Abstract
Background:
Chronic cognitive overload strains working memory and activates the body’s stress response via the hypothalamic–pituitary–adrenocortical axis, potentially causing stress-related health issues. Over-the-counter herbals, including lemon balm extract, are gaining popularity for stress management, possibly due to their lack of side effects.
Objectives:
To examine whether Zensera™, a lemon balm extract, produced mood and cognitive improvements throughout the day, 106 young healthy adults, with moderate stress complaints, were tested to compare the effects of 300 mg Zensera™ to a matched placebo during periods of cognitive overload.
Design:
An acute, parallel groups, randomised, double-blind, placebo-controlled design was implemented with cognitive function, mood, heart rate and blood pressure measured at baseline and at 1, 3 and 5 h post-treatment.
Methods:
Overall subjective calmness (primary aim) and fatigue (secondary) were captured at the beginning and end of the test day. Other transient mood measures were captured throughout the day, before and after high cognitive demand sessions.
Results:
For the primary outcome, no changes in overall calmness emerged. Zensera™ elicited improved performance on the hardest executive function trials at 5 h, and multiple regression confirmed that increased performance on the hardest attention network tasks was predicted by increased calmness and contentment at 5 h. Finally, Zensera™ helped restore feelings of transient calmness following the impact of cognitive demand, which was not seen for the placebo.
Conclusion:
Overall, these findings provide evidence for acute protective effects of Zensera™ in stressed adults, with benefits for challenging executive function tasks and enhanced calmness recovery following cognitive effort. Further research exploring the impact of Zensera™ for chronic stress is warranted.
Trial registration:
The study was registered on clinicaltrials.gov under the code NCT06183372: https://clinicaltrials.gov/study/NCT06183372) where the trial protocol and statistical analysis plan can be accessed.
Introduction
Cognitive overload occurs when the mental resources needed exceed task demands, causing stress and decreased performance.1,2 In such high-stress situations, the ability to remain calm, think clearly and maintain focused attention is crucial for effective decision-making. Over time, if cognitive demand remains elevated, it can diminish an individual’s ability to cope with stress, reduce motivation and impair overall functioning, potentially leading to long-term emotional and physical health problems.3–5 Mechanistically, the hypothalamic–pituitary–adrenocortical axis plays a critical role in the response to stress caused by the high demands of information retrieval on the working memory system.6–8 Overactivation of the hypothalamic–pituitary–adrenocortical axis is associated with stress-related health problems 9 , and it has become of increasing interest to address mild-to-moderate stress before it progresses to chronic levels.10–12 Side effects, such as drowsiness, are typically associated with pharmacological interventions, 13 and therefore, alternative remedies have gained increasing interest.14,15
Lemon balm (Melissa officinalis L.) contains a range of compounds, including polyphenols such as rosmarinic acid, essential oils and triterpenes.16–19 Numerous health benefits have been associated with lemon balm ingestion such as reducing anxiety and enhancing sleep and mood. Lemon balm acts on several neurochemical circuits, including GABAergic17,20–22 and cholinergic23–25 pathways, as well as having an indirect action on the hypothalamic–pituitary–adrenocortical axis via actions such as cortisol modulation.26–28 All of these pathways are associated with supporting a calm state, psychologically and/or physiologically. 29 Lemon balm appears safe and tolerable, given that vulnerable populations, including infants and hospitalised patients, report no side effects when administered up to 5000 mg daily for 20 days.30–33
Given the interaction between lemon balm and the hypothalamic–pituitary–adrenocortical axis, there is potential for lemon balm to benefit mood states during cognitive overload. For example, given that cognitive performance can worsen under acute stress,34–36 enhancing calmness may benefit cognitive performance. Several acute studies have investigated the effects of lemon balm supplementation on mood during cognitive tasks (see review, Mathews et al. 37 ). These studies measured the impact of lemon balm at various time points (from 1 up to 6 h following supplementation) and different doses (from 300 up to 1600 mg).38,39 Results from healthy adults without stress complaints were mixed, with no consistent effects on cognitive performance. In a review of this literature, Mathews et al. 37 observed that the effect of lemon balm on mood varied depending on the degree of cognitive stress. For example, in non-stressful situations, doses of 300 and 900 mg improved calmness, 38 but under more fatiguing conditions, which might be perceived as more stressful, only doses of 600 mg ameliorated negative mood effects; effects that were not seen when 300 mg was administered. 40 The absence of effects in this latter study could be because the participants were not in a general state of subjective stress. Recent reviews further acknowledge that it is unclear how lemon balm might buffer against stress or prevent stress-related cognitive impairment;37,41 however, it is notable that lemon balm may modulate GABAergic activity, which is associated with calmness and sleep benefits.17,42
In summary, there are some plausible mechanisms by which lemon balm may improve mood states, especially calmness, in a stressful environment such as during cognitive overload. However, the effect of lemon balm on mood outcomes and cognition during a state of cognitive overload, specifically in a moderately stressed population has not previously been investigated. Therefore, this study investigated whether Zensera™ (Givaudan Naturals, France), a lemon balm extract, can buffer against feelings of stress by inducing calmness during periods of cognitive demand in adults (18–40 years) with moderate subjective stress complaints. It was hypothesised that Zensera™ would be associated with improved levels of calmness and cognitive performance, with these effects hypothesised to be strongest at the 5 h timepoint when cognitive demand would be greatest due to general fatigue. A secondary aim was to explore whether changes in particular mood states were associated with cognitive performance.
Method
Study design
An acute, parallel groups, randomised, double-blind, placebo-controlled design was implemented. Cognitive function, mood, heart rate and blood pressure were measured at baseline (0 h, prior to treatment), and at 1, 3 and 5 h post treatment. The reporting of this study conforms to the CONSORT guidelines 43 (Supplemental Table S1).
Participants
One hundred and thirty participants aged 18–40 were recruited via opportunistic sampling between December 2023 and July 2024. GPower v3.1 was used to determine sample size, assuming a medium effect size of 0.5 for a mood effect39,40 indicated that 65 participants per intervention group were needed to detect a significant difference between groups with an alpha of 0.05 and 80% statistical power. Inclusion criteria required a Perceived Stress Scale score between 14 and 26, indicating moderate stress problems. 44 Exclusion criteria were any diagnosed disease or illness, including psychiatric or neurological conditions, consumption of any medication, consumption of any nutritional supplements not able to stop within 2 weeks of study commencement, and participation in any cognitive trial within the previous 3 months. A full list of the inclusion and exclusion criteria can be found in Supplemental Table S2. All participants were required to give informed consent before taking part.
Treatment
Participants were randomly allocated to the Zensera™ (lemon balm extract) or placebo using an established randomisation procedure. An independent researcher not involved in recruitment or data collection used a computerised random blocked randomisation. This consisted of random permuted blocks of equally balanced males and females weighted across Zensera™ and placebo conditions. Furthermore, this double blinding was maintained by the manufacturer (Givaudan Naturals), providing pre-labelled bottles. Both researchers conducting the study and participants were blinded until the end of the study period following completion of analysis. The Zensera™ and placebo capsules were identical in appearance and matched for weight containing 300 mg powder. The placebo contained 300 mg maltodextrin. A full composition of the active treatment and placebo is listed in Supplemental Tables S3 and S4.
Outcome measures
Mood measures
Subjective measures of overall calmness and mental fatigue were assessed using a nine-point Likert scale administered at the start and end of the test day. Transient measures of mood (Bond-Lader and Stress Visual Analogue Scale) were administered immediately before and immediately after each cognitive battery (0 h (baseline), 1, 3, and 5 h). The 16-item Bond-Lader Visual Analogue Scale (Bond-Lader 45 ) measured subjective calmness, alertness, and contentedness using 100 mm visual analogue scales anchored by various antonyms (e.g. Alert-Drowsy, Lethargic-Energetic) and then clustered into respective mood factors of calmness (average of 2 items), alertness (average of 9 items), contentedness (average of 5 items). Additionally, unlike the original Bond Lader scoring system where lower scores indicated improvement, here, all positive items were reverse-scored to reflect higher levels of calmness, alertness and contentedness. Participants also completed a further four Stress Visual Analogue Scales 46 that required them to rate their current levels of stress, anxiety, calmness and relaxation between ‘not at all’ and ‘extremely’ on a 100 mm line. Individual Stress Visual Analogue Scales were measured and then also combined into two scores, ‘stress/anxiety’ and ‘calm/relaxed’ with a higher score (average % along the line) representing more of the descriptor. Subjective Satisfaction 47 was measured using a two-part 4-item questionnaire rating participants’ satisfaction with their level of tiredness, mental overload and overall satisfaction from ‘not satisfied at all’ to ‘entirely satisfied’. The first part of the Subjective Satisfaction was utilised at the start of each cognitive battery (0, 1, 3 and 5 h) and asked how participants felt in the present moment whilst the second part of the Subjective Satisfaction was utilised following the intervention (prior to start of 1, 3 and 5 h cognitive battery) assessed their level of satisfaction following the treatment that had been administered.
Cognitive measures
E-Prime 3.0 48 running on a PC with a 15” screen was used to display the stimuli and record participant responses. Cognitive function was assessed with a computerised battery of tests administered in a fixed order and lasting approximately 50 min including the auditory verbal learning test, task switching task, modified attention network test, rapid visual information processing task, and paced auditory serial addition task.
The auditory verbal learning test provided a measure of episodic memory. Participants were required to listen to a recording of 15 words (list A) followed by a 2-min period to orally recall as many words as possible. An interference list (list B) was introduced after five recalls of List A, and participants were asked to recall this list only once. There was then a further free recall of list A (Recall 6) followed by a 10-min delay and then a final free recall of list A (Recall 7). Finally, participants were shown a list of 50 nouns, containing all the words from lists A and B plus an additional 20 filler words, and asked to identify only the words from list A. The full auditory verbal learning test procedure is outlined in Bell et al. 49 Dependent variables calculated were immediate recall (out of 15 words from first performance of List A), delayed recall (out of 15 words), total recall (out of 75 words), word learning (List A recall 5–List A recall 1) and word recognition (number of correct yes responses and no responses).
The task switching task and attention network test measured executive function. In the task switching task, participants viewed a circle divided into eight equal segments, with a bold horizontal line splitting the circle into top and bottom halves. Numbers 1–9 (excluding 5) were randomly presented in a clockwise direction in each segment for 3000 ms with an inter-stimulus interval of 1000 ms for 64 trials. For trials in the bottom half of the circle, participants were required to identify if the number was higher or lower than 5; whilst for trials in the top half of the circle, participants were required to identify if the number was odd or even. These tasks switched every four trials, with the switch sequence denoted as S1, S2, S3 and S4. Measures of accuracy and response time for correct responses (ms) for task and switch position were calculated (see Miller et al. 50 ). For the attention network test, participants respond to the direction of a centrally presented arrowhead by pressing the corresponding left and right arrow keys. Across multiple trials, the target stimulus was either flanked by arrows pointing in the same direction (congruent) or the opposite direction (incongruent). The number of flanking arrows also varied between trials (load). Two blocks of 40 trials were completed, on one of the two blocks to add additional cognitive load, participants were additionally required to perform a serial 3 s subtraction task where they orally counted backwards from a random three-digit number. The serial 3 performance was introduced to Attention Network Test. Accuracy and reaction time for the different trial types of congruency (congruent, incongruent, none), load (high, medium, low) and block (with & without concurrent serial 3 performance) were calculated.
Rapid Visual Information Processing Task measured sustained attention and numbers between 0 and 9 appearing sequentially in the centre of a computer screen. Each number was presented for 800 ms and participants were required to detect sequences of three odd or three even numbers by pressing the spacebar on the keyboard when the last number of a target sequence appeared. The dependent variables were reaction time and correct responses. The paced auditory serial addition task measured attention and memory.51,52 Here, the participant is auditorily presented with a single digit every 2 s and is required to complete a mental calculation to add each new digit to the one immediately prior to it saying the answer aloud. Researchers were present during this task to provide an additional element of social evaluation, thereby increasing the participant’s stress response.53–55 Sixty trials were presented over a 10–15 min period with 15 trials per block (four blocks in total), and the dependent variables were the number of correct responses.
Anthropometric measures were taken at the end of each cognitive battery and included measures of heart rate, systolic and diastolic blood pressure taken in duplicate measurements. 56
Procedure
Screening
All participants completed an online screening session where a number of demographic details were captured (height and weight to allow calculation of body mass index) and exercise habits. Importantly, the Perceived Stress Scale was completed where a score of 14–26 (indicating moderate stress levels) was required for entry to the study and randomisation to treatment. Participants were then invited to attend the laboratory at the University of Reading on two occasions separated by 1 week and up to a maximum of 9 days between visits in the event of rescheduling; the familiarisation visit and the test day (Figure 1).
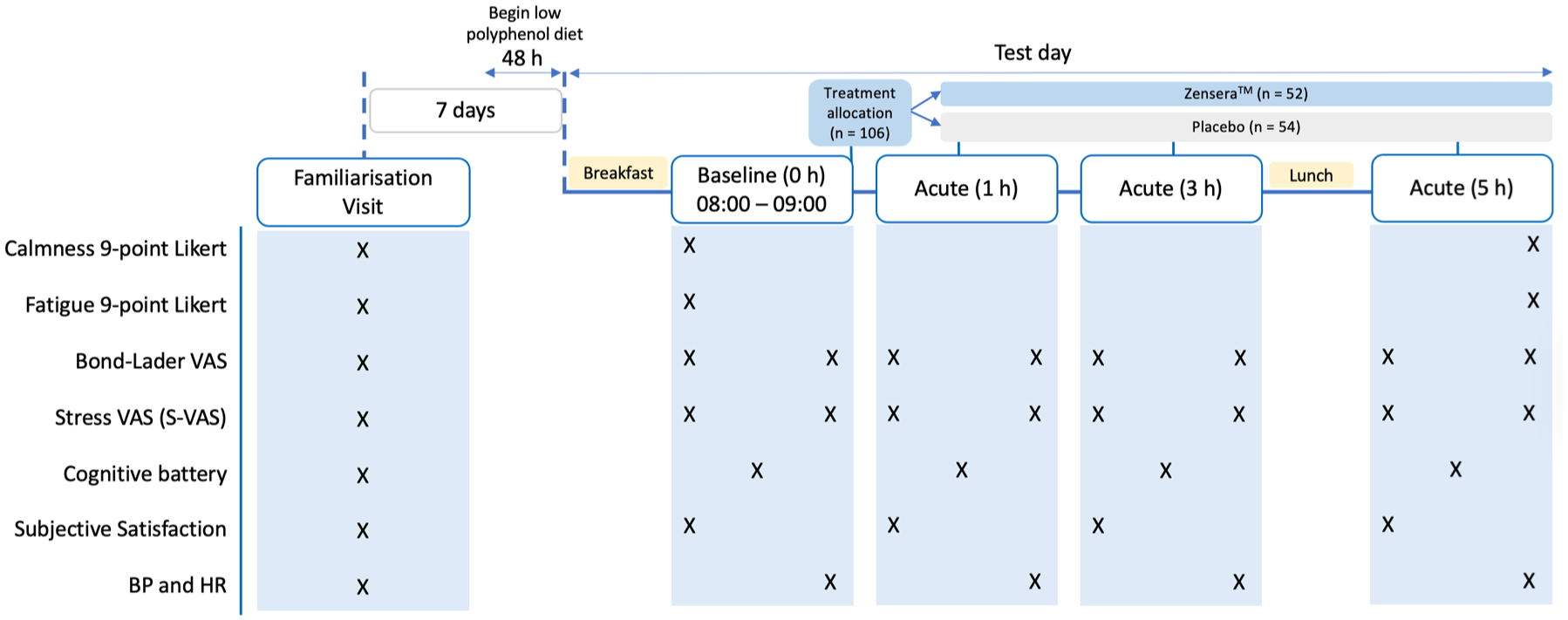
Lemcog study design and timeline of procedure on test day.
Familiarisation visit
Here, two full run-throughs of the task battery were completed to ensure that participants were habituated to the testing environment and procedures prior to starting the study test visits. Participants also had their height and weight measured to calculate Body Mass Index. In addition, participants completed the EPIC-Norfolk Food Frequency Questionnaire to record their habitual diet.
Test Visit
Given the known effects of other polyphenol-rich foods on cognitive performance, 57 48 h prior to each test visit, participants followed a low polyphenol diet requiring restriction of foods including fruits, vegetables, juices, chocolate, tea and coffee (caffeinated beverages were only restricted for 24 h). Compliance was recorded with a food diary. Participants were also required to be fasted for 12 h, as well as not consume any alcohol or perform exhaustive exercise within 24 h before the test visit. On arrival at the laboratory (at either 8 or 9 am), participants consumed a low polyphenol breakfast consisting of two croissants, consumed within 10 min. Following breakfast, participants completed their subjective ratings of overall calmness, mental fatigue, Bond Lader and Stress Visual Analogue Scales, Subjective Satisfaction part 1 (perception of satisfaction not associated with treatment) before completing the baseline cognitive battery. Bond Lader and Stress Visual Analogue Scale measures were collected again immediately after completing the cognitive tasks, alongside blood pressure measurements. The allocated treatment was consumed following completion of these baseline mood/cognitive measures. At 1, 3 and 5 h, participants completed a further battery of cognitive tasks, with Bond Lader, Stress Visual Analogue Scales collected before and after these tasks. In addition, both parts of Subjective Satisfaction (part 2 was in relation to perceived satisfaction of taking treatment) before 1, 3 and 5 h sessions. Finally, after completing the mood and cognitive task battery at 5 h, subjective ratings of overall calmness and mental fatigue were collected. A standardised lunch (cheese sandwich and crisps) was provided at the end of the 3 h cognitive battery. During all breaks, participants could leave the laboratory to resume normal daily activities except food and beverage consumption or any vigorous exercise, which was asked to be avoided.
Statistical analysis
A linear mixed model using an unstructured covariance matrix with Perceived Stress Scale as a covariate was applied. All data were analysed using R Studio v4 58 and using the lme4 package. 59 Session and Treatment Group were included as fixed factors in the model. Treatment included two levels (placebo and Zensera™), and Session included four levels (baseline (0 h), 1, 3 and 5 h), except for the stand-alone measures of overall calmness and mental fatigue for which there were only two sessions (before 0 h and after 5 h). For mood measures assessed pre- and post- the cognitive battery (Stress Visual Analogue Scale, Bond Lader, Subjective Satisfaction), a further fixed factor called Time was included with two levels (before and after the cognitive battery). Participants were included as a random factor as a way of controlling for non-independence of data from the same subjects. Post hoc pairwise comparisons were used to investigate any significant main effects and interactions of interest. A Bonferroni correction was applied to all multiple comparisons. Cohen’s d effect sizes with 95% CI (lower limit, upper limit) were also calculated using the emmeans package 60 for all significant pairwise comparisons between Zensera™ and placebo treatments. For all reaction time data, only correct responses were included. Responses faster than 100 ms were excluded as per previous studies. 49 For task switching task data responses slower than 3000 ms were excluded as recommended as per previous studies. 61 Z score analysis was used to identify outliers and values with an z score > 3.29 were removed prior analysis. 62 The assumptions for normality of each model were met (i.e. Q–Q plots of the residuals), despite some raw data violating normality, linear mixed models are generally robust to non-normality in the observed data. 63 Raw data from this project is available from the researchers upon reasonable request.
In addition, in light of the importance of high cognitive demand for the hypothesis, a secondary analysis of the cognitive data focused on the most difficult trials in each test, which were selected from the attention network test and task switching task. For the task switching task, S1 trials were considered the most difficult (these are the first segments of the circle where the task changes from high/low (below the line) and odd/even (above the line). For the attention network test, incongruent, high-load trials were considered the most difficult (which included arrows with the addition of oral serial 3’s). Finally, to investigate the association between mood state and cognitive outcomes, regressions were performed, separately for placebo and Zensera™, with cognitive performance at 5 h as the predicted variable and changes in mood state across the cognitive battery at 5 h as the predictors (Bond Lader calmness, Bond Lader alertness, Bond Lader contentedness, and stress visual analogue scales for calmness, relaxed, anxious and stressed scales). For this analysis, only the 5 h time point was chosen as this is the time point at which cognitive fatigue can be hypothesised to be at its greatest, and therefore, any cognitive benefits are most likely to be detected at this point. A similar rationale underpins only using performance data from the most difficult trials for the most challenging cognitive test in the battery (attention network test) for this regression analysis. Being selective for this analysis also reduces the number of exploratory regressions being undertaken, thus mitigating the risk of type 1 error.
Results
As shown in the CONSORT diagram in Figure 2, 106 participants completed the study. Participant demographic information is shown in Table 1. Paired t-tests identified a significant baseline difference in Perceived Stress Scale between groups, where the Zensera™ group had lower perceived stress compared to the placebo group (p = 0.037). As such, the Perceived Stress Scale was included as a covariate in all linear mixed models. Please note that Estimated Marginal Means with standard error and linear mixed model outputs (including F statistic and p values) for all outcome measures can be found in the Supplemental Tables S5 and S6. No other baseline differences between groups were identified. In addition, no adverse events were reported during the trial.

Consort diagram using CONSORT reported guidelines 43 of the participant sample at each stage of recruitment and participation in the Lemcog study.
Sample characteristics at enrolment.
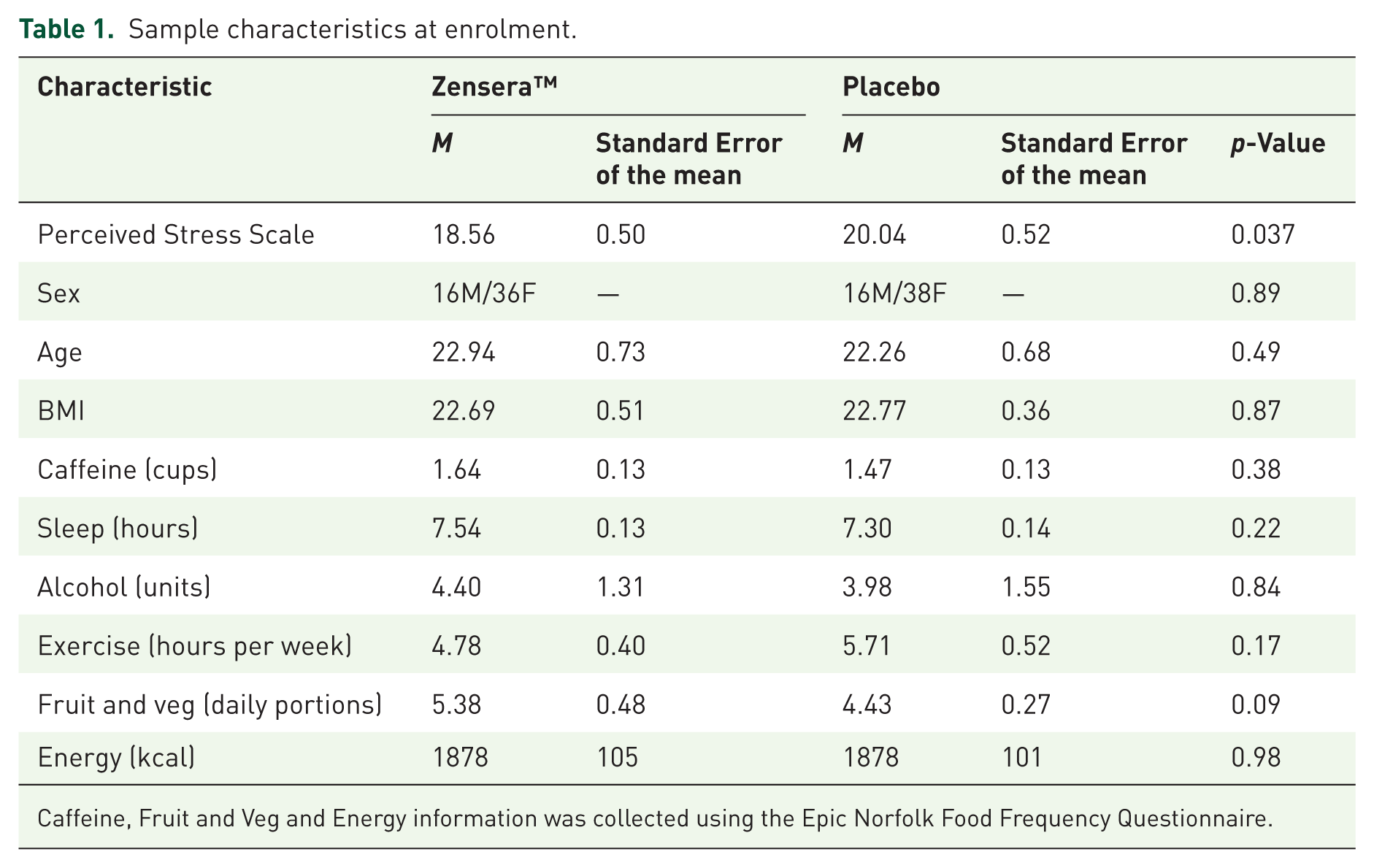
Caffeine, Fruit and Veg and Energy information was collected using the Epic Norfolk Food Frequency Questionnaire.
Primary outcome: Overall calmness
Contrary to the primary hypothesis, there were no treatment-related changes in calmness across the test day, and no Session × Treatment interactions.
Executive function
Regarding the Task Switching Task, Treatment (F[1,99.22] = 6.29, p = 0.013) and Session (F[3,296.81] = 10.1, p < .0001) significantly predicted overall task performance. Pairwise comparisons demonstrated significantly better performance for Zensera™ relative to placebo at 3 h (p = 0.014, d = 0.92, CI = 0.18, 1.66) and 5 h (p = 0.005, d = 1.07, CI = 0.32, 1.81), however, there was also a significant difference at baseline (p = 0.049, d = 0.74, CI = 0.002, 1.48). Despite this baseline difference, Zensera™ was also associated with an attenuation of the decline in performance across the test day, where performance only dropped relative to baseline at 5 h (p = 0.02, d = 0.6, CI = 0.019, 0.99). In contrast, performance in the placebo condition declined across the day, with significantly poorer performance compared to baseline at 3 h (p = 0.015, d = 0.6, CI = 0.21, 0.99) and 5 h (p < .0001, d = 0.92, CI = 0.52, 1.31), and poorer performance at 5 h relative to 1 h (p = 0.009, d = 0.63, CI = 0.24, 1.03), as shown in Figure 3. When considering performance on only the hardest trials (S1, switch trials), Session significantly predicted performance (F[3,707.10] = 19.81, p < 0.0001), whilst Treatment was trending (F[1,102.23] = 3.04, p = 0.08). Pairwise comparisons revealed that the Zensera™ group demonstrated better performance relative to placebo at 5 h (p = 0.026, d = 0.69, CI = 0.08, 1.29), as seen in Figure 4.

Total accuracy on switching task by Treatment and Session where a significant factor of Treatment (F[1,99.22] = 6.29, p = 0.013) and Session (F[3,296.81] = 10.1, p < 0.0001) where post hoc tests demonstrated higher performance in Zensera™ at 3 h (cp = 0.014) and 5 h (dp = 0.005) relative to placebo. A difference at 0 h was also evident (bp = 0.049). In addition, Zensera™ showed a decline in performance at 5 h relative to 0 h (*p = 0.02), whilst for the placebo group there was a significant decrease at 3 h (*p = 0.015) and 5 h (***p < 0.0001) relative to 0 h, and a decrease at 5 h relative to 1 h (**p = 0.009). Results are presented as estimated marginal means from respective linear mixed models (±SE), where letters represent between-group differences with a different letter indicating a significant difference between the Zensera™ and placebo. Within-group differences are represented by asterisks where a significant difference is indicated using *(p < 0.05), **(p < 0.01) and ***(p < 0.0001).

Hardest trials of executive function tasks by Treatment and Session. On task switching task switch trials (left panel), there was a significant effect of Session (F[3,707.10] = 19.81, p < 0.0001), and Treatment was trending (F[1,102.23] = 3.04, p = 0.08), showing higher performance in Zensera™ at 5 h relative to placebo (bp = 0.026). On the hardest trials of the attention network test (incongruent, high load, with serial 3’s) (right panel), there was a significant interaction for Treatment × Session (F[3,306.24] = 3.56, p = 0.014) with performance trending higher in Zensera™ at 5 h relative to placebo (bp = 0.053), and a significantly higher performance at 5 h relative to 0 h in the Zensera™ group (*p = 0.037). Results are presented as estimated marginal means from respective linear mixed models (±SE), where letters represent between-group differences, with a different letter indicating a significant or trending difference between the Zensera™ and placebo. Within-group differences are represented by asterisks, where a significant difference is indicated using *(p < 0.05).
Regarding the attention network test, analysis of the most cognitively demanding trials (high load, incongruent with the addition of Serial 3’s), showed a significant Treatment × Session interaction (F[3,306.24] = 3.56, p = 0.014). Pairwise comparisons revealed Zensera™ significantly improved in performance at 5 h compared to 0 h (p = 0.037, d = 0.54, CI = −0.93, −0.15), which was not seen for the placebo, and moreover, Zensera™ performance was trending as higher at 5 h compared to the placebo (p = 0.053, d = 0.6, −0.009, 1.19; Figure 4).
No significant effects of Treatment, Session or Treatment × Session interactions were seen for the auditory verbal learning test, rapid visual information processing task or paced auditory serial addition task.
Associations between changes in mood pre- to post-cognitive battery and attention network test performance on the most difficult trials at the 5 h session
As described in the analysis section, separate multiple regressions were carried out for Zensera™ and placebo to explore whether changes in mood predicted cognitive performance. Table 2 for the overall model for the placebo group was not significant (F(7,45) = 0.42, p = 0.88). Table 3 outlines the multiple regression findings for Zensera™, where the overall model was significant (F(7,43) = 2.96, p = 0.013), explaining 21% of the variance. A greater increase in Stress Visual Analogue Scale for calmness and Bond Lader contentedness after the battery at 5 h relative to before the battery was associated with better performance on attention network test hard trials (p = 0.002 and p = 0.02, respectively).
Changes in mood at 5 h as predictors of the hardest attention network test performance at 5 h for placebo.

Changes in mood at 5 h as predictors of the hardest attention network test performance at 5 h for Zensera™.

For p-Value’s the asterisks represent a significant difference where p < .05 (*) and p < .001 (**).
Secondary mood outcomes
Fatigue
As expected, participants showed significantly elevated ratings of fatigue at the end of the test day (Session; F[1,103.87] = 38.75, p < .0001) relative to 0 h. A trending Session × Treatment interaction was also detected (F[1,103.87] = 3.89, p = 0.051); however, post hoc tests did not reveal any significant differences between treatment groups.
Transient mood measures: Bond Lader and Stress Visual Analogue Scales
All p-values from the linear mixed model outcomes for Bond Lader and Stress Visual Analogue Scales are reported in Table 4. As expected, cognitive demand had a negative impact on mood as shown by significant reductions in contentedness, alertness, feeling relaxed and both measures of calmness, alongside increased feelings of anxiousness and stress after the cognitive battery relative to before.
Significance values from the 2 × 2 × 4 linear mixed model (time × treatment × session).

For p-Value’s the asterisks represent a significant difference where a trend (#0.056 and 0.054), p < .05 (*), p < .001 (**), and p < .0001 (***).
Additionally, a trending Time × Treatment interaction for Stress Visual Analogue Scale for calmness (F[1,714.35] = 3.65, p = 0.056) indicated that calmness was higher for Zensera™ relative to placebo immediately before the 1, 3 and 5 h batteries as shown in Figure 5 (although the post hocs were not significant). Given the reductions in calmness caused by the cognitive battery, this can be interpreted as Zensera™ enhancing the recovery in calmness prior to the next battery, which was not seen for placebo, see Figure 5 (although the after-to-before post-hoc tests were not significant).

Stress Visual Analogue Scale for calmness was measured before and after the cognitive battery by Treatment, Time and Session. A trending Time by Treatment interaction (F[1,714.35] = 3.65, p = 0.056) revealed Zensera™ was associated with greater calmness before the 1, 3 and 5 h batteries. Results are presented as estimated marginal means from respective linear mixed models (±SE).
Physiological measures
Blood pressure and heart rate
As shown in Figure 6, the placebo group had a significant increase in diastolic blood pressure between 0 and 5 h (p = 0.006, d = −0.7, CI = −1.12, −0.28) as supported by a significant Session × Treatment interaction (F[3,261.21] = 3.02, p = 0.03). Furthermore, diastolic blood pressure was trending lower in the Zensera™ group compared to the placebo (p = 0.059, d = −0.65, CI = −1.33, 0.03) at 5 h. Regardless of treatment, both groups demonstrated higher systolic blood pressure at the 5 h session relative to 0 h (Zensera™ p = 0.004, d = −0.75, CI = −1.18, −0.31; and placebo p = 0.016, d = -0.64, CI = −1.06, −0.22). Heart rate was found to drop at 1 h (Zensera™ p < .0001, d = 1.41, CI = 0.97, 1.85; placebo p < .0001, d = 1.26, CI = 0.83, 1.69) and 3 h (Zensera™ p < .0001, d = 1.49, CI = 1.05, 1.94; placebo p < .0001, d = 1.62, CI = 1.2, 2.06) compared to 0 h, regardless of treatment.
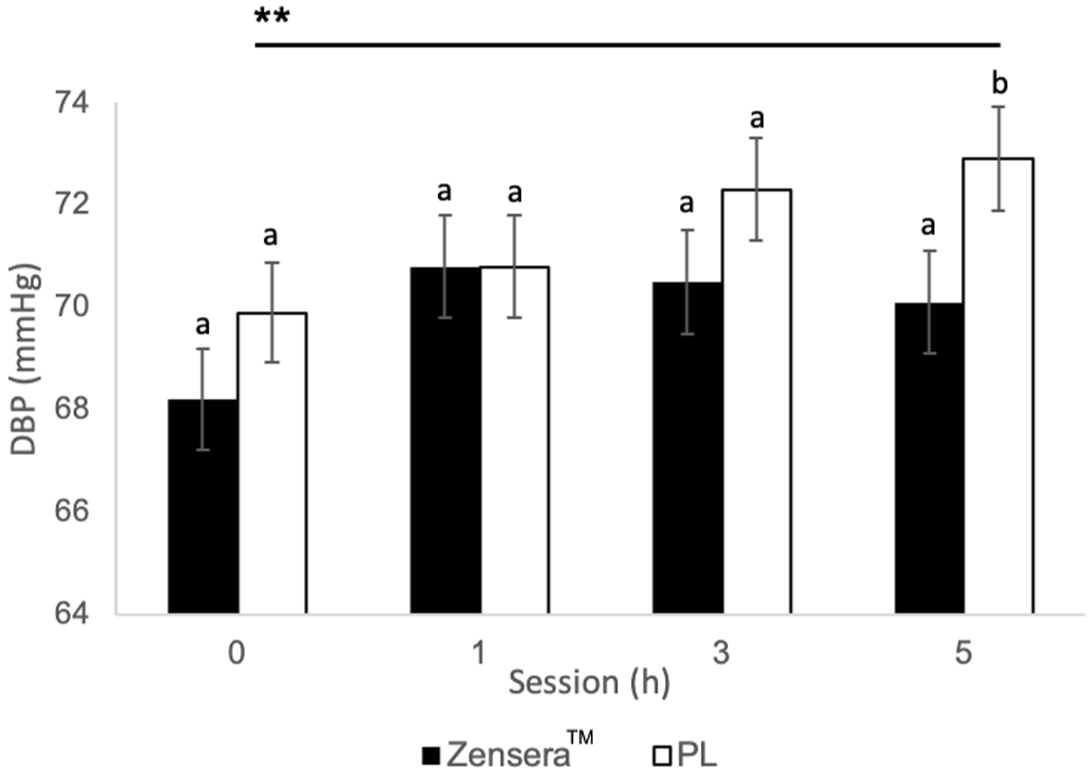
Diastolic blood pressure after the cognitive battery by Treatment and Session. Results revealed a significant effect of Session × Treatment (F[3,261.21] = 3.02, p = 0.03) showing increases in diastolic blood pressure was observed following placebo between 0 h and 5 h (**p = 0.006), and although no main effect of treatment, Zensera™ had a trend for lower diastolic blood pressure at 5 h relative to placebo (bp = 0.059). Results are presented as estimated marginal means from respective linear mixed models (±SE) where letters represent between-group differences, with a different letter indicating a trending difference between the Zensera™ and placebo. Within-group differences are represented by asterisks where a significant difference is indicated using **(p < 0.01).
Discussion
In the current study, a single dose of 300 mg Zensera™ was primarily investigated for its calming effects across the day during cognitive overload in healthy adults with moderate stress complaints. However, no changes in our primary outcome measure of overall calmness were observed. Interestingly, Zensera™-treated participants performed better relative to placebo on the hardest executive function trials of the attention network test and task switching task during the final cognitive battery of the day, and this may be mediated by Zensera™-induced increases in transient contentedness and calmness, notably a different measure to the primary outcome measure of overall calmness. Furthermore, there was tentative evidence for protective mood effects of Zensera™ indicated by a greater recovery in calmness between the cognitive batteries relative to placebo. Finally, Zensera™ also appeared to have cardiovascular benefits as shown by an attenuation of the increase in diastolic blood pressure shown at 5 h following placebo.
The primary purpose of including the cognitive task battery in the present study was to induce cognitive overload, and benefits to cognitive function were not necessarily expected, given mixed findings in the existing literature (see reviews, Mathews et al. 37 and Oliveira et al. 64 ). However, Zensera™ was found to improve performance on the most cognitively demanding executive function trials in both the attention network task (incongruent, high load trials with concurrent serial 3s) and the switching task (switch trials). Whilst previous studies have not reported benefits to executive function, 300 mg dose of lemon balm has previously been associated with improved mental arithmetic and speed of maths processing.28,40 Our results, therefore, may be expected given the importance of executive function skills of working memory, inhibition and shifting in the development of mathematics proficiency. 65 It may be that the systematic increase in cognitive load offered in these executive function tasks, particularly the attention network task with serial 3’s, allows for greater sensitivity to the effect of Zensera™. This finding also aligns with the wider nutrition cognition literature, where beneficial effects of phytonutrients are typically apparent under conditions of high cognitive load.66–68
The present study also adds to the growing volume of research demonstrating a calming effect of lemon balm and highlights that the timing of mood measurements, particularly in relation to cognitive overload, may be crucial in assessing Zensera™ efficacy. For example, effects may have gone undetected without the inclusion of mood assessments immediately before and after each cognitive battery. In addition to timing, the scale used for measuring calmness may also be important, as different results were found across the three measures of calmness utilised in the present study (overall calmness, Stress Visual Analogue Scale, Bond Lader). Given that different scales were used to measure calmness at different points of the test day in the present study, it is not possible to directly compare these measures in this context; however, these findings indicate the importance of identifying the most suitable measure of calmness at the outset, depending on the aims and hypotheses, which is likely to impact interpretations.
In contrast to much of the existing literature, the present study reports concurrent improvements in cognitive function following Zensera™ supplementation. Previous acute studies have reported poorer executive function 3 and 6 h post-intervention, 69 or improvements in both cognitive and mood outcomes but each at different doses and/or timepoints,28,40 making the previous findings challenging to interpret. In contrast, the present study illustrates greater performance on cognitively demanding executive function trials at 5 h compared to the placebo, and moreover, feelings of calmness and contentedness were associated with increases in cognitive performance for Zensera™ only, at 5 h. The data also suggest that Zensera™-induced mood effects are important in explaining the present cognitive benefits. At 5 h, the regression analysis showed that increased calmness and contentedness predicted better performance. As such, the present findings suggest that a 300 mg dose of Zensera™ may enhance cognitive performance under conditions of cognitive overload by facilitating a calm but focused mood state required for optimum cognitive function and by supporting a greater recovery in calmness during periods of rest between demanding tasks. In addition, it was also interesting to observe cardiovascular benefits. Diastolic blood pressure increased in the placebo group at 5 h, but not in the Zensera™ group. Both groups showed higher systolic blood pressure, likely due to stress and cognitive demand. Improvements in blood pressure with Zensera™ may require longer duration, higher doses, and populations with health issues, as seen in studies with type 2 diabetes (1000 mg for 12 weeks 70 ) and chronic angina (3000 mg for 8 weeks 71 ).
Limitations
Whilst the present trial has notable strengths including a robust randomised, placebo-controlled design, which controlled for polyphenol intake and included challenging cognitive tasks that did not produce ceiling effects, the current study is not without limitations. First, biochemical samples were not collected, and, therefore, the bioavailability of Zensera™ cannot be confirmed nor can potential mechanisms of action be determined. However, since participants consumed the acute intervention whilst in the presence of the researchers, we can infer that circulating components like rosmarinic acid were present, given that a known peak occurs 1 h post-absorption.28,72 Interestingly, we observed effects at 5 h, when bioavailability of rosmarinic acid in lemon balm has been shown to drop from its peak at 1 h. 72 This suggests that the observed effects may not depend solely on rosmarinic acid and that other components in this Zensera™ may have a prolonged impact on cognition and mood. Other lemon balm studies have also reported lasting effects up to 6 h after supplementation, though this may be attributed to the addition of valerian. 69 Whilst bioavailability data for lemon balm are limited, rosmarinic acid from spearmint was shown to be bioavailable in adults at 4 h. 73 Additionally, it is worth noting that we did not fully meet our recruitment target, whilst 130 participants were enrolled, only 106 participants fully completed the trial. Whilst our study sample size was relatively large, there was a predominance of female participants, and although there are no available studies for sex differences in humans, the efficacy of lemon balm as an anxiolytic has been shown to be gender dependent in rats, 74 and therefore sex may interact with the efficacy of lemon balm and undermine the generalisability of the findings. Finally, we must acknowledge that stress tests induced in a laboratory setting may not accurately reflect real-life stress, so the ecological validity of simulating cognitive overload should be interpreted with caution.
Conclusion
The present work supports the promising effects of Zensera™ on executive function later in the day during peak cognitive overload. A single dose of 300 mg Zensera™ was sufficient to induce beneficial acute effects for mood and cognitive measures. Zensera™ may help individuals under moderate stress perform better and feel better during challenging executive function tasks, particularly later in the day. Future studies should focus on the biological and psychological mechanisms involved in these effects, which will be important for harnessing the therapeutic benefits of lemon balm extract for cognition and mood.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253261415706 – Supplemental material for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress
Supplemental material, sj-docx-1-tpp-10.1177_20451253261415706 for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress by Imogen Mathews, Jessica Eastwood, Lynne Bell, Daniel Lamport, Romain Le Cozannet, Pascale Fanca-Berthon and Claire Williams in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253261415706 – Supplemental material for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress
Supplemental material, sj-docx-2-tpp-10.1177_20451253261415706 for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress by Imogen Mathews, Jessica Eastwood, Lynne Bell, Daniel Lamport, Romain Le Cozannet, Pascale Fanca-Berthon and Claire Williams in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-3-tpp-10.1177_20451253261415706 – Supplemental material for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress
Supplemental material, sj-docx-3-tpp-10.1177_20451253261415706 for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress by Imogen Mathews, Jessica Eastwood, Lynne Bell, Daniel Lamport, Romain Le Cozannet, Pascale Fanca-Berthon and Claire Williams in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-4-tpp-10.1177_20451253261415706 – Supplemental material for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress
Supplemental material, sj-docx-4-tpp-10.1177_20451253261415706 for The acute effects of Zensera™ (Melissa officinalis L.) extract on mood and cognitive performance during cognitive overload: a randomised placebo-controlled, double-blind study in healthy young adults with moderate subjective stress by Imogen Mathews, Jessica Eastwood, Lynne Bell, Daniel Lamport, Romain Le Cozannet, Pascale Fanca-Berthon and Claire Williams in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
