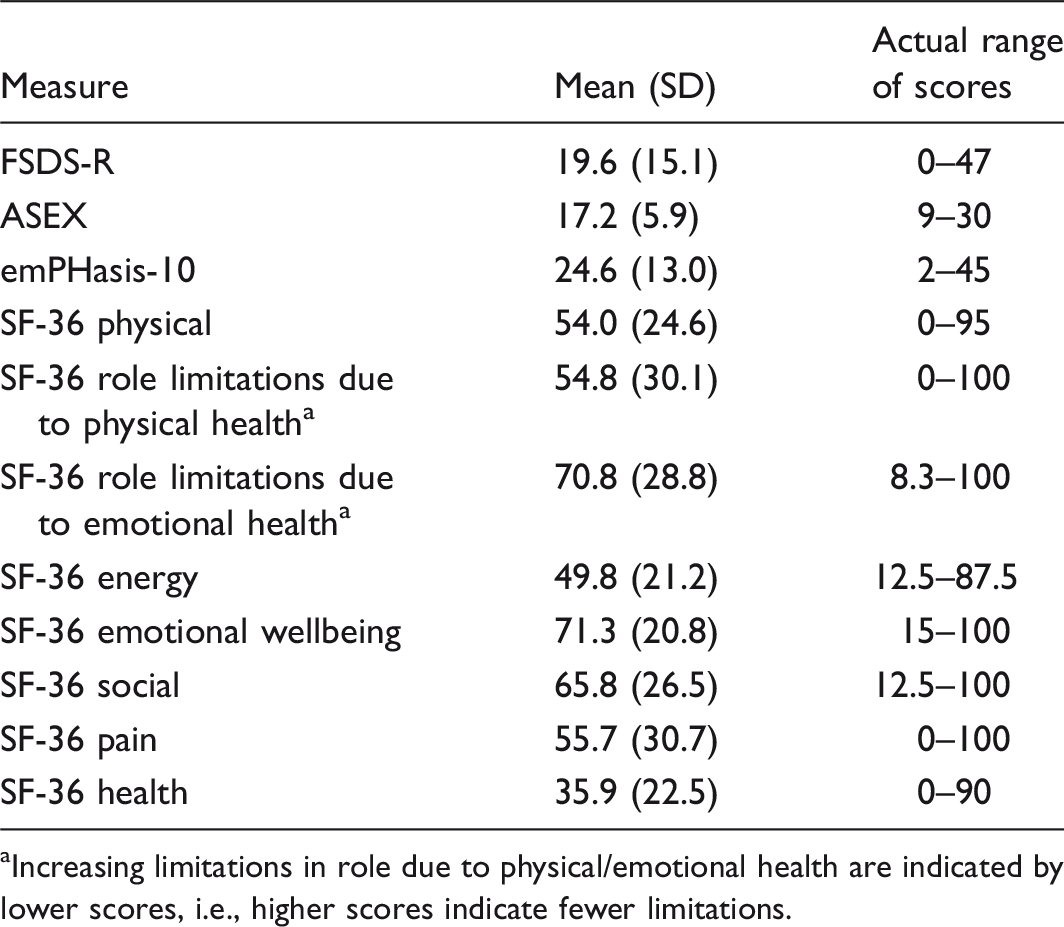
Increasing limitations in role due to physical/emotional health are indicated by lower scores, i.e., higher scores indicate fewer limitations.
As compared to another sample of patients with PH of varied etiologies, the emPHasis-10 sample mean of 24.6 (SD = 13.0) is comparable, though slightly lower (indicating less life disruption by PAH) in the current sample than reported by Yorke et al. (28.0, SD = 11.5);
17
this difference was not statistically different. When comparing the SF-36 subscale scores from our sample to those of the general population (mean = 50, SD = 10), our participants reported means for role limitations due to emotional problems (t(34) = 4.3, p < 0.001), emotional wellbeing (t(34) = 6.1, p < 0.001), and social functioning (t(34) = 3.5, p < 0.002) that are significantly higher than the population mean, indicating fewer difficulties and higher quality of life in our sample. That said, our participants reported lower quality of life only in the overall health domain (t(34) = 3.7, p < 0.001) of the SF-36. Figure 1 illustrates scale score comparisons between the SF-36 subscale means from our sample and means from other studies.
Radar plot comparing the SF-36 subscales means from the current sample of women with PAH (solid line) to US population norms (dashed line).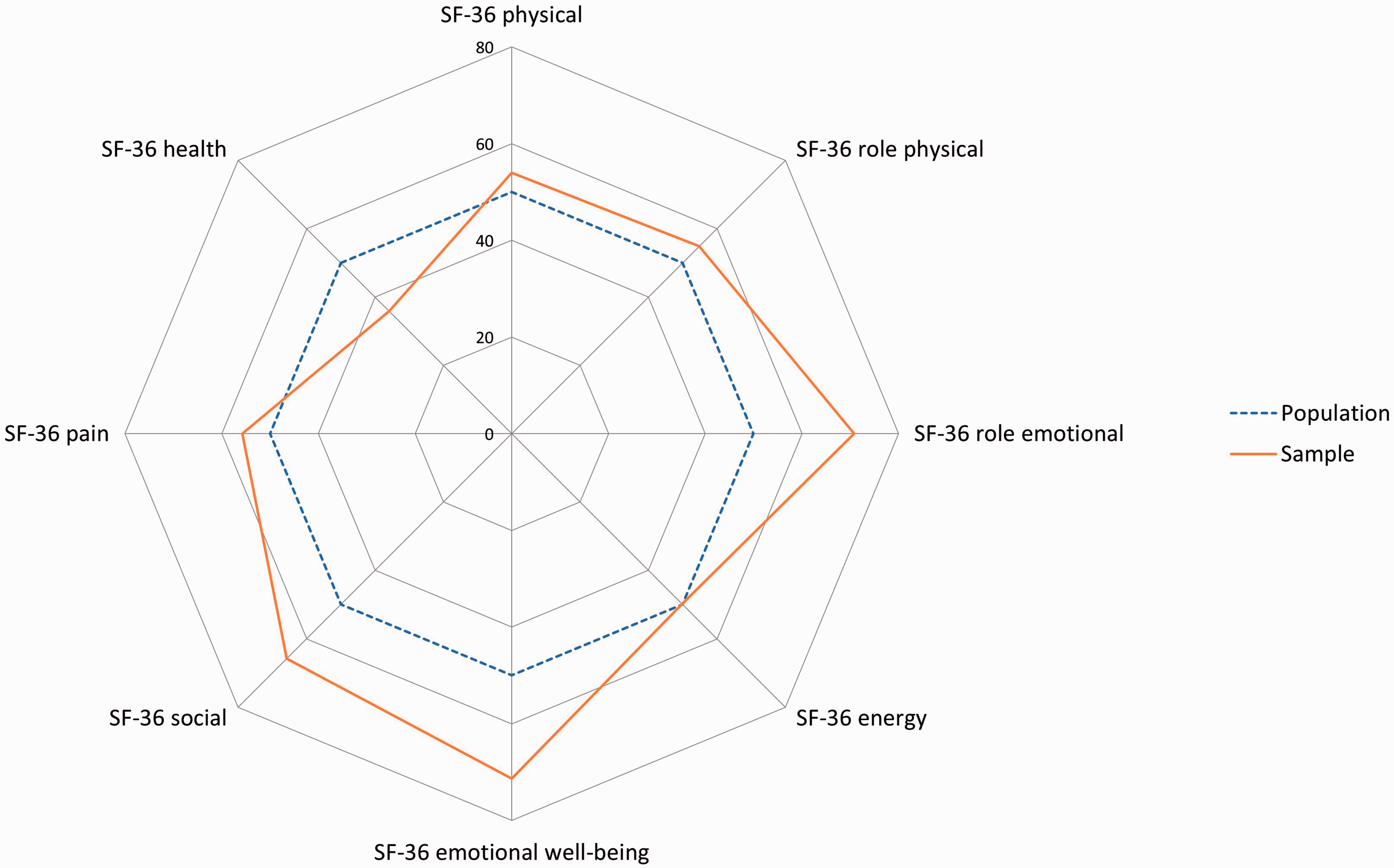
In terms of sexual functioning, our participants reported higher levels of sexual dysfunction on both the FSDS-R (19.6) and the ASEX (17.2) compared to control groups from the general population in previous studies (2.2 and 13.5, respectively).31,32 In fact, our participants endorsed sexual dysfunction on the FSDS-R and ASEX that approached levels similar to individuals with clinically diagnosed sexual dysfunction (28.4 and 20.3, respectively).
Relationships between scales within the sample
Correlation coefficients between scales.
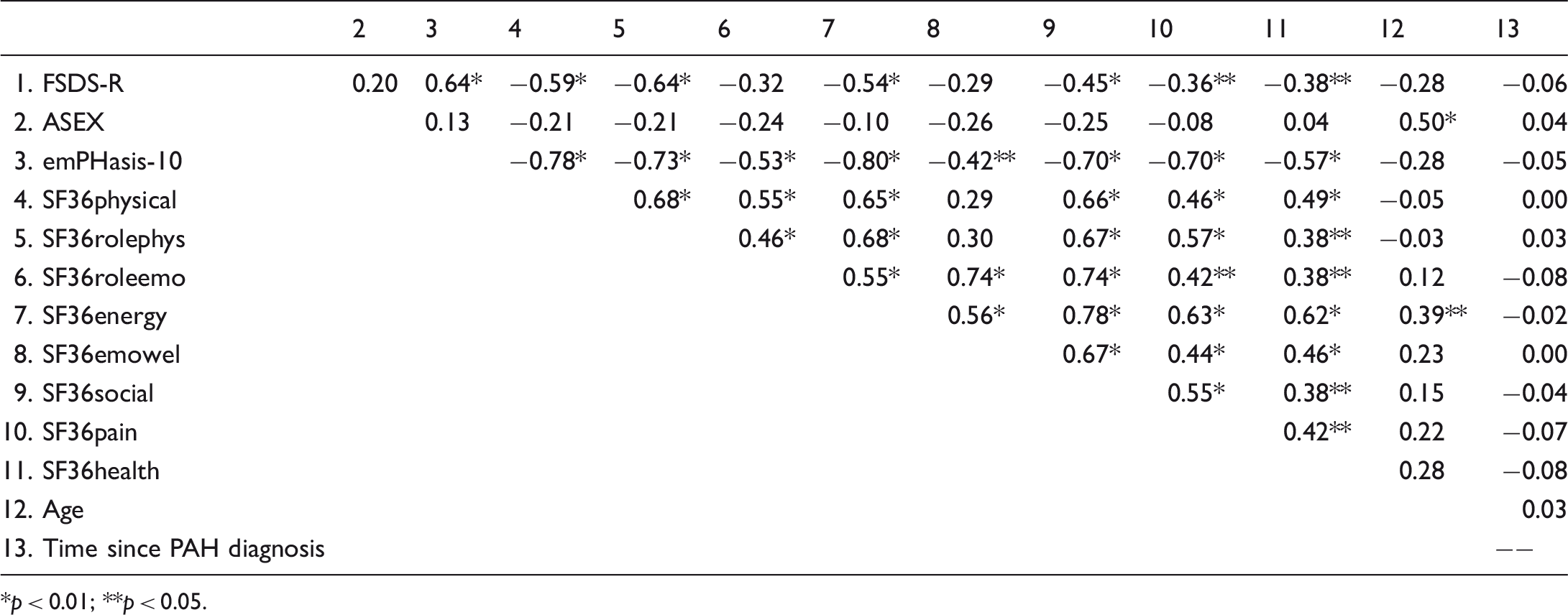
p < 0.01; **p < 0.05.
Functional Class
One-way analyses of variance (ANOVAs) were used to compare scale and subscale scores by WHO functional class. There were no significant main effects for the FSDS-R and ASEX survey scores by functional class.
ANOVA, means, standard deviations, F-test statistics, and p-value by functional class, n = 32.
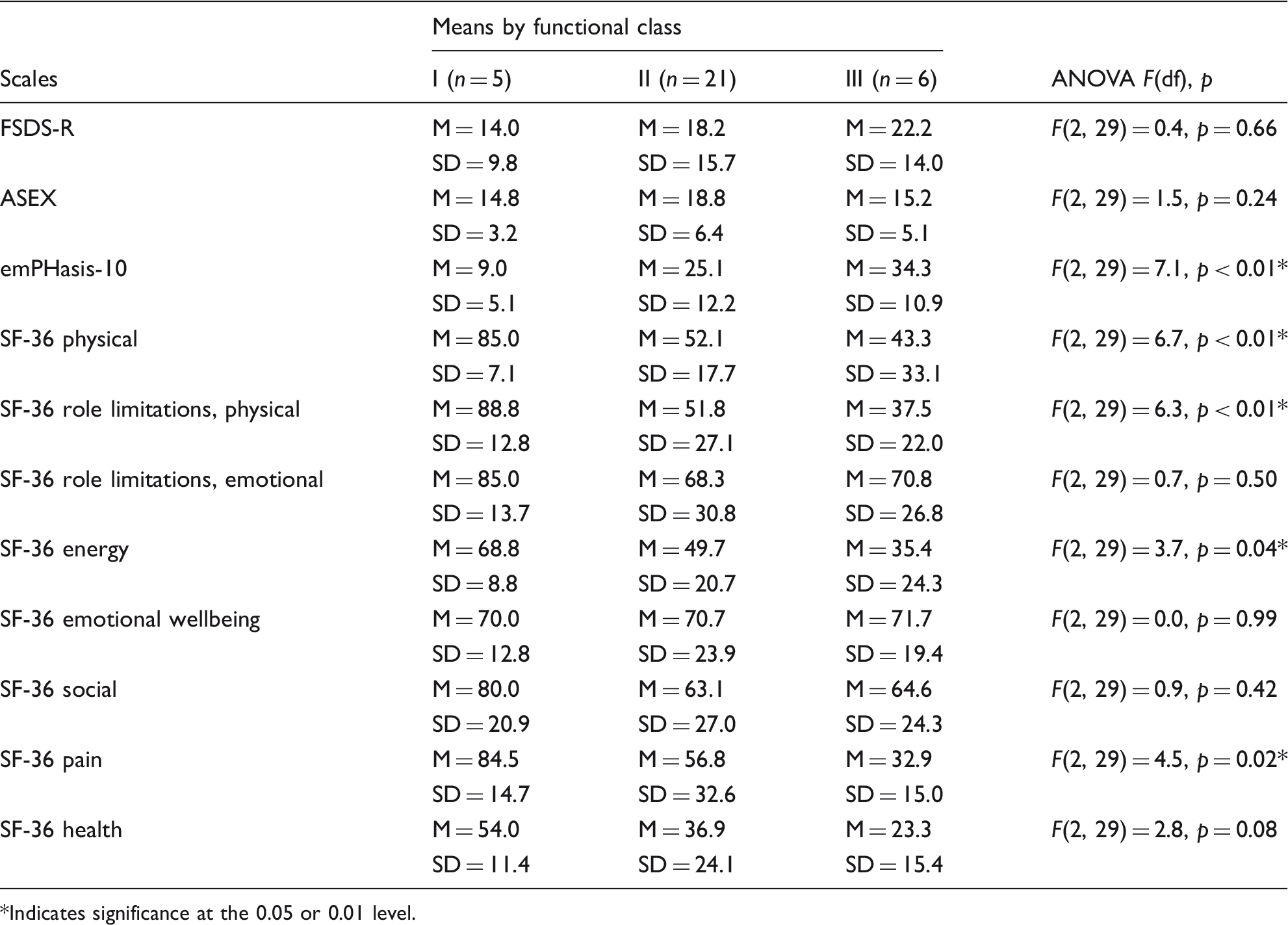
Indicates significance at the 0.05 or 0.01 level.
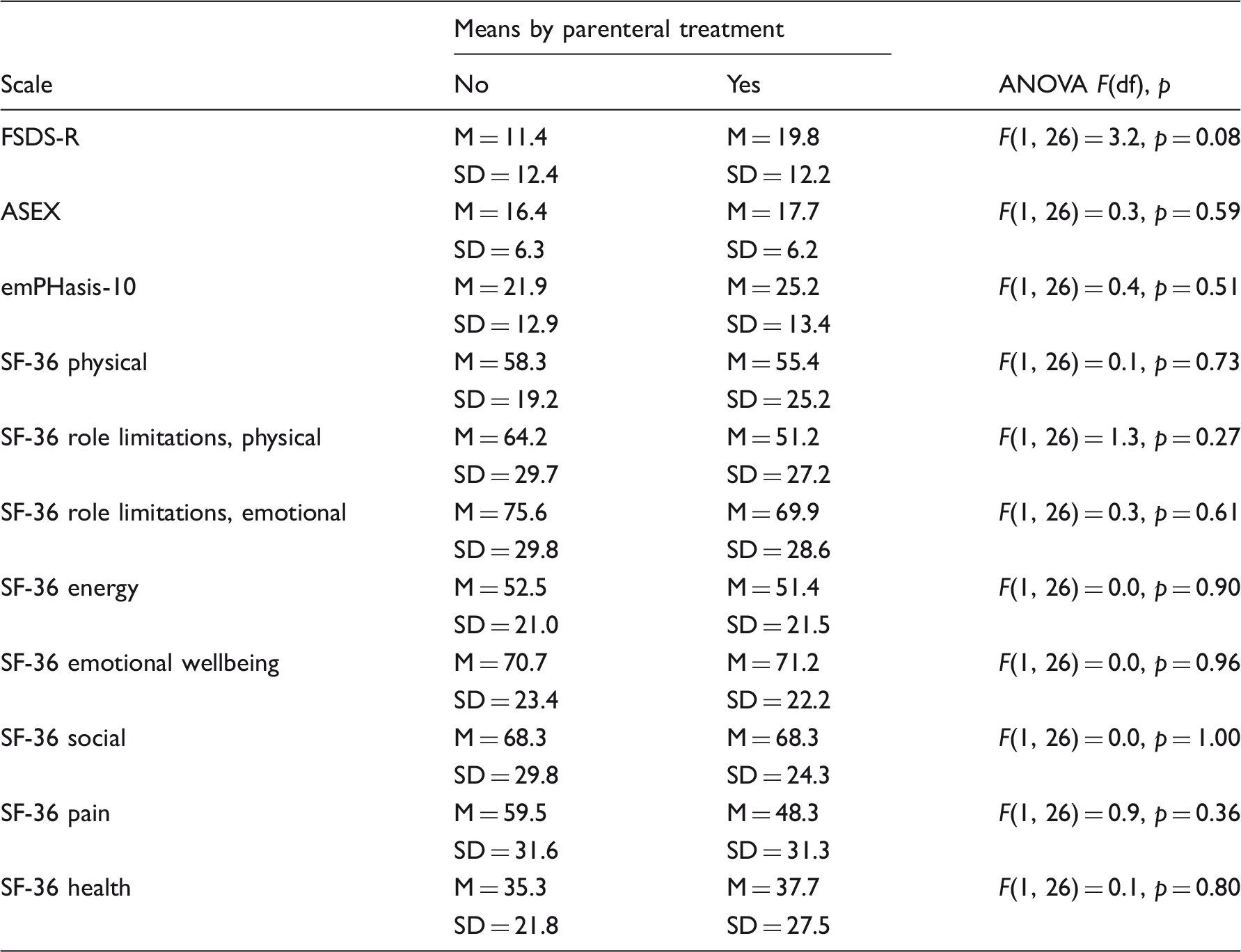
Treatment type
ANOVA, means, standard deviations, F-test statistics, and p-value by treatment type, n = 32.
Discussion
This formative study demonstrated that the two SHRQoL scales (FSDS-R and ASEX) had excellent internal reliability in our study sample. As hypothesized, we demonstrated that: (1) women with PAH appear to have reduced SHRQoL, at a level that approaches the diagnosis of sexual dysfunction, (2) FSDS-R but not ASEX was generally well correlated with both PAH-specific (emPHasis-10) and general HRQoL (SF-36) measures, and (3) the FSDS-R may discriminate patients on parenteral therapy (i.e., those treated with intravenous or subcutaneous therapy had greater levels of sexual anxiety and distress), a signal which was not detected with ASEX, emPHasis-10, or SF-36.
Participants in our sample had scores indicating greater impairment in sexual health than control populations. The mean FSDS-R and ASEX scores were well above those reported for control groups in previous studies, and were more consistent with mean scores found among clinical samples of individuals with sexual dysfunction.31,32 Interestingly, despite having impaired sexual functioning and related increases in distress, general HRQoL as assessed by the SF-36 was significantly better than population norms for the SF-36 in domains related to emotional and social functioning. These findings are consistent with a previously published pattern of greater functioning in the SF-36 mental health domain among PAH patients. 14 The study was conducted at the 2016 PHA conference; thus, this was a very engaged segment of the PAH patient population that was also physically able to travel. We did not enroll any participants with functional class IV symptoms and most participants were functional class II. That said, almost half (48%) were on parenteral therapies, which allowed us to note the absolute differences in SHRQoL in these patients. It is possible that this highly selected sample of patients feel a sense of connection, community, and control of their condition that is associated with higher than expected physical and mental health functioning. The disconnect between sexual dysfunction and emotional and social wellbeing may be explained by a degree of acceptance among these women that sexual activity is no longer possible. It is noteworthy that even among highly motivated and empowered conference attendees and despite supra-normal scores on general HRQoL surveys, deficits in sexual health and dysfunction emerged.
The discordance between worse than expected sexual health and dysfunction and better than expected emotional and social functioning domains in our study population may indicate that established HRQoL measures in PAH miss a major determinant of quality of life by not directly assessing SHRQoL and intimacy.10,11 Indeed, the scale score of the ASEX, for instance, captures sexual drive, arousal, penile erection/vaginal lubrication, ability to reach orgasm, and satisfaction from orgasm as a unidimensional construct. But it may be that in women with PAH sexual drive and arousal are not compromised, but their ability to reach orgasm such that they are satisfied is impeded by the physical (not emotional) limitations of PAH and/or the treatment modalities they rely on. The lack of correlation between the ASEX and the other measures captured here may be indicative of this disconnect. Additionally, ASEX, but not FSDS-R, was associated with age. It is possible that age-related changes in sexual arousal and behavior are most strongly affecting ASEX scores or that effects of PAH are difficult to measure using such a biologically-driven scale. The FSDS-R, in contrast, largely captures more psychosocial constructs in sexual health and functioning.
Our hypothesis that SHRQoL measures would be correlated with established HRQoL measures was generally supported, specifically by the outcomes from the FSDS-R. The FSDS-R is designed to capture anxiety and distress regarding sexual function in women, and given the likelihood of strong face validity, this association is perhaps not surprising. ASEX, however, was not correlated with FSDS-R or any HRQoL measure. Given the physiological nature of the ASEX scale it is possible that age-related changes in sexual functioning were more pronounced than changes related to PAH symptoms and/or treatment alone, 46 or that in our study sample of mostly functional class II patients, PAH had not progressed to the point of causing systemic effects which could interfere with sexual biologic function.
Taken together, these observations suggest that women with PAH have high rates of distress associated with sexual health, potentially impacted by PAH therapies, and not detected by currently utilized HRQoL measures. Contrary to our expectations, we did not find significant differences in sexual health measures between individuals of different functional classes or treatment modalities, though a trend in greater distress on the FSDS-R among those on parenteral therapy was noted. While it is possible that there are not statistically significant differences among these groups, we offer as an alternative hypothesis that the currently available scales may not adequately discern the specifics and nuances of sexual health and “dis-ease” in women with PAH, potentially resulting in a missed opportunity to improve quality of life in this patient population. Thus, we recommend further qualitative study of SHRQoL among patients with PAH with the goal of developing PAH-specific SHRQoL assessment tools that can be used in both research and clinical populations.
Finally, the observation that participants treated with parenteral prostacyclin analogues trended toward higher (worse) FSDS-R scores than those on other modes of therapy is suggestive of treatment-based concerns in functionality and warrants further study. Based on the results of this study, the FSDS-R may identify such patients better than the emPHasis-10 or SF-36 scales. Parenteral therapies remain the “gold standard” for higher risk patients,47–50 but require specialized training, labor-intensive care, monitoring, and typically a partner or caregiver to participate in administration in addition to the patient. Not only are the pumps and catheters burdensome, their interruption can be fatal. 51 In patients with advanced left heart failure who have left ventricular assist devices (LVADs), device-related issues frequently contribute to sexual dysfunction and SHRQoL, akin to our observations, and in one study the impact of LVADs on sexual health tracked with the development of depression, known to be common in PAH.27–29 While we did not quantify anxiety and depression in this study, mental health is frequently impacted in individuals with chronic diseases characterized by equipment and physical challenges,52,53 which suggests that these issues may need to be addressed concurrently in PAH. Given the fact that PAH treatments are challenging but reduce symptoms and prolong survival, it is plausible that treatment with parenteral therapy may negatively impact sexual health and patients’ engagement in sexual behaviors, but not affect (or at least beneficially offset) other aspects of HRQoL. Intriguingly, only a small proportion of patients (43%) receive parental therapies prior to their death and only half of patients undergo therapeutic escalation even when they are recognized to have severe disease. 54 While predictors of patients receiving inadequate therapy have not been studied, a major perceived barrier is patient refusal. Treatment-specific impacts on factors such as caregiver burden, interpersonal relationships, and sexual intimacy are entirely unstudied but may explain the trend noted here.
Study considerations
It is difficult to determine from quantitative data how participants conceptualize their sexual health and functioning in the context of their illness. We cannot know whether participants responded to the survey items relative to their functioning prior to becoming symptomatic or their expected functioning taking into account their health status and treatment. It may be that women with PAH do not view sex as an important and/or necessarily frequent occurrence for them, either due to the progression of the disease or other factors – and that they would then respond to such surveys from that perspective. We anticipate that qualitative data gathered and analyzed from in-depth interviews may provide further insight into the context in which women with PAH perceive both their SHRQoL and general HRQoL which may yield hypotheses for further exploration and intervention.
The results of the present study may not represent the prevalence and severity of sexual health, sexual dysfunction and HRQoL in the PAH population-at-large. As this was a formative study designed to gauge whether SHRQoL was an important aspect of wellness in this previously unstudied population, we intentionally chose the conference as our sampling population (despite potential issues with generalizability) to: (1) ensure engaged participants willing to discuss a sensitive and difficult topic; and (2) increase the likelihood that participants were sexually active.
While it would have been helpful to correlate our HRQoL data with medical record data for confirmation of diagnosis and treatment details, these data were not available in the context of the PHA conference research room. Additionally, due to a very small, homogenous sample of men with PAH, we have only analyzed and reported data from women. While PAH does disproportionately affect women (compared to men), we were unable to address how quality of life and sexual health and functioning impact men with PAH. This may be of great interest given men with PAH treated with PDE5i, a therapeutic class also approved for the treatment of erectile dysfunction in the general population, are more likely to achieve clinically relevant responses in physical and mental component scores of the SF-36 than women with PAH treated with PDE5i. 14
Additional comprehensive studies to capture a more representative sample and to collect a wider range of disease-relevant biomarkers are needed. While we have used previously reported HRQoL and sexual functioning scores in general and clinical populations as comparisons for quality of life ratings in our sample, future studies should collect data from matched controls to further isolate the effects of PAH and PAH therapies on these outcomes. However, this study provides the first preliminary evidence of significant sexual dysfunction and distress in PAH patients and identifies a strong rationale for further study.
SHRQoL is an important domain of quality of life to address in women with PAH. Further analysis and studies are warranted to explore how well patients’ SHRQoL needs and impairments are being measured, understood, addressed, and treated. By better understanding specific issues faced by patients with PAH, patients, their sexual partners, and their providers can work toward an improved quality of life, both for the patients themselves and their partners, who often play a central caregiving role in the patient’s selection of treatment regimens and treatment administration. In addition, improved quality of life and improved intimacy may further ameliorate symptoms of depression and anxiety that often accompany the disease.55,56
Footnotes
Acknowledgments
We would like to thank the study participants and the Pulmonary Hypertension Association, as well as the PHA Conference’s Research Room Leadership Committee for allowing us to conduct this research. Brittany M. Wickham was a student intern at Stonehill College, Department of Psychology, Easton, MA, and contributed to this article as part of her externship program; she is now at the Rhode Island Hospital, Bradley/Hasbro Research Center, Providence, RI.
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health (grant number P20GM1403652) and National Institutes of Health R01 HL141268.
