Abstract
Background:
Valproic acid (VPA)-induced hyperammonemia is a common condition whose clinical presentations can range from asymptomatic cases to severe hyperammonemic encephalopathy. This can happen at any stage of treatment and is difficult to predict, as it often occurs without any alteration in liver function tests.
Objectives:
We aimed at exploring the association between hyperammonemia and clinical/biochemical characteristics among inpatients with mental disorders.
Design:
This cross-sectional study was conducted on adults consecutively admitted to two acute psychiatric inpatient units in the Milan metropolitan area from October 2022 to October 2024. All participants were treated with oral VPA during hospitalization.
Methods:
Sociodemographic, clinical, and biochemical data were retrieved from clinical interviews and electronic chart reviews. Univariate analyses and multiple logistic regressions were used to assess the association between hyperammonemia and candidate variables.
Results:
We included 130 participants (mean age: 38.2 ± 15.3 years; males: 77.7%). Bipolar I (36.9%), personality (31.5%), and schizophrenia spectrum (20.8%) disorders were the most common diagnoses. Mean serum ammonium levels were 46.4 ± 17.5 μmol/L, with hyperammonemia observed in 52 (40.0%) participants. Univariate analyses estimated an association of hyperammonemia with male sex, the concomitant use of 2+ medications (besides VPA), treatment with antipsychotics, and a diagnosis of alcohol use disorder (AUD). No further clinical/biochemical features were associated with hyperammonemia, including serum VPA levels and daily dose of VPA. Regression models showed that the concomitant use of 2+ other medications (coeff. = 1.03, p = 0.018) and AUD (coeff. = 1.32, p = 0.018), but not male sex (p = 0.12) and the use of antipsychotics (p = 0.09), were associated with hyperammonemia.
Conclusion:
Our findings highlight the potential influence of AUD and polypharmacy on the risk of hyperammonemia in subjects treated with oral VPA. Additional studies are needed to confirm these associations and to test their potential causal role in hyperammonemia. Clinical implications for limiting polypharmacy in people treated with oral VPA should be considered.
Plain language summary
Background: This study explored the correlation between high ammonium levels (hyperammonemia) and clinical and biochemical factors in psychiatric patients taking valproic acid (VPA). Hyperammonemia is a common side effect of VPA, ranging from no symptoms to severe confusion, and can occur without changes in liver function tests. Methods: The study included 130 adults admitted to two psychiatric units in Milan from October 2022 to October 2024. All patients were receiving VPA during their hospitalization. The team collected medical histories, test results, and other health information, then analyzed potential correlations using statistical models. Results: The patients, mostly male (77.7%) with an average age of 38, were diagnosed mainly with bipolar I disorder (36.9%), personality disorders (31.5%), or schizophrenia-related conditions (20.8%). 52 (40.0%) had high ammonium levels. The analysis found that hyperammonemia was more common in patients taking two or more other medications along with VPA, those using antipsychotics, and individuals with alcohol use disorder (AUD). However, after adjusting for multiple factors, only AUD and taking multiple medications remained significantly linked to high ammonium levels. Interestingly, neither the daily dose of VPA nor blood VPA levels predicted hyperammonemia. This means that high ammonium levels can occur regardless of the amount of VPA in the body. Conclusions: The results highlight the risks of using many medications together and the impact of alcohol use in patients on VPA. Reducing the number of medications may help lower the chance of hyperammonemia. Additional studies are needed to confirm these associations and to test their potential causal role on hyperammonemia. These insights could guide doctors in improving the safety of VPA treatment, especially for patients with alcohol use issues or those on multiple medications.
Introduction
Valproic acid (VPA) is a wide-spectrum anticonvulsant medication that is commonly used in the treatment of epilepsy and migraine.1–4 In addition, it has shown efficacy in the treatment and prophylaxis of manic or mixed episodes in bipolar disorder, though it seems less effective for bipolar depression.5–7 VPA is also frequently off-label used in the management of mood instability, impulsivity, agitation, and aggressive behavior in mental health care settings across a broad range of diagnoses, including personality disorders and schizophrenia spectrum disorders. 4 Common side effects of VPA can include fatigue, weight gain, nausea, hair loss, and tremor. 8 However, VPA can also be associated—though less often—with hepatotoxicity, thrombocytopenia, and symptomatic hyperammonemia.8,9
Hyperammonemia refers to serum levels of ammonium—a byproduct of protein metabolism that is converted to urea in the liver—⩾50 μmol/L. 10 One of the key mechanisms of VPA-induced hyperammonemia seems to be the inhibiting effect of VPA on carbamoylphosphate synthetase-I and, consequently, on the urea cycle.11,12 This condition is often reversible upon discontinuation of the drug and can occur without changes in liver function tests and at any stage of treatment, even after long-term use. 13
While hyperammonemia may be asymptomatic in the majority of cases, it sometimes presents with neurological and gastrointestinal symptoms as well as hyperventilation, and might progress to hyperammonemic encephalopathy (HE).13,14 HE has been reported in 0.7%–22.2% of cases,13,14 with a variability that likely reflects differences in study design, population characteristics, and thresholds, as well as patients’ clinical management and follow-up. Hyperammonemia leads to elevated glutamine levels in the central nervous system, causing astrocyte swelling and cerebral edema. 14 The resulting clinical presentation can vary in severity, with ataxia, lethargy, and confusion in milder cases, while agitation, focal neurological deficits, miosis, and seizures have also been occasionally reported. 15 Progression to stupor, coma, and, ultimately, death has also been documented. 16 Previous evidence has demonstrated a number of conditions (hereditary or acquired) that may affect serum ammonium levels.10,17 Relevant factors include the total daily dosage of VPA, the concomitant use of other antiepileptic drugs and/or antipsychotics, and the overall use of multiple medications.17–19 Furthermore, female sex, older age, intellectual disability, body weight, liver disease, urea cycle disorders, vegetarian or vegan diets, hypercatabolic states (e.g., burns or trauma), and the intravenous administration of VPA have been associated with hyperammonemia.19–21 While some studies suggest a correlation between higher VPA plasma concentrations and the development of hyperammonemia, most cases occur within the drug’s therapeutic plasma levels. 16 Taken together, this evidence highlights the complexity and difficulties of predicting hyperammonemia.
Thus, we conducted a cross-sectional study based on a representative sample of inpatients with severe mental disorders treated with oral VPA to explore the association between hyperammonemia and several clinical and biochemical features, including VPA dosing and serum levels.
Methods
This cross-sectional observational study was designed and reported following the “Strengthening the Reporting of Observational studies in Epidemiology (STROBE)” statement. 22 It was carried out in accordance with the Declaration of Helsinki. 23 The study was part of the broader Northern Milan Area Cohort (NOMIAC) project. 24
Setting and inclusion criteria
We included any adults treated with oral VPA consecutively admitted from October 2022 to October 2024 to the two acute inpatient units (accounting for a total of 27 beds) of the Department of Mental Health and Addictions of the Nord Milano Health and Social Care Trust, which delivers mental health services to the 280,000 residents within a highly urbanized catchment area of the Metropolitan City of Milan. 25 Eligible individuals had to be: (i) aged ⩾18 years at the time of data collection; (ii) with stable doses of oral VPA for at least 5 days prior to serum VPA and ammonium measurement, allowing sufficient time to reach steady-state serum VPA concentrations; and (iii) serum VPA and ammonium levels measured at least once on the same day during the hospitalization. To avoid duplicate data and ensure consistency in the analysis, if serum VPA and ammonium levels were measured multiple times, only the first measurement was considered. Subjects with missing, incomplete, or not valid data on serum VPA and ammonium levels were excluded. For participants with multiple admissions, we used data from the first admission during the study period.
Data collection
We collected information on sociodemographic characteristics—such as age and sex—as well as on clinical features, that is, psychiatric diagnoses, lifetime diagnosis of alcohol (AUD) (F10.X) and/or substance use (SUD) disorders (F11–F19), and current pharmacotherapy. Pharmacotherapy was further categorized according to the use of two or more medications (hereafter also referred to as polypharmacy) and included the daily dosage of oral VPA. Comorbid organic conditions (hypertension, obesity, diabetes, and hepatopathy) were recorded alongside anthropometric parameters such as body mass index (BMI). Information on serum ammonium (μmol/L) and VPA levels (μg/mL), as well as on markers of liver, renal, and metabolic function, was retrieved from routinely performed biochemical serum analyses. Blood sampling was performed at 8 a.m. after an overnight fast. Blood samples for ammonium measurement were immediately placed in ice and promptly transported to the laboratory for analysis within 30 min of collection, as recommended. 26 According to the local laboratory standards, the normal reference range for serum ammonium level was 14.85–49.5 μmol/L, with hyperammonemia defined as a level exceeding 49.5 μmol/L, consistent with well-established definitions and cut-off values.27,28 This ensured a consistent and interpretable framework for categorization across the sample. Information was obtained from clinical interviews and assessments performed during participants’ hospitalization, and integrated with features retrieved from clinical records and chart review through the electronic hospital data management platform. They were entered into a custom-designed database. Information was managed in compliance with privacy regulations and anonymized to minimize the potential risk of identification.
Data analyses
Standard descriptive statistics—including count and percentage for categorical variables as well as mean with standard deviation (SD) or median with interquartile range (IQR) (according to data distribution) for continuous variables—were used to summarize sample characteristics. The normality of data distribution was assessed using the Shapiro-Wilk test. Univariate analyses were aimed at estimating the potential differences between individuals with and without serum ammonium level >49.5 µmol/L. Chi-square and Fisher’s exact tests were used for categorical variables, while Student’s T or Mann–Whitney U tests (consistently with data distribution) for continuous variables. In addition, to test for multicollinearity, we performed relevant diagnostics based on standard rules of thumb, examining both standard errors and the variance inflation factor (VIF < 10).
Multiple logistic regression models were performed to assess the association between hyperammonemia and candidate explanatory variables. These included predefined characteristics expected to influence ammonium levels, that is, age, sex, serum VPA level, and daily dosage of VPA, as well as any clinical or biochemical variables showing a p-value < 0.05 at univariate analyses. Potential overfitting issues were also considered based on the number of events per variable. Regression coefficients with their 95% confidence intervals (95% CIs) were used to estimate the effects of explanatory variables. Statistical significance was set at p < 0.05. For all predictor variables, including those not found to be statistically significant, 95% CIs and p-values were consistently reported. Data analyses were performed using Stata statistical software, release 18 (StataCorp LLC, 2023, College Station, TX, USA).
Results
Sample characteristics
Of the 679 people admitted to the two acute inpatient units between October 2022 and October 2024, 137 were treated with oral VPA during their hospitalization. Among them, while serum VPA levels were measured in all cases, information on serum ammonium levels was missing for seven cases. Thus, 130 subjects were ultimately included in the analysis. A flow diagram of the participants’ inclusion process is displayed in Figure 1.
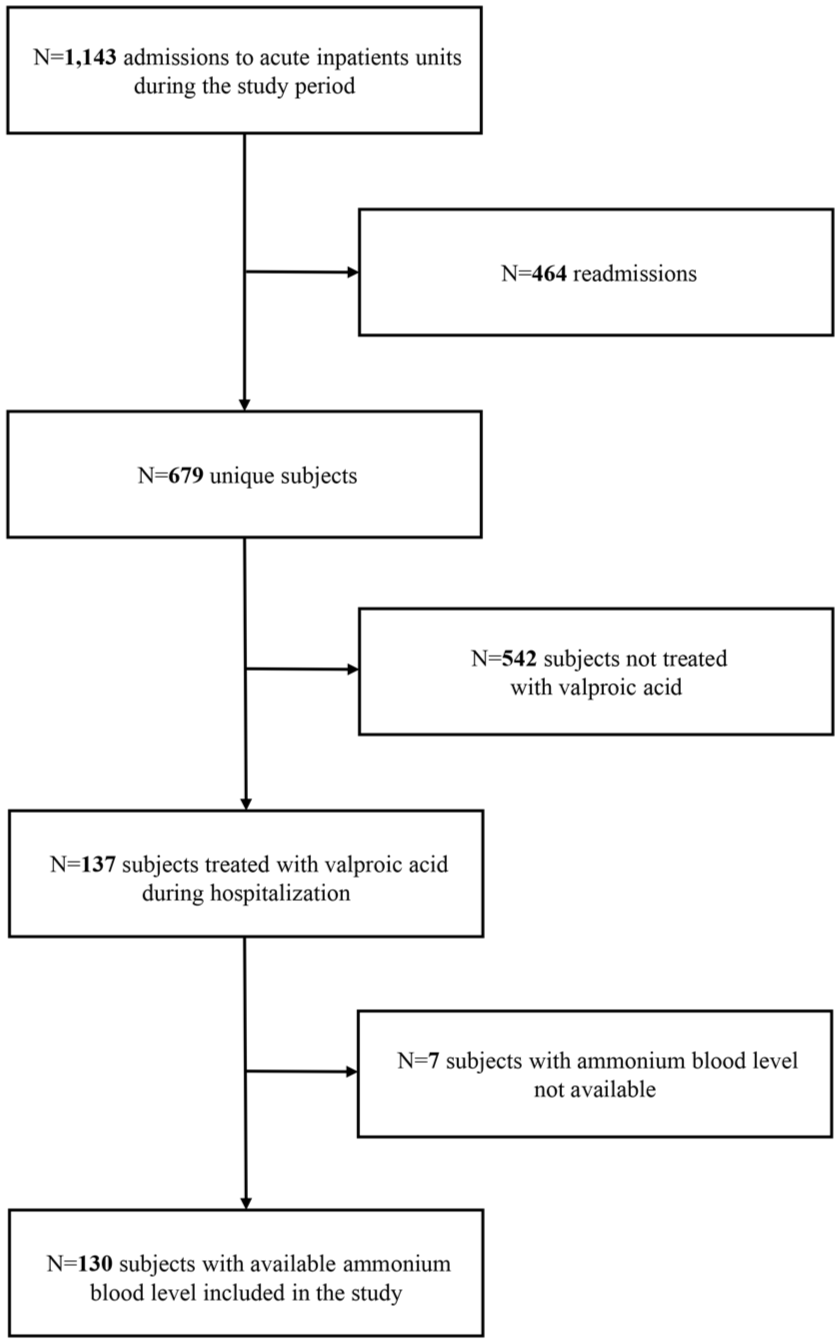
Flow diagram of participants’ inclusion process.
The mean age of study participants, including 101 (77.7%) males and 29 (22.3%) females, was 38.2 ± 15.3 years. The mean serum levels of VPA were 58.4 ± 20.4 μg/mL (min: 14 μg/mL—max: 130 μg/mL), while mean serum ammonium levels were 46.4 ± 17.5 μmol/L (min: 12.2 μmol/L—max: 106 μmol/L). There was no significant correlation between serum valproate (VPA) levels and serum ammonium levels (Pearson’s r = 0.05), suggesting a negligible linear association between the two variables. A scatter plot visualizing the relationship between VPA levels and ammonium levels is shown in Supplemental Figure 1.
Hyperammonemia was observed in 52 participants (40.0%). HE was not observed in any of the study participants.
Bipolar I disorder was the most common diagnosis (n = 48, 36.9%), followed by personality disorders (n = 41, 31.5%) and schizophrenia spectrum and other psychotic disorders (n = 27, 20.8%). Comorbid AUD and SUD were reported in 21 (16.2%) and 47 (36.2%) subjects, respectively.
Half of the sample (51.5%) was treated with two or more medications besides VPA, with antipsychotics (n = 111, 85.4%) and benzodiazepines (n = 114, 87.7%) as the most represented pharmacotherapies.
The sociodemographic and clinical characteristics of the overall sample are reported in Table 1, while the biochemical data are reported in Table 2.
Sociodemographic and clinical characteristics and differences between subjects with serum ammonium levels ⩽49.5 and >49.5 µmol/L.

z Values were obtained from Mann–Whitney U tests. χ² values were obtained from Chi-squared tests.
Fisher’s exact test’s p-value. The p-value from Fisher’s exact test was computed when a subgroup contained a small number of cases or non-cases (i.e., fewer than 10).
BMI, body mass index; IQR, interquartile range; N, n, number of subjects with data available for each variable; SD, standard deviation; VPA, valproic acid.
Biochemical serum levels and differences between subjects with serum ammonium levels ⩽49.5 and >49.5 µmol/L.
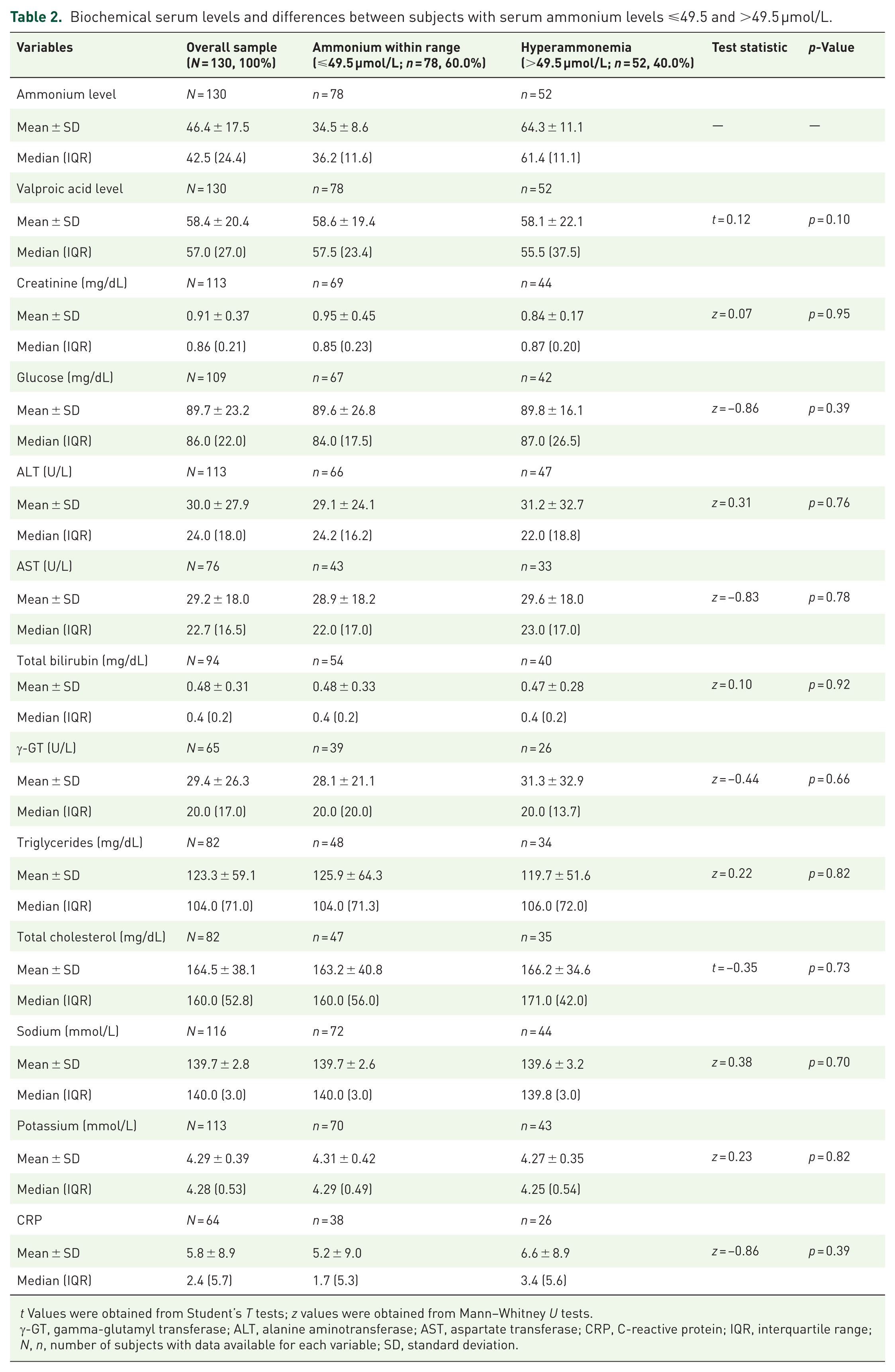
t Values were obtained from Student’s T tests; z values were obtained from Mann–Whitney U tests.
γ-GT, gamma-glutamyl transferase; ALT, alanine aminotransferase; AST, aspartate transferase; CRP, C-reactive protein; IQR, interquartile range; N, n, number of subjects with data available for each variable; SD, standard deviation.
Factors associated with hyperammonemia: Univariate analyses
Univariate analyses showed that subjects with hyperammonemia, compared to those with serum ammonium levels within range, were more likely to be males (χ2 = 3.91, p = 0.048), diagnosed with comorbid AUD (p = 0.030), concomitantly using two or more medications (besides VPA) (χ2 = 4.93, p = 0.026), and concurrently treated with antipsychotics (p = 0.023). On the other hand, serum VPA level (p = 0.10) and daily dosage of VPA (p = 0.55) were not associated with hyperammonemia. No other biochemical variables, including blood markers of liver dysfunction (alanine aminotransferase, aspartate transferase, gamma-glutamyl transferase, and total bilirubin), were associated with a serum ammonium level >49.5 μmol/L.
The characteristics of each subgroup according to the presence of hyperammonemia and the results of univariate comparisons are reported in Tables 1 and 2.
Factors associated with hyperammonemia: Multivariable logistic regression model
The multivariable logistic regression model, including age, sex, serum VPA level, and daily dosage of VPA, as well as variables associated with hyperammonemia at the univariate level (p < 0.05), confirmed the relationship between serum ammonium level >49.5 μmol/L and the concomitant use of two or more medications (coeff. = 1.03, p = 0.018). Moreover, hyperammonemia was more frequent in people with a diagnosis of AUD (coeff. = 1.32, p = 0.018). No association with male sex (p = 0.12) and antipsychotic use (p = 0.09) emerged. No evidence of collinearity between variables was observed. The results of the regression analysis are reported in Table 3.
Correlates of hyperammonemia in people treated with valproic acid: multivariable logistic regression model.

For a 1-unit change.
Versus female gender.
Versus without AUD.
Versus not on antipsychotics.
Versus < 2 medications.
Logarithmic scale.
95% CI, 95% confidence interval; AUD, alcohol use disorder; SE, standard error; VPA, valproic acid.
Discussion
Interpretation of findings
The aim of this study was to investigate sociodemographic, clinical, and biochemical correlates of VPA-induced hyperammonemia among 130 adults consecutively admitted for psychiatric inpatient care. Hyperammonemia was observed in 52 subjects (40.0%), which is slightly lower than the mean prevalence estimates of 47.4% reported in previous cross-sectional studies (e.g., Chicharro et al., 2007). 29 However, this prevalence varies considerably across studies, likely reflecting differences in study design, population characteristics, and diagnostic thresholds. 16 Notably, VPA-induced hyperammonemia is more extensively described in neurological conditions, while it is less documented in populations affected by mental disorders.30,31 Consistently, no cases of HE were observed in our sample, in line with the relatively few reports of this condition in psychiatric disorders compared to those reported when treating epilepsy. 13 Moreover, the close clinical monitoring provided in our inpatient units, including regular assessment of VPA and ammonium levels and the early administration of lactulose in most cases of hyperammonemia, may have prevented the development of HE in our sample. In this regard, it should also be considered that the risk of developing hyperammonemia symptoms and HE seems influenced by prolonged exposure to elevated ammonium levels.13,14
The most relevant finding from our study is the association between hyperammonemia and treatment with two or more medications besides VPA, as well as with comorbid AUD. Polypharmacy represents a risk factor for adverse drug reactions in general,32,33 while the use of different compounds may be necessary for managing complex medical conditions, this practice can significantly increase the likelihood of potential drug-drug interactions, affecting the pharmacokinetics and the pharmacodynamics of the prescribed medications.33,34 In line with our findings, some available evidence suggests that VPA-induced hyperammonemia may actually be more common in subjects on polypharmacy.19,35,36 Although different mechanisms may be involved, the increased pharmacological hepatic burden impairing ammonium detoxification might be the main culprit. 19 Indeed, VPA is metabolized via mitochondrial beta-oxidation and hepatic glucuronidation.37,38 During treatment with multiple medications, other drugs competing for these pathways can lead to the formation of toxic metabolites, such as 2-propyl-4-pentenoic acid, which finally impairs ammonium clearance. 39 Moreover, the overall metabolic capability of the liver can be saturated, thus reducing its efficiency in clearing ammonium. 40 Finally, polypharmacy may exacerbate underlying conditions such as liver dysfunction or deficiencies in essential nutrients, particularly carnitine, primarily due to metabolism disruption, malabsorption, and loss of appetite.41,42 The effect of VPA on carnitine homeostasis is equally well documented:43–45 VPA is indeed able to inhibit the enzyme γ-butyrobetaine hydroxylase, determining a reduction in carnitine synthesis, and to potentiate the urinary excretion of amino acids. 43 Such reduction in carnitine levels, resulting in the cytosolic accumulation of unoxidized fatty acyl-coenzyme A molecules (which are believed to inhibit the urea cycle), can impair ammonium excretion,46,47 thus potentially representing a mechanism involved in the development of hyperammonemia in subjects treated with VPA.
Consistently, the association between AUD and hyperammonemia is not surprising. AUD is known to produce a wide spectrum of hepatic impairments, from steatosis and inflammation to complete loss of liver function. 48 Both acute and chronic alcohol consumption can lead to malnutrition by decreasing dietary caloric intake, impairing protein synthesis and secretion, and hindering the absorption of essential nutrients. These effects ultimately result in reduced free and total carnitine levels.49,50 Thus, hepatic dysfunction, combined with malnutrition-related deficits, may play a key role in disrupting ammonium metabolism.42,48 Our findings suggest that AUD could therefore be a factor contributing to the development of hyperammonemia in individuals treated with VPA.
Clinical implications
In clinical practice, the measurement of ammonium concentrations in asymptomatic patients treated with VPA is not routinely recommended, being seemingly unnecessary or even potentially leading to misdiagnoses.29,51 On the other hand, the identification of specific factors associated with increased blood ammonium may favor the implementation of targeted monitoring practices to prevent symptomatic hyperammonemia and, ultimately, HE. The association between polypharmacy, comorbid AUD, and hyperammonemia uncovered by our study emphasizes the need for clinicians to be aware of the potential risks underlying complex medical conditions, especially given the high prevalence of both polypharmacy and comorbid AUD in people with severe mental disorders.34,52–54 In such cases, traditional disease-oriented practice may prove inadequate.33,34 To overcome this gap and ensure effective prevention, a personalized approach should be prioritized, focusing on key areas such as the identification of at-risk patients and the implementation of robust monitoring and follow-up strategies.32,55
Limitations
Some limitations should be acknowledged when interpreting our findings. First, the cross-sectional design of our study did not allow us to make any inference on the causal relationship between the explored clinical factors and hyperammonemia. Second, since our study was based on a relatively limited number of participants without any sample size estimation, we should not rule out further associations between putative variables and hyperammonemia possibly not detected by our analyses (i.e., the use of specific drug classes). In addition, the relatively rough variable “polypharmacy” may introduce the potential for residual confounding, preventing the specificity needed to meaningfully inform clinical practice in the context of VPA-associated hyperammonemia. Future analyses with more detailed medication classification (e.g., focusing on drug classes known to affect ammonium metabolism or hepatic function) would be valuable in better delineating these interactions. Third, the lack of information on additional potential confounders, such as prior VPA use duration, recent dose adjustments, and medication adherence, somewhat limits our conclusions. Similarly, additional factors potentially contributing to the development of hyperammonemia, such as genetic polymorphisms affecting urea cycle enzymes, nutritional status, and underlying metabolic disorders, 56 could not be assessed. Further investigation into these variables is needed to delve deeper into the pathophysiology of VPA-induced hyperammonemia. Finally, our study assumed just a categorical perspective for hyperammonemia and covariates, such as AUD and SUD, since our sample size did not allow us to consider their duration and severity. Additional studies are needed to analyze whether putative clinical variables correlate with hyperammonemia severity.
Conclusion
Our study provides additional insights into factors associated with VPA-induced hyperammonemia and the need for careful monitoring with particular regard to complex conditions, including comorbid AUD and the prescription of multiple psychotropic medications. Identifying relevant correlates of hyperammonemia may be useful in clinical practice, particularly considering the complexity of predicting this condition. Additional studies are needed to test the generalizability of our findings and to explore the potential causal role of these and other clinical features on VPA-induced hyperammonemia.
Supplemental Material
sj-tif-1-tpp-10.1177_20451253251385730 – Supplemental material for Clinical and biochemical factors associated with hyperammonemia in inpatients with mental disorders treated with valproic acid: a cross-sectional analysis
Supplemental material, sj-tif-1-tpp-10.1177_20451253251385730 for Clinical and biochemical factors associated with hyperammonemia in inpatients with mental disorders treated with valproic acid: a cross-sectional analysis by Ilaria Riboldi, Daniele Cavaleri, Marco Morreale, Cristina Crocamo, Francesco Bartoli and Giuseppe Carrà in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
