Abstract
One of the most important challenges in the management of patients with schizophrenia is to ensure adherence to antipsychotic treatment. The contribution of long-acting injectables (LAI) is undeniable in this matter, but there are still some unmet medical needs not covered by these drugs (e.g. quick onset of action for patients with acute exacerbation of schizophrenia). This article summarises the pharmacokinetics, efficacy and safety of Risperidone ISM (in situ microparticles). The aim of this review is to provide information about the potential uses of this new LAI formulation of risperidone for the treatment of schizophrenia, contextualising and diving into the published evidence. Risperidone ISM shows a rapid release which allows achieving within 12 h risperidone active moiety levels similar to those observed in the steady-state for oral risperidone treatment, achieving a mean average concentration of 38.63 ng/mL. The plasma concentration of active moiety achieved by Risperidone ISM comes with a predictable dopamine D2 receptor occupancy above 65% throughout the 28-day dosing period, which is accepted as a threshold for the efficacy of the antipsychotic treatment. This can be associated with the positive efficacy findings throughout its clinical development. In the short term, it provides an early and progressive reduction of symptoms in adult patients with acute exacerbation of schizophrenia without the need for loading doses or oral risperidone supplementation, which could contribute to reinforcing the therapeutic alliance between the patient and the psychiatrist. In addition, long-term treatment was effective, safe and well tolerated regardless of the initial disease severity or whether patients were previously treated with Risperidone ISM during an acute exacerbation or switched from stable doses of oral risperidone. Improvement and maintenance of personal and social functioning and health-related quality of life were observed in each setting, respectively. All these findings endorse Risperidone ISM as a useful and valuable treatment for the acute and maintenance management of patients with schizophrenia.
Introduction
Poor adherence to oral antipsychotics is one of the most important issues in the treatment of schizophrenia,1–3 and could be considered the Achilles’ heel of the efficacy of these antipsychotics. Indeed, it has been associated with relapse and worsening of long-term functional and mental health outcomes.1–3 The association between poor adherence and negative outcomes for many antipsychotics seems to lie within the dopamine D2 receptor occupancy (D2RO) achieved by them, which should range from 65% to 80% to achieve efficacy while minimising the odds of extrapyramidal symptoms (EPSs) such as akathisia. 4 When treatment adherence fails, the risk of symptom worsening increases as D2RO decreases. 5 For this reason, it is essential to ensure adherence to treatment to prevent relapses, which is one of the most important challenges in the management of these kinds of patients. Long-acting injectable (LAI) antipsychotics are capable of decreasing all-cause discontinuation, hospitalisation rates, as well as mortality in relation to oral antipsychotics.6–10 Thus, for LAI antipsychotics, it is also important to ensure that the drug is delivered timely and reliably to achieve the target D2RO of 65%–80% for optimal antipsychotic effect and minimal side effects. 11 Risperidone ISM (in situ microparticles) is a new intramuscular (IM) LAI of risperidone for monthly (every 28 days) administration without the need for loading doses or oral risperidone supplementation. This formulation combines the well-known clinical efficacy of second-generation antipsychotic risperidone with an innovative mechanism of release based on the ISM technology. This technology enables to formation of an in situ solid and stable polymeric matrix system containing risperidone. The drug is reconstituted to an injectable suspension that, after IM injection into either the gluteal or deltoid muscle, precipitates in situ to form a matrix by solvent diffusion to body fluids. This matrix biodegrades slowly, providing a sustained and controlled drug release for up to 1 month. 12 This technology allows for therapeutic plasma levels to be reached rapidly, and maintained over the time interval between administrations, enabling the extended delivery of compounds from the first day of treatment.13,14
In December 2021, the Committee for Medicinal Products for Human Use (CHMP) issued a positive opinion, recommending the granting of a marketing authorisation for this medicinal product, intended for the treatment of schizophrenia in adults. A few months later, in February 2022, the European Commission authorised the commercialisation of Risperidone ISM for the treatment of schizophrenia in adults for whom tolerability and effectiveness have been established with oral risperidone. 15
This article summarises the pharmacokinetics (PK), efficacy and safety of Risperidone ISM. The aim of this review is to provide information about the potential uses of this new LAI formulation of risperidone for the treatment of schizophrenia, contextualising and diving into the published evidence. It is based on previously conducted studies and does not contain any new studies with human participants or animals performed by the authors.
Methods
A literature search was conducted in September 2023 on the US National Library of Medicine’s PubMed.gov database (https://www.ncbi.nlm.nih.gov/pubmed/) using the word ‘Risperidone ISM’. Only full-text articles written in the English language with available abstracts were considered, none of them were excluded and there were no other filters. Taking these premises into account, the search yielded 11 final references. The results were reviewed by the authors for information regarding Risperidone ISM. In addition, 10 conference abstracts were also found through Ovid and Google Scholar search as well as in the main European congresses of schizophrenia held since 2020 (European College of Neuropsychopharmacology [ECNP] Congress, European Congress of Psychiatry of the European Psychiatric Association, or European Conference on Schizophrenia Research [ECSR]), which covers other scientific sources different from that of the published papers. An additional search on clinical trials on ClinicalTrials.gov provided seven records (Table 1). No other literature search was conducted in other databases for the aim of this review.
Clinical trials of Risperidone ISM, as registered at ClinicalTrials.gov.

ISM, in situ microparticles.
Chemistry
Risperidone is a psychotropic agent belonging to the chemical class of benzisoxazole derivatives. 16 It has a strong affinity for dopaminergic D2 and serotoninergic 5HT2 receptors acting as antagonists, which gives risperidone the possibility of improving both positive and negative symptoms of schizophrenia and reducing the incidence of extrapyramidal adverse effects. Hence, its lack of affinity for muscarinic receptors reduces the induction of anticholinergic adverse effects common to most antipsychotic drugs.17,18
As the predominant risperidone metabolite (9-OH-risperidone) has a pharmacological activity similar to risperidone, the antipsychotic active moiety is considered the sum of risperidone plus 9-OH risperidone.
Formulation
The product is available as an integral Drug Device Combination product, containing a powder pre-filled syringe (cyclic olefin polymer syringe with a nozzle cap and plunger stopper composed of chlorobutyl rubber covered with polytetrafluoroethylene) and a solvent pre-filled syringe (cyclic olefin polymer syringe with a tip cap composed of chlorobutyl rubber, and a plunger stopper composed of bromobutyl rubber covered with ethylene-tetrafluoroethylene copolymer). 19
The ISM formulation of risperidone consists of a monthly (every 28 days) multi-component kit ready to reconstitute and administer by IM injection as a homogeneous, viscous, flowable suspension. The kit comprises: 19
- Powder pre-filled syringe containing a sterile powder mixture of risperidone United States Pharmacopeia (USP)/European Pharmacopoeia (Ph. Eur.) grade and biocompatible/biodegradable polylactic and glycolic acid copolymer.
- Solvent pre-filled syringe containing sterile dimethyl sulfoxide USP/Ph. Eur. grade which dissolves the copolymer, but risperidone only partially, giving rise to a suspension for injection.
- Two sterile safety hypodermic needles:
○ One 20G needle × 2” for injection into the gluteus muscle. ○ One 21G needle × 1” for injection into the deltoid muscle.
Pharmacokinetics
To know the benefits associated with Risperidone ISM, it is first necessary to understand the PK characteristics of this LAI. The PK have been mainly evaluated in four phase I studies13,20,21 and one phase II study. 14
The first clinical study conducted was aimed at evaluating the PK, safety and tolerability of two single-ascending doses (25 and 37.5 mg) of Risperidone ISM in 17 healthy volunteers. This study reported no tolerability or safety concerns, achieving mean active moiety plasma concentrations at 2 h and at 30 days post-administration of 4.37 and 2.90 ng/mL, respectively, after the 25 mg injection, as well as 10.43 and 7.34 ng/mL after the 37.5 mg injection. 20 Apart from demonstrating that Risperidone ISM was safe and well tolerated, the subsequent PK PRISMA-1 (phase I) and PRISMA-2 (phase II) studies13,14 confirmed that therapeutic levels were achieved within the first hours after drug administration and the levels were maintained throughout the 4-week dosing period without any loading dose or oral supplementation, regardless of the injection site and without accumulation of the active moiety throughout treatment. 14 In addition, the 75 mg and 100 mg doses tested in these trials were recommended for further studies to evaluate efficacy and safety.13,14
The phase I BORIS study is one of the last PK trials conducted, which evaluated the steady-state comparative bioavailability, safety and tolerability of Risperidone ISM and oral risperidone. 21 In this study, the mean steady-state concentration versus time profiles for risperidone, 9-OH risperidone and active moiety were characterised in stable patients with schizophrenia after repeated oral administration of risperidone 4 mg once daily for 7 days followed by 4 monthly (once every 4 weeks) IM doses of Risperidone ISM 100 mg. The first dose of Risperidone ISM was administered 24 h after the last oral dose, without any washout period. From the first measurement after the first injection (12 h), mean active moiety plasma concentrations achieved similar levels to those observed with the oral treatment in the steady-state and were maintained above the therapeutic threshold (defined as above 7.5 ng/mL 22 ) throughout the dosing period, with a mean Cmin of 24.12 ng/mL during the first month (28 days) of treatment. 21 Besides, the mean (standard deviation (SD)) steady-state plasma concentrations for risperidone active moiety attained after the fourth monthly dose of Risperidone ISM 100 mg were within the steady-state Cmin ss − Cmax ss range observed for oral risperidone in this study, with a mean Cave of 38.63 ng/mL in the steady state 21 (Figure 1).
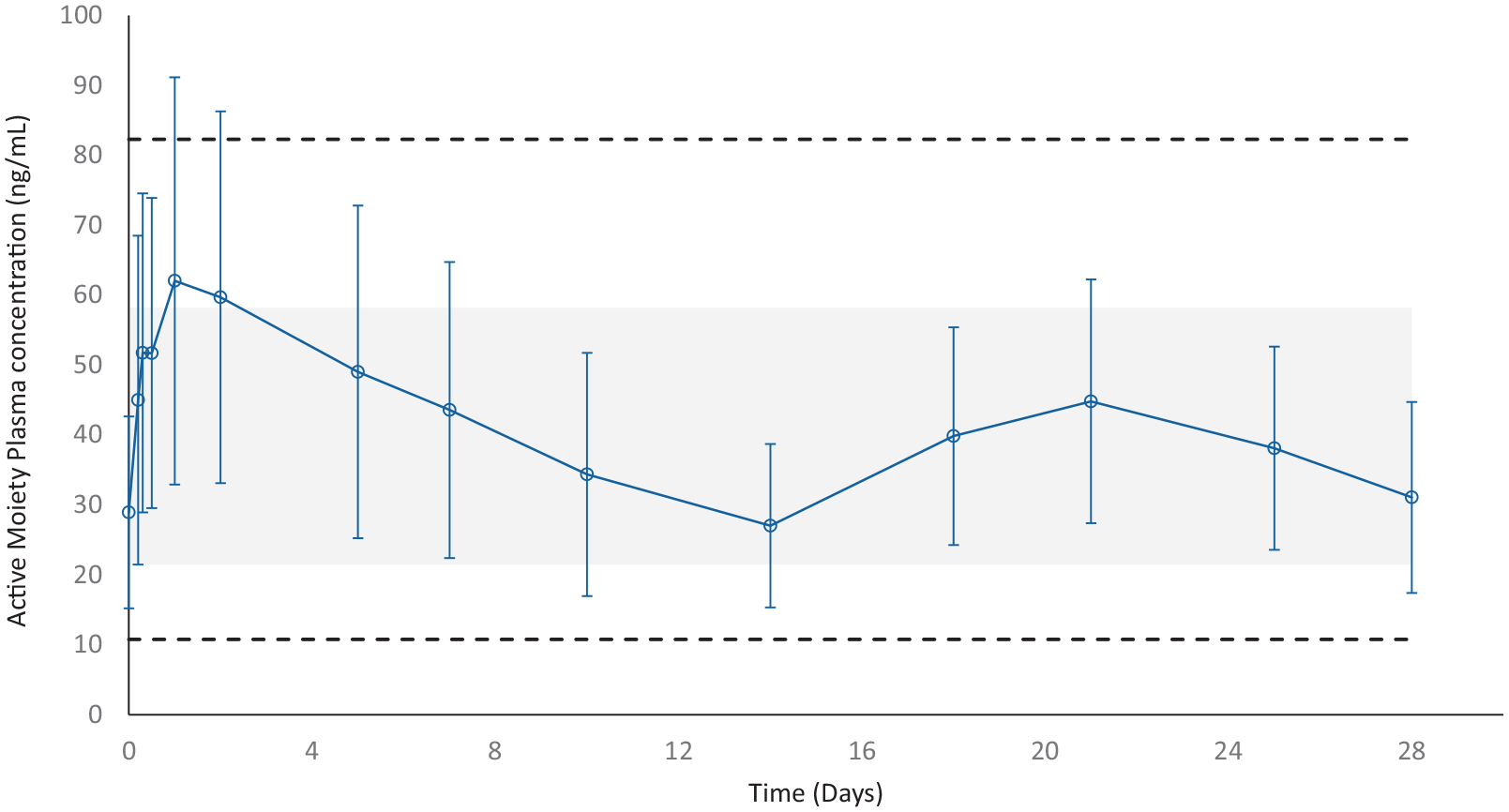
Mean (±SD) steady-state plasma concentrations versus time profiles for risperidone active moiety after the fourth monthly dose of Risperidone ISM 100 mg (PK population).
The results of this study demonstrate that a direct switch is possible 24 h after oral risperidone 4 mg/day to Risperidone ISM 100 mg, without the need for loading doses or oral risperidone supplementation while maintaining steady-state concentrations of the active moiety. In addition, there was no time lag in achieving plasma concentration comparable with the oral formulation after the first dose of Risperidone ISM. This means an important advantage because other atypical LAIs (monthly) antipsychotics, such as paliperidone palmitate (PP), require a loading dose to attain steady-state concentrations when switching from oral treatment to the injectable formulations since they exhibit a more delayed release profile, which results in the need for loading doses and/or oral supplementation upon initiation of treatment.23,24 Besides, with the two ways to initiate treatment with aripiprazole monthly or aripiprazole lauroxil, not only a loading dose but also an oral aripiprazole supplementation is also needed to achieve and maintain therapeutic concentrations during initiation of therapy.24,25 Furthermore, with Risperidone ISM the desired drug levels similar to those achieved with steady-state oral risperidone are maintained throughout the 28-day dosing period (Figure 1). Additionally, safety and tolerability data, along with the PK findings of this study provide further assurances that switching from oral risperidone to Risperidone ISM IM injection treatment is well tolerated and adequate to maintain steady-state active moiety levels throughout the first month and beyond. 21 Similar results were obtained in the twin study BORIS-2, which was conducted after switching from an oral risperidone sourced from a European country. 26
D2RO is considered a key driver of the clinical efficacy and safety of antipsychotic treatment in schizophrenia and is the main proven mechanism of action of risperidone, and therefore its efficacy or lack thereof has to be attributed to it. It is well accepted that D2RO should lie between 65% and 80% for an optimal antipsychotic effect and minimal extrapyramidal side effects, whereby the likelihood of clinical response increased significantly when D2RO exceeded the lower threshold of 65%.4,5 However, D2RO is not the only parameter determining clinical improvement, but knowledge of other parameters and their impact on clinical improvement has not been described as precisely as it has been done for D2 receptors. Additionally, no other pharmacodynamic parameter has obtained a better correlation with improvement in some clinical metrics as the D2RO, particularly striatal occupancy and positive symptoms. 27 And though clinical and preclinical research may have been lacking due to overconfidence in the overall implication of this receptor, there has been some research on the matter, but still no better correlation between any other pathophysiological markers and clinical efficacy (measured by any of the metrics or in any of the different aspects of this disease), at least to the best of our knowledge.
Simulations of D2RO time profiles comparing monthly Risperidone ISM to monthly PP 28 or oral risperidone 29 were performed. They showed that Risperidone ISM is more adequate than monthly PP to achieve and maintain D2RO above 65% as it provides a longer average time of D2RO above the 65% threshold during the first 4 months of treatment. 28 In addition, a similar average time of D2RO < 65% for Risperidone ISM than oral risperidone was obtained in a hypothetical scenario of perfect adherence. Additionally, the simulations predicted a higher average time above 65% D2RO than oral risperidone in realistic clinical scenarios of medium or bad adherence. 29
Efficacy, safety and tolerability in acute schizophrenia
The short-term efficacy, safety and tolerability of Risperidone ISM were demonstrated in the PRISMA-3 clinical trial, a double-blind (DB), randomised, placebo-controlled study conducted in 438 patients with an acute exacerbation of schizophrenia. 30 The participants who had a Positive and Negative Syndrome Scale (PANSS) total score between 80 and 120 at the baseline visit were randomised to receive three IM injections of Risperidone ISM (75 mg or 100 mg) or placebo (1:1:1) for 12 treatment weeks. The primary efficacy outcome was a change in the PANSS 31 total score from baseline to week 12 (Day 85). The key secondary efficacy outcome was a change from baseline in the Clinical Global Impressions-Severity scale (CGI-S) 32 score. Other secondary endpoints evaluated were the Clinical Global Impression-Improvements (CGI-I) 32 score, PANSS subscales score and overall response rate (defined as PANSS total score ⩾30% decrease from baseline to endpoint or CGI-I score of 2 or 1 at endpoint), among others. Safety and tolerability were also assessed.
Superiority of the active treatment over placebo was shown for the primary efficacy outcome (PANSS total score mean change from baseline to Day 85), with a statistically significant advantage of both 75 mg and 100 mg doses of Risperidone ISM versus placebo (Figure 2(a)), with placebo-adjusted differences (standard error, SE) of −13.0 (2.19), (p < 0.0001), and −13.3 (2.21), (p < 0.0001), respectively (Table 2). The key secondary efficacy variable (CGI-S improvement from baseline to Day 85) was also superior to placebo for both doses of Risperidone ISM with a statistically significant mean change of −0.7 (0.13); p < 0.0001, for both doses (Table 2). The improvement for both efficacy outcomes was observed as early as 8 days after the first injection.

PANSS-30 total score: (a) LSM change from baseline at each time point in the double-blind study PRIMA-3 and (b) Mean (SD) total score at each time point in the open-label extension of the PRISMA-3 study.
Efficacy assessment at endpoint (mean change from baseline versus placebo) in the main published studies of Risperidone ISM.

SE.
SD.
CGI-I, Clinical Global Impression-Improvements; CGI-S, Clinical Global Impressions-Severity scale; ECSR, European Conference on Schizophrenia Research; ISM, in situ microparticles; NA, not available; PANSS, Positive and Negative Syndrome Scale; SD, standard deviation; SE, standard error.
In addition, significant differences versus placebo in the mean change from baseline were observed at each assessment time point since Day 8 for PANSS Positive subscale, and Day 15 for PANSS Negative subscale in both Risperidone ISM groups, and since Day 8 for the General Psychopathology subscale in the Risperidone ISM 100 mg group. 30
For the CGI-I scale, differences from placebo were significant at each assessment point from Day 8 and beyond for both Risperidone ISM doses, with placebo-adjusted least square mean (LSM) difference (SE) at Day 85 of −0.8 (0.14) for 75 mg, and −0.7 (0.14) for 100 mg (p < 0.0001, in both groups). 30
For the overall response rate, the difference versus placebo was 39.2% (95% confidence interval (CI): 27.5–49.2) for the 75 mg dose, and 33.8% (95% CI: 22.0–43.8) for the 100 mg dose (p < 0.0001, for both groups) at the end of treatment, which means a number needed-to-treat of three for both doses. 30 Besides, the differences versus placebo were already significant from Day 8 for the 100 mg dose (11.8%; 95% CI: 3.3–20.5; p = 0.005) and from Day 15 for the 75 mg dose (22.0%; 95% CI: 12.1–31.5; p < 0.0001). 30
The effect sizes were also calculated for the change from baseline of the PANSS total score, PANSS subscales (positive, negative and general psychopathology) scores, CGI-S and CGI-I scores in the 75 mg and 100 mg dose groups. 33 Cohen’s d effect size (ES) was determined by calculating the mean difference between the groups and then dividing the result by the pooled SD, with ES = 0.2 being small, ES = 0.5 medium and ES = 0.8 large. 34 The observed ESs for both doses of Risperidone ISM versus placebo were between medium and large relative effects (0.5–0.8). Furthermore, the ES for the CGI-I and PANSS Positive subscale in patients treated with 75 mg and 100 mg, respectively, almost reached 0.8, while for the General Psychopathology subscale, it was 0.72 in both doses. Similarly, the PANSS Negative subscale ES approached medium in size with both doses (Figure 3).

Effect sizes for change from baseline on each efficacy assessment conducted in PRISMA-3 study.
The efficacy of Risperidone ISM was also evaluated in a sub-analysis of the PRISMA-3 study, which included patients with the most severe psychotic symptoms (baseline PANSS total score ⩾95). 35 A total of 213 patients met this criterion, with a global mean (SD) baseline PANSS total and CGI-S scores of 102.1 (5.54) and 5.1 (0.49), respectively, with 93% of patients rated as markedly or severely ill (CGI-S = 5 or 6). The PANSS total score (mean difference, 95% CI) improved significantly from baseline to Day 85 with both doses of Risperidone ISM with LSM difference (SE) versus placebo of −13.8 (2.90); p < 0.0001, and −15.6 (2.91); (p < 0.0001), respectively (Table 2). The statistically significant improvements were achieved by Days 8 and 15 for the 100 mg and 75 mg doses of Risperidone ISM, respectively. Likewise, significant differences versus placebo in the mean change from baseline for all three PANSS subscales (positive, negative and general psychopathology) were observed at Day 85 with both doses of Risperidone ISM (−3.9 (0.97), p < 0.0001 and −5.1 (0.97), p < 0.0001 for 75 mg and 100 mg, respectively); (−2.5 (0.79), p = 0.002 and −2.7 (0.80), p < 0.0001 for 75 mg and 100 mg, respectively); (−7.1 (1.54), p < 0.0001 and −7.7 (−1.55), p < 0.0001 for 75 mg and 100 mg, respectively) (Table 2).
In this sub-analysis, the CGI-S score LSM difference versus placebo ((SE), 95% CI) from baseline to Day 85 was: −0.8 (0.17) (−1.1 to −0.4; p < 0.0001) for 75 mg and −0.8 (0.18) (−1.2 to −0.5; p < 0.0001), for 100 mg group. LSM change since Day 8 was significantly greater with both doses of Risperidone ISM compared with placebo.
In a post-hoc analysis, a validated short version of the 30-item PANSS scale, the PANSS-6 scale, 36 was used for evaluating the short-term efficacy of Risperidone ISM in patients enrolled in the PRISMA-3 study and to confirm if results obtained with this abbreviated scale were consistent with those previously obtained with the PANSS-30 scale in the pivotal study. 37 For this analysis, the six items included in the PANSS-6 were selected from the data originally obtained with the PANSS-30, that is, three positive (P) and three negative (N) symptoms: P1-Delusions; P2-Conceptual disorganisation; P3-Hallucinations; N1-Blunted affect; N4-Social withdrawal; and N6-Lack of spontaneity and flow of conversation. 36 A statistically significant difference was observed in both Risperidone ISM 75 mg and 100 mg treatment groups versus placebo, on both the PANSS-6 and PANSS-30 scores when assessing the mean change from baseline to Day 85 (Table 3). A statistically significant improvement in PANSS-6 score mean change against placebo was shown as early as Day 8 for Risperidone ISM 100 mg (LSM difference, 95% CI: −1.3, −1.9 to −0.7; p < 0.001), and from Day 15 for Risperidone ISM 75 mg (LSM difference, 95% CI: −1.7, −2.5 to −1.0; p < 0.001). These differences remained statistically significant until the end of the study (Figure 4).
Mean change in PANSS-6 and PANSS-30 scores from baseline to Day 85 in the phase III study PRISMA-3.

Source: Originally published by Correll et al. 37
CI, Confidence interval; LSM, least square mean; PANSS, Positive and Negative Syndrome Scale; SD, standard deviation.
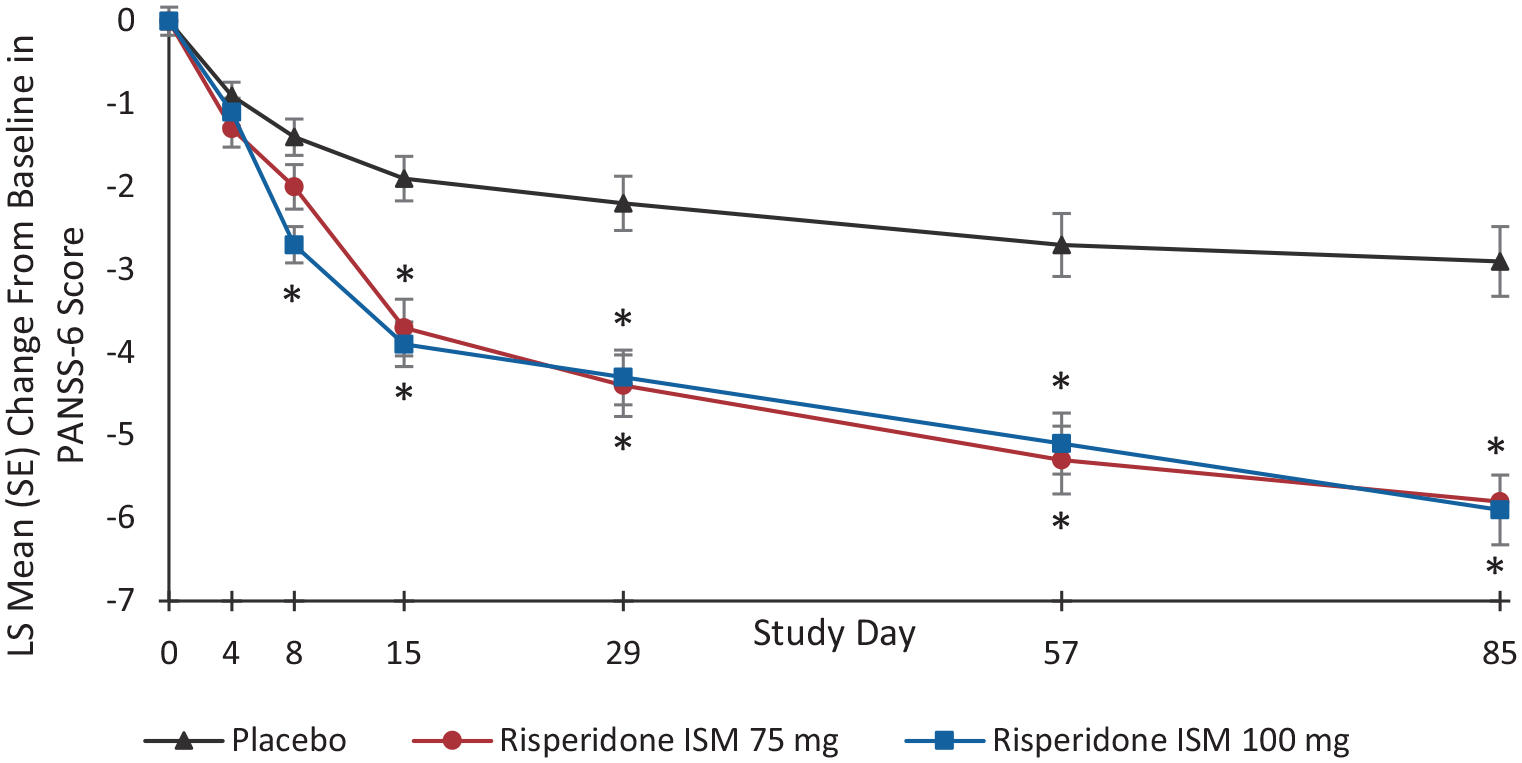
LSM change from baseline in PANSS-6 score at each time point.
Regarding safety and tolerability in the acute setting, at least one treatment-related treatment-emergent adverse event (TEAE) was reported in 41.7% and 52.7% of participants in the study PRISMA-3, who received 75 and 100 mg of Risperidone ISM, respectively, (vs placebo with 21.8%). The most frequently reported drug-related TEAEs in both Risperidone ISM groups (75 and 100 mg) were blood prolactin increase (9.0% and 14.4%), hyperprolactinaemia (5.6% and 8.9%), akathisia (3.5% and 7.7%), headache (6.3% and 3.4%) and weight increase (3.5% and 4.1%). Neither new nor unexpected relevant safety information was recorded and discontinuations due to TEAE were low (7.5% with placebo, 4.2% with Risperidone ISM 75 mg and 6.2% with Risperidone ISM 100 mg group).
Prolactin values decreased in the placebo group from baseline to end of treatment, whereas they increased in both Risperidone ISM groups, with mean (SD) endpoint prolactin levels of 220.6 (257.4) mIU/L with placebo, 875.4 (1080.7) mIU/L with Risperidone ISM 75 mg and 904.8 (810.6) mIU/L with Risperidone ISM 100 mg.
Injection site pain was assessed using a 0–10 Visual Analogue Scale (VAS) score, considering “no pain” on the left end (=0) of the scale and the “worst pain” on the right end of the scale (=10). A median pain value of 2 was reported in all study groups. In 12 (8.3%) and 14 (9.6%) patients treated with 75 mg and 100 mg, respectively, an injection site reaction (ISR) was seen, and redness (6.2%) and swelling (1.8%) were the most frequently reported. 30
The Abnormal Involuntary Movement Scale (AIMS), 38 Barnes Akathisia Rating Scale (BARS) 39 and Simpson-Angus Scale (SAS) 40 were used to assess EPS, observing that treatment groups were comparable and relevant changes were not observed from baseline to the end of treatment in any treatment group (Table 4). 30
Mean change from baseline to end of treatment in the EPS-related scores over the 12-week DB and 1-year OLE phases of PRISMA-3 study.

Source: Originally published by Filts et al. 48
AIMS, Abnormal Involuntary Movement Scale; BARS, Barnes Akathisia Rating Scale; DB, Double-Blind; EPS, extrapyramidal symptom; ISM, in situ microparticles; OLE, Open-Label Extension; SAS, Simpson-Angus Scale; SD, Standard deviation.
All these short-term data demonstrate that monthly IM administration of Risperidone ISM 75 mg or 100 mg can be an effective therapeutic strategy for patients with schizophrenia suffering an exacerbation with severe or moderate psychotic symptoms. The fact that the onset of significant improvement in PANSS total and CGI-S scores mean change was shown as early as Day 8 after the first injection of the 100 mg dose and was maintained until the end of treatment period indicates that Risperidone ISM could address an unmet medical need. It could be used early as antipsychotic therapy upon admission of adult patients with acute exacerbation of schizophrenia for a rapid and effective reduction of severe or moderate psychotic symptoms without the need for oral supplementation or loading doses. In fact, Risperidone ISM obtained a higher placebo-adjusted reduction of the PANSS total score than the SC LAI risperidone in a similar acute schizophrenia study. 41
Likewise, consistent results were demonstrated in the PANSS positive, negative and General Psychopathology subscales. For the PANSS positive and negative subscales, statistical differences in mean change were also obtained as early as Day 8 and Day 15, respectively, demonstrating that Risperidone ISM may rapidly resolve not only positive symptoms but also negative symptoms. These results contrast with those obtained with a different long-acting risperidone, where negative scores were not significantly different across the treatment and placebo groups. 41
These findings, together with the ESs obtained, demonstrate that both Risperidone ISM doses have a clinically meaningful effect on symptom improvement in moderately to severely ill patients suffering an acute exacerbation of schizophrenia, without the need for oral antipsychotic supplementation or loading doses. This observation, along with the statistically significant results previously published, further strengthens the evidence for Risperidone ISM as a new monthly injectable treatment option with the necessary features to cover the existing unmet medical need for an LAI antipsychotic with a quick onset of action for adult patients with acute exacerbation of schizophrenia.
Furthermore, PANSS-6 results were consistent with the previously published PRISMA-3 study based on the PANSS-30 score, showing that Risperidone ISM provides rapid and progressive reduction of symptoms in patients with acute exacerbation of schizophrenia.30,37 Since the brief PANSS-6 rating scale can be administered using much less time compared to the complete PANSS-3042,43 it is an important potential tool for assessing the efficacy of antipsychotics, including Risperidone ISM, and for use in clinical practice, which can contribute to the improvement of the therapeutic alliance between patients and health professionals.
In addition, the effect shown in the markedly to severely ill subpopulation (⩾95 PANSS total score at baseline) further demonstrates the robust efficacy of Risperidone ISM, particularly with the dose of 100 mg, which at Day 85 achieved a particularly large reduction over placebo of 15.6 points in PANSS total score, as well as a rapid improvement of positive and negative symptoms. This finding provides further evidence of the potential value of Risperidone ISM even for the more severely ill population who may need to be hospitalised due to the seriousness of their symptoms.
Both doses of Risperidone ISM were generally well tolerated, and the adverse events observed during the 12-week treatment period were consistent with those reported in previous studies,13,14 as well as with those expected for oral and LAI risperidone at therapeutic doses. 44 The rate of discontinuation due to TEAE was lower in Risperidone ISM treatment groups than in placebo. No remarkable differences between treatment groups were seen in pain score, and EPS were similar between placebo and Risperidone ISM treatment groups. Even though the events related to prolactin increase were included in the most frequent TEAEs, their incidence and the prolactin levels reported in this short-term study were similar to those observed with other risperidone formulations. 45
On the other hand, further examinations under real clinical conditions of established effects are worthwhile once a new medicinal product is approved. Accordingly, a multicountry non-interventional study is ongoing. 46 The RESHAPE study will provide real-world data on the use of Risperidone ISM for the treatment of adult patients who are hospitalised due to a schizophrenia relapse and the way these patients are managed under daily clinical practice conditions by psychiatrists from several European countries.
Long-term efficacy, safety and tolerability as maintenance treatment in schizophrenia
A multicentre OLE of the PRISMA-3 study was conducted to assess the long-term efficacy and safety of Risperidone ISM in patients with schizophrenia, independent of the stability degree. Therefore, long-term treatment with Risperidone ISM was evaluated in patients either coming from the placebo or Risperidone ISM arms of the acute 12-week study or de novo enrolled patients stabilised on oral risperidone. 47 Eligible placebo (unstable) and Risperidone ISM (stabilised) rollover patients from the previous 12-week DB phase as well as de novo stable patients received once-a-month IM injections of Risperidone ISM 75 or 100 mg for 12 months. The long-term efficacy assessment included the PANSS, CGI-S and CGI-I scales. Safety evaluation involved TEAEs, ISRs, laboratory tests and several safety scales.
A total of 215 patients entered the study (55 unstable, 119 stabilised and 41 stable patients), and most of them (74.9%) completed it. The discontinuation rates were similar across the study subgroups, and the primary reason for discontinuation was withdrawal of consent (12.1%). PANSS total scores decreased from baseline to the endpoint in all groups, with a mean (SD) PANSS total score of 58.7 (10.68), 57.6 (10.87) and 57.0 (8.13) in the unstable, stabilised and stable patient groups, respectively, at the end of treatment (Figure 2(b)). The greatest decrease was shown for unstable and stabilised patients, reporting at study Day 365 a mean (SD) PANSS total score reduction of −20.9 (14.40) and −9.8 (13.88), respectively (Table 2). Similarly, PANSS subscale (Positive, Negative and General Psychopathology) scores decreased also from baseline to the endpoint in all study groups, with the largest decreases observed also in the unstable patients: −6.3 (5.05) for Positive, −3.7 (4.58) for Negative and −10.9 (7.95) for General Psychopathology subscales, respectively (Table 2). Improvement from baseline to 12 months was also shown for CGI-S and CGI-I scores in both unstable and stabilised patients; the CGI-S and CGI-I scores remained almost unchanged in the stable group (Table 2).
The safety and tolerability of Risperidone ISM were also evaluated in the 12-month OLE. At least one treatment-related TEAE was reported in 39.1% of participants, the most frequent being headache (12.1%), hyperprolactinaemia (9.8%), asthenia (5.1%), weight increase (4.2%), insomnia (4.2%) and akathisia (3.7%). Seven (3.3%) patients had treatment-related TEAEs which led to drug discontinuation. Four of them received Risperidone ISM 75 mg and three received 100 mg.
From a total of 2355 Risperidone ISM injections evaluated, 8 (0.3%) cases of ISR were reported, and the injection site pain score after IM administration was low (below two points from a 0 to 10 VAS) throughout the study. Overall relapse was low, with a rate of 10.7% (95% CI, 6.9%–15.6%), and specifically, only 4.2% of patients required rehospitalisation due to an exacerbation of psychotic symptoms. 47
EPS-related events were evaluated by using the AIMS, BARS and SAS, as well as by the Standardised MedDRA Queries Analysis in a subsequent post-hoc analysis of ‘extrapyramidal syndrome’ (broad search) from TEAEs. 48 Use of concomitant anticholinergic and beta-blocking agents (excluding non-EPS reasons) was also assessed in this subsequent analysis. Altogether, nine (4.2%) patients reported EPS-related TEAEs (i.e. akathisia, extrapyramidal disorder, restlessness, tremor), leading to treatment discontinuation in two cases: due to akathisia in one (0.5%) patient and due to an extrapyramidal disorder in the other (0.5%). Five patients (2.3%) received anticholinergic agents and three (1.4%) beta-blocking agents. The scores obtained using all three scales were comparable between the treatment groups and no relevant changes from baseline to the end of treatment were observed in any of them (Table 4).
To compare Risperidone ISM as a maintenance therapy to other common once-monthly LAI formulations such as aripiprazole monohydrate once-monthly (AOM) and PP, a matching-adjusted indirect comparison (MAIC) was performed for the safety and tolerability endpoints of EPS incidence and use of anticholinergic agents. The MAIC was performed after a systematic literature review to identify treatment studies reporting safety and tolerability for LAI antipsychotic therapies, and to generate comparative evidence for safety and tolerability endpoints between trials. 49 The analysis showed that the incidence of EPS was lower in patients receiving Risperidone ISM than in those receiving PP (odds ratio, OR (95% CI): 0.63 (0.29, 1.38), p = 0.253) or AOM (OR (95% CI): 0.25 (0.12, 0.53), p < 0.001). In addition, use of anticholinergic agents for relieving EPS was also significantly lower in Risperidone ISM patients than in PP patients (OR (95% CI): 0.29 (0.10, 0.83), p = 0.021) or AOM (OR (95% CI): 0.01 (0.003, 0.06), p < 0.001).
Moreover, 12-lead electrocardiograms (ECGs) were performed as scheduled during the OLE study, and treatment-related cardiovascular events were also evaluated.47,50 Absolute values for QT intervals were automatically corrected for heart rate by the ECG machine using both Bazett’s formula (QTcB) and Fridericia’s formula (QTcF). Of the 215 patients receiving Risperidone ISM (75 mg or 100 mg) for 12 months, 3 (1.4%) patients had treatment-related cardiovascular events, but no one was removed from the study for this cause (Table 5). No notable differences were observed between the treatment groups.
Patients with treatment-related cardiovascular adverse events and CFB at endpoint on QT parameters reported over the 12-week DB and 1-year OLE phases of PRISMA-3 study.

Source: Originally published by Litman et al. 50
CFB, change from baseline; DB, double-blind; NA, not applicable; OLE, open-label extension.
One of the most frequent TEAEs reported in the OLE phase of the PRISMA-3 study was hyperprolactinaemia (9.8%). However, only 2 (0.9%) patients stopped treatment with Risperidone ISM because of treatment-related hyperprolactinemia, one of them due to libido decrease and the other due to gynaecomastia). 47
Long-term efficacy of both doses of Risperidone ISM (75 mg and 100 mg), as well as its safety and tolerability over 12 months of treatment, was demonstrated in the extension phase of PRISMA-3 study and the subsequent post-hoc analyses conducted. It should be noted that a very low rate of adverse events leading to treatment discontinuation was reported during the study. In addition, the percentage of patients who completed the study was high (74.9%) and higher than in similar long-term studies with other LAIs, such as biweekly risperidone (50%) or PP (41%), 51 or with monthly subcutaneous LAI risperidone (47%) 52 or aripiprazole lauroxil (68%). 53 As it has been highlighted in a previous publication, the PK profile of this new IM risperidone and its optimised binding kinetics at the D2 receptor 54 may be contributing to an increased tolerability and consequently to the high completion rate observed in this study. 47 In addition, a low incidence of EPS-related TEAEs and a low percentage of patients using beta-blocking or anticholinergic agents were observed during the study. Considering currently available evidence, 55 the prescription of anticholinergic agents must be prudent and only when absolutely needed to control EPS to avoid its side effects. 56 Therefore, Risperidone ISM could be also useful in this context to reduce anticholinergic prescription. Furthermore, the MAIC study confirmed this favourable safety and tolerability profile in terms of EPS incidence and anticholinergic agent use, in comparison to other antipsychotic therapies used for the treatment of patients with schizophrenia in the maintenance setting. 49 Thus, Risperidone ISM showed a good EPS tolerability profile, despite the rapid achievement of therapeutic plasma levels. On the other hand, neither new nor unexpected cardiovascular findings were observed and the changes from baseline in the QT interval were small and not clinically significant, with a very low incidence of cardiovascular-related adverse events. In summary, Risperidone ISM showed also a favourable cardiovascular safety profile.
It should be noted that, despite the expected increase in prolactin levels, interestingly only two patients (0.9%) experienced hyperprolactinaemia-related adverse events that led to treatment discontinuation (one libido decrease and one gynaecomastia), which also confirms the good tolerability profile of Risperidone ISM.
Despite this, caution is needed because a 12-month study does not completely rule out longer-term side effects; however, considering that the active ingredient of this new medicine is risperidone, a very well-known antipsychotic, an abnormally high frequency of adverse effects beyond 12 months is not to be expected, but rather the opposite, considering the safety results observed. 47
Patient well-being and social functioning
Social functioning is considered an integral outcome parameter to evaluate the success of the treatment of schizophrenia. 57 Thus, one of the secondary objectives of the PRISMA-3 study included establishing health-related quality of life (HRQL) and social functioning in patients treated with Risperidone ISM. Social functioning was assessed by the clinician-administered Personal and Social Performance Scale (PSP) 58 and HRQL through the patient-reported 20-item Subjective Well-Being Under Neuroleptics Treatment (SWN-20) scale. 59 Additionally, to assess the short and long-term effects of Risperidone ISM on clinician-administered personal and social functioning and patient-reported subjective well-being, an analysis based on data obtained from PRISMA-3 study was also conducted. 60 In the short-term analysis, the Risperidone ISM group contained pooled data on Risperidone ISM 75 mg + Risperidone ISM 100 mg, and in the long-term analysis, the three patient groups (unstable, stabilised and stable) also contained pooled data on Risperidone ISM 75 mg + Risperidone ISM 100 mg.
Risperidone ISM significantly improved PSP total score from baseline to Day 85 versus placebo with mean change total score (95% CI) of 10.7 (9–12) compared to 4.8 (3–7) for placebo (p < 0.0001) (Figure 5), showing also a significantly greater improvement (p < 0.05) than placebo in all domains of the PSP (except for “Self-Care”) based on clinicians’ assessment. 60 The statistically significant improvement was present from the first measurement time point (Day 29) (Figure 5). SWN-20-measured HRQL also increased on average in the short-term treatment with Risperidone ISM, where the mean (95% CI) change was slightly higher in the active treatment group than in the placebo group at the end of the study (Day 85) (8.6 (7–10) vs 5.6 (3–8); p = 0.0771). 60
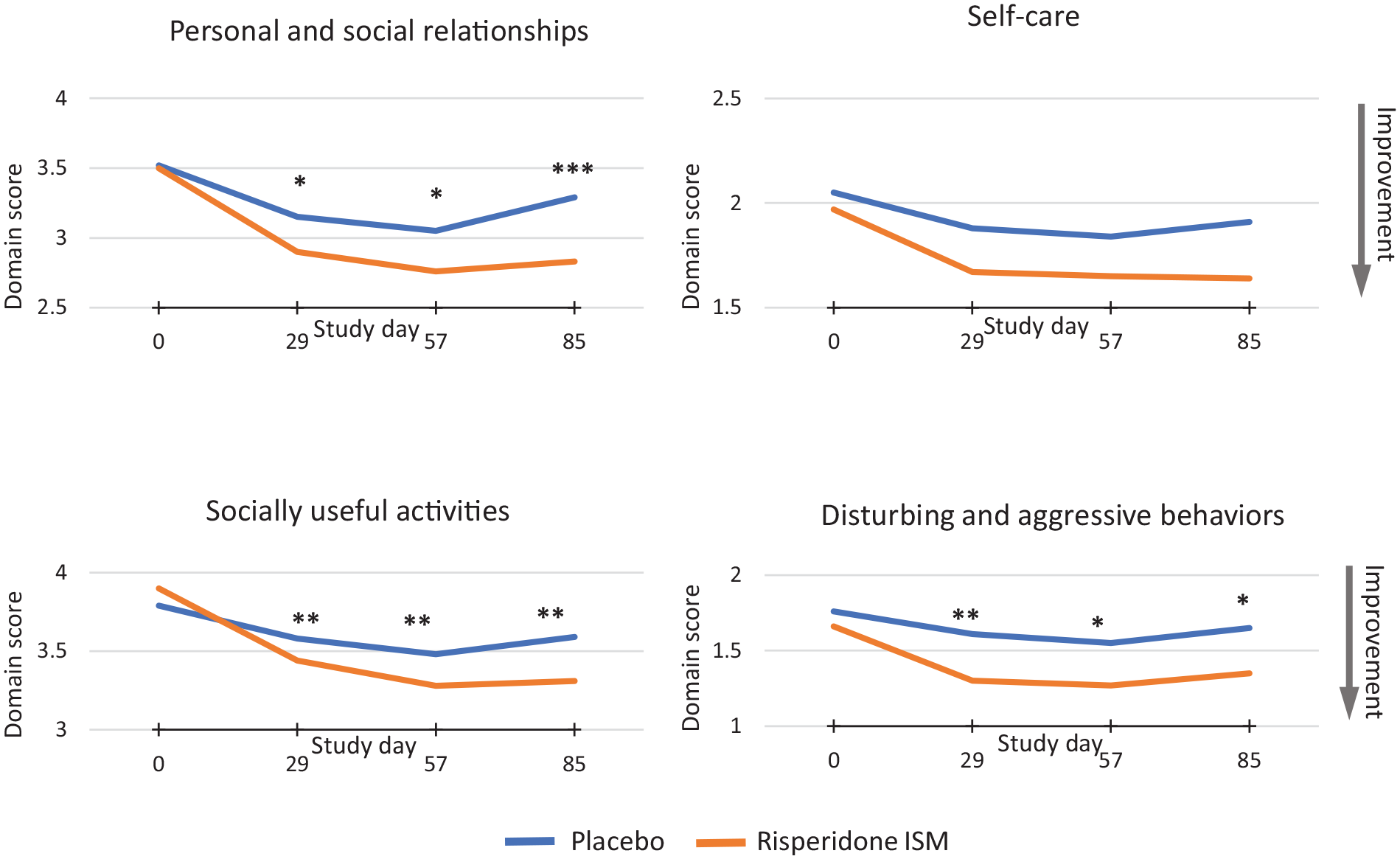
Mean domain scores of the PSP scale at each assessment point during the DB phase.
Likewise, a significant improvement was observed for PSP scores during the long-term treatment in patients transitioning from the DB phase of the study (unstable and stabilised), achieving a mean (95% CI) score of 66.9 (64–70) and 68.2 (66–70) at Week 52, respectively. The stable patients maintained a steady PSP total score throughout the long-term treatment obtaining a mean (95% CI) PSP total score of 73.31 (71–76) at Week 52.
Regarding SWN-20, a mean improvement from baseline to Week 52 was observed for all three groups with a total score mean (95% CI) change of 8.08 (4–12), 4.16 (2–7) and 1.94 (−1 to 5) in the unstable, stabilised and stable patients, respectively. Although the differences were statistically significant in unstable and stabilised groups, the largest improvement was observed in the first, where statistically significant improvements in SWN- 20 total scores were seen at all time points (Weeks 12, 24 and 52) from baseline. 60
Improvement in functioning in patients with acute exacerbation of schizophrenia is a key factor for reintegrating them to their normal life 61 and, in this sense, Risperidone ISM can play a major role in the achievement of this goal in this population. Indeed, a significant improvement in the level of functioning over time, achieved as early as 4 weeks after starting the treatment, and a similar trend regarding HRQL, measured by PSP and SWN-20 scales, was shown in patients treated with Risperidone ISM. Although a period of 12 weeks could have been too short to obtain definitive results about the possible effects of Risperidone ISM on functioning or HRQL, this new LAI has shown a better PSP result compared with that published for other similar LAIs in the short-term treatment.62,63 On the other hand, the long-term benefits of this drug were also observed, especially in the unstable rollover participants from the DB phase of the study PRISMA-3 but also in patients who were clinically stable with oral risperidone upon entering the long-term study (de novo participants), which maintained the same level of functioning and well-being until the end of the study (12 months), confirming that Risperidone ISM could be an effective and useful treatment for adult patients switching from oral risperidone. 60
Despite the benefits of LAIs, there is some controversy around them (e.g. the long-term prophylactic use, cumulative effects of antipsychotics on physical health and brain structure) 64 and the advantages of these drugs must be weighed against their side effects. Even so, most guidelines and experts recognise the importance of antipsychotic medicinal products for the treatment of schizophrenia.65–67 In fact, long-term treatment with antipsychotics is recommended by clinical practice guidelines65,68 not only because it prevents relapses69,70 and rehospitalisations or mortality,10,71,72 but also because patients benefit in terms of quality of life, functioning and sustained remission. Nowadays, these parameters are the main therapeutic objectives, shifting from the control of acute psychotic and behavioural symptoms to long-term goals such as improving psychosocial functioning and quality of life with the aim of attaining both clinical and personal recovery of the patients. Risperidone ISM could also contribute to improving and maintaining these parameters.
Conclusion
Considering the results of the above-mentioned studies, Risperidone ISM presents PK characteristics that allow a fast release providing sustained therapeutic levels of active drug from the first day, conferring the possibility of directly switching from the oral treatment without loading doses or oral supplementation, in contrast to other currently available LAI second-generation antipsychotics, which do not reach therapeutic levels rapidly, and thus they meet such requirements.23,24,73
The mean average concentration of 38.63 ng/mL attained in the steady state by Risperidone ISM 100 mg is observed in parallel with a consistent D2RO above 65%, which is associated with the clinical efficacy of the antipsychotic treatment for schizophrenia. Predictable achievement of D2RO above this threshold is in line with the observed robust efficacy and safety profile of Risperidone ISM in both clinical settings of patients, those with an acute exacerbation of schizophrenia symptoms, including those with more severe psychotic symptoms, as well as stable patients during the maintenance treatment. Therefore, Risperidone ISM can be used for the clinical management of the acute phase of schizophrenia to rapidly control symptoms, but it is also effective and well tolerated as long-term maintenance therapy. 47 Additionally, this advantage may facilitate the ongoing relationship and therapeutic alliance between patients and psychiatrists that are essential for reliable monitoring of the patient’s progress, 74 favourably affecting the patient’s attitude towards treatment, reducing potential negative perceptions and improving adherence to therapy.
At present, a limited number of studies has been conducted with Risperidone ISM and, therefore, further research would be advisable, such as head-to-head comparisons, meta-analysis or real-world evidence; in fact, one multinational non-interventional study is currently ongoing, 46 and its results will contribute to bringing knowledge of this new medicinal product into routine clinical practice.
In conclusion, Risperidone ISM has been designed to fulfil certain unmet medical needs that other LAIs cannot cover yet. Thus, it could be used as an antipsychotic therapy upon admission of adult patients with acute exacerbation of schizophrenia for an early and effective reduction of psychotic symptoms as well as a maintenance treatment once the patients have been stabilised because it: (I) provides therapeutic levels within the first day of treatment; (II) provides stable and predictable plasma levels from the first dose; (III) predictably achieves D2RO above the threshold for clinical efficacy of 65%; (IV) shows an early efficacy with a rapid and sustained reduction of symptoms (from Day 8 of treatment); (V) has a good safety and tolerability profile in the long term, with a low rate of adverse events, including prolactin-related, EPS and cardiovascular events; (VI) it is easy to use as it does not require concomitant oral supplementation, nor loading doses; just administering a single dose 24 h after the last oral risperidone intake or 2 weeks after the last dose of biweekly risperidone.
Therefore, for all the above-mentioned reasons, Risperidone ISM could be a useful treatment for the clinical management of all phases of schizophrenia.
