Abstract
Background:
Antipsychotic medications are associated with weight gain and metabolic derangement. However, comprehensive evidence for the efficacy of co-commenced pharmacological treatments to mitigate initial weight gain is limited. Metformin has been shown to be effective in reducing weight among people on antipsychotic medications who are already overweight, but the potential benefits of metformin co-commencement in mitigating antipsychotic-induced weight gain has not been systematically reviewed.
Method:
We conducted a systematic review of PubMed, EMBASE, PsychInfo, CINAHL, the Cochrane database, and China National Knowledge Infrastructure from inception to 18 November 2023. We undertook a meta-analysis of concomitant commencement of metformin versus placebo for attenuation of weight gain and metabolic syndrome for people with schizophrenia commencing a new antipsychotic.
Results:
Fourteen studies from Australia, United States, Venezuela, and China with 1126 participants were included. We found that metformin was superior to placebo in terms of attenuating weight gain (−3.12 kg, 95% CI −4.22 to −2.01 kg). Metformin also significantly attenuated derangement of fasting glucose levels, total cholesterol, and total triglyceride levels. Sensitivity analysis on study quality, duration, and antipsychotic agent did not impact the results. Meta-analysis was also conducted on adverse drug reactions (ADR) reported in each study which showed no significant difference in ADR incidence between metformin and placebo groups. Subgroup analysis on antipsychotic-naïve participants and participants switching to new antipsychotic did not impact the results.
Conclusion:
Metformin led to statistically significant and clinically meaningful attenuation of weight gain as well as attenuation of several other metabolic parameters when commenced concomitantly with antipsychotic medications. Co-commencement of metformin with antipsychotic medications, where tolerated, should be considered in the clinical setting with aim to improve long-term cardiometabolic outcomes for patients with long-term need of antipsychotic treatments.
Introduction
Schizophrenia is associated with a 20-year reduction in lifespan compared to the general population. 1 This is driven largely by increased risk of cardiometabolic disease. 1 Risk factors include poor diet, sedentary behavior, smoking, socioeconomic status, reduced use of healthcare and genetic associations between schizophrenia and glucose dysregulation. However, an additional major cause of cardiometabolic disease includes the metabolic adverse drug reactions (ADR) associated with antipsychotic medications. 2
Antipsychotic medications, notably quetiapine, clozapine, olanzapine, risperidone and paliperidone which are all commonly used, have a high risk of metabolic side effects including weight gain, glucose homeostasis dysregulation and dyslipidemia. 2 Clozapine, the most effective antipsychotic for positive symptoms of treatment refractory schizophrenia, 3 is associated with a 6% weight gain in the first year after commencement, 4 and ongoing weight gain for at least 5 years. 5
The prevalence of metabolic syndrome is concerningly high, with over half of people with an diagnosis of schizophrenia in Australia meeting criteria for the syndrome. 6 The prevalence of metabolic syndrome varies according to the medication prescribed, with a prevalence of 28.2% associated with olanzapine, 27.9% with risperidone and 51.9% with clozapine. 7 Metabolic derangement was also found to continue throughout the course of treatment, with the highest rate of weight gain within 6 months of commencing antipsychotic medication. 7 Over and above the cardiometabolic impact, weight gain can compromise treatment adherence leading to psychosis relapse. 8
Metformin, a biguanide antihyperglycemic medication, is the first-line treatment for type 2 diabetes mellitus globally. It has been shown to be effective when used ‘off-indication’ for reduction of weight gain among people receiving antipsychotic medications, who are already overweight or obese.9,10 However, excess weight is hard to lose once gained. While lifestyle interventions can be used to address weight gain, uptake can be challenging and the impact of these interventions often takes time to be observed, as behavioral change can be a slow process.2,11 Therefore, lifestyle interventions alone are insufficient to counteract the rapid and severe weight gain and metabolic disturbance that occurs upon commencement of antipsychotic medication.
There is an urgent need for evidence-based treatments that can prevent weight gain at the time of antipsychotic initiation. We conducted a systematic review and meta-analysis for randomized controlled trials (RCTs) to assess the impact of metformin commenced at time of new antipsychotic initiation to assess its impact on attenuation of weight gain.
Method
Protocol and registration
This meta-analysis was registered with PROSPERO (registration number: CRD42023393377), and performed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses. All data included were previously published in RCTs with ethical approval.
Eligibility criteria:
We included RCTs that investigated the impacts of metformin on adults, who were initiated on antipsychotic medications. It was a requirement that studies specified that metformin was commenced within 7 days of starting a new antipsychotic medication. Studies were required to report baseline and endpoint values, or mean change for parameters measured. These parameters included body weight, body mass index (BMI), and other metabolic parameters, such as fasting blood glucose level (BGL), high-density lipoprotein (HDL), low-density lipoprotein (LDL), total cholesterol, triglycerides, waist circumference, and waist-hip ratio.
Exclusion criteria included studies where participants who were not commenced on a new antipsychotic and metformin concomitantly, had pre-existing metabolic syndrome, type 2 diabetes, or had significant systemic comorbidities.
Search strategy
We searched PubMed, EMBASE, PsychInfo, CINAHL, COCHRANE database, and China National Knowledge Infrastructure for relevant studies up to 18 November 2023. Studies were limited to humans. There were no limits on language. Search terms are listed in Supplemental Appendix 2.
Study selection
Studies were included if they were RCTs. Participants had to be between 18 and 65 years of age, were commenced metformin concomitantly with a new antipsychotic medication, and did not have existing diagnoses of diabetes mellitus or metabolic syndrome. The studies were required to have a metformin intervention arm, and a control arm with or without placebo. Outcome data must have included at least one of weight, BMI or another relevant metabolic parameter including BGL, total cholesterol level, total triglycerides, HDL, LDL, waist circumference, and waist hip ratio.
All studies identified were screened at the title and abstract level by three authors (OY, ML, or TL). Studies that met the inclusion criteria on title and abstract review, or that could not be excluded were reviewed at full text level. Attempts were made to contact corresponding authors of included studies in cases where information was missing. Prisma Flow Diagram is included in Supplemental Appendix 1.
Data collection process
Data extraction was conducted by three independent researchers (OY, ML, and TL). All discrepancies during all stages of study selection, data extraction, and quality assessment were resolved by re-checking source papers. Data analysis was conducted by three authors (OY, ML, and DS).
Data items
Data was extracted on year, country, setting, trial duration, diagnostic tool, gender of participants, mean age of participants, number of participants in control and intervention group, dosage of metformin, whether participants were newly commenced on antipsychotics, as well as the type and dosage of antipsychotics prescribed.
We also extracted data on the following outcomes of interest: body weight, BMI, waist circumference, waist hip ratio, fasting blood glucose, blood insulin levels, triglycerides, HDL, LDL, cholesterol level, and blood pressure.
Outcomes
Where multiple outcome time points were reported in the study, data for the final outcome time point was used. The primary outcome was change in body weight in kilograms. Secondary outcomes were BMI, fasting BGL, HDL, LDL, total cholesterol, triglycerides, waist circumference, and waist-hip ratio.
Study quality
Quality of included studies were assessed using the Joanna Briggs Institute (JBI) Critical Appraisal Tool for Assessment of Risk of Bias for RCTs. Studies were deemed to be of high quality if achieved a score of nine and above out of thirteen. Quality assessment table is included in Supplemental Appendix 12.
Statistical analysis
We used Review Manager version 5 for Windows for meta-analyses where at least two studies had usable data for the outcome of interest. Where possible, outcomes were presented as a mean difference. We used random effects models as we were unable to assume homogeneity among studies. Meta-regression and publication bias analysis was conducted in Comprehensive Meta-Analysis version 4 for Windows.
We conducted sensitivity analyses on study quality, lifetime antipsychotic-naïve participants, participants who were switched to a new antipsychotic, and whether or not the study had a placebo for control group. We undertook a meta regression on dosage of metformin. We explored heterogeneity using the I2 statistic, with I2 > 50% indicating significant heterogeneity. We investigated publication bias using the Egger’s test (Supplemental Appendix 10). Mean body weight for all participants was calculated using a weighted mean, based on the sum of each study group’s mean baseline body weight multiplied by the number of people in each group, divided by the total number of participants. Mean % change in body weight was calculated using the mean difference in change in weight between metformin and control groups divided by the mean body weight.
Mean net change in body weight for metformin and control groups was calculated by subtracting mean baseline body weight from the mean endpoint body weight, then performing a weighted mean calculation for the outcome value, which was multiplied by the number of people in each group, divided by the total number of participants (Figure 1).
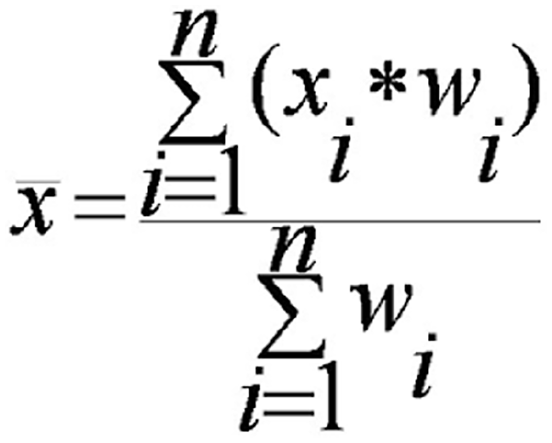
Formula for calculating weighted mean.
Results
Included studies
Following removal of duplicates, 6130 articles were screened at title and abstract level, of which 136 were selected for review at full text level. A total of 14 full text articles met inclusion criteria12–25 (PRISMA Diagram in Supplemental Appendix 1). Studies excluded at full text level, with rationale, are supplied in Supplemental Appendix 11.
Study characteristics
There were 1126 participants with a mean age of 31.35 years (SD 6.68), and 49.86% were male. There was no statistically significant difference between participants on metformin and those in the control arm in terms of age, sex nor mean baseline body weight (Supplemental Appendix 13–Appendix 15). Mean study duration was 15 weeks. The atypical antipsychotics used included risperidone (n = 2),18,25 olanzapine (n = 11),12–17,20–24 and clozapine (n = 1). 19 The maximum dosage of metformin used ranged from 500 mg daily to 2000 mg daily.
In all studies metformin was commenced within a week of commencing a new antipsychotic medication.
Five studies were conducted exclusively in an inpatient setting.13,18,22,24,25 The remaining nine studies were conducted in a mixed hospital and community setting.12,14–17,19–21,23 One study was from Australia, 19 another from Venezuela, 12 one from the United States, 17 and eleven were from mainland China.13–16,18,20–25
The overall study quality was fair; out of the 14 studies, 9 were rated to be of high quality. Measurement of outcome, missing data, and use of statistical analysis were adequately described in all papers. Lack of blinding was the most common issue (Table 1).
Table of included studies.

CCMD, The Chinese Classification of Mental Disorders; DSM, Diagnostic and Statistical Manual of Mental Disorders; ICD, International Classification of Diseases; RCT, Randomised Controlled Trial; yo, year-old.
Body weight
Ten studies had usable data for change in body weight with 709 participants. The mean change in body weight was +2.74 kg (SD 2.55) for the metformin group, and +6.19 kg (SD 3.61) for the control group. There was statistically significant attenuation of body weight gain among the metformin group (−3.12 kg, 95% CI −4.22 to −2.01 kg, p < 0.001, I2 = 58%). With a mean combined baseline body weight of 61.54 kg (SD 12.38 kg), this suggests that metformin was associated with an attenuation of 5.1% of body weight change (Supplemental Appendix 15).
When restricted to studies with participants who were antipsychotic-naïve (n = 560, k = 6), the attenuation of body weight was also significantly greater in the metformin group (−3.28 kg, 95% CI −4.76 to −1.81 kg, p < 0.001, I2 = 74%). The mean baseline body weight for participants who were antipsychotic-naïve was 60.10 kg (SD 2.9), suggesting that metformin was associated with an attenuation of 5.4% of body weight gain (Supplemental Appendix 15). The mean change in body weight was +2.81 kg (SD 2.92) for the intervention group (on metformin), and +6.43 kg (SD 4.59) for the control group.
When restricted to studies with patients who were switched to a new antipsychotic medication (n = 149, k = 4), the attenuation of body weight was again statistically significantly greater in the metformin group (−3.15 kg, 95% CI −4.86 to −1.44 kg, p < 0.001, I2 = 0%). The mean baseline body weight for these participants was 67.94 kg (SD 15.95) (Supplemental Appendix 15), suggesting that metformin was associated with an attenuation of 4.64% of body weight. The mean change in body weight was+2.49 kg (SD 2.30) for the intervention group (on metformin), and +5.26 kg (SD 1.36) for the control group.
Subgroup analysis was conducted on the baseline weight for participants who were antipsychotic-naïve, and participants switching to a new antipsychotic. There was no statistically significant difference between the subgroups for all baseline parameters including body weight (p = 0.89), percentage of male participants (p = 0.87) and age of participants (p = 0.23) (Figure 2, Supplemental Appendix 3).

Meta-analysis figure for weight.
BMI
Twelve studies had usable data for BMI with 394 participants in intervention group and 513 participants in control group. BMI at time of follow-up was statistically significantly lower for the metformin co-commencement group (−1.33 kg/m2, 95% CI −1.81 to −0.84 kg/m2, p < 0.001, I2 = 73%).
Change in other metabolic parameters
Blood glucose level
Fourteen studies had usable data for BGL, with 468 participants in the intervention group and 658 participants in the control group. At time of follow-up, the intervention group had 0.64 mg/dL statistically significantly lower fasting blood glucose than the control group (95% CI −0.89 to −0.39 mg/dL, p < 0.001, I2 = 93%).
HDL
Five studies had usable data for HDL, with 155 participants in the intervention group and 150 participants in the control group. At time of follow-up, there was no statistically significant difference between the intervention and control group.
LDL
Five studies had usable data for LDL, with 155 participants in the intervention group and 150 participants in the control group. At time of follow-up, there was no statistically significant difference between the intervention and control group.
Total cholesterol
Nine studies had usable data on total cholesterol, with 427 participants in the intervention group and 424 participants in the control group. At time of follow-up, people on metformin had 0.53 mmol/L statistically significantly lower total cholesterol than people in the control group (95% CI −0.78 to −0.29 mmol/L, p < 0.001, I2 = 67%).
Triglycerides
Ten studies had usable data on triglycerides, with 435 participants in the intervention group and 429 participants in the control group. At time of follow-up, participants on metformin had 0.25 mmol/L statistically significantly lower triglycerides than participants in control group (95% CI −0.40 to −0.11 mmol/L, p = <0.001, I2 = 65%).
Waist circumference
Six studies had usable data on waist circumference, with 75 participants in the intervention group and 262 the participants in control group. At time of follow-up, the difference between the intervention group and control group was not statistically significant (Table 2).
Summary table of outcomes.

Mean combined baseline weight 61.54 kg, mean combined endpoint weight 65.62 kg.
BGL, Blood glucose level; BMI, body mass index; HDL, high density lipoprotein; LDL, low density lipoprotein.
Subgroup analysis
We conducted subgroup analyses on two subgroups – the antipsychotic-naïve group, and the group which was switched to a new antipsychotic. There was no statistically significant difference between the two subgroups for all outcomes of interest and baseline parameters (Supplemental Appendix 3). We found that the metformin group in both subgroups had statistically significant attenuation of body weight gain and BMI, as well as statistically significantly lower blood glucose and lipid (cholesterol and triglyceride) than the control group (Supplemental Appendix 4 and Appendix 5).
Sensitivity analyses
Restricting the analyses to higher quality studies or those with specified antipsychotic-free periods did not alter the results for any outcomes of interest discussed above (Supplemental Appendix 7). There was also no difference to the statistical significance of outcomes when analyses were conducted on studies that used olanzapine or clozapine (Supplemental Appendix 7). However, when restricted to only studies that used Risperidone (k = 2), the outcome for change in body weight (p = 0.09) and change in BGL (p = 0.14) was no longer statistically significant (Table 3, Supplemental Appendix 7).
Sensitivity analyses outcome for weight.

There was no statistically significant difference between metformin and control groups for incidence of ADRs including nausea, extrapyramidal symptoms, sedation, constipation, tachycardia, dry mouth, headache, hypotension, dizziness, abdominal pain, insomnia, and agitation. There was no statistically significant difference in dropouts between metformin and control groups for either ADRs or all other reasons. Table of meta-analysis outcomes for ADRs and dropouts are supplied in Supplemental Appendix 8.
Metformin dose did not statistically significantly impact on change in weight between intervention and control groups in the meta-regression (Supplemental Appendix 9).
Publication bias
Egger’s regression showed no evidence of publication bias (p = 0.7) (Supplemental Appendix 10).
Discussion
This systematic review and meta-analysis of RCTs focused on the impact of metformin on weight gain prevention when commenced concomitantly with a new antipsychotic. This analysis demonstrated that metformin significantly attenuated weight gain by more than 5% of baseline and BMI among patients initiating antipsychotic treatment. We further categorized participants into two subgroups – the antipsychotic-naïve group, and the group switched to a new antipsychotic. The weight attenuation effect of metformin was similar in both subgroups However, the attenuated weight gain in metformin group where participants were antipsychotic-naïve exceeded the threshold for clinically meaningful weight differences of 5%, while the attenuation of weight gain in the group switching to a new antipsychotic was 4.6%. We also noted a blood glucose and blood lipid lowering effect of metformin in both subgroups.
Although we undertook a meta-regression by dose of metformin, we did not find a statistically significant impact of dose on change in weight. One previous study suggested that 1000 mg metformin may be effective in attenuating antipsychotic-related weight gain. 26 However, the same study found no effect in weight change with metformin 500 mg. Further studies are necessary to clarify whether the weight attenuation effect of metformin is dosage dependent. 27
Further, metformin did not appear to impact HDL and LDL cholesterol, nor waist circumference. HDL cholesterol has been shown to be less sensitive to change with pharmacotherapy. 28 Our findings extend upon previous studies that show improvements in metabolic derangement with metformin use in patients with clozapine-associated obesity. 29 In conjunction with our findings, there is promising evidence to suggest that metformin also has a favorable impact on lipid metabolism. 27
Our findings are in keeping with two recent retrospective cohort studies which found that use of metformin attenuated weight gain among people newly commenced on clozapine.4,29 Our findings are also in keeping with recent Cochrane reviews demonstrating that metformin may have a role in preventing antipsychotic-related weight gain. 26 Previous systematic reviews have found metformin to be effective for reducing prevalent antipsychotic-induced obesity.10,26
Our study had several key strengths. We undertook a rigorous systematic search across multiple international databases, and used strict eligibility criteria to ensure the inclusion of studies where metformin was commenced around the time of antipsychotic initiation. We explored the effects of potential confounders and effect modifiers, such as study quality, prior antipsychotic use, type of antipsychotics, and the presence of placebo. We found no change in the statistical significance of weight change for metformin groups. We also explored the effects of metformin dosage on weight and other relevant metabolic parameters.
Our study had several limitations. Heterogeneity was observed across several outcomes, suggesting potential variability among included studies. As such our results should be treated with caution. There was also a paucity of studies that used antipsychotics other than olanzapine and clozapine, which may have affected the statistical significance of the sensitivity analysis outcome, when restricted to each individual antipsychotic medication. A large portion of the studies were conducted in mainland China, which might limit the generalizability of results to other populations, as indicated by the low mean body weight of participants at inclusion (61.54 kg). Further, most participants included at the start of study were not overweight (mean BMI = 23.17) and remained within the normal weight range by the end of the study. This may not accurately reflect populations of other ethnic backgrounds. Metabolic syndrome sub-component thresholds such as waist circumference vary between ethnic groups. Four studies did not specify the length of the antipsychotic free period prior to commencement of a new antispsychotic which may confound results. Furthermore, the lack of blinding in many studies might have introduced bias. Finally, none of the studies considered or specified the potential impacts diet and exercise may have on the outcomes. This is notable given 9 out of 14 studies were conducted in outpatient settings.
Among people who are obese but not on antipsychotic medications, the efficacy of metformin for weight loss has been mixed. 30 However, among people on antipsychotic medications, metformin should be considered a first-line pharmacological therapy to attenuate antipsychotic-induced obesity. 31 Although not ensuring a weight neutral intervention, our findings suggest that metformin may also have a role in primary prevention of antipsychotic weight gain. Glucagon-like peptide-1 receptor agonists (GLP-1RAs) have shown promise for weight loss among people with existing antipsychotic induced obesity. 32 There may also be a role for GLP-1RAs in prevention of weight gain at time of antipsychotic initiation; however no trials have been conducted to date. Currently, GLP-1RAs are costly and therefore may not be readily accessible to the general public. There remains a paucity of high quality co-commencement studies of antipsychotics and other pharmacotherapies that could attenuate body weight gain and metabolic syndrome.
Conclusion
We found that metformin can clinically and statistically significantly attenuate weight gain and other metabolic changes in individuals starting antipsychotic medications. For clinicians, patients and their carers, this supports the consideration of adding metformin as an adjunctive therapy when initiating antipsychotic treatment. Such an approach can reduce the cardiometabolic risks associated with these medications, potentially improving treatment adherence and overall patient outcomes. For policymakers, these findings should guide treatment protocols or recommendations, emphasizing the importance of prevention of weight gain to improve the cardiometabolic health of people living with schizophrenia.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253241255476 – Supplemental material for Metformin co-commencement at time of antipsychotic initiation for attenuation of weight gain: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tpp-10.1177_20451253241255476 for Metformin co-commencement at time of antipsychotic initiation for attenuation of weight gain: a systematic review and meta-analysis by Ou Yu, Mengyao Lu, Terence K. Y. Lai, Margaret Hahn, Sri Mahavir Agarwal, Brian O’Donoghue, Bjørn H. Ebdrup and Dan Siskind in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
