Abstract
Background:
Long-acting injectable (LAI) antipsychotics can improve treatment adherence in patients with schizophrenia. Despite their benefits, LAIs are underused in China compared to other countries. Little real-world evidence describes the impact of switching from oral to LAI antipsychotics on adherence and healthcare utilization in clinical practice in China, which could help address this gap.
Objectives:
To understand utilization of LAI and to assess the impact of switching from oral to LAI antipsychotics on adherence and healthcare utilization.
Design:
This is a retrospective, 1-year mirror-image study using electronic health records (2012–2019) from a psychiatric specialized hospital and the psychiatry department in a general hospital in China. The observation period was 1 year before and after LAI initiation in patients already receiving oral antipsychotics.
Methods:
Adult patients (aged 18–65) who initiated LAIs after receiving oral antipsychotics, with schizophrenia diagnosis at least 12 months before LAI initiation were included. The date of LAI initiation was designated as the index date. Adherence to antipsychotics was assessed by the proportion of days covered. Schizophrenia-related healthcare utilization comprised the percentage of patients who had admissions, the duration of inpatient stays, the number of inpatient visits, and outpatient visits. Wilcoxon signed-rank test and McNemar’s test were used for before-after comparison.
Results:
Overall, 98 and 59 eligible patients were included in two hospitals, respectively. Treatment adherence (proportion of days covered) after switching increased significantly from 46% to 61% (p < 0.01) and 32% to 58% (p < 0.01), respectively. The frequency of hospital admissions (and cumulative admission days) reduced from 10.2% to 4.1% (6 days to 2 days, p = 0.11), and 55.9% to 10.2% (11 days to 2 days, p < 0.01), respectively. Outpatient visits increased from 5 to 6 visits (p = 0.10), and 7 to 9 visits (p < 0.01), respectively.
Conclusion:
Consistent benefits of LAIs in enhancing treatment adherence and optimizing healthcare utilization were observed in two representative hospitals having different clinical settings and patient characteristics.
Plain Language Summary
Why was the study done?
Many schizophrenia patients struggle to consistently take their oral antipsychotic medications, which can lead to poor health outcomes. Long-acting injectable antipsychotics (LAIs) may help improve adherence by reducing the frequency of medication doses required, but there’s limited real-world evidence of their impact in Chinese clinical settings.
What did the researchers do?
Researchers analyzed electronic health records from two major hospitals in China, examining data from 2012 to 2019. They focused on adults aged 18 to 65 who switched from oral antipsychotic medications to LAIs. These patients had been receiving care for at least a year before and after the switch. The study compared treatment adherence and healthcare utilization one-year before and after this transition.
What did the researchers find?
The study found that after switching to LAIs, patients’ adherence to treatment significantly improved. At one hospital, adherence increased from 46% to 61%, and at another, it rose from 32% to 58%. Additionally, the number of hospital admissions and the total days spent in the hospital decreased, while outpatient visits slightly increased. These changes suggest that patients switching to LAIs may benefit in treatment adherence and healthcare engagement.
What do the findings mean?
These findings indicate that LAIs can be an effective option for improving treatment adherence in schizophrenia patients in China. By helping patients stick to their treatment regimes, LAIs may contribute to better health outcomes.
Introduction
Schizophrenia is a severe and chronic mental disorder characterized by psychotic symptoms such as hallucinations, delusions, and disorganized speech, negative symptoms such as decreased motivation and diminished expressiveness, and cognitive deficits including impaired executive functions, memory, and mental processing speed. 1 The disease severely affects social functioning, employment, undermines the quality of life of suffers and their family members, and results in a considerable socioeconomic burden. Patients with schizophrenia have higher rates of comorbidities and a significantly reduced lifespan compared to the general population. 1
Antipsychotic drugs are an important aspect of treatment, and their long-term use is recommended for all individuals with schizophrenia. 2 Antipsychotic treatment is associated with reduced risk of relapse, reduced hospitalization, and reduced suicidal behavior. However, available medications have side effects that reduce their tolerability and have limited effectiveness on negative symptoms. 3 These features, potentially alongside poor disease awareness, lack of insight, poor social supports, fear of stigmatization, and possible financial constraints, can all lead to poor adherence to treatment. Adherence to oral antipsychotics is estimated to be around 50%, 3 and poor adherence is one of the strongest risk factors for relapse. 4
Long-acting injectable (LAI) antipsychotics act by extending the dosing interval and prolonging the duration of therapeutic drug levels and are recommended in many countries for patients with a history of poor adherence.2,3 Real-world studies have shown that compared to oral antipsychotics, LAI antipsychotic drugs can improve treatment adherence, reduce the risk of relapse, reduce hospitalizations, lower mortality, reduce healthcare utilization and associated costs, and improve patient satisfaction with treatment.5–9
In China, the life-time prevalence of schizophrenia is 0.6%, with a higher prevalence between the ages of 18 and 34 years. 10 The Chinese Schizophrenia Expert Consensus Group recommends LAI for use in all stages of schizophrenia, stating that the earlier they are used, the more benefits patients may experience. 11 However, despite the increase in popularity of LAIs and their promising potential in improving schizophrenia management, the utilization of LAIs in China is among the lowest across 15 Asian countries, with a rate of less than 1% among schizophrenia patients, creating a significant gap in treatment strategy. 12 There is little real-world evidence describing the impact of switching from oral to LAI antipsychotics in clinical practice in China. Understanding these effects may help address the underuse and demonstrate the local benefits of LAIs. In this study, we aim to leverage data from electronic health records (EHRs) collected from two major mental health institutions in China to understand utilization and treatment patterns of LAI and to assess the impact of switching from oral to LAI antipsychotics on adherence and healthcare utilization.
Methods
Study design and population
This was a retrospective, observational, multi-center study utilizing a mirror-image design. All antipsychotic medications were identified from prescription data. The index date (or mirror point, T0) was defined as the date of LAI treatment initiation. Fixed time periods 12 months before and after the index date were designated as the pre-LAI and post-LAI periods, respectively. LAI initiation was defined as the issuance of the first LAI prescription, with no prior LAI prescriptions (of any type) at pre-LAI period. All LAIs (listed in Table S1) and oral antipsychotics were considered in the study (first- or second-generation).
All eligible adult patients aged between 18 and 65 years with a confirmed diagnosis of schizophrenia (International Classification of Diseases version 10 (ICD-10) code F20.X) who had a schizophrenia diagnosis at least 12 months before the initiation of LAIs between 2013 and 2018,1–3 were included in the study. Patients were excluded if they did not receive an oral antipsychotic during the pre-LAI period, had fewer than four medical visits of any type in either the pre- or post-LAI periods, or if they were known to be non-responsive or to have treatment resistance (prescribed clozapine during the pre-LAI period). The reporting of this study conforms to the STROBE statement. 13
Data source
The study used EHR databases between 2012 and 2019 from two major regional mental health institutions in China: Peking University Sixth Hospital (PKU6H), a specialized psychiatric hospital, and the psychiatry department of the Xijing Hospital, Air Force Medical University (XJH). Both institutions are first-class, tier III hospitals that provide tertiary medical services to patients with psychiatric diseases from all over China.14,15 No referral is needed to attend either hospital.
The research databases contain unique de-identified patient information including demographic characteristics, diagnoses as ICD-10 codes for outpatient visits and inpatient admissions, discharge information for inpatient services (e.g., date, medication administration), and prescription-fill data including start date, medication name, quantity, and days of supply at both inpatient and outpatient settings. Data held in the Hospital Information System (HIS) were transformed into integrated structured EHR research databases with data verification to ensure consistency with the original HIS databases.
Study outcomes
Treatment adherence before and after T0 was assessed using the Proportion of Days Covered (PDC).16,17 The PDC is defined as the number of non-overlapping days covered by any prescriptions of antipsychotics captured during the pre-LAI and post-LAI period (including oral or LAI, first- or second-generation), divided by the total number of days during the respective fixed time period (12 months).
Good adherence was defined as PDC ⩾ 80%.16,17
In practice, hospital admission is considered as failure of previous treatment. 18 Thus, special considerations were applied to account for the hospital admission (Figure S1). In the primary analysis, T0 was placed 14 days after LAI initiation, based on evidence that antipsychotics usually achieve a satisfactory therapeutic response within 2 weeks after initiation, and that on average LAIs reaches peak plasma concentrations approximately 13 days after initiation.19,20 Schizophrenia-related healthcare utilization was assessed before and after T0. Healthcare utilization was defined as the number of hospital outpatient visits, hospital admissions, and cumulative duration of inpatient stay and proportion of patients having hospital admission.
Demographics information at index date, including age, sex, and clinical features including hospital admissions prior to T0, and past mental health-related diagnoses and medications were captured. The number of prescriptions of LAI antipsychotics at follow-up was summarized.
Statistical analysis
Demographic and clinical characteristics and antipsychotic use of the study patients were summarized using descriptive statistics. Frequencies with percentages were used for categorical variables; means with standard deviations (SD), medians with interquartile ranges (IQR), and ranges were used for continuous variables.
Due to the non-normal distribution of the continuous variables, comparisons of study outcomes between the pre-LAI and post-LAI periods used non-parametric Wilcoxon signed-rank tests due to the paired nature of the data. Exact McNemar’s tests were used for matched-pairs dichotomous outcomes. A statistically significant difference between the pre-LAI and post-LAI periods was defined as a p-value < 0.05. No correction for multiple testing was employed. A sample size of 40 is sufficient to achieve 80% power for comparisons of treatment adherence (i.e., PDC) between the pre-LAI and post-LAI periods in each hospital (additional details are provided in the Supplemental Materials).
Sensitivity analyses assessed alternative placement of T0 around inpatient LAI initiation by placing T0 at 7 days after LAI initiation (Figure S1). Additional sensitivity analyses used an alternative, less stringent threshold of number of visits4,5 (at least two at both the pre- and post-LAI period), and an alternative observation period of 6 months around the mirror point. Sub-group analysis by LAI type (first- or second-generation) and visit type (outpatient or inpatient) at index date was conducted as applicable.
All analyses were conducted using SAS version 9.2 (SAS, Cary, NC, USA).
Results
Patient population and LAI utilization
Between 2012 and 2019, there were 25,466 patients with schizophrenia in the PKU6H EHR database and 20,989 in the XJH database. Of these, 1127 and 332, respectively, were prescribed LAI antipsychotics. In total, 98 patients at PKU6H and 59 at XJH were included in the analysis (Figure 1).

Patient selection flowchart.
In the PKU6H, the mean (SD) age of patients was 33.9 years (11.7), 45.9% were male, and 7.1% of patients initiated LAIs during a schizophrenia-related hospital admission. Past mental health-related diagnoses within 90 days prior to the index date were present in 68.4% of patients, of whom 85.1% had other schizotypal and delusional disorder, and 22.4% had major depressive disorder. Among 48.0% of patients with past mental health-related medications, 51.1% were prescribed anxiolytics, and 40.4% were prescribed antidepressants (Table 1). In patients prescribed LAI antipsychotics, 38.8% were initiated on first-generation LAIs of whom 81.6% received haloperidol decanoate. There were 61.2% initiated on second-generation LAIs, of whom 78.3% received paliperidone palmitate. The mean (SD) number of LAI prescriptions was 7.7 (5.8) times, with 30.6% of patients had >10 prescription (Table S2).
Patient demographic characteristics and clinical features at baseline.
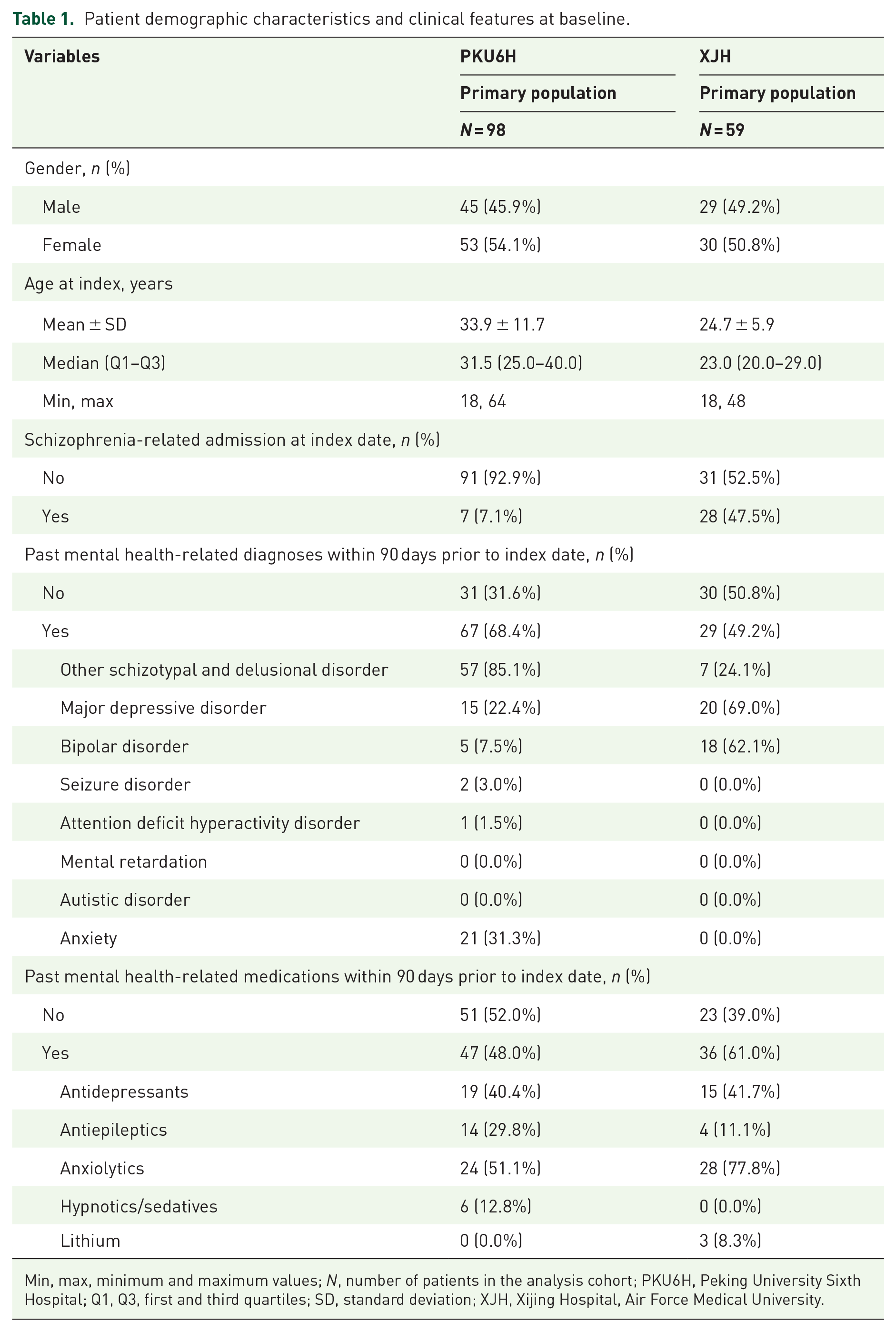
Min, max, minimum and maximum values; N, number of patients in the analysis cohort; PKU6H, Peking University Sixth Hospital; Q1, Q3, first and third quartiles; SD, standard deviation; XJH, Xijing Hospital, Air Force Medical University.
In the XJH, the mean (SD) age of patients was 24.7 years (5.9), 49.2% were male, and 47.5% of patients initiated LAIs during a schizophrenia-related hospital admission. Past mental health-related diagnoses were present in 49.2% of patients, of whom 69.0% had major depressive disorder, 62.1% had bipolar disorder, and 49.2% had other schizotypal and delusional disorder. Among 61.0% of patients with past mental health-related medications, 77.8% were prescribed anxiolytics, and 41.7% were prescribed antidepressants (Table 1). At the index date, all patients at XJH were initiated on paliperidone palmitate. The mean (SD) number of LAI prescriptions were 6.9 (4.3) times, with 32.2% of patients had >10 prescription (Table S2).
Treatment adherence
In each hospital, the proportion of patients with PDC ⩾ 80% was significantly higher in the post-LAI period compared to the pre-LAI period (Table 2). At PKU6H, the proportion with PDC ⩾ 80% was 12.2% in the pre-LAI period and 34.7% in the post-LAI period (p < 0.01). At XJH, the proportion was 1.7% and 23.7%, respectively (p < 0.01).
Treatment adherence and healthcare utilization in pre-LAI and post-LAI periods.
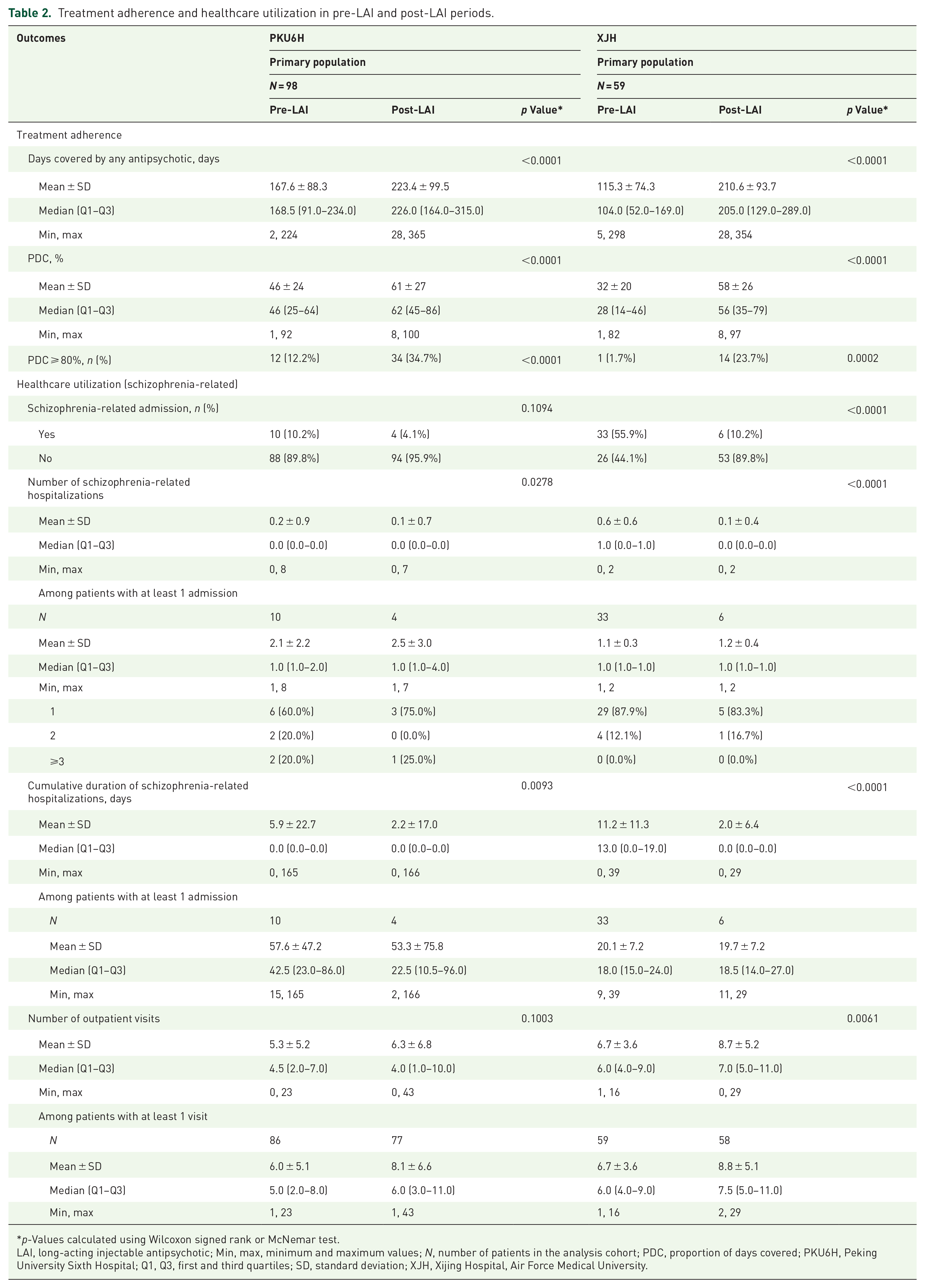
p-Values calculated using Wilcoxon signed rank or McNemar test.
LAI, long-acting injectable antipsychotic; Min, max, minimum and maximum values; N, number of patients in the analysis cohort; PDC, proportion of days covered; PKU6H, Peking University Sixth Hospital; Q1, Q3, first and third quartiles; SD, standard deviation; XJH, Xijing Hospital, Air Force Medical University.
Healthcare utilization
At PKU6H, the percentage of patients who had schizophrenia-related hospital admissions was 10.2% in the pre-LAI period compared with 4.1% in the post-LAI period (p = 0.11). Concurrently, the mean cumulative number of days spent in hospital decreased from 5.9 days in the pre-LAI period to 2.2 days (p < 0.01), and the number of outpatient visits increased from 5.3 times to 6.3 times (p = 0.11). At XJH, the percentage of patients who had schizophrenia-related hospital admissions was 55.9% in the pre-LAI period compared with 10.2% in the post-LAI period (p < 0.01). The mean cumulative number of days spent in hospital decreased from 11.2 days to 2.0 days (p < 0.01), and the number of outpatient visits increased from 6.7 times to 8.7 times (p < 0.01), respectively (Table 2).
Sensitivity analyses
Sensitivity analyses of different T0 placement gave similar results to the main analysis in terms of reductions in hospital admissions and healthcare utilization between the pre-LAI and post-LAI period (Table 3).
Sensitivity analyses for treatment adherence and healthcare utilization in the pre-LAI and post-LAI periods.

p-Values calculated using Wilcoxon signed rank or McNemar test.
LAI, long-acting injectable antipsychotic; Min, max, minimum and maximum values; N, number of patients in the analysis cohort; PDC, proportion of days covered; PKU6H, Peking University Sixth Hospital; Q1, Q3, first and third quartiles, SD, standard deviation; XJH, Xijing Hospital, Air Force Medical University.
Additional sensitivity analyses of patient selection criteria identified 129 eligible patients at PKU6H and 85 at XJH using the alternative requirement of at least two visits of any types at both pre-LAI and post-LAI period, respectively, and 127 and 91 eligible patients, using the alterative pre-LAI and post-LAI observation period of 6 months (Table 3), respectively. The trends of change observed between the pre-LAI and post-LAI periods observed in both sensitivity analyses were consistent with the main analysis, except for the number of outpatient visits, which remained similar over a 6-month observation period.
Subgroup analysis by type of LAI at index and admission status at index
No first-generation LAI antipsychotics were observed at XJH. In the sub-group analysis conducted for PKU6H, patients initiated on first- and second-generation LAI antipsychotics were similar in terms of demographic characteristics and history of schizophrenia admissions in the pre-LAI period. On the other hand, PKU6H had 7 (8%) patients who were initiated on LAI antipsychotics in an inpatient setting at index, whereas at XJH, patients who began LAI treatment in an inpatient setting tended to be more often female, slightly older, and had a higher prevalence of past mental health related diagnoses and medications at baseline compared to those who initiated LAI antipsychotics in an outpatient setting (Tables S3 and S4).
The sub-group analysis of patients at PKU6H suggested similar benefits in terms of improving treatment adherence, lowering hospital admission rates, and healthcare utilization in patients initiated on either first- and second-generation LAI. Specifically, the proportion of patients with PDC ⩾ 80% increased significantly while a decreasing trend of hospital admission and increasing trend of the number of outpatient visits was observed. Similarly, subgroup analysis by index admission shared similar trend with the primary analysis for patients initiated on LAI in both inpatient and outpatient settings, with the changes being more evident for those initiated LAI in the inpatient setting, especially based on data from XJH. However, it is important to note that further evaluation on the inpatient subgroup at PKU6H was limited due to the small sample size (Tables S5–S7).
Discussion
Our study examined LAI utilization in two Chinese hospitals, providing further evidence for the significant clinical benefits of these medications in a real-world setting. There are currently several LAI antipsychotics approved in China for the treatment of schizophrenia; however, drug availability varies across different regions 21 and only second-generation LAIs were available at XJH. The benefits of LAI in improving treatment adherence, and lowering healthcare utilization were consistently observed across the two study hospitals. In both hospitals, the PDC increased significantly after LAI initiation, accompanied by a significant decrease in the number of hospital stays and lowered cumulative days of admission. By contrast, an increasing trend of the number of outpatient visits was observed, possibly due to the need for regular administration of the LAI antipsychotic. The use of LAIs may increase the frequency of contacts between patients and the mental health center, potentially creating a more collaborative and cohesive relationship between patients and doctors, promoting greater adherence and better control of relapses. Regular evaluation and timely adjustment of treatment could improve outcomes, ultimately supporting individuals with schizophrenia by aiding their reintegration into society and resumption of social functioning.
Our results in the Chinese population are consistent with worldwide results from two meta-analyses showing superiority of LAIs over oral antipsychotics in rates of hospitalization,22,23 and evidence of reduced healthcare utilization associated with LAI compared to oral antipsychotics. 21 One study in China showed that compared to oral antipsychotics, LAIs were associated with reduced indirect costs, no significant increase in total costs, and a reduction in the number of hospitalizations and duration of hospitalization. 21 In the subgroup analysis, both first-generation and second-generation LAIs enhanced treatment adherence and reduced overall healthcare utilization. Similarly, initiating LAIs in both inpatient and outpatient settings showed comparable advantages, with a tentative trend toward more significant benefits for those who began treatment in an inpatient setting. However, we emphasize that these subgroup analyses are exploratory and constrained by sample size. Thus, additional studies are needed to confirm these findings.
Compared to patients enrolled from PKU6H, patients with schizophrenia at XJH tended to be younger, had substantially more hospital admissions in the year prior to the index date, and had fewer mental health-related diagnoses but more mental health-related medications. Some of these differences may reflect different clinical settings (dedicated psychiatric hospital vs psychiatric department in a general hospital), and that PKU6H has more patients travelling to it from other provinces. As observed previously,21,24 the age of patients with schizophrenia attending both hospitals was lower than observed in other countries including the United Kingdom, United States, and South Korea.19,25,26 Since the age of schizophrenia onset appears to be similar worldwide,27,28 this difference could reflect Chinese guidelines recommending early initiation of LAI treatment, or other aspects relating to treatment choice not identified in our study.
Despite observing improved treatment adherence following the initiation of LAI antipsychotics, our results indicate that the adoption of LAIs among schizophrenia patients remains low. This finding is consistent with several previous studies on Chinese populations and aligns with another study reporting that China ranks among the lowest in LAI usage rates among 15 Asian countries.12,24,29 The relatively small proportion of patients initiating LAI treatment in our study reflects these broader trends of inadequate treatment and highlights challenges related to the availability, accessibility, and acceptability of mental health care in China. Additionally, a recent nationally representative epidemiological study on depression treatment found that only 9.5% of participants with 12-month depressive disorders received treatment, with a mere 0.5% treated adequately. 30 This observation underscores the common challenges in the mental health management in China. Moreover, our results hinted long-term adherence could be suboptimal in our patient populations, indicating significant remaining challenges in improving treatment adherence. Lower adherence in Chinese patients compared with elsewhere is noteworthy, 24 and points to a need for additional research to understand underlying reasons and investigate the effectiveness of potential interventions that could improve disease control in Chinese patients. As noted in the proposal of upcoming Chinese guidelines for the management of schizophrenia, 31 treatment adherence is intricately linked to drug efficacy and safety, as well as the alignment of perspectives between doctors and patients. We advocate for increased attention to treatment adherence in schizophrenia management and urge comprehensive actions such as enhanced education about the disease and its treatment for both physicians and patients.
This study strengthens the growing body of evidence supporting the clinical utility of LAIs, particularly through our observation of improved treatment adherence and reduced healthcare utilization in Chinese hospitals. This aligns with recent practical guidelines that emphasize LAIs’ role in improving adherence and reducing readmission rates for schizophrenia. 32 Beyond these benefits, recent research suggests LAIs offer broader clinical advantages. A meta-analysis indicates potential benefits in acute schizophrenia treatment with fewer side effects, advocating for earlier LAI use thus improving long-term outcomes. 33 Furthermore, Stahl and Djokic highlight that LAIs offer benefits beyond improving adherence. They suggest that LAIs may help differentiate treatment failures caused by subtherapeutic plasma levels. By ensuring consistent medication delivery, LAIs can maintain stable pharmacokinetic levels and help identify patients who are mislabeled as “treatment-resistant” due to nonadherence or inadequate dosing. 34 The clinical value of LAIs extends to other severe mental illnesses, as demonstrated by Bartoli et al., 35 who showed LAIs effective in improving clinical outcomes, including reduced hospitalizations and relapses in bipolar disorder. Collectively, this evidence, including our findings, underscores the critical role of LAIs in improving medication adherence, reducing hospitalization burdens, and facilitating proactive clinical management for schizophrenia and other mental illnesses.
Strengths of the study are the use of data from two large, regional hospitals from different geographic areas in China with catchment of patients with differing characteristics, offering reasonable representativeness, and reflecting clinical practice. Applying alternative placements of the mirror point increases comparability with existing literature.
Limitation
There are several limitations of the study. First, the mirror image design lacks an independent control group, necessitating caution in interpreting results, particularly in directly comparing the effectiveness of LAIs with oral antipsychotics. Instead, our focus was on assessing the impact of switching to LAIs on treatment adherence and healthcare utilization to reveal their real-world benefits among patients received LAIs. While the mirror image design controls for within-patient variability, it does not account for time-varying effects, which was partly addressed by employing shorter observation periods. It is important to note that the latter may result in overestimation of the improvement particularly among those had poor adherence histories. Similarly, selection bias may exist as patients who switch to LAIs may differ systematically from those who do not, potentially influencing outcome estimations. In addition, the retrospective nature of our study posed limitations in systematically gathering data on other potential confounding variables, such as symptom severity, quality of life, psychosocial interventions, and external social determinants, which may limit the understanding of the broader impact of switching to LAIs. Another potential limitation is that healthcare visits might be underestimated because patients could seek care in other health care facilities. Requiring evidence of health-care encounters as an exclusion criterion helped to mitigate this issue,2,3 as did use of alternative observation periods and sensitivity analyses that obtained consistent results. The generalizability of the results to all LAIs may be limited by the availability of LAIs as well as the number of eligible patients at the hospitals under study. Patients aged over 65 years were not included in our study because of the potential confounding effect of multiple co-morbidities on treatment adherence.36,37 The study cohorts were therefore comprised mainly of younger adults, which may limit its representativeness in older age groups. Another limitation of our study is the challenge associated with determining the optimal placement of T0. We placed T0 at 14 days to account for when LAIs reach peak plasma concentration. Although our sensitivity analyses explored alternative, shorter placements for T0 and consistently showed benefits of LAIs, it is important to acknowledge that the attribution of bed days to the post-LAI period may reflect inefficacies of prior oral antipsychotic treatment or recovery from acute episodes. Consequently, our findings may not fully capture the benefits of LAIs. In addition, our approach with mirror-image design necessitated including only those patients with an established history of schizophrenia, thus excluding those who began LAI treatment shortly after diagnosis. This exclusion limits our findings to patients transitioning from oral medications to LAIs and may not reflect outcomes for those who start LAI treatment earlier in their clinical course. Finally, despite the low use rate of LAIs in China, and our effort in including two large mental healthcare providers in China, it is important to acknowledge that our results may be potentially limited by the small sample size, future study is recommended to validate our findings under broader settings in terms of patient characteristics and available LAIs. Future research should incorporate larger, more diverse cohorts to address these limitations and further explore the impact of LAIs on clinical outcomes, considering confounding variables and regression to the mean phenomenon.
Conclusion
In conclusion, our study demonstrates that LAIs provide notable benefits, including improved adherence and reduced healthcare utilization in Chinese populations with schizophrenia. These findings are consistent with observations in other countries, underlining the universal effectiveness of LAIs despite diverse patient characteristics and clinical settings. Given the global trend of LAIs in improving clinical outcomes, and their currently low usage rate in China, our results advocate for the integration of LAIs into standard practice to optimize treatment outcomes. This integration is particularly relevant in varied clinical settings ranging from specialized mental health centers to general hospitals, where patient characteristics and healthcare resources may differ, yet substantial improvements with LAIs are observed.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251360400 – Supplemental material for Effect of long-acting injectable antipsychotics on treatment adherence and healthcare utilization in Chinese patients with schizophrenia: a mirror-image study
Supplemental material, sj-docx-1-tpp-10.1177_20451253251360400 for Effect of long-acting injectable antipsychotics on treatment adherence and healthcare utilization in Chinese patients with schizophrenia: a mirror-image study by Lei Zhang, Tao Wu, Jingwen Li, Chengxiang Du, Rui Chi, Kun Jiang, Hong Qiu, Yea-Jen Hsu, Wentian Dong, Huaning Wang and Tianmei Si in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253251360400 – Supplemental material for Effect of long-acting injectable antipsychotics on treatment adherence and healthcare utilization in Chinese patients with schizophrenia: a mirror-image study
Supplemental material, sj-docx-2-tpp-10.1177_20451253251360400 for Effect of long-acting injectable antipsychotics on treatment adherence and healthcare utilization in Chinese patients with schizophrenia: a mirror-image study by Lei Zhang, Tao Wu, Jingwen Li, Chengxiang Du, Rui Chi, Kun Jiang, Hong Qiu, Yea-Jen Hsu, Wentian Dong, Huaning Wang and Tianmei Si in Therapeutic Advances in Psychopharmacology
Footnotes
Appendix
Acknowledgements
We thank Yanhua Yang from the Xijing Hospital information department for her contribution to database extraction, cleaning, and validation. We thank Tong Li for her contribution in literature research. We thank Joanne Wolter (independent on behalf of Global Epidemiology, Janssen Research & Development) for medical writing support and production assistance.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

