Abstract
Antidepressants are commonly prescribed for children and adolescents, but their discontinuation may result in withdrawal symptoms, though rarely described for this age group in the literature. Systematically examining the types and prevalence of withdrawal syndromes in children and adolescents in accordance with PRISMA guidelines is the aim of the present paper. A search of PubMed, Embase, Web of Science, and Cochrane Library was conducted from inception to December 2024. Inclusion criteria were: clinical populations; subjects aged 2–18; antidepressant discontinuation; assessment of withdrawal symptoms; randomized controlled trials (RCTs) or observational studies; rate/type of withdrawal symptoms. A total of 6227 citations were screened, leading to 12 studies, encompassing 690 participants aged 2–18 years, with eight RCTs and four observational studies. The analysis revealed common withdrawal symptoms across various antidepressant classes, predominantly affecting the central nervous and gastrointestinal systems. Symptoms such as nausea (14.6%–22.0%), headache (14.6%–15.0%), diarrhea (10.0%–22.0%), depression (10%), inner tension (44%), anxiety/worry (44%), and cough (33%) were frequently observed, especially after discontinuation of selective serotonin reuptake inhibitors. Such symptoms clustered in new withdrawal symptoms according to a diagnostic classification of withdrawal syndromes at discontinuation of antidepressants used in adults. Withdrawal symptoms and withdrawal syndromes following antidepressant discontinuation in children and adolescents are still neglected but pose significant clinical challenges, often resembling or exacerbating underlying conditions. Future research is needed, as well as a systematic assessment of these symptoms in the clinical realm (registered in OSF DOI: https://doi.org/10.17605/OSF.IO/GYQ9Z).
Plain language summary
Introduction
Antidepressant medications are widely prescribed for treating mental disorders in children and adolescents. 1 Despite their widespread use, there is still ongoing debate and controversy surrounding their effectiveness and safety.2,3 In addition, withdrawal symptoms following discontinuation, decrease, or switch are a problem understudied and underestimated.4–9 Recent research showed that in adults, withdrawal symptoms are more prevalent than expected, potentially affecting up to 56% of patients who discontinue antidepressants. 10 No similar data is available for children and adolescents. Understanding the nature, prevalence, and management of antidepressant withdrawal symptoms in the young population has become increasingly important as prescription rates continue to rise. 1
Withdrawal syndromes encompass a range of physical and psychological symptoms that emerge following discontinuation, decrease, or switch of the drug.7,8,11 Cosci and Chouinard 7 identified three types of withdrawal syndromes associated with antidepressants for adults: new withdrawal symptoms, rebound symptoms, and persistent post-withdrawal disorders. New symptoms are novel symptoms to the patient, while rebound symptoms involve a rapid recurrence of the original symptoms, but more intense than before treatment. In contrast, persistent post-withdrawal disorders manifest as long-lasting, severe, potentially irreversible, potentially including new symptoms and/or rebound symptoms. This underscores the significance of considering the long-term effects of these medications, 12 in children and adolescents as well.
Since research has focused on adults,8,10,11,13 a significant gap in understanding of how these symptoms manifest in children and adolescents is evident.7,14 Identifying and managing withdrawal in younger individuals can be challenging, given the ambiguity in symptom communication and the heightened disruption of daily activities, including school attendance, social interactions, and family dynamics. 15 Moreover, distinguishing between withdrawal and relapse and identifying specific withdrawal syndromes is crucial in younger populations 16 as treatment approaches differ significantly depending on the underlying cause.7,8,11
Given these challenges and the increasing recognition of the importance of appropriate management of withdrawal symptoms, there is a clear need for a systematic review of the current evidence on withdrawal related to antidepressant discontinuation, decrease, or switch in children and adolescents. This is the aim of the present systematic review of the literature, which also made an attempt to apply the diagnostic criteria proposed for adults by Cosci and Chouinard 7 to illustrate withdrawal after decrease, discontinuation, or switch of antidepressants in children and adolescents.
Methods
The protocol was registered with the Open Science Framework (OSF registration DOI: https://doi.org/10.17605/OSF.IO/GYQ9Z), and the paper reported in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines.17,18
Eligible articles included English language papers published in peer-reviewed journals reporting data on clinical populations of children and/or adolescents (2–18 years) who decreased, discontinued, or switched antidepressant medications and in which assessment of withdrawal symptoms was performed. 19
We included studies of various designs: randomized controlled trials (RCTs), open investigations, retrospective studies, and observational studies. Studies with fewer than 10 subjects, conducted in nonclinical samples, or studies conducted in sample exploration from patients’ websites were excluded. Studies had to describe the rate and type of withdrawal symptoms.
The Cochrane Library, PubMed, Embase, and Web of Science were comprehensively searched from inception to December 2024. Search terms were “withdrawal/discontinuation/rebound/tapering” combined using the Boolean “AND” operator with “antidepressive agents/Selective Serotonin Reuptake Inhibitors/SSRI/citalopram/escitalopram/fluoxetine/fluvoxamine/paroxetine/sertraline/fluoxetine/dapoxetina/vortioxetine/vilazodone/zimeldine/Serotonin and Noradrenaline Reuptake Inhibitors/SNRI/venlafaxine/desvenlafaxine/duloxetine/levomilnacipran/milnacipran/nefazodone/ammuxetine/TCA/amineptin/amitriptyline/amoxapine/clomipramine/cyclobenzaprine/desipramine/desmethyldoxepin/dibenzepin/dothiepin/doxepin/imipramine/iprindole/lofepramine/melitracene/metapramine/nortriptyline/noxiptilin/opipramol/protriptyline/tianeptine/trimipramine/setiptiline/mianserine/mirtazapine/maprotiline/bupropion/agomelatine/esketamine/ketamine/trazodone/monoamine oxidase inhibitors/MAOI/chlorphenamidine/iproniazid/isocarboxazid/moclobemide/pargyline/phenelzine/selegiline/tranylcypromine” (Supplemental eAppendix A reports the search strategy used in PubMed with MeSH terms). A manual search of reference lists from all articles selected, for full-text reviews, and relevant reviews was done.
Data extraction and quality assessment
A standardized data extraction form was developed to extract relevant data which include reference, settings, primary diagnoses, classes of drugs, treatments, duration of the treatment, assessment tools, discontinuation method, and withdrawal symptoms. After data extraction, studies were grouped according to classes (e.g., SSRIs). The methodological quality of studies was assessed independently by JW and FC via the Joanna Biggs Institutes’ Critical Appraisal (JBI) tools (https://jbi.global/critical-appraisal-tools). For RCTs, the JBI considers 13 items in five dimensions: selection and allocation, administration of intervention/exposure, assessment, detection, and measurement of the outcome, participant retention, and statistical conclusion validity. For observational studies, the JBI evaluates quality based on 10 items across four dimensions: confounding, selection, and information bias, and the importance of clear reporting. Scores were calculated on a four-point Likert scale (“yes, no, unclear, not applicable”) with a maximum sum score of 13 or 10 (Munn et al., 2021). 20 The sum score was converted into a percentage; over 66% were considered low, between 44% and 65% as moderate, and < 44% as high risk of bias.
Titles and abstracts were screened by two reviewers (F.C. and J.W.). Articles appearing potentially relevant were retrieved, and the two reviewers independently assessed each of the full reports, arriving at a consensus regarding eligibility.
When information about methods or results was omitted, the corresponding author was contacted to obtain the missing information. In case of the suspect of duplicate publications, the authors of the reports were contacted to receive details, and only the publication with the largest sample size was retained.
Since new withdrawal symptoms have been described in the literature in several ways and using different words, a categorization previously used for antidepressant discontinuation symptoms was adopted.6,7,21 Based on the withdrawal classification proposed for adults by Chouinard, Chouinard, and Cosci,7,21 the unpleasant post-drug symptoms observed in the studies included in the present review were classified, encompassing general, cardiovascular, gastrointestinal, genitourinary, sensory-related, neuro-muscular, sexual, and central nervous system, further delineated as neurological, cognitive, affective, psychotic, behavioral, and sleep-related symptoms.
Results
A total of 6227 citations were identified, resulting in 252 unique items after deduplication and abstract review. Among these, 240 studies were excluded for not meeting the inclusion criteria. Thus, a total of 12 studies were included (see Figure 1), with a combined total of 690 participants, ranging in age from 2 to 18 years. The studies were eight RCTs, which substituted a placebo for an active drug, aiming at testing the necessity of treatment with the active drug to forestall relapse, and four observational studies. The studies involved various classes of antidepressants, including SSRIs (n = 6), Serotonin Noradrenaline Reuptake Inhibitors (SNRIs) (n = 1), and tricyclics (TCAs) (n = 1) across eight RCTs. In addition, three observational studies focused on TCAs (n = 2) and SSRIs (n = 1), while another study sourced data from the World Health Organization database. The studies assessed withdrawal symptoms following antidepressant discontinuation with follow-up duration up to 8 months via the use of tools standardized to collect side effects (e.g., Side-Effects Checklist (SEC); Common Adverse and Side Effects Scale (CASES); the electronic Columbia–Suicide Severity Rating Scale (C-SSRS); Serious adverse event (SAE)) or via general clinical inquiry. No studies used standardized tools specifically devoted to assessing withdrawal symptoms. Data on withdrawal symptoms were incidental findings. Regarding quality assessment, one observational study 30 showed poor quality. A summary of study characteristics, including sample size and JBI scores, is presented in Tables 1 and 2.
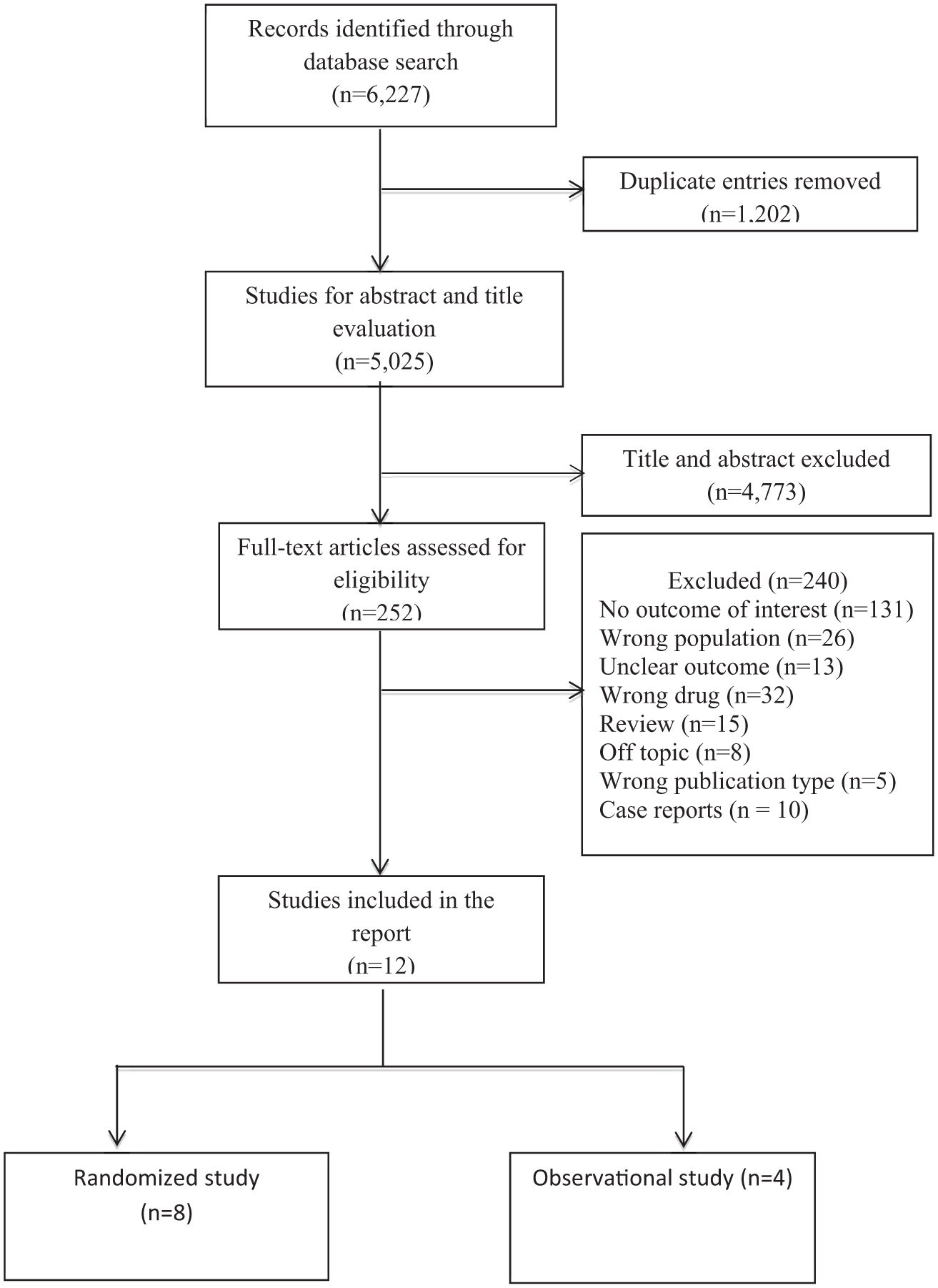
Flow chart of included studies.
Randomized studies reporting withdrawals following discontinuation of antidepressants.

ADHD, attention-deficit/hyperactivity disorder; CASES, Common Adverse and Side Effects Scale; CIT, citalopram; C-SSRS, the electronic Columbia–Suicide Severity Rating Scale; ESC, escitalopram; FLX, fluoxetine; ICSR, Individual Case Safety Reports; IPM, imipramine; JBI, Joanna Biggs Institutes’ Critical Appraisal Checklist; JF, juvenile fibromyalgia; MDD, major depressive disorder; MedDRA, Medical Dictionary for Regulatory Activities; MIP, milnacipran; NA, not available; NT, nortriptyline; PBO, placebo; PXT, paroxetine; SAE, Serious adverse event; SEC, Side-Effects Checklist; SNRIs, serotonin-norepinephrine reuptake inhibitors; SRT, sertraline; SSRIs, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants; WHO, World Health Organization; yrs, years.
Observational studies reporting withdrawals following discontinuation of antidepressants.

F, female; hrs, hours; IPM, imipramine; JBI, Joanna Biggs Institutes’ Critical Appraisal Checklist; M, male; MDD, major depressive disorder; mo, months; NA, not available; NT, nortriptyline; PXT, paroxetine; yrs, years.
Withdrawal symptoms based on results of randomized discontinuation trials
In eight randomized controlled discontinuation trials, patients were randomly allocated to a placebo control group following a period of systematic antidepressant therapy lasting typically more than 12 weeks. This design, originally implemented to test if relapse occurred absence of the drug, offers a systematic means to investigate withdrawal symptoms, particularly when they exhibit significantly higher prevalence in the placebo group relative to the active drug group. These studies focused on SSRIs, SNRIs, and TCAs. A total of 21 withdrawal symptoms involving multiple systems were observed, with the central nervous system (CNS) and the gastrointestinal system being most commonly involved. We here illustrate the type and rate of withdrawal symptoms observed for the different classes of antidepressants.
Selective serotonin reuptake inhibitors
Four randomized controlled discontinuation trials yielded data on 21 symptoms, showing a higher prevalence of withdrawal symptoms in the placebo group previously treated with SSRIs (fluoxetine = 1 or citalopram = 1 or escitalopram = 1, or sertraline = 1). Among these symptoms, nausea (14.6%–22.0%) emerged as the primary in three trials, followed by headache (14.6%–15.0%), diarrhea (10.0%–22.0%), depression (10%), inner tension (44%), anxiety/worry (44%), cough (33%) reported in two trials. Additional 14 symptoms were general (i.e., dizziness, runny nose, dry mouth, rash/skin irritation, and pyrexia), CNS manifestations (i.e., myalgia, abnormal/involuntary movements, nervousness, and hypersomnia), gastrointestinal symptoms (i.e., weight gain, weight loss, increased appetite, and decreased appetite), or referred to the cardiovascular system (i.e., palpitations).
One study on paroxetine reported that withdrawal symptoms were significantly higher in the placebo-substituted group compared to the active medication group, with rates of 11.2% and 8.4%, respectively. Another study with fluoxetine exposure reported similar rates in both groups during the discontinuation treatment, although they were assessed via general inquiry about nonspecific physical symptoms since the last visit.
Serotonin noradrenaline reuptake inhibitors
In a milnacipran discontinuation study, withdrawal symptoms were more frequently observed in participants substituted with a placebo compared to those receiving the drug (66.7% vs 42.9%). Headache was the most prevalent withdrawal symptom, with a higher incidence in the placebo group (16.7%) than in the active treatment group (0%).
Tricyclic antidepressants
In a nortriptyline discontinuation study for attention-deficit hyperactivity disorder (ADHD), the total score and irritability score of the ADHD scale increased significantly within 1 week after nortriptyline was discontinued.
Based on withdrawal classification proposed for adults by Chouinard, Chouinard, and Cosci,7,21 the unpleasant post-drug symptoms observed in the eight studies can be classified by system, including: (1) CNS: feeling disinterested detached, inner tension, nervousness, anxiety/worry, hypersomnia, abnormal and involuntary movements; (2) gastrointestinal: nausea/vomiting, diarrhea, weight gain, weight loss, increased appetite, decreased appetite, dry mouth; (3) cardiovascular: palpitations and dizziness; (4) neuromuscular: myalgia; (5) general: headache, runny nose, rash/skin irritation, temperature dysregulation, cough. As illustrated in Table 3, most withdrawal symptoms that occur in children and adolescents are not significantly different from those in adults, except for cough and temperature dysregulation. Cough was documented in two randomized controlled discontinuation studies involving sertraline and citalopram24,25 and was observed in seven subjects. Temperature dysregulation was noted in four subjects in a recent multicenter randomized controlled discontinuation study with escitalopram. 26
Withdrawal symptoms after discontinuation of antidepressants in children and adolescents.

One study exposed to milnacipran withdrawal reported a higher incidence of headache.
SSRIs, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants.
Across the eight studies, the randomized discontinuation study design provided systematic observation of new onset symptoms and worsening of prior mental symptoms after randomization to the discontinuation group. The study design also provided systematic observation in the long-term follow-up, which could be divided into new withdrawal symptoms, rebound symptoms, and persistent post-withdrawal disorders according to published criteria7,21 (see Supplemental eTables 1 and 2). After rapid switching from escitalopram or nortriptyline to placebo, the adverse post-drug symptoms observed with higher prevalence in the placebo group compared to the active drug group fall within the diagnostic criteria for new withdrawal symptoms.26,29 In addition, two controlled discontinuation studies demonstrated an exacerbation of symptom scores shortly after switching from antidepressants to placebos, offering evidence of potential rebound symptoms upon discontinuing escitalopram 26 and nortriptyline. 29 During a 36-week follow-up period post-switch from escitalopram to placebo, adverse events in the placebo arm (11, 26.8%) were almost twice as high as those in the escitalopram arm (6, 15.4%), 26 a discrepancy not attributable to escitalopram pharmacokinetics or pharmacodynamics but suggestive of persistent post-withdrawal disorders. In a study in which patients responding to 16 weeks of paroxetine were randomized to continue paroxetine or to placebo for 16 additional weeks, subjects taking placebo with obsessive-compulsive disorder comorbid with ADHD, tic disorder, or oppositional defiant disorder had a significantly higher rate of recorded relapse compared to those without such a comorbidity; however, no difference was observed among those who continued paroxetine, thus suggesting the group switched to placebo suffered persistent post-withdrawal disorders subsequent to paroxetine discontinuation. 28 In a third study, children and adolescents 7–18 years of age with major depressive disorder were treated openly with fluoxetine. Those who had an adequate response after 12 weeks (indicated by a Clinical Global Impression improvement score of 1 or 2 and a decrease of at least 50% in Children’s Depression Rating Scale-Revised score) were randomly assigned to receive fluoxetine or placebo for an additional 6 months. During the continuation phase, clinical manifestations that occurred in the placebo group resulted in hospitalization, two of them due to nonspecified physical symptoms; a third condition was represented by a suicidal attempt. This suggests that discontinuation of fluoxetine could further the occurrence of these conditions and produce persistent post-withdrawal disorders. 27
Withdrawal symptoms based on results of observational studies
Two observational studies during imipramine and nortriptyline tapering noted a significant increase in symptom frequency,30,31 further supporting the occurrence of withdrawal symptoms. Typically, 20% of patients with major depressive disorder (n = 30) experienced six symptoms (i.e., withdrawal, anger, slowness, stomachache, nausea, and emesis) when tapering imipramine. 30 Another study compared the symptom differences of patients with imipramine before treatment, during treatment, and after dose decrease. Seven out of nine groups of symptoms had statistically significant differences after dose decrease, which manifested as epigastric pain, abdominal pain, nausea, vomiting, decreased appetite, drowsiness/fatigue, apathy/withdrawal, tearfulness, agitation, and headaches. 31 In addition, 14.3% of patients taking paroxetine experienced more adverse events during tapering. 32 Quilichini et al. found a withdrawal syndrome risk of 2.02 (1.45–2.82) for patients aged 6–11 years and 2.42 (2.05–2.86) for those aged 12–17 years based on the World Health Organization’s Global Individual Case Safety Reports database, although specific symptoms were not provided (see Table 2). 22
Based on the diagnostic criteria of the withdrawal classification proposed for adults by Chouinard, Chouinard, and Cosci,7,21 new withdrawal symptoms could be diagnosed in two studies after nortriptyline 30 or imipramine 31 tapering, since clinical manifestations described in these studies30,31 matched with the clinical description included in the mentioned diagnostic criteria.
Discussion
The systematic review focuses on children and adolescents and indicates that withdrawal symptoms can manifest with different types of antidepressants, that is, SSRIs (citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, and sertraline), SNRI (bupropion), and TCAs (desipramine, imipramine, nortriptyline, and amitriptyline), referring to classifications used for adults.7,21
From the research side, investigating evidence of withdrawal symptoms in studies designed to research something else, as it is the case for studies included in the present review, implies that incidental findings must be reinterpreted in the light of probable withdrawal effects.
Symptoms identified as withdrawal ones in children and adolescents comprised a wide array of clinical manifestations (see Table 3), which included diarrhea, inner tension, myalgia, tachycardia, and hypersomnia. These symptoms were documented in trials and aligned with those seen in adults.7,21,33 Rash, skin irritation, dry mouth, weight changes, cough, increased appetite, weight gain, and fever were still observed in trials but were not reported in adults. Thus, children and adolescents might have withdrawal manifestations that do not fully overlap with those observed in adults, needing exploration and assessment.
A solid clinical competence in recognizing withdrawal phenomena and later formulating the correct diagnosis is needed. Apparently, recognizing withdrawal symptoms and syndromes is highly challenging for clinicians since they may not fit with the diagnostic criteria described by the current standard nosography. In addition, the standard clinical interview tends nowadays to focus on type, dosage, duration, and efficacy of treatments received, missing a crucial point: being watchful for iatrogenesis, which implies assessing adverse drug reactions related to tolerance. 34 Iatrogenesis includes withdrawal symptoms and syndromes. Distinguishing between disease symptoms and treatment-induced effects is challenging, 35 as well as distinguishing between disease symptoms and reactive distress over treatment when withdrawal occurs. Overall, patients experiencing withdrawal symptoms or syndromes are at risk of being misdiagnosed, mistreated, and of developing chronicity or additional clinical manifestations. Given the limited efficacy of SSRIs in youth, 36 high placebo response rates, 37 the early and late emergence of adverse effects, 38 along with our discussion of withdrawal issues, concerns rise regarding the presumed risks and benefits of antidepressant use in children and adolescents. In this framework, psychological treatments, particularly cognitive-behavioral therapy, should be considered as first-line treatment option, having shown promising results. 39 Similarly, neuromodulation techniques, like repetitive transcranial magnetic stimulation (rTMS), should be considered first when major depression occurs. 40
Limitations and strengths
The present findings should be interpreted in the light of several limitations. The studies here included, as well as those available in the literature, used tools not specifically designed to assess withdrawal symptoms, which may lead to their underestimation. The identification of withdrawal symptoms would necessitate a thorough examination of potential symptomatology (see Table 3), using observer interviews and/or self-rating tools such as the semi-structured interview of the Diagnostic Clinical Interview for Drug Withdrawal 14,41,42 and the Discontinuation-Emergent Signs and Symptoms (DESS), 43 respectively. Unfortunately, these tools were developed only for adults. Second, the available literature is scarce; we examined eight RCTs and four observational studies, thus the generalizability of results is limited. Third, in some research, withdrawal symptoms were incidental findings in the general clinical inquiry, or the withdrawal symptom assessment method was not specified.
Besides such limitations, the present review is the first attempt to systematically sum up the literature on RCTs and observational studies collecting data on withdrawal symptoms in children and adolescents. The present findings give the opportunity to shade some light on a widely neglected topic of high clinical impact.
Conclusion
Withdrawal at antidepressant discontinuation is a major challenge in research and clinical practice. There are still major difficulties in distinguishing symptoms of the disease and those induced by the treatment, and the information available from RCTs and observational studies is limited. The pressing issue is that young patients presenting withdrawal symptoms or syndromes are at risk of being misdiagnosed, thus mistreated.
There are important issues that need to be explored, and we have tools such as the Discontinuation-Emergent Signs and Symptoms and the Diagnostic Clinical Interview for Drug Withdrawal 1—New Symptoms of SSRI and SNRI, which need to be developed in a version suitable for children or adolescents. Neurobiological studies to identify why certain patients develop withdrawal syndromes, and others do not, are urgently needed. Longitudinal studies exploring the occurrence, clinical features, and neurobiological correlates of withdrawal syndromes are warranted, as well as RCTs considering the possibility that withdrawal syndromes may be confounded with relapses (or treatment-resistance) in long-term drug/placebo continuation studies. The present findings aim at encouraging the flourishing of this field of research, taking advantage of available methodological knowledge to design highly rigorous methods.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251404780 – Supplemental material for Acute and persistent withdrawal syndromes following discontinuation of antidepressants in children and adolescents: a systematic review
Supplemental material, sj-docx-1-tpp-10.1177_20451253251404780 for Acute and persistent withdrawal syndromes following discontinuation of antidepressants in children and adolescents: a systematic review by Jianjun Wang and Fiammetta Cosci in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253251404780 – Supplemental material for Acute and persistent withdrawal syndromes following discontinuation of antidepressants in children and adolescents: a systematic review
Supplemental material, sj-docx-2-tpp-10.1177_20451253251404780 for Acute and persistent withdrawal syndromes following discontinuation of antidepressants in children and adolescents: a systematic review by Jianjun Wang and Fiammetta Cosci in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
