Abstract
Neuroleptic malignant syndrome (NMS) is a rare illness that results from reactions to antipsychotics. However, the diagnosis of NMS is challenging due to its atypical clinical presentation and unclear pathogenesis. We report the case of a patient with NMS induced by irregular use of antipsychotics, especially risperidone (RSP). He had typical hyperthermia, muscle rigidity and rhabdomyolysis, which led to renal impairment. We carefully analysed the mechanism by which NMS occurred in this patient. An interesting aspect of the case is the synergistic involvement of risperidone, antidepressants, opioids and stress. Because of these complex predisposing factors, it is difficult to completely rule out the diagnosis of malignant hyperthermia (MH). In addition, the rare phenomenon of elevated lipase and amylase was observed in this patient.
Introduction
Neuroleptic malignant syndrome (NMS) is a rare, severe illness associated with antipsychotics. Its clinical manifestations include muscle tonicity, tremor, fever, sympathetic nervous system dysregulation, altered mental status, and elevated creatine kinase (CK). 1 Risperidone-induced NMS has been reported, but interestingly, in the case, we observed abnormal elevations of lipase and amylase in addition to the typical clinical presentation. His unique clinical symptoms and interesting pathogenesis are worthy of consideration by clinicians.
Case presentation
A 35-year-old male was admitted to the hospital for painless fibroscopy to determine the nature of a pulmonary nodule. He had a 10-year history of depression and was taking multiple antipsychotics intermittently. He was not taking antipsychotic medication in the month prior to his admission and sometimes be irritable. Given that he has a bidirectional affective disorder. After consulting with a physician at the mental health centre, we prescribed antipsychotics for the patient, including risperidone (2 mg/day), paroxetine (20 mg/day), and lorazepam (2 mg/day). After two days, the patient had painless fiberoptic bronchoscopy. During the surgery, sufentanil (8 µg), isoproterenol (150 mg) and succinylcholine (120 mg) were given as induction of anaesthesia, and the procedure went smoothly. After the surgery, he felt muscle aches and pain, and his limbs were stiff and immobile. During the neurological examination of the patient, we found increased muscle tone in the extremities. No pathological signs or hyperactive tendon reflexes were elicited. Stiffness of the extremities was gradually relieved after approximately two hours. Approximately 24 hours later, the patient developed a high fever and chills with a temperature of 41 °C. The patient had an elevated creatine kinase (CK) level of 713 IU/L. Myoglobin (Myo) and lactate dehydrogenase (LDH) were also significantly higher than before (Table 1). Subsequently, the patient’s course entered the anuric phase of acute renal failure within 72 hours.
Experimental results in patients with neuroleptic malignant syndrome.
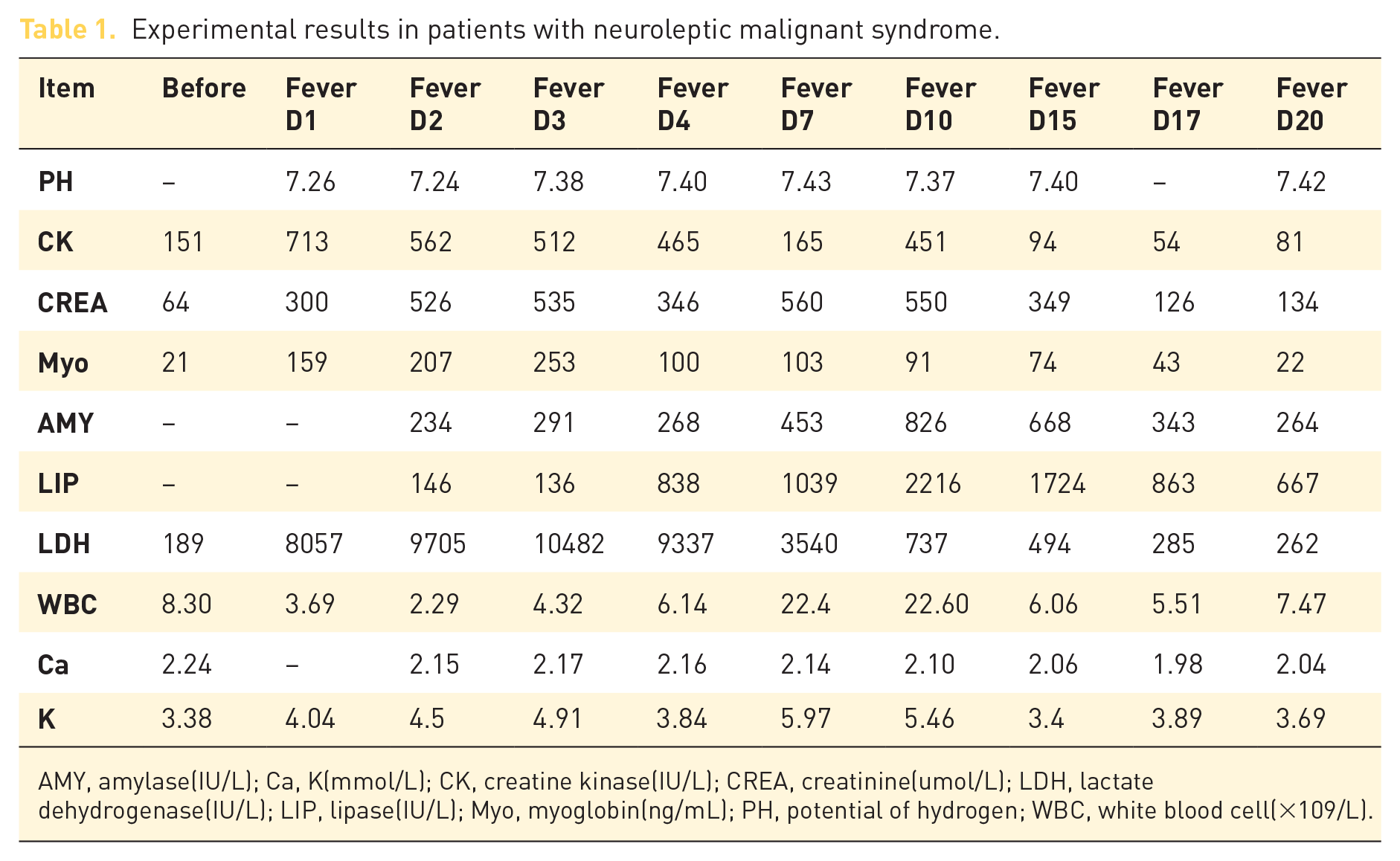
AMY, amylase(IU/L); Ca, K(mmol/L); CK, creatine kinase(IU/L); CREA, creatinine(umol/L); LDH, lactate dehydrogenase(IU/L); LIP, lipase(IU/L); Myo, myoglobin(ng/mL); PH, potential of hydrogen; WBC, white blood cell(×109/L).
No pathogenic bacteria were detected in blood or respiratory secretions. Therefore, we stopped all medications that could have triggered the disease, including antipsychotics. Also, we collected the patient’s blood for malignant hyperthermia(MH) susceptibility gene testing. The results showed that the two common MH susceptibility genes, RYR1 and CACNA1 S, were negative. Then, the patient was treated with continuous renal replacement therapy (CRRT) and other treatments to maintain organs function. We also observed abnormally elevated lipase (LIP; up to 2216 IU/L) and amylase (AMY; up to 826 IU/L) in the patient. However, the patient did not present with abdominal pain, and the abdominal CT was normal. We still prohibited the patient from eating and provided parenteral nutrition replacement therapy. After one month, the patient recovered and was restarted on oral clonazepam (3 mg/day) and duloxetine hydrochloride (120 mg/day), and thus far, no adverse reactions have been observed.
Discussion
NMS is challenging to diagnose and manage. To a large extent, this is because the clinical symptoms are atypical and the pathogenesis is not yet clear. In addition, NMS needs to be distinguished from serotonin syndrome (SS) and malignant hyperthermia (MH).
On the basis of the patient’s clinical presentation, we were inclined to diagnose this patient with NMS, but we could not completely rule out a diagnosis of MH. The reasons are as follows. First, the patient was taking risperidone. Risperidone binds to dopamine D2 receptors, resulting in a decrease in dopamine in the body. 2 Second, in this relatively hypodopaminergic state, the patient received paroxetine and the opioid sufentanil for general anaesthesia. Both of these drugs will increase the levels of free 5-hydroxytryptamine (5-HT) in the body. 3 Although excess 5-HT in the body may cause serotonin syndrome, the patient never showed paroxysmal or hyperreflexia and therefore did not meet Hunter’s criteria for the diagnosis of serotonin syndrome. 4 MH is a genetic disease inherited in an autosomal-dominant pattern. The patient’s Clinical Grading Scale (CGS) 5 score was 30, which was rated as a ‘high likelihood of MH’. The patient tested negative for the RYR1 and CACNA1 S mutation loci in the MH susceptibility test. Both the CGS score and the genetic susceptibility test findings suggest that the patient was less likely to have MH. Of course, the ‘gold standard’ diagnostic test for MH susceptibility is the caffeine-flurane skeletal muscle contraction test. 6 Patients with negative genetic results should be offered contracture testing to confirm their MH-negative status. Therefore, the possibility of a diagnosis of MH cannot be completely ruled out in this patient.
Acute pancreatitis is one of the rare and serious complications of antipsychotics. Studies reveal that among atypical antipsychotics, olanzapine and clozapine are associated with increased chances of induced pancreatitis, 7 whereas risperidone is the least likely to cause acute pancreatitis. The duration of acute pancreatitis varies, occurring in most patients within 6 months of the initiation or adjustment of their dose of antipsychotic medication. It has been suggested that the development of acute pancreatitis may be related to the effect of antipsychotic drugs on the metabolism of glucose, triglycerides, insulin and leptin. A study 8 found that long-term use of risperidone results in significantly elevated cholesterol, triglycerides, and blood sugar. In addition, genetic polymorphisms can also affect drug metabolism and impact steady-state serum. Furthermore, the use of multiple (2 or more) antipsychotics has been identified as a major contributor to antipsychotic-induced pancreatitis.
Conclusion
The question we would like to raise through this case is that some other drugs, such as paroxetine and fentanyl, in combination with antidopaminergic drugs, in addition to known antidopaminergic drugs, can exacerbate the occurrence of NMS. In addition to the common clinical manifestations of high fever, psychiatric abnormalities, elevated CK, and renal failure, one needs to be alert for the development of asymptomatic pancreatitis. A better understanding of the etiology and clinical manifestations of NMS was achieved, and the time to initiate treatment was subsequently reduced. This will effectively improve the cure rate for NMS and reduce criticality and mortality.
Footnotes
Ethics statement and patient consent
Our study did not require an ethical board approval because it did not contain human or animal experiments. The patient provided a written informed consent to publish his case.
Author contribution(s)
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Central Guide Place-free Exploration Project, Sichuan Provincial Department of Science and Technology (2020ZYD005).
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
