Abstract
Symptomatic patients with residual pulmonary perfusion defects or vascular lesions but no pulmonary hypertension at rest are diagnosed with chronic thromboembolic disease (CTED). Balloon pulmonary angioplasty (BPA) is an emerging treatment for patients with inoperable chronic thromboembolic pulmonary hypertension (CTEPH), but data regarding the safety and efficacy of BPA in patients with CTED are lacking. We report a prospective series of ten consecutive patients with CTED who underwent 35 BPA interventions (median of four per patient) at two German institutions. All patients underwent a comprehensive diagnostic workup at baseline and 24 weeks after their last intervention. BPA was safe, with one pulmonary vascular injury and subsequent self-limiting pulmonary bleeding as the only complication (2.9% of the interventions, 10% of the patients). After the procedures, World Health Organization functional class, 6-min walking distance, pulmonary vascular resistance, and pulmonary arterial compliance improved, and NT-proBNP concentrations declined in 9/10 patients. BPA may be a new treatment option for carefully selected patients with CTED. A larger, prospective, international registry is required to confirm these results.
Keywords
Chronic thromboembolic pulmonary hypertension (CTEPH) is defined as symptomatic pulmonary hypertension (PH) due to non-resolving pulmonary emboli with a mean pulmonary arterial pressure (mPAP) of at least 25 mmHg, normal pulmonary arterial wedge pressure (PAWP; ≤15 mmHg), and perfusion defects that persist despite at least three months of anticoagulation. 1 It is assumed that PH develops due to an obstruction of at least 40–60% of the pulmonary vasculature, 2 but the role of the ensuing small-vessel pulmonary vascular remodeling remains unclear.3,4 In addition, patients may be symptomatic with persistent pulmonary perfusion defects but no PH at rest. These patients are referred to as having chronic thromboembolic disease (CTED). 5
Surgical pulmonary endarterectomy (PEA) is the gold-standard treatment for CTEPH and is potentially curative.6,7 Successful PEA has also been reported in patients with CTED, with perioperative mortality rates in the range of 0–0.9%.6,8,9 Due to the low risk of the procedure, PEA surgery is the treatment of choice for operable, symptomatic patients with CTED. 10
Approximately one-third of all CTEPH patients are inoperable, however, mostly due to peripheral localization of pulmonary vascular obstructions. 11 These findings can probably be extrapolated to CTED patients. Currently, an increasing number of inoperable CTEPH patients are treated with balloon pulmonary angioplasty (BPA) in addition to targeted medical therapy.12–17 Hence, BPA may also be considered a treatment option for patients with inoperable CTED, 11 but data on the application of BPA in this population are lacking.
Here, we present the results of the first ten consecutive CTED patients who underwent BPA in Germany.
Methods
Patient selection
The two centers participating in this study (Hannover Medical School and the Kerckhoff Clinic, Bad Nauheim) hold weekly conferences of a multidisciplinary team consisting of experienced PEA surgeons, interventional radiologists, cardiologists and/or pulmonologists, and anesthesiologists, if required, to discuss particular cases. 15 CTED was diagnosed in symptomatic patients in at least World Health Organization (WHO) functional class (FC) II with pulmonary vascular lesions on computed tomography (CT) or PA angiography but with mPAP < 25 mmHg at rest. 5 Patients were deemed inoperable based on a comprehensive assessment of imaging findings for peripherally located lesions that were not surgically approachable. 18 Per protocol, PH-targeted medication was not introduced before BPA.
All patients were informed in detail about the innovative nature of the procedure, including potential risks and benefits, and gave written consent to participate. The ethics committees of both institutions approved this prospective observational study.
Clinical assessment
As previously described, 15 all patients underwent standardized assessment before the first BPA and six months after the last intervention. Assessment included WHO FC, 6-min walking distance (6MWD) with Borg dyspnea index, pulmonary function testing including lung diffusion capacity for carbon monoxide (DLCO), serum levels of creatinine (with calculation of the estimated creatinine clearance) and of the N-terminal fragment of pro-brain natriuretic peptide (NT-proBNP), echocardiography, and right heart catheterization to determine right atrial pressure, pulmonary arterial pressures, PAWP, cardiac output (CO), cardiac index (CI), pulmonary vascular resistance (PVR), pulmonary arterial compliance (PAC), and mixed-venous oxygen saturation (SvO2). Cardiac magnetic resonance imaging (MRI) was used in the majority of patients to assess right ventricular (RV) function.
Balloon pulmonary angioplasty
BPA was performed as a staged procedure, with treatment of a limited number of pulmonary segments during each session. All procedures were performed in conscious patients with local anesthesia. The two centers used a common standard procedure: 15 a detailed plan for all interventions was established before the first session using angiographic und CT findings with the aim to treat all target lesions. Short-term web-like stenoses and long-segment obstructions in sub- and sub-subsegmental pulmonary arterial branches were considered as target lesions. Using a femoral or jugular access, a sheath was placed in the pulmonary artery and a guiding catheter was inserted into the target segmental arteries. The guidewire was then inserted into the target subsegmental branches, which were subsequently dilated by multiple balloon inflations. Based on the measured vessel diameter in CT and angiography slightly undersized balloons were used in all cases to avoid pulmonary arterial rupture. A final pulmonary angiogram documented the post-procedural morphologic result. In Hannover, selective C-Arm CT was also used to localize the target lesions. 17
Statistical analysis
All data for continuous variables are expressed as mean ± SD or as median and interquartile range (IQR), as appropriate. Categorical variables are reported as number and percentage. Continuous variables were compared using the Wilcoxon signed-rank test. Within-subject comparisons are made across repeated observations without correction for multiple comparisons. The CTED cohort data were distributed parametrically, as determined by the Kolmogorov–Smirnov test. All statistical tests were performed with SPSS software, version 22.0. A two-tailed P value <0.05 was considered to be statistically significant.
Results
Baseline characteristics, effects of BPA, and procedures
Characteristics of patients at time of inclusion.
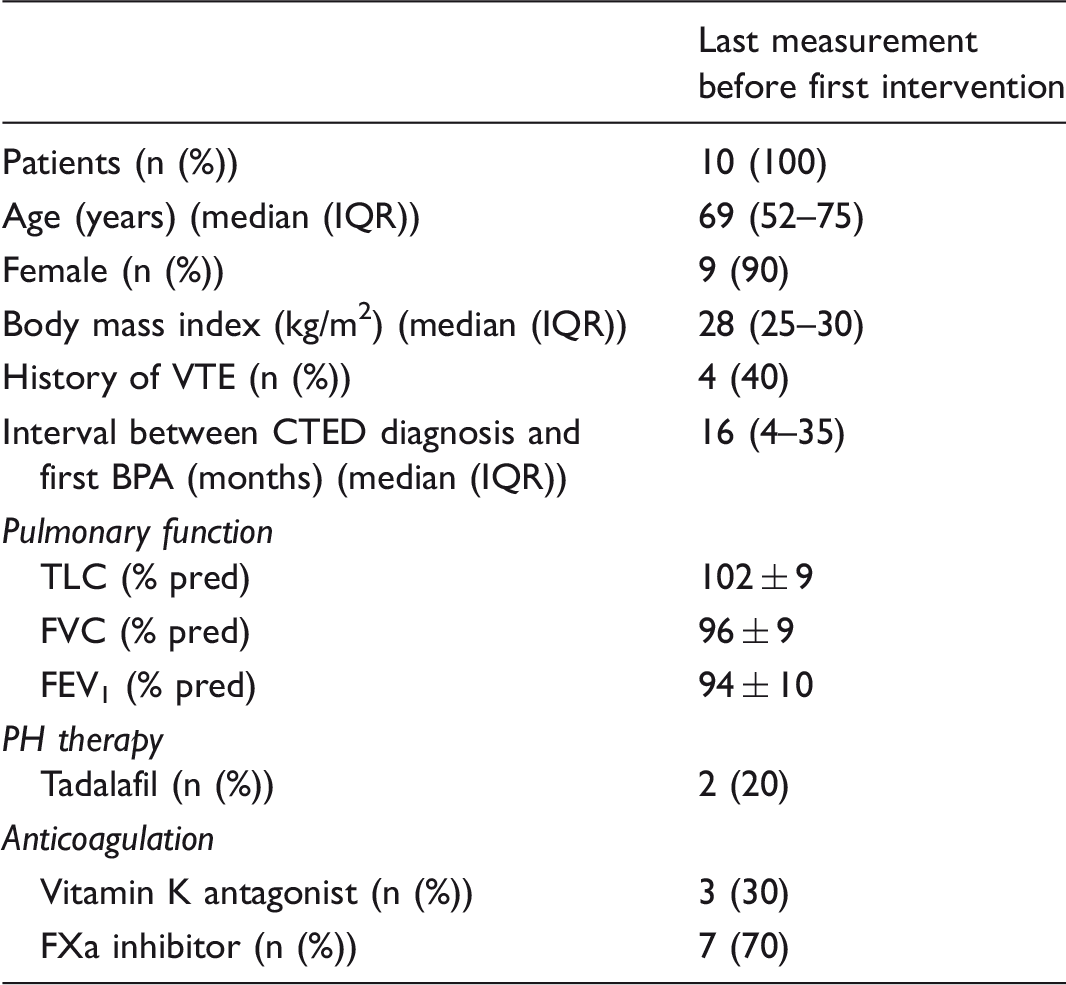
Values are given as mean ± SD unless otherwise indicated.
IQR, interquartile range; VTE, venous thromboembolism; CTED; chronic thromboembolic pulmonary disease; BPA, balloon pulmonary angioplasty; TLC, total lung capacity; FVC, forced vital capacity; FEV1, forced expiratory volume in 1 s.
All patients were treated with anticoagulants at least three months before diagnosis. Despite mPAP < 25 mmHg at the time of diagnosis, two patients received PH-targeted medical therapy with the phosphodiesterase 5 inhibitor sildenafil that was introduced at least three months before baseline assessment and kept unchanged during the study. Neither of these patients experienced improvement of their physical capacity under medication.
A total number of 35 interventions were performed: ten in Hannover and 25 in Bad Nauheim. In both centers, the median number of interventions per patient was four (range = 1–5). The median number of vessels targeted in all interventions was ten (range = 2–14). The median duration from first BPA to the 24-week follow-up assessment was 11 months (IQR = 9–14 months).
Treatment response
Changes from baseline to week 24.
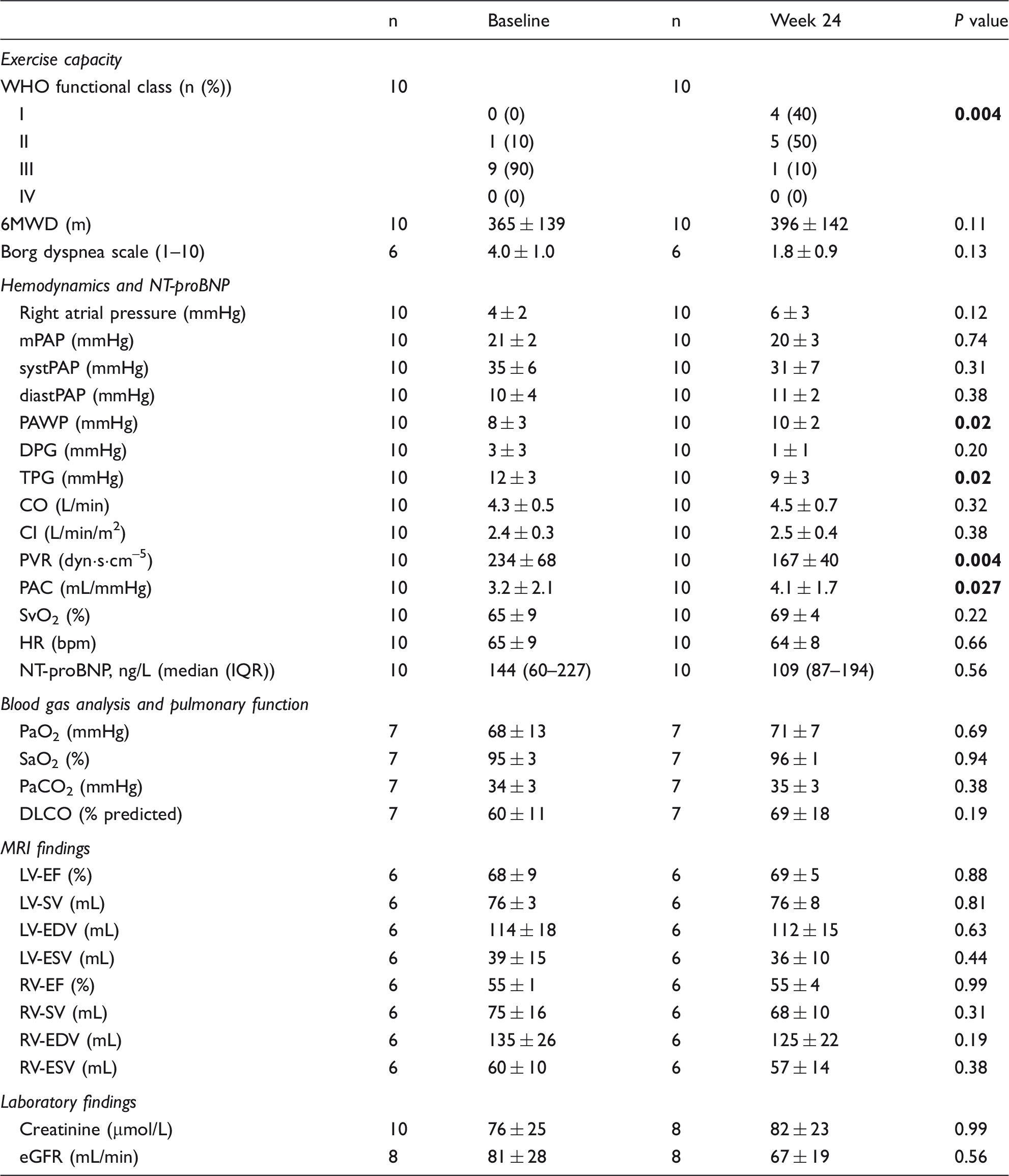
Values are given as mean ± SD unless otherwise indicated.
WHO, World Health Organization; mPAP, mean pulmonary artery pressure; systPAP, systolic pulmonary artery pressure; diastPAP, diastolic pulmonary artery pressure; PAWP, pulmonary arterial wedge pressure; DPG, diastolic pressure gradient; TPG, transpulmonary gradient; CO, cardiac output; CI, cardiac index; PVR, pulmonary vascular resistance; PAC, pulmonary arterial compliance; SvO2, mixed venous oxygen saturation; HR, heart rate; NT-proBNP, N-terminal fragment of pro-brain natriuretic peptide; PaO2, partial pressure of oxygen; SaO2, oxygen saturation; PaCO2, partial pressure of carbon dioxide; DLCO, lung diffusion capacity for carbon monoxide; MRI, magnetic resonance imaging; LV, left ventricular; RV, right ventricular; EF, ejection fraction; SV; stroke volume; EDV, end diastolic volume; ESV, end systolic volume; eGFR, estimated glomerular filtration rate based on serum creatinine.
Complications
One procedure-related complication (Hannover, n = 0; Bad Nauheim, n = 1) occurred during the 35 interventions (2.9% of all interventions, 10% of the patients). This adverse event was caused by wire perforation of the pulmonary vasculature, resulting in parenchymal bleeding with mild hemoptysis; intervention was not required. All of the patients were alive at the end of the observation period.
Discussion
The observations presented here focus on the use of BPA in inoperable symptomatic CTED patients. We demonstrate individual improvements in exercise capacity (6MWD, WHO FC) and pulmonary hemodynamics (PVR, PAC). Furthermore, we show that BPA is feasible and relatively safe in this particular group of patients.
CTED was only described recently and there are only a few reports addressing treatment of these patients. In 13 years, only 42 out of 1019 patients undergoing PEA in a UK cohort were diagnosed with CTED. 8 PEA programs worldwide have shown excellent results, with functional improvements and low mortality rates.5,7–9 Taboada et al. reported improvements in WHO FC, 6MWD, and PVR in 39 CTED patients before and six months after PEA: 20 patients were in WHO FC II and 22 in FC III before PEA; 16 patients were in WHO FC I, 21 in II, and two in FC III after PEA. 8 The 6MWD improved from 372 m to 413 m, and the PVR at rest decreased from 164 dynes to 128 dynes. These findings are in line with van Kan et al. who described changes in physical capacity and pulmonary hemodynamics in nine CTED patients one year after PEA: all patients were regrouped into WHO FC I after PEA and the 6MWD improved from 517 m to 548 m. 19 Therefore, PEA has become an accepted therapeutic strategy for symptomatic CTED patients, who potentially represent the lower border of the wide spectrum of CTEPH patients. 10 Based on experiences with CTEPH, however, it can be speculated that around one-third of all CTED patients are deemed inoperable. Previous data indicated that BPA may be feasible and effective in patients with lower mPAP. 19
Our preliminary results suggest that BPA may be an effective interventional treatment for patients with inoperable CTED, leading to improvements in WHO FC and pulmonary hemodynamics in the majority of the patients. The mean PVR reduction of 67 dynes*s/cm5 is comparable to the results published for PEA procedures.7–9 Furthermore, we observed a distinct improvement in PAC, which is believed to be one of the most important markers of prognosis in P(A)H patients. 20 However, its meaning in CTED remains unclear.
In our study, 6/10 patients increased their 6MWD and 7/10 patients had a lower NT-proBNP level 6 months after treatment. Overall, however, the changes in 6MWD and NT-proBNP were not statistically significant, potentially due to the low number of patients studied. Nevertheless, 9/10 patients had an improvement in WHO FC, a change that was significant for the cohort as a whole. MRI revealed that RV function was not affected by treatment; these findings before and after treatment. However, these findings could be explained by the absence of PH in this particular group of patients.
The small number of patients enrolled in this study is a major limitation that must be considered. In addition, there was no control group and long-term data are missing. The findings are nevertheless sufficient to demonstrate improvement in physical capacity and PVR. Furthermore, our data may provide a realistic perspective on the treatment of CTED patients in referral centers with an established surgical and interventional program for CTEPH.
Little is known about CTED and it is still being discussed whether it is an early stage in the development of CTEPH. PEA is an accepted surgical treatment for CTED patients, but there is no established treatment for inoperable patients. Our observation is the first to describe the effects of BPA in this particular group of patients. Improvements in physical capacity as well as pulmonary vascular resistance were achieved without severe complications. These data add important new information for the treatment of inoperable CTED patients. However, we have no data concerning prognostic implications of BPA in inoperable CTED patients. The development of a comprehensive therapeutic concept for CTED patients will require additional data and individualized decision-making based on multidisciplinary experience.
Individual effects of balloon pulmonary angioplasty on mPAP, PVR, functional capacity, 6MWD, and NT-proBNP. NT-proBNP, N-terminal pro-brain natriuretic peptide; PVR, pulmonary vascular resistance; PAC, pulmonary arterial compliance.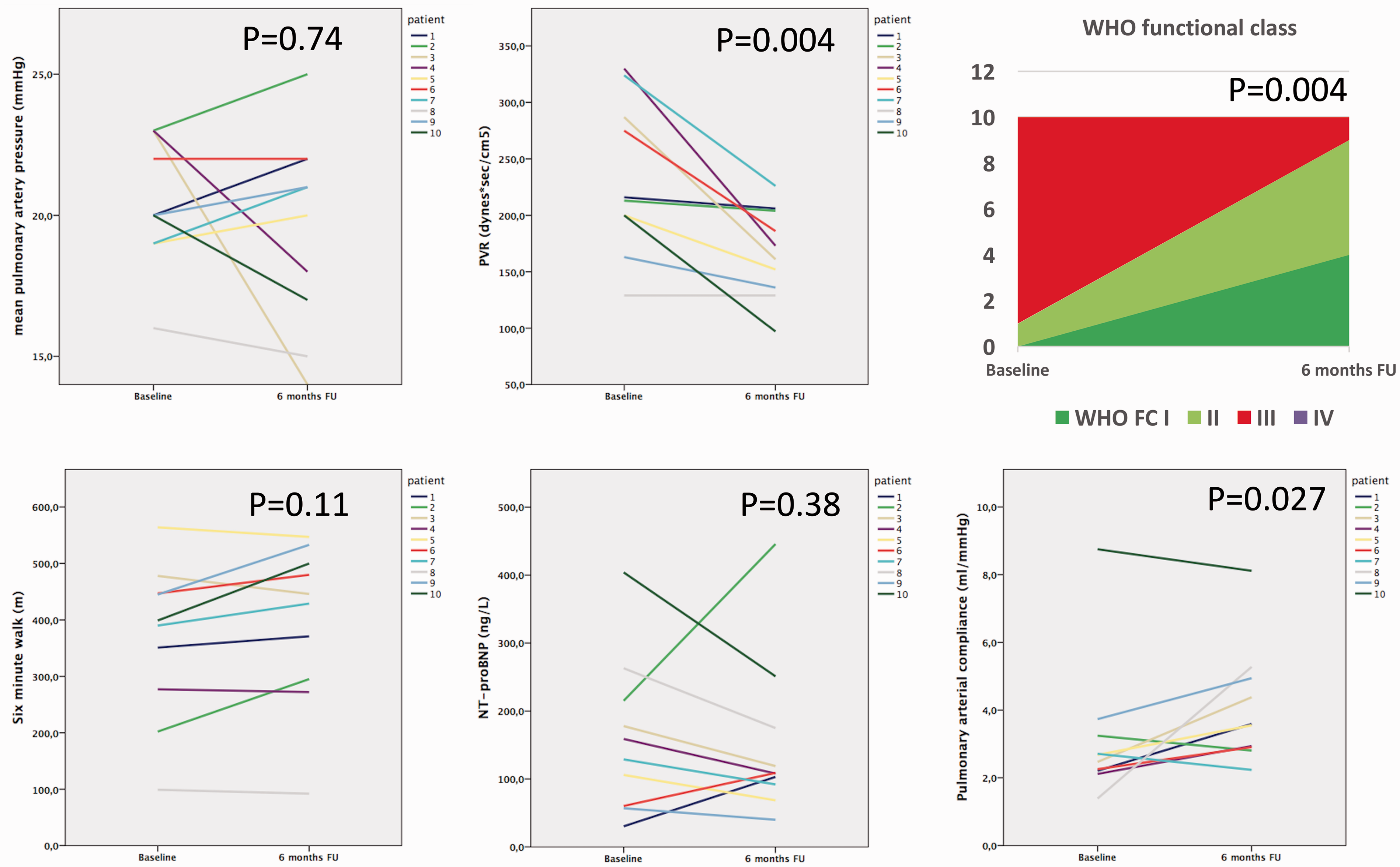
Footnotes
Acknowledgments
The authors thank Elizabeth Martinson, PhD, from the KHFI Editorial Office for her editorial assistance.
Conflict of interest
The author(s) declare the following conflicts of interest: CB Wiedenroth has received speaker fees or consultant honoraria from Actelion, Bayer AG, BTG, MSD, and Pfizer. KM Olsson has received speaker fees from Actelion, Bayer, GSK, Pfizer, and United Therapeutics. S Guth has received speaker fees from Actelion, Bayer, GSK, and Pfizer. E Mayer has received speaker fees and/or honoraria for consultations from Actelion, Bayer, GSK, MSD, and Pfizer. HA Ghofrani has reported receiving fees for serving as a board member for Bellerophon Pulse Technologies, Medscape, OMT, UCB Celltech, and Web MD Global; receiving consultancy fees and fees for serving on a steering committee for Actelion Pharmaceuticals, Bayer, Gilead Sciences, GlaxoSmithKline, Merck, Novartis, and Pfizer; receiving lecture fees from Actelion Pharmaceuticals, Bayer, GlaxoSmithKline, Merck, Novartis, and Pfizer; and receiving grant support from Actelion Pharmaceuticals, Bayer, Novartis, and Pfizer. BC Meyer has received speaker fees from Bayer, MSD, and Siemens Healthineers. C Liebetrau has received speaker fees from Abbott, Astra Zeneca, Bayer, Berlin-Chemie, Boehringer Ingelheim, Daiichi Sankyo, Elixir Medical, and Pfizer. The rest of the authors have nothing to disclose.
Funding
This study was funded by the German Centre of Lung Research (DZL).
