Abstract
Background:
Cariprazine, a novel antipsychotic drug, is a partial agonist of dopamine D2/D3 receptors with preferential binding to the D3 receptor. There has been an increasing interest in cariprazine augmentation to clozapine; however, the evidence thus far has been only limited to case reports and case series.
Objectives:
To evaluate the efficacy and safety of the augmentation of clozapine with cariprazine in patients with sub-optimal treatment response.
Methods:
Demographic and clinical information of the study population were collected from the electronic records and PANSS scale administered at baseline and 3 months. Tolerability and discontinuation reasons where applicable were also recorded.
Results:
Ten patients (four men and six women) with a mean age of 36.5 years (range = 26–45) were included. Reasons for cariprazine initiation included inadequate treatment response, persistent negative symptoms and/or tolerability issues with clozapine or previous augmentation options. Two patients discontinued cariprazine within the first 6 weeks due to restlessness and poor response, respectively. There was a significant reduction in the median total PANSS score from baseline to 3 months (from 59 to 22.5, p < 0.05), median positive PANSS score (from 11.5 to 5.5, p < 0.05) and in the median negative PANSS score (from 15.5 to 3, p < 0.05) which correspond to a 48%, 33.8% and 65.8% mean score reduction, respectively.
Conclusion:
This is the first pilot study evaluating the effectiveness of clozapine augmentation. The preliminary evidence suggests that this may be a safe and effective practice in patients failing to adequately respond to or tolerate clozapine and/or previous augmentation strategies.
Keywords
Introduction
Cariprazine is a novel D2/D3 partial agonist, with preferential binding to D3 receptors, that has shown robust efficacy in clinical trials. Cariprazine has been widely used in treating psychotic and mood disorders 1 and has also demonstrated superiority over risperidone in the treatment of predominant negative symptoms in schizophrenia.2,3 Furthermore, due to its favourable tolerability profile, cariprazine is often used as a first-line treatment for patients with early psychosis and as a viable alternative to other antipsychotics causing troublesome side effects. 4 In addition, some emerging empirical evidence suggests that it may have a role in the management of treatment-resistant psychosis. 5
Treatment resistance in this patient population remains a challenge 6 with up to 60% even failing to respond to clozapine, 7 the most effective antipsychotic to date for treatment-refractory schizophrenia. 8 In addition, although individual patients with primary negative symptoms may benefit from clozapine treatment in clinical practice, its overall efficacy appears limited in addressing these type of symptoms,9–11 which are some of the most debilitating symptoms in schizophrenia significantly affecting patients’ quality of life and psychosocial functioning. 12
Inadequate response and/or poor tolerability to monotherapy (particularly metabolic side effects) has often turned clinicians to antipsychotic polypharmacy, despite the lack of convincing or consistent evidence on its safety and efficacy. 13 However, augmentation strategies with partial agonists appear to become increasingly popular mediated but not limited by their favourable tolerability. For example, aripiprazole augmentation has been shown to mitigate the cardiometabolic risks of clozapine and may be even effective in reducing positive symptoms. 14 What is more, a recent systematic review and meta-analysis conducted by Guinart and Correll 15 found significant improvements in negative symptoms with aripiprazole augmentation to clozapine.
Similarly, it has been suggested that other partial agonists, such as cariprazine, may be beneficial as an adjunctive treatment to clozapine, particularly when there are prominent negative symptoms. 4 Thus far, only a few case studies16–18 have been reported in favour of the cariprazine and clozapine combination; however, there has not been any larger studies conducted to date. Hence, the aim of this pilot study is to assess the efficacy and safety of cariprazine augmentation to clozapine in patients with suboptimal treatment response.
Methods
Ten patients were prospectively enrolled in this pilot study conducted within a large mental health provider in West London, UK. The study population included adults with any diagnosis receiving cariprazine as an adjunctive treatment to existing clozapine.
Demographic and clinical information of the study population was gathered from the electronic records. Information on previous treatments and the reasons to switch to cariprazine were also collected, alongside information around tolerability and, where applicable, discontinuation reasons. Finally, treatment response was measured using the Positive and Negative Syndrome Scale (PANSS) at baseline and at 3 months. An interval scale of 0 to 6 (instead of a standard 1 to 7 scale) was utilised to measure individual PANSS items. 19 We opted for the 0 to 6 Likert-type scale based on the issues previously raised by Leucht et al., 19 Obermeier et al. 20 and Thompson et al. 21 regarding the underestimation of treatment response when the classical scoring system (1–7) is used in particular when percent change scores need to be calculated, and it has been argued that overall the PANSS should be rescaled. The above authors recommend that 30 minimum points of the total score (including 7 points for positive and 7 points for negative points correspondingly) need to be subtracted when calculating percentage reduction from baseline in order to transform the scale from interval to ratio, which is not relevant in our study with 0 to 6 Likert-type scale use.
Apart from the usual descriptive statistics, Wilcoxon signed-rank test was applied to assess the differences in the median PANSS scores before and after an adjunctive treatment (p-value of 0.05 considered as significant). The percentage decrease in PANSS scores was calculated using the following formula: [(Baseline PANSS score – PANSS score at 3 months)/baseline PANSS score × 100]. All statistical analyses were performed using the statistical software package SPSS 23.0 version.
Results
As shown in Table 1, a total of 10 patients (four males and six females) were included in this pilot study with a mean (SD) age of 36.5 (6.4) years. Ninety percentage of patients had a psychotic illness (six had schizophrenia, three had schizoaffective disorder), and one had emotionally unstable personality disorder with autism. Reasons for cariprazine initiation included inadequate treatment response, persistent negative symptoms, clozapine induced side effects and tolerability issues with previous augmentation strategies. The majority of patients (70%) were on other combination treatments prior to adding cariprazine to clozapine and three were on clozapine monotherapy.
Demographics, clinical characteristics, treatment and response.

ASD, autism spectrum disorder; CLO, clozapine; CPZ, cariprazine; Dx, disorder; EUPD, emotionally unstable personality disorder; PANSS, Positive and Negative Syndrome Scale; PD, personality disorder; SE, side effects; Sx, symptoms; TR, treatment resistance.
Most patients (80%) tolerated cariprazine without any significant concerns and adhered to treatment throughout the observed period. One patient (case 4) developed dizziness on doses above 3 mg; however, this has rapidly improved following dose optimisation. Two patients discontinued treatment between 4 and 6 weeks: case 9 – due to poor response (although the dose was only titrated up to 1.5 mg daily without any further increase) and case 10 discontinued due to restlessness. The remaining patients tolerated the adjunctive treatment well at a median dose of 4.5 mg (min 1.5 mg–max 6 mg).
With regard to the treatment efficacy, there were significant differences in the positive (p = 0.046), negative (0.018) and total (0.012) PANSS scores from baseline to 3 months (Table 2). As shown in Figure 1, the median (min–max) positive PANSS score significantly dropped from 11.5 (0–20, n = 8) at baseline to 5.5 (0–16, n = 8) at 3 months. There was also a significant reduction in the median negative PANSS score from 15.5 (4–35) to 3 (0–28) and the median total PANSS score from 59 (12–102) to 22.5 (4–93). The mean percentage decrease in positive PANSS scores was 33.8%, negative PANSS scores – 65.8% and total PANSS scores – 48%. As demonstrated in Figure 2, the largest and the most prominent percentage reduction across most individual cases can be seen in negative scores with less pronounced reductions in positive and total scores.
Positive, negative, general psychopathological symptoms, total PANSS scores and percentage reduction.

PANSS, Positive and Negative Syndrome Scale.
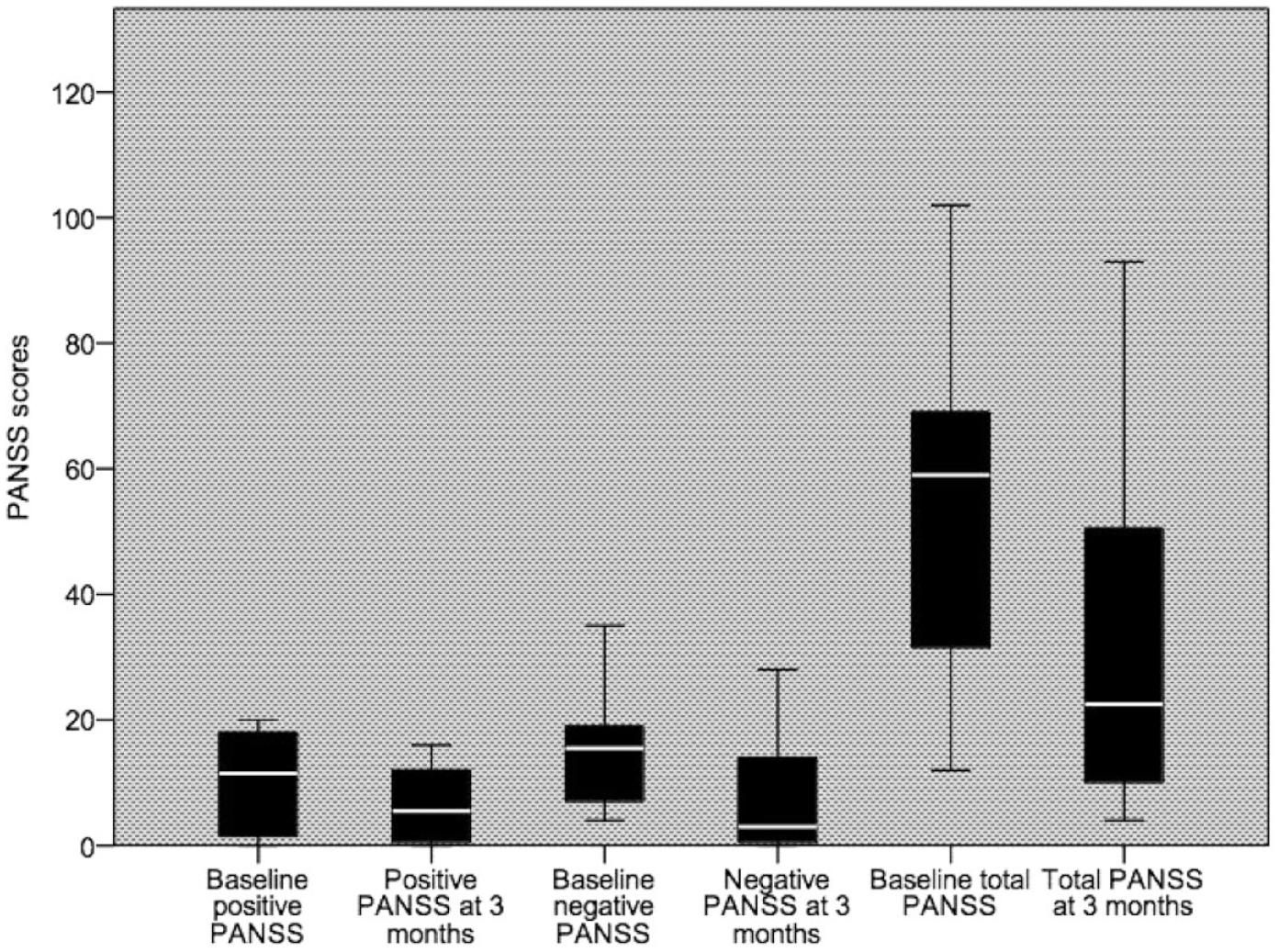
PANSS scores at baseline and at 3 months (median, min–max).

Percentage reduction in PANSS scores from baseline to 3 months.
Discussion
To date, this is the first prospective study to assess the efficacy and tolerability of cariprazine augmentation in patients treated with clozapine. The results have shown a notable effect on symptom improvement, as demonstrated by a reduction across the main PANSS domains from baseline to 3 months, particularly for negative symptoms.
Negative symptoms in schizophrenia remain an unmet treatment target, as the currently approved treatments, including clozapine, mostly treat positive symptoms and/or address secondary negative symptoms.22,23 A recent meta-analysis by Mizuno et al. 11 concluded that clozapine is not superior to other antipsychotics in improving negative symptoms as observed in studies both with strictly-defined treatment-resistant schizophrenia (g = 0.22, p = 0.135) and other non-refractory schizophrenia (g = 0.07, p = 0.262). Growing evidence supports the use of partial D2 agonists such as aripiprazole in combination with clozapine in alleviating not only adverse effects and positive but potentially also to some extent addressing negative symptoms.13,15 To this end, there has been an increasing interest in cariprazine augmentation; however, the evidence thus far has been only limited to case reports and case series.17,18 Nevertheless, and in line with our findings, they too found a marked reduction in negative symptoms in patients treated with clozapine and cariprazine combination. Furthermore, in some cases, the improvements across all PANSS domains allowed clinicians to reduce the dose of clozapine 17 or completely discontinue it leaving a patient on cariprazine monotherapy. 5 Consistent with our study, another case report by Gesi et al. 16 also described a considerable improvement in both positive and negative symptoms following cariprazine and clozapine combination.
Having said that, it is interesting to note that the patient with EUPD did not show any decrease in positive and negative PANSS scores and only a minimal reduction in the total PANSS score, although the use of antipsychotics including clozapine in EUPD continues to be largely debated. 24 On the other hand, a patient with schizoaffective disorder has shown significant improvements across all PANSS domains, which could perhaps be explained by cariprazine’s unique pharmacodynamic profile (D3/D2 receptor partial agonism, 5-HT1A receptor partial agonism, 5-HT2B and 5-HT2A receptor antagonism) and potential mood enhancing and stabilising properties. 25
Only one patient discontinued cariprazine due to adverse effects, that is, feelings of restlessness on a dose of 1.5 mg. Although this particular patient had failed to tolerate low doses of a number of other psychotropic medication previously, newer antipsychotics such as partial agonists and lurasidone have been associated with higher rates of agitation restlessness and akathisia. 26 Cariprazine augmentation was very well tolerated by the rest of the group including where previous treatment options were changed due to side effects or augmentation had been sought because higher doses of clozapine could not be considered due to side effects including due to weight gain or worsening of metabolic parameters. A meta-analysis and systematic review by Mizuno et al. 27 showed that aripiprazole could reduce patients’ weight by 2.13 kg [95% CI: (−2.87, −1.39)] when added to clozapine. A reversal or improvement of the metabolic syndrome with cariprazine 28 has been reported in the literature including when added on to clozapine.17,18 Even though we did not systematically measure any metabolic parameters in our study, individual improvements were noted and the potential beneficial effects of clozapine–cariprazine combination on weight control and metabolic parameters should be explored in future longitudinal studies.
Main limitations of the study include its small sample size, short period to follow-up and heterogeneity of the data, including broad inclusion criteria and potential confounding factors, for example, diagnostic variability and different degree of symptom severity. Furthermore, the findings must be interpreted cautiously as some patients already had relatively low scores at baseline, which could have led to an overestimation of the mean percentage reduction
Conclusion
This is the first prospective pilot study to assess cariprazine augmentation to clozapine. Despite the small number of patients and brief treatment duration, it provides preliminary evidence that this may be a safe and effective practice in patients failing to adequately respond to clozapine and/or not able to tolerate previous augmentation strategies. This study adds to a growing body of evidence supporting the use of cariprazine in reducing not only positive but also negative symptoms in treatment-resistant psychosis. However, further research is needed in this area, preferably involving studies with a randomised, controlled design and adequate sample size.
