Abstract
Background:
Treatment-resistant depression (TRD) remains a major clinical challenge, and long-term real-world data on intranasal esketamine are limited.
Objectives:
To describe long-term dosing patterns and clinical outcomes associated with intranasal esketamine in routine clinical practice.
Design:
Single-center observational cohort study with descriptive analyses.
Methods:
We analyzed 45 patients with TRD treated with intranasal esketamine in a naturalistic setting, with follow-up extending up to 260 weeks. Esketamine was used adjunctively with ongoing oral antidepressants. Outcomes included the Montgomery–Åsberg Depression Rating Scale (MADRS), the Young Mania Rating Scale (YMRS), and the Glasgow Antipsychotic Side-effect Scale (GASS). Treatment patterns were summarized using person-time (person-weeks).
Results:
Baseline MADRS was 40.0 (SD 4.63). Mean MADRS decreased to 22.9 (SD 7.99) at week 4 and to 9.70 (SD 5.35) at week 52, with mean scores remaining around 9–10 at later time points among patients with available follow-up. Eight patients (17.8%) discontinued treatment (lack of efficacy
Conclusion:
In a real-world TRD cohort with complex comorbidities, intranasal esketamine used adjunctively was associated with sustained symptom improvement and a favorable long-term safety profile among patients who continued treatment, with dosing evolving from induction to individualized maintenance schedules. Findings are limited by the observational design, concomitant treatments, and survivor bias.
Trial registration:
Not applicable.
Keywords
Introduction
Major depressive disorder (MDD) is a prevalent and disabling illness; however, a large number of patients fail to achieve complete remission and functional capacity restoration despite undergoing evidence-based therapeutic interventions. 1 Clinical research has demonstrated that approximately 30%–40% of MDD patients have poor responses to initial antidepressant treatment, whereas as many as 60%–70% fail to achieve complete remission. Furthermore, approximately 10% of individuals with depression persist in exhibiting enduring depressive symptoms despite undergoing various treatment interventions. 2 As the count of unsuccessful therapies rises, the likelihood of attaining remission markedly diminishes, thereby complicating long-term treatment strategies.
The functional and clinical disability of treatment-resistant depression (TRD) is significant. Studies have demonstrated that up to 75% of individuals with TRD are profoundly disabled with severe impairment in work, social relationships, and overall functioning. The continuation of depressive symptoms is linked with a higher risk of relapse, suicidal ideation, and long-term disability. 3 Findings from the STAR*D trial point out that following two unsuccessful antidepressant treatments, the probability of remission falls below 14%, thereby underscoring the urgent need for more efficacious and individualized treatment strategies. Because of these concerns, contemporary recommendations favor a move to an integrative and patient-oriented therapeutic practice. This entails not just optimizing pharmacological practice but also integrating psychotherapy, neuromodulation, and lifestyle interventions to improve general health and well-being.
The cross-sectional study by Jaffe et al. 4 provides compelling evidence that conventional treatments for MDD yield suboptimal results, particularly in patients with TRD.
The total sample included 52,060 survey respondents, among whom 3308 were identified as having MDD. Within this subgroup, 622 individuals (18.8%) met the criteria for TRD, defined as patients who had failed to respond to at least two different antidepressant treatments during their current moderate-to-severe depressive episode.
Findings revealed that conventional antidepressant treatments, primarily those targeting the monoaminergic system, fail to provide adequate and sustained clinical benefits in up to 70% of TRD patients. Furthermore, TRD was associated with significantly greater functional impairment, reduced quality of life, and increased healthcare utilization, including higher hospitalization and emergency visit rates compared to both nTRD patients and the general population.5–7
Certain second-generation antipsychotics—including aripiprazole, brexpiprazole, cariprazine, and quetiapine XR—have shown efficacy as adjunctive therapies for partial responders. Beyond pharmacological treatments, neuromodulation techniques such as repetitive transcranial stimulation (rTMS) and electroconvulsive therapy have gained recognition in TRD management. rTMS, including accelerated theta-burst protocols, is FDA approved for TRD.8,9
Intranasal esketamine, developed following encouraging findings with intravenous ketamine,10,11 has been studied as a viable option for TRD when used alongside an antidepressant and was approved by regulatory authorities in Italy in 2019, increasing its availability as a therapeutic option for TRD. 12
Concurrently, research is ongoing to develop novel pharmacological approaches, such as newer pharmacological strategies targeting novel mechanisms N-methyl-D-aspartate (NMDA) receptor modulators, amino-3-hydroxy-5-methyl-4-isoxazole propionic acid (AMPA) potentiators, and kappa opioid receptor antagonists in advanced clinical trials. 13 Among these approaches, R-ketamine, an enantiomer of ketamine, was identified as a possible alternative to (S)-ketamine. 14 Its presumptive mechanism of action is thought to involve NMDA receptor antagonism, along with other non-NMDA mechanisms, which cumulatively afford its rapid and sustained activity across neurological and psychiatric disorders. 14
While these developments highlight ongoing efforts to find more effective and better-tolerated therapeutic strategies, a pressing need remains to bridge the gap between clinical trials and everyday clinical practice in real-life conditions.
Three pivotal short-term efficacy trials (TRANSFORM-1, -2, -3) evaluated esketamine nasal spray plus a newly initiated oral antidepressant versus placebo nasal spray plus oral antidepressant in patients with treatment-resistant depression. Results were mixed: TRANSFORM-1 did not achieve statistical significance for the primary endpoint (
Randomized withdrawal trial SUSTAIN-1 showed that continued therapy with esketamine seemed to extend time to relapse compared with switching to placebo. The open-label extension study SUSTAIN-2 supported the tolerability and potential advantages of esketamine for up to 1 year, while SUSTAIN-3 provided extended follow-up for up to 4 years, which showed a good safety profile.18,19 Conversely, ESCAPE-TRD represented a phase IIIb, rater-blinded, randomized trial that rigorously tested the efficacy of esketamine nasal spray in relation to quetiapine XR, both with an adjunct oral antidepressant, in TRD individuals.20,21 The results indicated a higher remission rate at 8 weeks (27.1% compared to 17.6%), with an increased rate of prolonged remission at 32 weeks in the esketamine group, thereby supporting its efficacy in both acute and maintenance treatment phases. 22
The REAL-ESK study 23 represents the first large study in Italy to examine whether esketamine nasal spray works well and safely, with 116 TRD patients examined at 1 month and 3 months, using reliable outcome measurements (MADRS, HAMD-21, HAM-A). At 3 months, 64.2% of patients showed clinical response (⩾50% reduction in depression severity), while 40.6% seemed to achieve remission (absence of substantial depressive symptoms). Response trajectories showed heterogeneity in time to clinical improvement, with some patients exhibiting a rapid onset of therapeutic effect while others showed delayed response patterns. The adverse event profile was predominantly mild to moderate in severity and consistent with previously published safety data, with no reported incidents of substance misuse, abuse potential, or dependence.
Despite these encouraging results, the query on long-term treatment outcomes within everyday clinical practice is yet to be answered. 24 Given the mixed results from controlled trials—with only one of three pivotal TRANSFORM studies achieving its primary endpoint—and questions about the clinical significance of observed effects in controlled settings, 25 long-term real-world evidence is particularly important for informing clinical decision-making. Observational studies, while unable to establish causality or isolate treatment effects, can provide complementary evidence about outcomes in more diverse, complex patient populations treated in routine practice settings, where treatment resistance may be more severe and comorbidities more prevalent than in controlled trial samples.
Specifically, data on the effectiveness and safety of esketamine in patients with common medical and psychiatric comorbidities, as seen in real-world clinical practice, are limited. 26 Addressing this gap in knowledge, the current study aims to explore both individuals and overall sample treatment patterns and outcomes trajectory in 45 patients with treatment-resistant depression, with follow-up to 5 years to pilot further investigation with more sophisticated statistical tools in the very next future.
Materials and methods
Study design and population
As an exploratory investigation, this study employs descriptive statistics to characterize the sample, treatment implementation, and outcomes without inferential hypothesis testing. The descriptive approach serves two key purposes: (1) to provide clinicians with preliminary real-world information about esketamine use in complex patients typically excluded from controlled trials, and (2) to identify patterns that can guide the design of more rigorous inferential analyses to be conducted on this expanding dataset as additional observations are collected. This naturalistic framework allows for an initial exploration of long-term treatment strategies, potential efficacy signals, and safety considerations in routine clinical practice.
This preliminary observational study provides a descriptive analysis of treatment patterns and outcomes in patients diagnosed with treatment-resistant depression (TRD) undergoing esketamine therapy in naturalistic clinical settings. A total of 45 patients were included, with a follow-up period extending up to 5 years. No a priori sample size calculation was performed for this observational study. The sample size represents all eligible patients initiating esketamine during the recruitment period. Patients were enrolled through rolling recruitment from March 18, 2019, to September 3, 2024, with final data collection on November 6, 2024.
Patients were recruited through convenience sampling from the outpatient psychiatry service. Treating psychiatrists identified potentially eligible patients with treatment-resistant depression and assessed eligibility. The decision to offer esketamine was made by the treating psychiatrist based on individual clinical judgment, considering severity, treatment resistance, functional impairment, and patient preference. No systematic protocol was used to determine which eligible patients were offered esketamine versus other options, representing potential selection bias. This convenience sampling may have resulted in the selection of patients with particular characteristics that limit generalizability.
Each patient’s baseline (Week 0) represents their individual treatment initiation date, not a fixed calendar date. Consequently, the maximum potential follow-up duration varied by enrollment date: patients enrolled in 2019–2020 could complete up to 5 years of observation, while those enrolled later had proportionally shorter maximum follow-up opportunities. The varying number of patients with available data at later time points reflects both actual treatment discontinuation and “administrative censoring” (insufficient elapsed time for later-enrolled patients to reach those time points before study closure). Patients were followed through regular clinical encounters for esketamine administration and routine clinical assessment. Standardized rating scales were administered during prespecified timing encounters at baseline and 11 follow-up time points (1, 2, 3, 4, 8, 13, 26, 39, 52, 104, 156, 208, and 260 weeks). Clinical assessments were conducted in a hospital setting, including both inpatients admitted to psychiatric wards and outpatients attending scheduled evaluations. The study was carried out at the University Hospital of Siena, a tertiary care academic center.
The reporting of this observational study conforms to the STROBE statement. 27
Inclusion and exclusion criteria
Patients aged 16–75 years (with guardian consent for minors) with DSM-5 major depressive disorder meeting treatment-resistant depression criteria were eligible. Treatment-resistant depression was defined as an inadequate response to at least two antidepressant trials of adequate duration and dosage. “Adequate trial” meant: (1) minimum 6 weeks at therapeutic dose, (2) dose within or above therapeutic range per prescribing guidelines, (3) reasonable adherence, and (4) <50% symptom improvement. Therapeutic drug monitoring was not systematically performed. All patients had documented SSRI trials, with most also failing SNRIs and other classes. Median previous trials: 3 (range 2–8). Medical exclusions: aneurysmal vascular disease, intracerebral hemorrhage history, uncontrolled hypertension, recent MI or unstable cardiac disease, and severe hepatic impairment. Psychiatric exclusions: active psychotic disorder (except psychotic depression), active substance use disorder (within 3 months), and acute suicidal ideation requiring hospitalization. Practical exclusions: inability to attend visits, pregnancy/breastfeeding, and lack of consent capacity.
Baseline assessments
Demographic and clinical characteristics were recorded at baseline, including age, gender, body mass index (BMI), employment status, and psychiatric comorbidities. The Montgomery-Åsberg Depression Rating Scale (MADRS) was used to assess depression severity, with additional measures for suicidality and psychiatric history. Concurrent psychotropic medication use, including antidepressants, mood stabilizers, antipsychotics, benzodiazepines, and GABA analogs, was documented.
Esketamine treatment protocol
Esketamine was administered intranasally as an adjunctive treatment to ongoing antidepressant therapy, following standard clinical guidelines. The initial dose was determined based on patient characteristics, with most patients receiving 56 mg per session. Dose adjustments were made based on clinical response and tolerability, with the possibility of increasing to 84 mg or decreasing to 28 mg if necessary. Treatment frequency varied over time, with an induction phase consisting of twice-weekly administrations followed by individualized maintenance schedules.
Follow-up and treatment patterns analysis
Person-time is an epidemiological concept that measures both the number of persons and their time contribution to a study, accounting for varying follow-up durations. In this analysis, person-weeks represent the cumulative time patients spent on treatment, allowing for comparison of treatment patterns while adjusting for different observation periods. The metric is calculated by summing the weeks each patient spent on treatment, providing a more accurate representation of treatment patterns than simple patient counts. Patients were followed for a total of 4607 person-weeks, with individual follow-up durations ranging from 1 to 260 weeks. Esketamine dosing frequency and cumulative weekly doses were recorded throughout the follow-up period. A detailed analysis of administration frequency trends was conducted, highlighting transitions from intensive induction dosing to extended-interval maintenance schedules.
Outcome measures
The primary outcome measure was the change in MADRS scores over time. Descriptive statistics (mean, standard deviation, median, range, and quartiles) were calculated for MADRS scores at each assessment time point (baseline, 1, 2, 3, 4, 8, 13, 26, 39, 52, 104, 156, 208, and 260 weeks). Individual MADRS trajectories were plotted alongside mean values to illustrate response patterns and heterogeneity in treatment effects.
Safety and tolerability assessment
Safety monitoring combined systematic assessment tools with clinical observation. The Glasgow Antipsychotic Side-effect Scale (GASS; range 0–22, higher scores indicating more side effects) was administered at each follow-up time point. The GASS assesses common side effects of antipsychotic medications, including extrapyramidal symptoms, sedation, weight gain, and sexual dysfunction. Given that 60% of patients were receiving concurrent antipsychotic medications, GASS was selected to monitor potential additive or synergistic side effects in this polypharmacy context.
However, GASS does not adequately capture esketamine’s characteristic adverse effects, including dissociation, perceptual disturbances, and blood pressure elevations.
The Young Mania Rating Scale (YMRS; range 0–60, higher scores indicating more severe manic symptoms) was used to monitor for potential mood elevation or treatment-emergent hypomanic/manic symptoms, given concerns about ketamine’s effects on bipolar spectrum patients.
Esketamine-specific safety parameters monitored through clinical assessment but not systematically quantified using validated scales included: (1) dissociative symptoms during and immediately following each administration, documented qualitatively in clinical notes when severe or distressing; (2) blood pressure measured before and after each administration per protocol; and (3) common adverse effects including nausea, dizziness, and headache elicited through clinical interview. All adverse events leading to treatment discontinuation or dose modifications were documented.
Statistical analysis
This study employed purely descriptive statistical approaches to characterize the sample and summarize treatment patterns and outcomes. No inferential statistical testing or hypothesis testing was performed, in alignment with the exploratory and descriptive aims of this preliminary investigation.
Baseline demographic and clinical characteristics were presented using frequencies and percentages for categorical variables (gender, comorbidities, medication use), while continuous variables were summarized using means, standard deviations, medians, ranges, and quartiles as appropriate (age, BMI, MADRS scores). Treatment patterns were analyzed using absolute and relative frequencies, with person-time calculations (person-weeks) employed to account for varying follow-up durations when describing treatment intensity and dosing strategies.
Temporal changes in MADRS, YMRS, and GASS scores were visualized through line graphs displaying individual trajectories alongside mean values at each time point, with descriptive statistics (mean, SD, min, max, quartiles) calculated for each assessment interval. Side effect patterns were further characterized through frequency distributions and percentage calculations to identify predominant adverse effect categories.
We did not perform inferential statistical tests (e.g., paired
All analyses and visualizations were conducted using R statistical software (version 4.1).
Results
Patient enrollment, retention, and reasons for discontinuation
Of the 45 patients who initiated esketamine treatment, 8 patients (17.8%) discontinued treatment during the observation period. Reasons for discontinuation were as follows: lack of efficacy (
The number of patients contributing data at each assessment time point was as follows: baseline (
No substantial differences in baseline demographics or clinical characteristics were observed between patients who discontinued treatment (
Demographic and clinical data
Table 1 reports the number of patients for each time point of observation (since the date of recruiting). The sample demonstrated atypical sex distribution with male predominance (68.9% male vs 31.1% female), contrasting with typical female predominance in major depression studies. Age ranged from 17 to 75 years (mean 52.6 ± 14.2 years); the single patient aged 17 was within one month of the 18th birthday and included with guardian consent per institutional ethics protocol. Body mass index (BMI) analysis revealed a mean of 25.5 (SD = 3.73) and a median of 26, with a range from 17 to 33. Employment status analysis showed that 38 patients (84.4%) maintained employment, while 7 (15.6%) were unemployed at baseline. Our cohort of 45 patients was followed for a total of 4607 person-weeks, with individual follow-up durations ranging from 1 to 260 weeks (5 years). The median follow-up duration was 52 weeks, with a mean of 102 weeks (SD = 85.7), indicating a right-skewed distribution of observation periods. Since 37 patients (82.2%) were still actively receiving esketamine at the end of the observation period, observed follow-up times predominantly reflect administrative censoring rather than completed treatment courses, and summary statistics of treatment duration would underestimate the true length of therapy. This extended follow-up period provided a robust framework for assessing both short-term treatment responses and long-term outcomes. Baseline sociodemographic characteristics are summarized in Table 2.
Number of patients for each time point.

Baseline sociodemographic and clinical characteristics.

MADRS, Montgomery–Åsberg Depression Rating Scale; YMRS, Young Mania Rating Scale.
The baseline clinical characteristics of the study population revealed a high burden of psychiatric and somatic comorbidities, reflecting the complexity of treatment-resistant depression. Baseline assessments of MADRS score revealed severe depressive symptomatology, with a mean score of 40.0 (SD = 4.63) and a relatively narrow distribution (IQR: 38.0–43.0), indicating a homogeneously severe clinical population at treatment initiation. Regarding baseline YMRS, patients exhibited low levels of manic symptoms, with a mean score of 12.0 (SD = 2.33).
Extensive polypharmacy was observed, with
Esketamine dosage and administration protocol
Analysis of initial dosing patterns showed both adherence to and variation from recommended protocols, with clinical judgment guiding individualization. The majority of patients (
The weekly esketamine dose is calculated by multiplying the single administered dose (28, 56, or 84 mg) by the frequency of administration per week. For example, a patient receiving 56 mg twice weekly would have a total weekly dose of 112 mg, while a patient receiving 84 mg once weekly would have a total weekly dose of 84 mg. This calculation provides a standardized way to compare treatment intensity across different dosing frequencies.
Analysis of the temporal pattern of weekly dosing (Figure 1) reveals several distinct phases in treatment implementation. During the first month (weeks 1–4), most patients (80.0%–82.5%) received high-intensity treatment with weekly doses exceeding 84 mg, reflecting the initial induction phase of twice-weekly administration. A smaller proportion (16.7%–20.0%) received moderate doses of 28–56 mg weekly, while very few patients started with lower-dosing regimens.

Distribution of weekly dose over time.
A notable transition occurred around week 8, where the distribution began to shift toward lower weekly doses. By week 13, 62.2% of patients were receiving 28 mg weekly, while 29.7% maintained moderate dosing (28–56 mg/week), and only 5.4% continued high-intensity treatment. This trend toward dose reduction continued steadily, with 78.4% of patients on low-dose maintenance by week 26 and 86.5% by week 39.
Long-term follow-up data showed further consolidation of this pattern. By week 52, 91.9% of patients were maintained on 28 mg weekly, and this proportion reached 100% by week 156 (3 years) and remained stable through year 5.
Analysis of the administration frequency distribution (Figure 2) reveals that monthly dosing was predominant, accounting for 2693 person-weeks (58.5% of total observation time). Biweekly dosing represented the second most common frequency with 1108 person-weeks (24.1%), likely reflecting an intermediate maintenance phase for many patients. Administration every 3 weeks accounted for 377 person-weeks (8.2%).
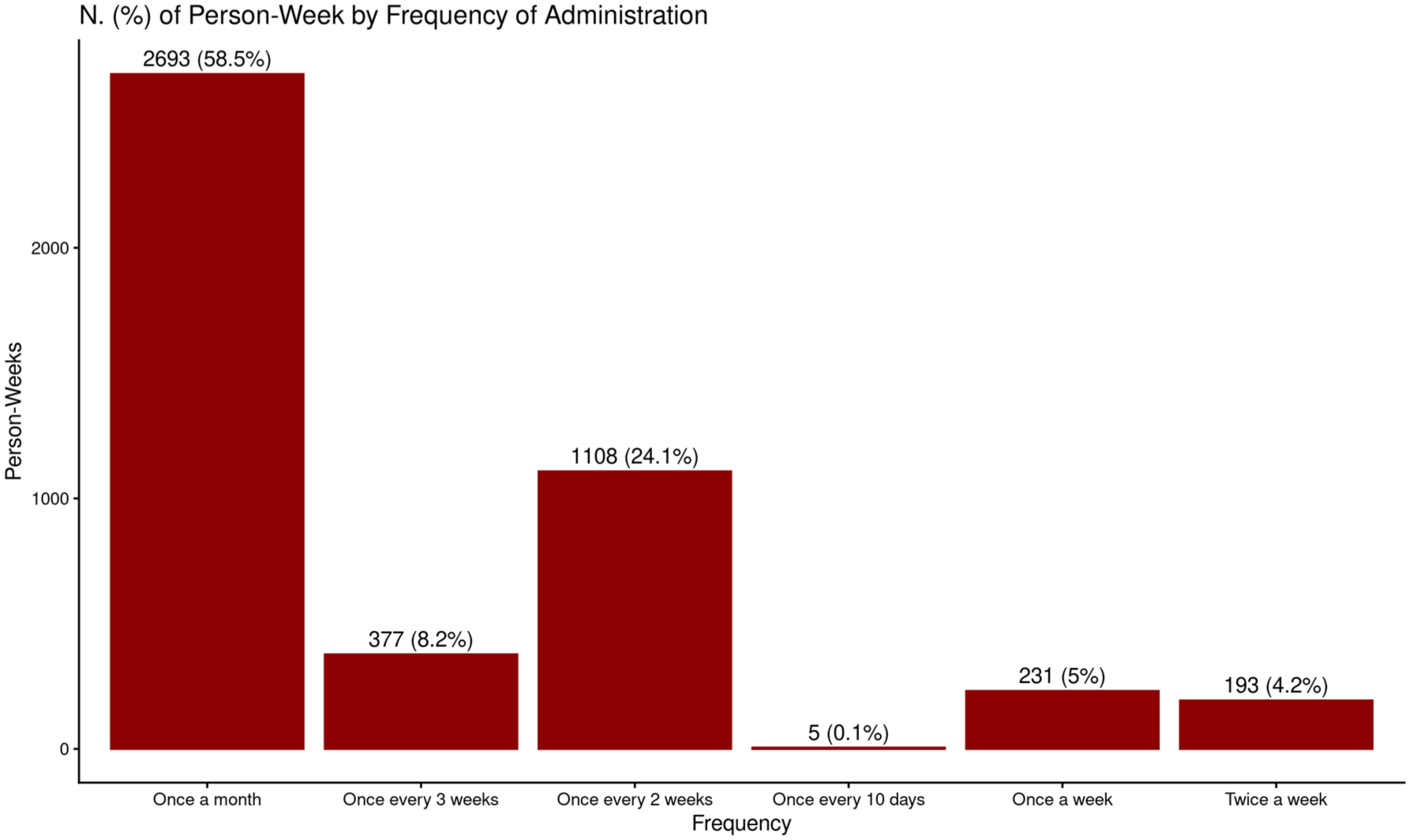
Count and percentage of person-week by weekly dose.
More frequent administration patterns were less common in terms of cumulative person-time: weekly dosing represented 231 person-weeks (5.0%), while twice-weekly administration accounted for 193 person-weeks (4.2%). The least common frequency was administration every 10 days, accounting for only 5 person-weeks (0.1%).
Clinical efficacy
The temporal evolution of depressive symptoms demonstrated a marked initial improvement. By week 4, mean MADRS scores decreased substantially to 22.9 (SD = 7.99), representing a 42.8% reduction from baseline. This robust early response continued through the first 3 months, with mean scores reaching 15.6 (SD = 6.37) at week 13. Further improvement was observed at 6 months (mean = 13.2, SD = 5.61), with scores stabilizing around the 1-year mark at 9.70 (SD = 5.35). Notably, these therapeutic gains were maintained through the extended follow-up period, with mean scores remaining stable between 9 and 10 points through years 2–5, though with progressively smaller sample sizes.
Individual trajectories, represented by the light blue lines in the figure, reveal considerable heterogeneity in response patterns, particularly during the first month of treatment. While most patients exhibited rapid initial improvement, the slopes of decline varied substantially. Some individuals demonstrated dramatic early responses, while others showed more gradual improvement. Despite this early variability, the majority of trajectories converged toward lower severity scores by month 3, with relatively stable long-term outcomes. The visualization of individual patterns suggests that while response timing may vary, sustained improvement was achievable across diverse response patterns (Figure 3).

MADRS score individual trajectories and mean values over time.
There was a progressive reduction in score variability over time, with the standard deviation decreasing from 7.99 at week 4 to 3.01 at the 5-year mark (week 260, mean = 9.33,
Side effects profile
Three patients (6.7%) discontinued treatment due to adverse effects. Specific adverse events were as follows: (1) severe headache and dissociative symptoms at 2 weeks (
Among patients who continued treatment, transient dissociative symptoms, blood pressure elevations (managed through monitoring), dizziness, and nausea were commonly observed during acute administration periods but were generally tolerated and did not lead to discontinuation. These common but tolerated adverse effects were not systematically quantified using validated measures.
At baseline, patients showed minimal manic symptoms (mean YMRS = 12.0, SD = 2.33), likely reflecting general psychomotor activation associated with severe depression rather than true manic features. During the initial treatment phase, YMRS scores showed a gradual decline rather than elevation, reaching a mean of 8.98 (SD = 2.03) by week 4. This downward trend continued steadily, with scores decreasing to 5.65 (SD = 1.74) at 3 months and further reducing to 3.76 (SD = 1.40) by 6 months. The long-term follow-up revealed sustained stability in YMRS scores, with means consistently below 3 points after the first year (2.54 at week 52, declining to 1.33 at week 260). Individual trajectories, while showing some variability in the early weeks, demonstrated remarkable consistency in their downward trend, with no cases of significant manic elevation observed throughout the follow-up period. Descriptively, we observed variance compression in YMRS scores over time, with standard deviations decreasing from 2.33 at baseline (

YMRS score individual trajectories and mean values over time.
Formal side effect assessment utilized the Glasgow Antipsychotic Side-effect Scale (GASS; range 0–22) and Young Mania Rating Scale (YMRS; range 0–60). GASS was selected given high rates of concurrent antipsychotic use (60%), but does not capture esketamine-specific adverse effects (dissociation, blood pressure changes), limiting the comprehensiveness of our tolerability characterization. YMRS monitored for potential mood elevation or treatment-emergent manic symptoms. Our analysis of side effects using the GASS revealed a favorable long-term tolerability profile (Figure 5). At baseline, patients demonstrated a relatively low side effect burden (mean GASS = 2.60, SD = 2.86, median = 2.0), which remained stable during the initial treatment phase (weeks 1–4, median consistently at 2.0 despite slight fluctuations in means). The distribution of GASS scores was notably right-skewed throughout the study period, with a substantial proportion of patients reporting minimal side effects, while a smaller subset experienced more pronounced adverse reactions. This skewness makes the median a more appropriate measure of central tendency than the mean, which is disproportionately influenced by the few higher values.
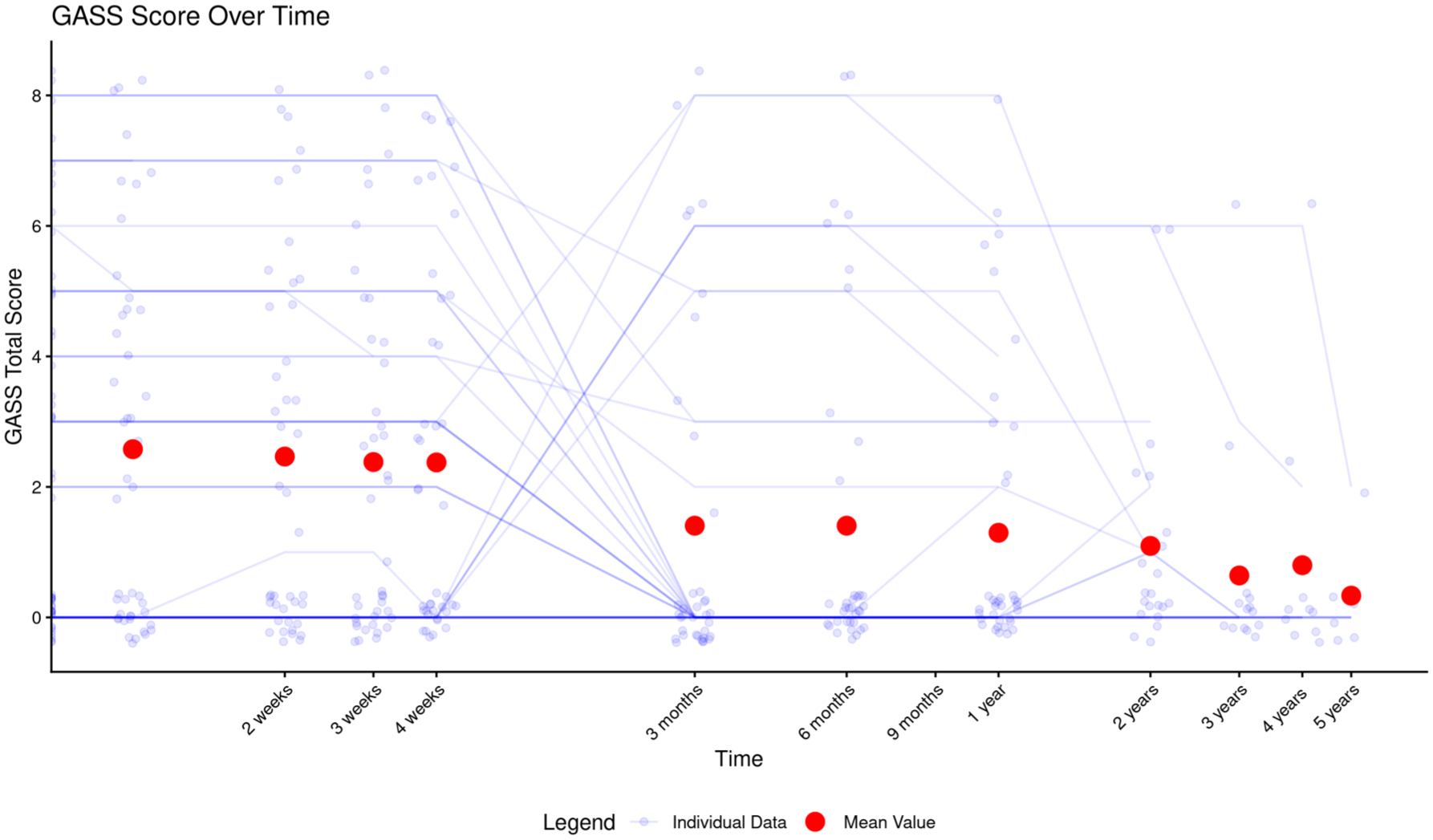
GASS score individual trajectories and mean values over time.
A notable pattern emerged between week 4 and week 13, where individual trajectories revealed considerable crossing of paths—some patients showing improvement, while others experienced transient worsening of side effects—before converging toward lower scores. This heterogeneity in individual responses during the transition from acute to maintenance treatment would have been obscured in analyses focused solely on aggregate measures. Despite this initial variability, a significant improvement was observed by week 13, with median GASS scores decreasing to 0 and mean scores to 1.41 (SD = 2.55).
The long-term follow-up demonstrated further improvements in tolerability. By week 52, the mean GASS score had decreased to 1.30 (SD = 2.27), with continued reduction through extended follow-up (mean = 0.333, SD = 0.816 at week 260). The distribution of scores showed that while many patients experienced no side effects (median = 0 from week 13 onward), there was persistent individual variability in susceptibility to adverse effects, as evidenced by maximum scores of 6–8 points through the first 2 years. This pattern of predominantly zero values with occasional higher scores creates zero-inflated distributions at later time points, further complicating traditional statistical approaches that assume normality.
Discussion
This real-world study provides valuable evidence on the use of intranasal esketamine in patients with treatment-resistant depression (TRD), including those with complex psychiatric and medical comorbidities, complementing the existing empirical literature. 28
Our treatment approach, with esketamine predominantly used as adjunctive therapy to ongoing antidepressants (82% of patients), aligns with regulatory approval conditions in Europe and North America, where esketamine is indicated as adjunctive treatment rather than monotherapy. This mirrors the design of pivotal trials (TRANSFORM, SUSTAIN) that evaluated esketamine plus antidepressant versus placebo plus antidepressant, and reflects typical real-world implementation, in which esketamine is added to inadequate-response regimens rather than replacing existing treatments. Despite these difficulties, esketamine was associated with sustained clinical improvement in patients who continued treatment as shown by a decrease from a 40.0 baseline MADRS score to around 9–10 after 1 year, with the therapeutic effect maintained for a maximum of 5 years. Moreover, it is noteworthy that the mean MADRS score at baseline was 40, whereas in most clinical trials it is approximately 35. This likely reflects the fact that, in our real-world setting, esketamine was preferentially selected for patients presenting with particularly severe depressive symptoms.
A critical consideration for interpreting our findings is that the observed clinical improvements occurred in the context of polypharmacy, with 82.2% of patients receiving concurrent antidepressants and 60%–64% receiving other psychotropic medications (mood stabilizers, antipsychotics). This treatment context has several important implications. First, observed improvements reflect the combined effect of the entire treatment regimen rather than esketamine in isolation, and without a control group receiving identical concurrent treatments, we cannot determine the specific contribution of esketamine to outcomes. Second, our treatment approach aligns with both regulatory approval (esketamine as adjunctive therapy to antidepressants) and real-world clinical practice, making our findings generalizable to typical implementation contexts. Third, this confounding underscores why our purely descriptive approach—rather than causal or efficacy claims—is methodologically appropriate. We characterize real-world outcomes when esketamine is added to existing treatment regimens, providing clinically useful prognostic information, but we cannot establish esketamine-specific efficacy.
The high rate of concurrent psychotropic medications also reflects the clinical complexity and treatment resistance of our cohort. Many patients required multiple agents to manage severe depression, comorbid anxiety or personality disorders, previous partial responses, or medication-related side effects. This polypharmacy, while complicating attribution of treatment effects, accurately represents the patient population most likely to receive esketamine in routine clinical practice: those with complex, treatment-resistant presentations requiring multifaceted pharmacological approaches.
Methodological considerations
Our purely descriptive analytical approach, while limiting inferential claims, was chosen deliberately given the methodological challenges inherent in this preliminary investigation. Standard inferential tests (e.g., paired
The observed heteroskedasticity, floor effects, and non-normal distributions at later time points further violate assumptions of parametric tests, potentially leading to biased estimates and invalid inferences.32,33
More sophisticated approaches—such as mixed-effects models or Bayesian hierarchical models incorporating item response theory (IRT) to model scale-level measurement characteristics 34 —would be needed for confirmatory analyses. We are developing Bayesian IRT models to properly handle ordinal scale properties, longitudinal dependencies, and missing data; these will be presented in future work.
For this preliminary report, transparent descriptive presentation allows examination of individual trajectory heterogeneity that would be obscured by population-averaged effects. 35 Our visualization revealed substantial individual variation in response patterns—with some patients showing rapid improvement while others demonstrated gradual changes—underscoring the importance of personalized treatment approaches. 36 These individual differences, while complicating group-level statistical inference, provide clinically meaningful information for treatment individualization that aggregate analyses might mask.
Our findings must be interpreted within the context of mixed and debated evidence from randomized controlled trials. The failure of two of three TRANSFORM trials to achieve statistical significance,15,17 combined with questions about the clinical meaningfulness of statistically significant findings in TRANSFORM-2,25,37 indicates that esketamine’s efficacy profile remains uncertain. Our observational data cannot resolve this uncertainty, as we lack a control group and cannot isolate esketamine effects from concurrent treatments (82% receiving antidepressants). However, our findings provide complementary real-world evidence that, in routine clinical practice where esketamine is added to existing regimens in complex treatment-resistant patients, 82% of patients continue treatment with observed clinical improvements over extended follow-up. This pragmatic effectiveness information—while not establishing esketamine-specific efficacy—may inform shared decision-making about treatment trials in similar patient populations.
Strengths
Strengths of this study include the naturalistic clinical cohort characterized by a high burden of psychiatric and medical comorbidities, capturing patients who are frequently excluded from randomized controlled trials. This enhances the ecological validity of the findings and provides clinically relevant insights into the real-world implementation of intranasal esketamine in treatment-resistant depression.
A major strength is the extended follow-up duration of up to 5 years, which allows characterization of long-term treatment patterns, dosing evolution, and clinical outcomes that remain largely unexplored in the existing literature. Unlike controlled trials with fixed protocols and limited observation windows, this study reflects routine clinical decision-making, including individualized maintenance strategies over time.
The use of a person-time (person-weeks) analytical framework represents an additional methodological strength, enabling an accurate description of treatment exposure while accounting for heterogeneous follow-up durations resulting from rolling recruitment. Furthermore, the visualization of individual symptom trajectories alongside group-level descriptive statistics allows detailed characterization of response heterogeneity. This approach highlights clinically meaningful differences in response timing and durability that would be obscured by analyses focused solely on mean effects.
Together, these strengths provide complementary real-world evidence on the long-term use of intranasal esketamine in complex patients with treatment-resistant depression and offer a valuable foundation for future hypothesis-driven and inferential investigations.
Limitations
Several important limitations warrant acknowledgment. First, while the rolling recruitment design reflects real-world clinical practice, treatment discontinuation introduces survivor bias. Although the discontinuation rate was relatively modest (17.8%), patients who discontinued treatment due to lack of efficacy or intolerable adverse effects likely experienced less favorable outcomes than those who continued treatment. As outcomes after treatment discontinuation were not systematically assessed, the present findings primarily characterize outcomes among treatment continuers (82.2% of initiators) rather than providing population-level intent-to-treat effectiveness estimates. This may result in overestimation of clinical benefit and underestimation of adverse effects.
Second, heterogeneity in follow-up duration resulting from rolling recruitment limits the interpretability and generalizability of long-term outcomes. Data from later time points (years 3–5) reflect a progressively smaller subset of early-enrolled patients, such that long-term estimates primarily represent treatment continuers rather than the full cohort.
Third, the single-center observational design and absence of a control group preclude causal inference regarding esketamine-specific efficacy. Esketamine was administered adjunctively with ongoing antidepressants and, in many cases, additional psychotropic medications, including mood stabilizers and antipsychotics. Consequently, observed clinical improvements reflect the combined effect of the entire treatment regimen rather than esketamine in isolation. It is not possible to disentangle the effects of esketamine from synergistic pharmacological interactions, delayed treatment effects, spontaneous symptom fluctuation, or increased clinical attention. This confounding is inherent to naturalistic observational designs and underscores the rationale for adopting a purely descriptive analytical approach in this study, which aims to characterize real-world outcomes rather than establish efficacy.
Fourth, safety assessment has important methodological limitations. The GASS, selected due to the high prevalence of concurrent antipsychotic use, does not adequately capture esketamine-specific adverse effects, such as dissociative symptoms, perceptual disturbances, and blood pressure changes. Validated dissociation measures, including the Clinician-Administered Dissociative States Scale, were not employed, and many adverse effects were monitored through clinical observation and spontaneous reporting rather than systematic elicitation using validated instruments. This incomplete safety characterization limits direct comparison with controlled trial safety profiles and underscores the need for esketamine-specific tolerability measures in future observational research.
Fifth, convenience sampling and clinician-directed treatment selection introduce potential selection bias. The atypical male predominance observed in the sample may reflect referral patterns, unmeasured clinical factors, or chance, and limits generalizability to broader treatment-resistant depression populations.
Finally, documentation of prior treatment adequacy has limitations. Therapeutic drug monitoring was not systematically performed, and information regarding prior treatment duration and adherence partially relied on patient self-report. As a result, some cases classified as treatment-resistant depression may have reflected suboptimal exposure to previous antidepressant trials.
Conclusion
This observational study offers several notable strengths, including its focus on a naturalistic cohort with high psychiatric and medical comorbidities and an extended follow-up period of up to 5 years. The exploratory, descriptive nature of our analysis serves a dual purpose: primarily to guide subsequent inferential approaches on this dataset, and to provide valuable preliminary clinical insights in an area where long-term naturalistic data are scarce. While not designed for hypothesis testing, our findings illuminate treatment patterns and outcomes in real-world settings that complement controlled trial data, capturing the complexity of clinical decision-making in routine practice, which may inform clinical considerations. Our descriptive analytical approach with visualization of individual trajectories reveals response heterogeneity and temporal patterns that would be obscured by aggregate analyses alone, highlighting methodological limitations in the existing literature where mean-based analyses predominate.
Our exploratory findings suggest that more sophisticated methodological approaches may be necessary to fully capture and model the complex patterns observed in long-term psychiatric treatment data. Future research would benefit from larger prospective studies with matched control groups, pre-specified analytical plans that account for the distributional properties of psychiatric rating scales, and statistical approaches that can accommodate both group-level trends and individual response trajectories.
Footnotes
Acknowledgements
This work has received support from the European Union’s Horizon Europe research and innovation programme under grant agreement No. 101095436 (OPADE). The OPADE study is registered at ClinicalTrials.gov (NCT06550037). The published OPADE protocol was also considered as relevant background for the present work. Funded by the European Union. Views and opinions expressed are however those of the authors only and do not necessarily reflect those of the European Union. Neither the European Union nor the granting authority can be held responsible for them.
