Abstract
Background:
Clozapine is the gold-standard medication for treatment-resistant schizophrenia (TRS) yet its initiation is often delayed.
Objective:
To examine whether earlier initiation of clozapine in TRS is associated with lower Clinical Global Impression – Severity (CGI-S) scores at 2 years.
Methods:
This was a retrospective cohort study from electronic health records of patients with first adequate trial of clozapine at the South London and Maudsley mental health service between 1 January 2007 and 31 December 2016. Dates of illness onset and clozapine commencement were manually extracted from anonymised case notes. CGI-S scores were rated blind to illness duration. Ordinal logistic regression was used to describe the association between illness duration at baseline and CGI-S outcome score at 2 years, following adjustment for CGI-S start score and other key covariates.
Results:
Among the 401 patients included, there was an association between illness duration and CGI-S outcome score with a 4% increase in the odds of a higher (worse) outcome CGI-S score per year of illness [adjusted odds ratio (AOR) = 1.04; 95% confidence interval (CI): 1.01–1.06]. The association between illness duration and clozapine response was most marked at less than 4 years illness duration. There were too few clozapine initiations within the first 2 years of illness to draw any conclusions about early clozapine initiation.
Conclusion:
Initiation of clozapine within 2–4 years of psychotic illness onset offers the best outcome for TRS, but the advantage, if any, of earlier initiation is unclear from these data.
Introduction
Schizophrenia most commonly manifests in late adolescence or early adult life, a time of significant growth in social and role functioning. Severe mental illness occurring in late adolescence and early adulthood can result in significant personal, family, and societal burden. Early intervention may improve outcomes, and the ‘critical period hypothesis’ 1 argues that improving long-term trajectory depends on the prompt initiation of effective interventions during a critical window, potentially lasting 3–5 years after illness onset.2–7 While some people with schizophrenia develop a relatively mild illness and recover most or all of their premorbid functioning, outcomes vary, and around 25% of patients are found to be treatment resistant.8–11 Treatment-resistant schizophrenia (TRS) is typically defined as a failure to respond to two antipsychotic trials at an adequate dose for an adequate duration. 12 Clozapine has long been the gold-standard medication for TRS, 13 and its superiority has been confirmed in randomised controlled trials (RCTs) and meta-analyses thereof14,15 as well as in a number of large pharmacoepidemiological studies.16–21 However, despite the clear rationale for clozapine, its use continues to be delayed, often for decades.10,22–26 Non-evidence-based treatments are frequently trialled ahead of clozapine, including prescribing antipsychotic drugs above their licenced limits and antipsychotic poly-pharmacy;24,27 both approaches are associated with potential for increased risk of adverse effects and questionable benefit.24,28
First episode treatment studies indicate that antipsychotic medication may be more effective when given earlier in the course of illness, with lower doses required for first episode schizophrenia compared with treatment of relapse.29–31 If clozapine were to be used earlier in the course of TRS, it is possible that it would be more effective than if its use is delayed. There is increasing interest in the concept of clozapine delay (time from onset of treatment resistance to treatment with clozapine) 32 and recent observational studies have found a relationship between duration of clozapine delay and outcome.33,34 However, there is evidence that treatment resistance is most often present from illness onset 9 and that a substantial proportion of patients may be treatment resistant on grounds of having persistent psychotic symptoms but fail to meet the typical TRS threshold due to not being prescribed two antipsychotic medications. 10 Also the point at which different patients would meet TRS criteria is likely to vary substantially depending on the duration of each antipsychotic treatment they receive. For these reasons, the interval between the onset of psychotic symptoms to introduction of clozapine may be more clinically relevant than the interval between reaching criteria for treatment resistance and clozapine initiation.
This study examines whether time from onset of psychotic symptoms to commencement of clozapine is associated with degree of response to clozapine. In keeping with the ‘critical period hypothesis’, we predict that earlier treatment with clozapine will be associated with a greater effect.
Material and methods
The study was a retrospective cohort study using data from the South London and Maudsley NHS Foundation Trust (subseuqently referred to as the Maudsley in the paper) case register, which comprises complete anonymized patient electronic records from 1 January 2007 onwards. Data can be accessed by researchers using the Clinical Records Interactive Search (CRIS) system for which methodology has been described elsewhere.35,36 The Maudsley serves a population of approximately 1.2 million people from the London boroughs of Lambeth, Croydon, Lewisham, and Southwark.
Sample identification
Searches using a combination of structured data and free text were used in order to identify all patients aged between 18 and 65 years who may have initiated clozapine within the trust. Structured data fields used were the medication table from the patient record, which records drug name, start and stop dates, but is often incomplete, supplemented by information from the trust pharmacy databases which records dates and quantities of clozapine dispensed. In addition, a natural language-processing application was built using general architecture for text engineering (GATE) (for description of methodology see Hayes et al. 37 ) to search free text for instances of clozapine with contextual information indicating actual use of clozapine at that time.
Patients were included in the initial sample if the first clozapine instance was recorded between 1 January 2007 and 31 December 2016. They were excluded if their first clozapine instance was under the National Psychosis unit, as this is a specialist tertiary service focussing on treatment refractory or medically complex patients, drawn from a national catchment area, with follow-up typically outside the trust.
Data extraction
Records were manually searched by reading progress notes and correspondence. Clozapine start and stop dates were recorded to identify all patients who had their first adequate trial of clozapine during the defined study period.
A subset of patients had already been included in a previous data set of first clozapine use, 38 and for these patients, clozapine start dates were taken from the existing database.
An adequate trial was defined as clozapine treatment duration of least 6 weeks in line with current National Institute for Health and Care Excellence (NICE) guidance for prescription of antipsychotic medication. 39 To determine whether this was a first clozapine trial, notes and correspondence were screened for any reference to previous use of clozapine. If clozapine had been prescribed previously, the patient was excluded unless it was apparent that the clozapine had been given for less than 6 weeks. If patients under the care of the Maudsley subsequently commenced clozapine at a non-Maudsley site, they were included if they remained under care-coordination by Maudsley clinicians.
For each patient identified as having their first adequate clozapine trial, progress notes and correspondence were reviewed to ascertain whether they were still under Maudsley services and taking clozapine at 2 years. Patients who had discontinued clozapine and had not restarted within 2 years were excluded from the study, as were patients who had moved out of area or had died during this time period.
Date of first onset of psychosis was identified by examination of clinical notes and correspondence. Notes were scrutinised from first contact with the Maudsley onwards until a record of first date of contact with mental health services for a psychotic episode was found. Dates were recorded to the nearest month. A random number generator was used to assign a month if only the year of onset of psychosis was available.
Patients were excluded if the year of onset of psychosis was not recorded.
A summary of the sample identification process is provided below in Figure 1.

Identification of sample.
Outcome
The outcome variable used in the study was the degree of illness severity at 2 years as measured by the Clinical Global Impression – Severity scale (CGI-S). 40 (for copy of the scale, see Appendix 1 Supplementary Material). CGI-S is rated from 1 to 7, with lower scores indicating lower levels of symptomatology and a CGI-S score of 1 meaning that no symptoms of illness are present. CGI-S was assessed retrospectively by reviewing patient records. Scores were assessed at both the start and end of the study period, so that CGI-S outcome scores could be adjusted for start scores in the analysis.
Ratings were carried out by an experienced consultant psychiatrist blind to illness duration (RJ). Start and outcome CGI-S scores were rated at different sittings and using separate searches, with records for outcome scores restricted to the time period 6 months before and 6 months after the 2-year endpoint.
Predictor variables
The primary predictor variable for the study was the duration of psychotic illness prior to commencement of clozapine. This was obtained by subtracting illness start date from date of first clozapine prescription.
Additional predictor variables included:
Age at first presentation with psychosis.
Sex.
Ethnicity (UK census categories collapsed into four groups reflecting demographics of catchment area – White, Black Caribbean, Black Other, Mixed/Other).
Deprivation score, obtained by linkage of location variable (LSO A11) to Index of Multiple Deprivation 2015 (IMD15) 41 where a higher score indicates a greater level of deprivation.
Coded ICD-10 substance misuse diagnosis (F10–F19).
Clozapine start date (by 2.5-year increments) to account for cohort effects during the 10-year inclusion period.
Number of medical hospital admissions during the follow-up period (0.1 or >1) as an indicator of medical co-morbidity.
Statistical analysis
Stata, version 15, was used for all analyses. 42
Ordinal logistic regression was carried out to test for an association between duration of illness prior to clozapine and CGI-S outcome score. The results were displayed as odds ratios (ORs) to indicate the ratio of the odds at any cut-off of being in a higher versus lower CGI-S outcome score as the predictor variable changed. Two regression models were conducted, the first using illness duration as a continuous variable (time in years) and the second where duration was presented as a categorical variable (illness duration 0–2 years, 2–4 years, 4–6 years, 6–8 years, 8–10 years, 10–15 years, and greater than 15 years). Both models were adjusted for illness severity at baseline (CGI-S baseline scores), age at illness onset, deprivation score, gender, substance disorder, ethnicity, clozapine start date, and medical admissions during follow-up. We compared the Akaike Information Criterion (AIC) 43 statistic to choose which to use as our primary model.
Results
Of the 407 patients included in the study, outcome data were available for 401 patients. The remaining six patients did not have sufficient notes available to complete either a pre- or post-CGI score and were excluded from the analysis.
Baseline characteristics of the sample are shown in Table 1.
Baseline characteristics of sample (n = 407).
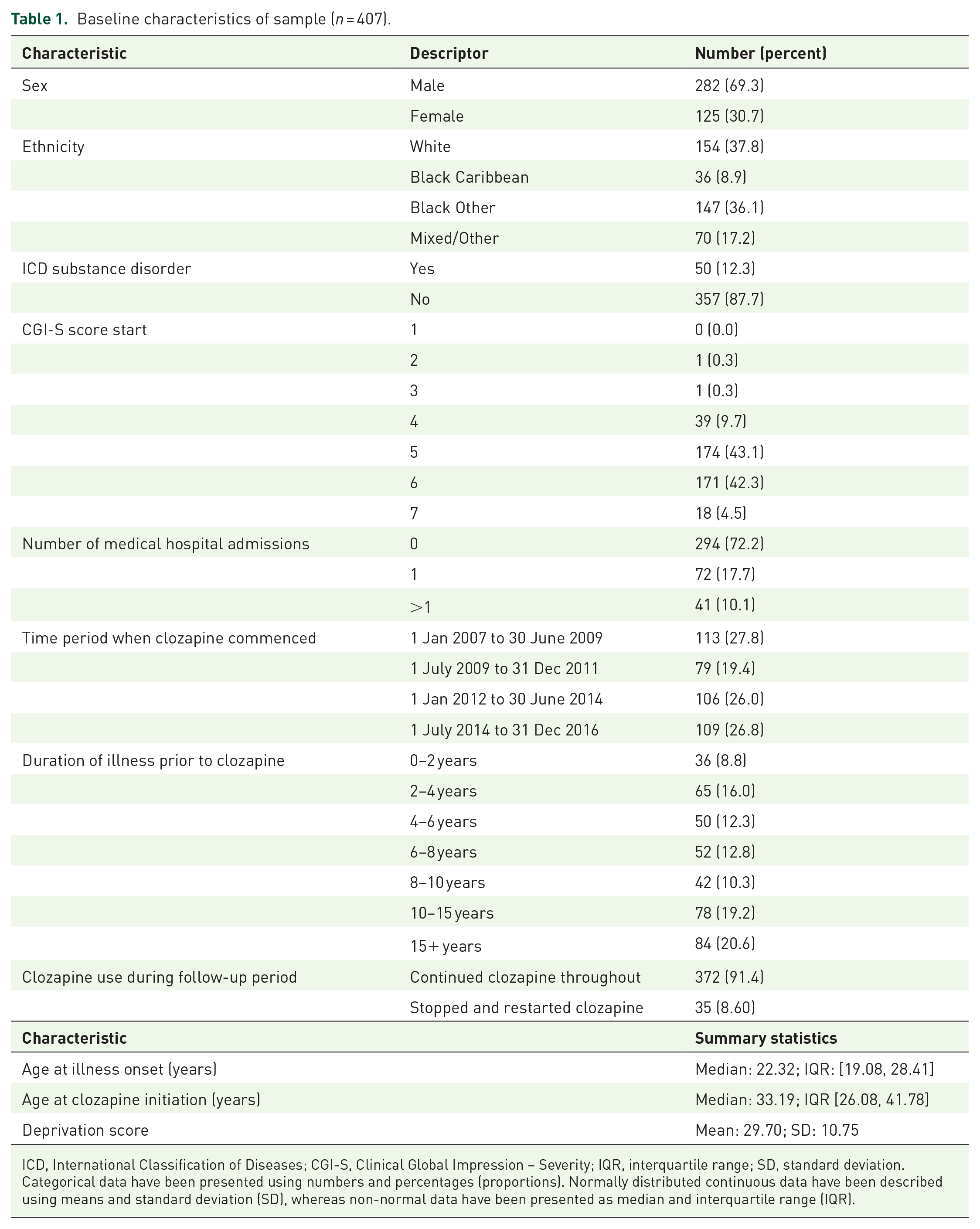
ICD, International Classification of Diseases; CGI-S, Clinical Global Impression – Severity; IQR, interquartile range; SD, standard deviation.
Categorical data have been presented using numbers and percentages (proportions). Normally distributed continuous data have been described using means and standard deviation (SD), whereas non-normal data have been presented as median and interquartile range (IQR).
The median duration of illness prior to clozapine was 8 years (range: 3 months to 44.5 years). The frequency distribution of duration of illness is shown in Figure 2. Most patients (89.9%) had CGI-S scores of 5 or above (5 = markedly unwell) at the time of clozapine commencement. Starting scores did not vary significantly with duration of illness (Figure 3). When treated as a continuous variable CGI-S outcome scores were on average 1.87 points lower than starting scores [paired t-test t = 31.56, df = 400, 95% confidence interval [CI]: (1.75, 1.99)].

Histogram of duration of illness prior to starting clozapine (years).

Mean CGI-S starting scores by duration of psychotic illness.
Ordinal logistic regression analysis showed an association between illness duration and CGI-S outcome score. AIC scores were 1140.98 for the model using duration of illness as a continuous variable and 1153.98 for the model using duration of illness as a categorical variable, indicating that the continuous model gave a better fit to the data after accounting for parsimony.
The results for the continuous model are shown in Table 2. There was a significant association between the duration of illness prior to clozapine and CGI-S outcome score at 2 years (fully adjusted OR: 1.04 (1.01–1.06)) indicating increased odds of a higher (worse) CGI-S outcome score as illness duration increased).
Model A: Illness duration as a continuous variable. Ordinal logistic regression of illness duration prior to clozapine and CGI-S outcome scores adjusted for age at illness onset, deprivation score, sex, co-morbid substance disorder, ethnicity, clozapine start date and medical admissions during follow-up.

CGI-S, Clinical Global Impression – Severity score; OR, odds ratio.
Significant result.
The regression using a categorical variable is included in Supplementary Table 1.
A plot of change in CGI-S score (treated as a continuous variable (CGI-S start – CGI-S outcome)) against illness duration indicated that the largest change in CGI-S score occurred with an illness duration of 2–4 years with a gradual reduction of effect as illness duration increased further (Figure 4). There appeared to be a reduced effect when clozapine was started earlier than 2 years illness duration; however, there were only 36 patients in this category.
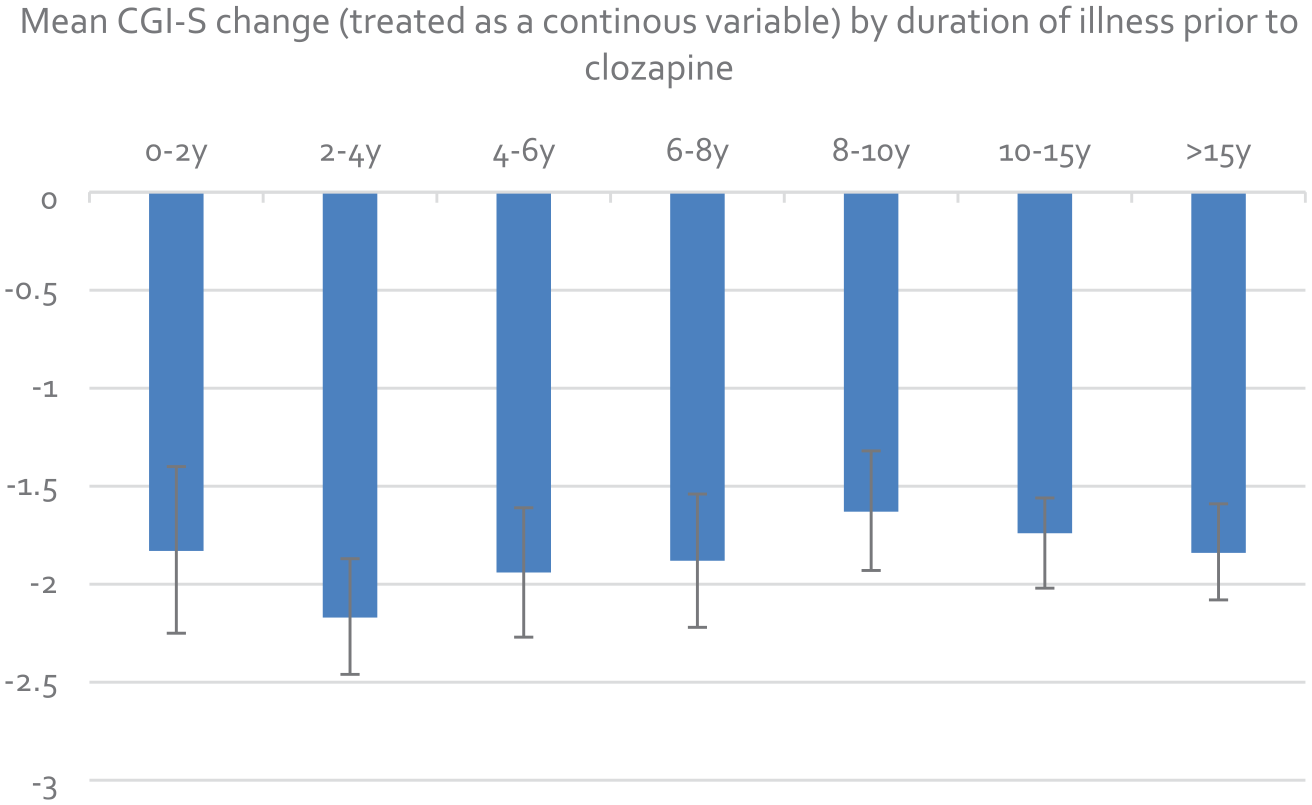
Mean CGI-S change by duration of psychotic illness
Discussion
The study demonstrated a significant association between duration of psychotic illness prior to clozapine and severity of illness at follow-up in patients who remained alive and were still taking clozapine under Maudsley services at 2 years. Overall, the analysis showed that the odds of a higher (worse) rather than lower (better) outcome CGI-S score increased by 4% per year of psychosis prior to clozapine. The CI indicated that the likely effect of illness duration on the odds was between 1% and 6% per year.
There was a lack of a clear association between starting clozapine earlier than 2 years and improved outcome. While it is possible that clozapine is less effective when started this early, this finding could be due to the small sample size in this category, or could reflect a degree of confounding by indication, with more seriously unwell patients with limited prospects of recovery more likely to be offered clozapine earlier in the course of their illness.
The results are in keeping with a recent meta-analysis of observational studies which suggested that delaying clozapine may lead to poorer response. 44 Studies with comparable methodology include Üçok et al. 33 who analysed retrospective case records of 162 patients with TRS and found a significant association between shorter length of clozapine delay and better response and Yoshimura et al. 34 who published similar findings for a sample of 90 patients with receiver operating characteristics (ROC) curve analysis indicating that 2.8 years delay from TRS diagnosis provided the best predictive cutoff for response.
The key strengths of our study are its larger size and also its generalisability, being a representative sample from an epidemiological clinical population.36,45 By using duration of illness as the predictor variable rather than duration of treatment resistance or clozapine delay, the results may be more easily replicated and more applicable to current service models in which resources are weighted towards the early years of psychosis. Similarly, the use of direct rather than indirect clinical information in determining clinical response is a strength, with CGI-S being chosen as a well-established tool for assessing overall illness severity with good face validity. The application of the CGI rating scale by an experienced consultant psychiatrist provided scores with good clinical utility as close as possible to those that would be obtained by seeing the patient in real time.
The study has some clear limitations. The results are applicable only to patients who survived and remained on clozapine for at least a 2-year period and cannot be applied to patients who for whatever reason discontinued clozapine, who are likely to have had a less-favourable treatment response. The use of CGI-S scores, applied retrospectively, is also a limitation in terms of the reliance on sufficient data being recorded in case notes to make an accurate assessment. In addition, CGI-S is not a continuous variable and therefore non-integer values have little meaning. This is not an issue in the regression analysis, but the use of change scores in Figure 4 needs to be interpreted with caution.
The wide range in duration of illness prior to clozapine increases the likelihood of survivor biases in older patients. While people who have lived for 10–15 years with schizophrenia might be expected to have less severe illness than those who have died; on the contrary, those who have responded well to treatment may have been discharged to their GPs affecting the severity of disease/case-mix in the patients included in this cohort. Although the extent of these biases could not be measured, CGI-S start scores did not reduce with age in the sample, suggesting that the overall effect of survivor bias was limited. Also there was a clear cohort effect which affected data quality, with newer patients having more complete records of their first psychotic episode; for this reason, the time period in which clozapine was commenced was controlled for in the analysis. However, additional factors were not able to be controlled for, such as duration of psychosis prior to referral to mental health services and the presence of negative symptoms. It is plausible that poor prognostic factors, such as prominent negative symptoms, may have led to clozapine being delayed in some patients, as highlighted in a recent systematic review of clozapine delay 32 and may account for the results obtained. Patients who are likely to respond well to antipsychotic treatment in general (i.e. those with prominent positive symptoms) may achieve a better response if treatment is given early in the course of schizophrenia, and this may also be the case with clozapine. 46
The time period from which clinical records were available for the study (2007 onwards) coincided with the national roll out of early intervention services in the United Kingdom, and therefore, patients who started clozapine early in the course of their illness were often under the care of the Maudsley early intervention teams. Others were in the forensic system receiving intensive rehabilitation. It is quite possible that psychosocial support aspects of these services contributed to the improved outcomes seen with clozapine in the 2- to 4-year illness duration category. A longer duration of follow-up would be required to see if clinical improvements following clozapine were sustained following transfer to generic services. However, whether or not clozapine is intrinsically more effective when started earlier or whether the therapeutic environment in which it is used is key, there appears to be clear benefit in starting clozapine before a pattern of severe enduring mental illness is set.
Overall, the results support the hypothesis that earlier clozapine initiation may be more effective in improving CGI-S scores, as beyond the first 4 years, a clear pattern of diminishing effect over time did emerge, with 2–4 years appearing to be the optimum period to commence clozapine. The use of clozapine during this time may improve outcomes by enabling patients to engage more in their recovery and rehabilitation, so that they have a better prospect of retaining or regaining a good level of functioning. Clozapine may also be intrinsically more effective if started earlier, and there is the possibility, albeit speculative, that it may be disease modifying if it results in a change of trajectory of disease, for example, by halting course to a deficit state. In recent years, there has been much interest in the role of inflammation in the pathogenesis of schizophrenia and promising trials of anti-inflammatory and immunomodulatory drug treatments. 47 Clozapine itself is known to have far-reaching immunomodulatory effects 48 which may account for its unique antipsychotic efficacy in TRS.
The low numbers of patients prescribed clozapine within the first 2 years in our sample is in keeping with clozapine prescribing elsewhere in the United Kingdom. A recent evaluation of prescribing patterns in the National Eden (National Evaluation of the Development and Impact of Early Intervention services) data looked at rates of treatment resistance and pathways to clozapine prescribing in a first episode psychosis sample of 1027 patients. 10 While the rate of treatment resistance over the course of 1-year follow-up was found to be 18.1%, there was a much lower rate of clozapine prescribing (2.4%) during the same time period. Likewise, the UK National Clinical Audit of Psychosis continues to show that clozapine is only offered to approximately 50% of patients in early intervention services who meet criteria for clozapine, 49 reflecting missed opportunities to establish patients on clozapine and improve their prognosis.
Reasons behind clozapine delay may be multiple but include inadequate knowledge and skills of prescribers. 25 Reluctance to prescribe may be well justified when there are legitimate concerns about adverse effects. However, other commonly cited reasons not to use clozapine, such as a belief that an individual would be too chaotic to comply with a clozapine regime, may stem from a lack of knowledge of its effectiveness, since adherence commonly improves on clozapine. Another barrier may be tolerance of incomplete response to antipsychotic medication, particularly in patients below threshold for acute admission. Clinicians, particularly in early-intervention services, have a responsibility to consider clozapine as soon as it is apparent that a patient is not responding adequately to first-line treatments.
Conclusion
This study provides further evidence that earlier use of clozapine may be more effective in TRS. Clozapine prescription continues to be delayed across the United Kingdom. Reasons for clozapine delay should be explored and addressed to enable patients to benefit more from clozapine.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253221103353 – Supplemental material for Duration of prior psychotic illness and clozapine response: a retrospective observational study using electronic health records
Supplemental material, sj-docx-1-tpp-10.1177_20451253221103353 for Duration of prior psychotic illness and clozapine response: a retrospective observational study using electronic health records by Rowena Jones, Rachel Upthegrove, Malcolm J. Price, Megan Pritchard, Joht Singh Chandan, Sophie Legge and James H. MacCabe in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253221103353 – Supplemental material for Duration of prior psychotic illness and clozapine response: a retrospective observational study using electronic health records
Supplemental material, sj-docx-2-tpp-10.1177_20451253221103353 for Duration of prior psychotic illness and clozapine response: a retrospective observational study using electronic health records by Rowena Jones, Rachel Upthegrove, Malcolm J. Price, Megan Pritchard, Joht Singh Chandan, Sophie Legge and James H. MacCabe in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
Thankyou to Debbie Cummings, CRIS administrator, NIHR Maudsley Biomedical Research Centre
Ethics approval and consent to participate
Patient consent was not required for the study as it was a retrospective analysis of anonymised patient care records using the Maudsley Clinical Records Interactive Search (CRIS) system. CRIS has been approved by Oxfordshire Research Ethics Committee as an anonymised data resource for secondary analysis (08/H0606/71). The study was approved by the NIHR BRIC CRIS oversight committee (application no 1112).
Availability of data and materials
contact
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: MJP is supported by the NIHR Birmingham Biomedical Research Centre. This paper represents independent research part funded by the National Institute for Health Research (NIHR) Maudsley Biomedical Research Centre at South London and Maudsley NHS Foundation Trust and King’s College London. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
