Abstract
Numerous case reports of acute pancreatitis (AP) induced by olanzapine have been published. Little is, however, known about the clinical features of olanzapine-induced AP. The aim of the study was to explore the clinical characteristics of olanzapine-induced AP. We collected literature on AP cases induced by olanzapine from 1996 to April 2021 for retrospective analysis in Chinese and English. The median time to onset of olanzapine-induced acute pancreatic symptoms was 12 (range = 0.86–216) weeks in 25 patients. The clinical features of AP range from asymptomatic elevation of blood amylase/lipase levels to digestive system symptoms (abdominal pain, vomiting, and nausea) and even death in a small number of patients. Laboratory tests showed varying degrees of elevated serum amylase and lipase levels, along with high blood sugar and high triglyceride levels in some patients. Computed tomography showed acute edematous pancreatitis, acute hemorrhagic pancreatitis, and acute necrotizing pancreatitis in the patients. The patients’ symptoms were completely relieved and high triglyceride levels gradually returned to normal levels after olanzapine was stopped. Some patients with hyperglycemia still needed hypoglycemic therapy. AP is a rare adverse effect of olanzapine. Clinicians should be aware of such complications and monitor pancreatin.
Introduction
Olanzapine is an atypical antipsychotic agent of the thienobenzodiazepine class and was initially approved in 1996 by the US Food and Drug Administration (FDA) for the treatment of schizophrenia, bipolar mania, bipolar depression, and treatment-resistant depression. 1 Olanzapine exerts pharmacological effects by antagonizing the dopamine D2 receptor 5-hydroxytryptamine (5-HT) (especially subtypes 2A, 2B, 2C, and 6). 2 The consensus of experts from Japan on the pharmacological treatment of schizophrenia and the American Psychiatric Association’s treatment practice guidelines for patients with schizophrenia still recommend olanzapine as the first-line drug for the treatment of schizophrenia.3,4
Olanzapine is available as ordinary tablets, orally disintegrating tablets (ODTs), long-acting injectable (LAI) formulations, and a fixed-dose combination with fluoxetine. Adverse effects of olanzapine include drowsiness, dry mouth, dizziness, restlessness, constipation, stomach upset, weight gain, increased appetite, tremor, and potentially serious extrapyramidal signs and symptoms. 5
Acute pancreatitis is one of the serious complications of atypical antipsychotics from case reports and pharmacovigilance. Koller et al. 6 studied antipsychotic-related pancreatitis and noted that 69 of 192 patients (63%) with pancreatitis were considered to be cases caused by olanzapine. Since 1996, numerous case reports of acute pancreatitis induced by olanzapine have been published. To our knowledge, little is known about the clinical features of olanzapine-induced acute pancreatitis. The purpose of this literature review is to analyze and discuss the clinical characteristics of olanzapine-induced acute pancreatitis and provide a reference for the safe and reasonable clinical application of olanzapine.
Methods
We searched the database in Chinese and English from 1996 to April 2021, including Wanfang Data, the China National Knowledge Infrastructure (CNKI), Chinese VIP, PubMed/Medline, Embase, the Web of Knowledge, OVID, Elsevier, Springer Link, and the Cochrane Library databases, and identified the literature regarding acute pancreatitis induced by olanzapine by the search terms ‘olanzapine’, ‘pancreatitis’, ‘lipase’, ‘amylase’, and ‘abdominal pain’.
A case report and case analysis of olanzapine-induced acute pancreatitis were included as a preliminary study. Olanzapine-induced acute pancreatitis meets Atlanta diagnostic criteria. We excluded duplicate literature, reviews, mechanistic studies, animal studies, and non-full-text literature.
According to the Atlanta classification of the diagnostic criteria for acute pancreatitis, the diagnosis of acute pancreatitis requires two of the following three features: (1) acute, sudden, continuous, epigastric pain often radiating to the back; (2) serum lipase activity (or amylase activity) at least 3 times greater than the upper limit of normal; and (3) characteristic findings of acute pancreatitis on contrast-enhanced computed tomography (CECT), and less commonly, through magnetic resonance imaging (MRI) or transabdominal ultrasonography. 7 The definition of severity of acute pancreatitis was used as follows: (1) mild acute pancreatitis (MAP): no organ failure and no local or systemic complications; (2) moderately severe acute pancreatitis (MSAP): organ failure that resolves within 48 h (transient organ failure) and/or local or systemic complications without persistent organ failure; and (3) severe acute pancreatitis (SAP): persistent single or multiple organ failure (>48 h). 7
Two researchers independently conducted a preliminary screening of the literature according to the inclusion and exclusion criteria, and then the group discussed the included literature. A self-designed data extraction table was used to extract the following information of the patients: nationality, sex, age, primary disease, underlying diseases, olanzapine application, combined medication, acute pancreatitis occurrence, treatment, and prognosis.
Statistical analysis
Statistical analyses were performed using SPSS 22.0 (SPSS Inc., Chicago, IL, USA). The results are expressed as medians with ranges and percentages (%). We used the Naranjo Adverse Reaction Score Scale to evaluate the causality of olanzapine-induced acute pancreatitis. 8
Results
Clinical presentation
We initially screened 172 studies. After careful review by two independent authors, case reports with incomplete data were excluded, and only 22 studies were included, with a total of 25 patients (17 males and eight females) in the study (Table 1). The 22 studies were all case reports with relatively complete data, and large-scale studies of pancreatitis and acute pancreatitis induced by other drugs were excluded. The median age of these patients was 36 (range = 17–72) years. Among these patients, nine patients were from Asia, nine patients were from Europe, six patients were from North America, and one patient was from Oceania. The dose range of olanzapine was 5~30 mg, including 5 mg in four patients (18.2%), 10 mg in 11 patients (50.0%), 20 mg in five patients (22.7%), and 30 mg in two patients (9.1%). Olanzapine was mainly used to treat schizophrenia in 16 patients (64.0%), depression in five patients (20.0%), and bipolar disorder in four patients (16.0%). The median time to onset of symptoms of acute pancreatitis was 12 (range = 0.86–216) weeks. One patient (4.0%) had preexisting pancreatitis and diabetes.9,10 Eight patients (32.0%) had high-risk factors for pancreatitis, including six patients (24.0%) who consumed alcohol, four patients (16.0%) who smoked, one patient (4.0%) with hypertriglyceridemia, and one patient (4.0%) with drug abuse. Five patients (20.0%) of these 25 patients had other underlying diseases at the same time, and eight patients (32.0%) were using other drugs. Three patients complained of weight gain.11–13 Two patients had acute pancreatitis with ketoacidosis.12,13 One patient was infected with COVID-19. 14
Characteristics of the 25 included patients.

BMI, body mass index.
Represents the number of patients out of 25 total patients for whom information regarding this particular parameter was provided.
Median (minimum, maximum).
Clinical presentation
The clinical manifestations of olanzapine-induced acute pancreatitis are summarized in Table 2. Abdominal pain in 22 patients (88.0%) was the most common clinical symptom of acute pancreatitis, followed by vomiting in eight patients (32.0%) and nausea in seven patients (28.0%). Three patients (12.0%) presented poor appetite. One patient developed fever, sore throat, cough, headache, and tachycardia. Three patients (12.0%) showed only hyperamylasemia. An analysis of the 22 patients with acute pancreatitis induced by olanzapine showed that 10 patients (45.5%) developed MAP, three patients (13.6%) developed MSAP, and nine patients (40.9%) developed SAP.
Clinical information on the 25 included patients.
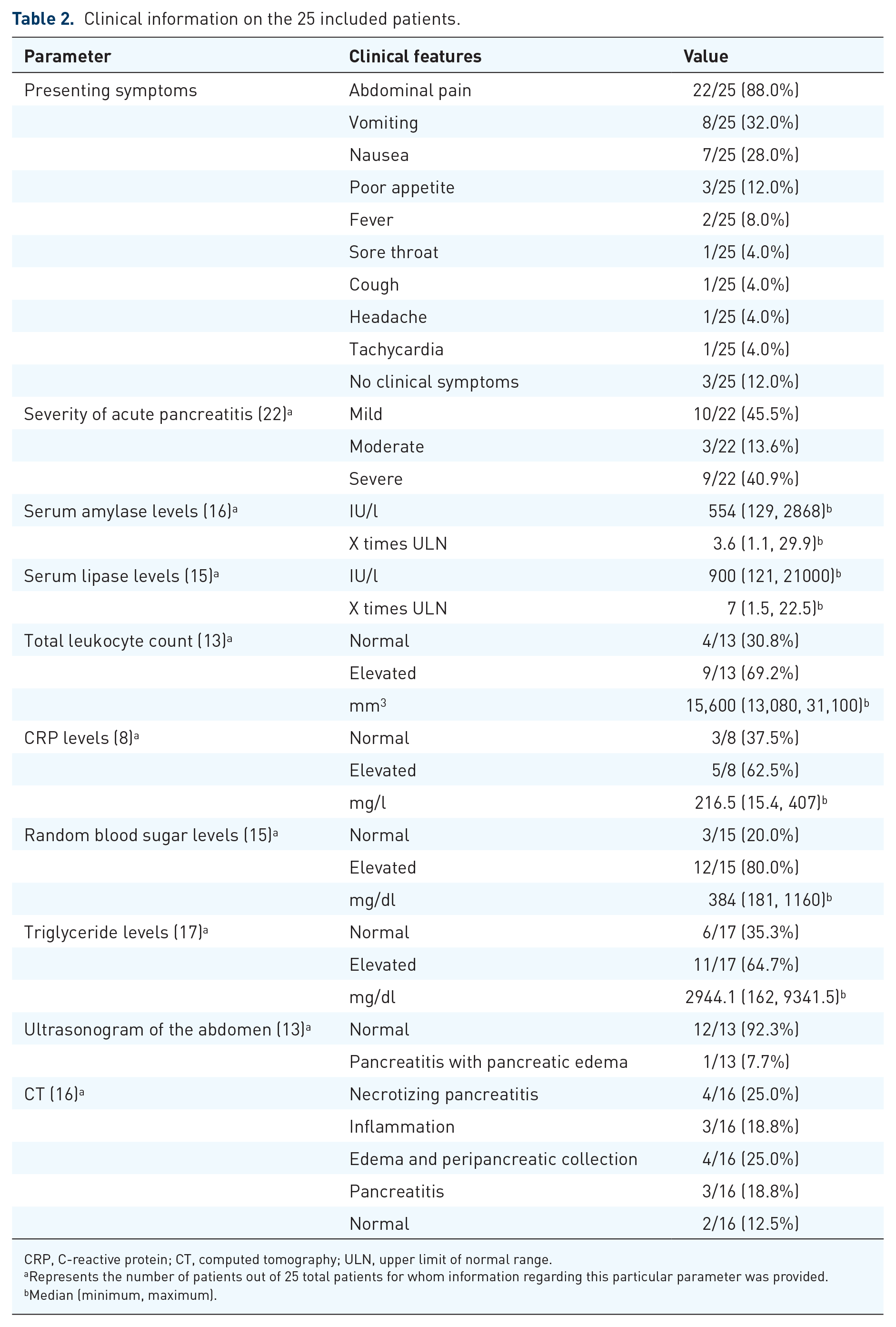
CRP, C-reactive protein; CT, computed tomography; ULN, upper limit of normal range.
Represents the number of patients out of 25 total patients for whom information regarding this particular parameter was provided.
Median (minimum, maximum).
Treatment and prognosis of the 25 included patients.
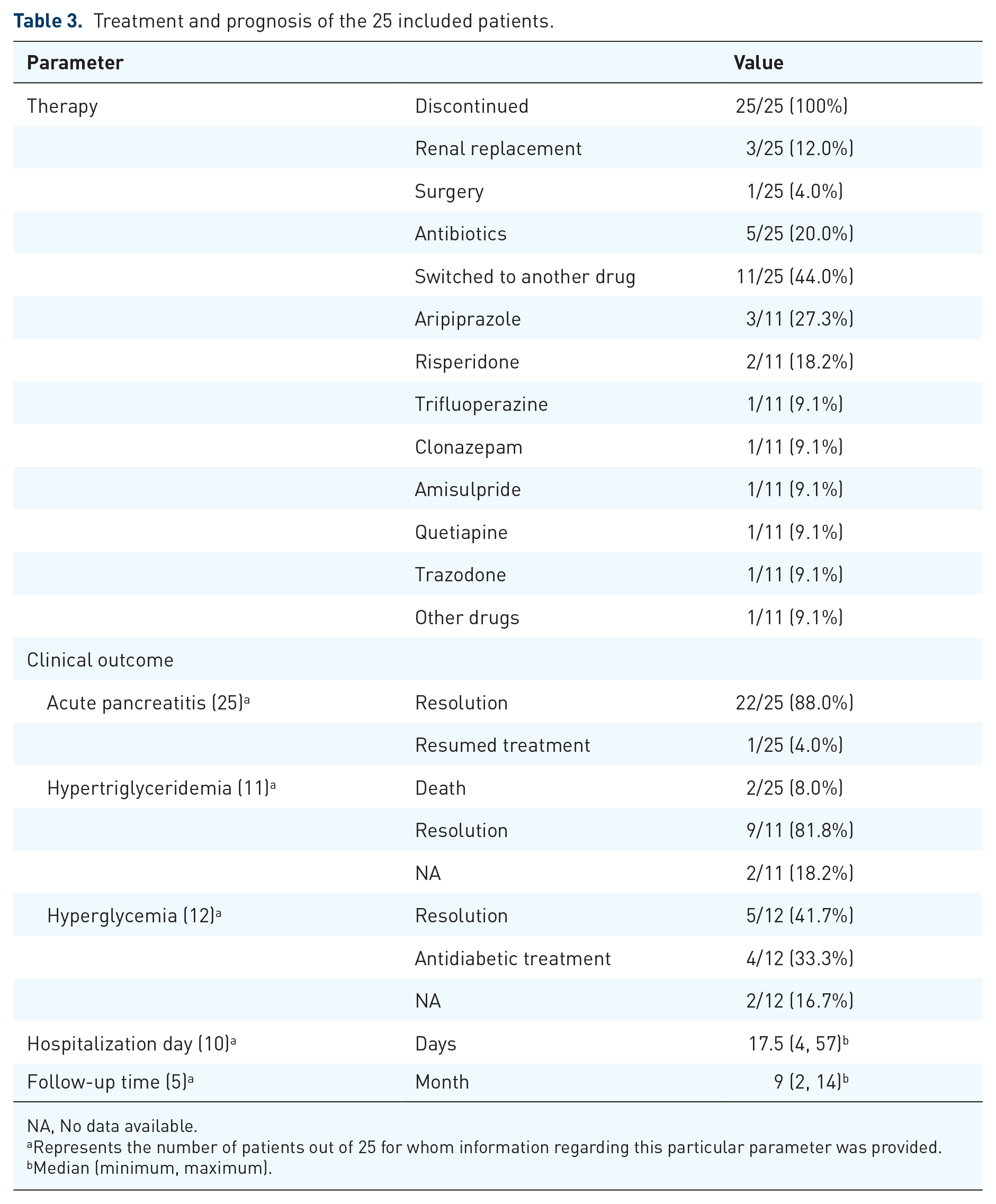
NA, No data available.
Represents the number of patients out of 25 for whom information regarding this particular parameter was provided.
Median (minimum, maximum).
Laboratory tests
The laboratory test results are summarized in Table 2. The median value of serum amylase levels was 554 (range = 129–2868) IU/l in 16 patients and the median value of serum lipase levels was 900 (range = 121–21,000) IU/l in 15 patients. According to the normal reference range of amylase/lipase levels in various laboratories, the median values of amylase/lipase levels that exceeded the upper limit of the normal range (ULN) are 3.6 (1.1, 29.9) and 7 (1.5, 22.5), respectively. Elevated white blood cell counts were observed in nine patients (69.2%), with a median value of 15,600 (range = 13,080–331,100) mm3. Among the eight reported patients, C-reactive protein (CRP) levels were elevated in five patients (62.5%) and the median value was 216.5 (range = 15.4–407) mg/l. The median blood glucose level was 384 (range = 181–1160) mg/dl in 11 patients (78.6%). Eleven (64.7%) of the 17 patients reported having elevated triglyceride levels, with a median value of 2944.1 (range = 162–9341.5) mg/dl.
Imaging examination
The imaging examination results are summarized in Table 2. Thirteen patients underwent abdominal ultrasound examination and only one patient showed pancreatitis with pancreatic edema. The computed tomography (CT) scans of 16 patients showed necrotizing pancreatitis in four patients (25.0%), inflammation in three patients (18.8%), edema and peripancreatic collection in four patients (25.0%), pancreatitis in three patients (18.8%), and normal pancreatitis in two patients (12.5%).
Treatment
The treatment and prognosis of these 25 patients are summarized in Table 3. All patients immediately stopped olanzapine and were given conservative management. Among them, three patients (12.0%) were given renal replacement therapy, one patient (4.0%) required surgery, and five patients (20.0%) were given antibiotics. Twenty-two patients (88.0%) recovered completely from their symptoms with a median hospital stay of 17.5 days (4, 57). Patients had no further recurrence of acute pancreatitis without continued olanzapine at a median follow-up time of 9 (range = 2–14) months. One patient (4.0%) died of peritonitis secondary to pancreatitis and another patient (4.0%) eventually died of severe cerebral edema and modest hemorrhagic pancreatitis due to fatal hyponatremia, hyperglycemia, and acute pancreatitis associated with psychotropic drug polypharmacy.15,16 Eleven patients switched to other antipsychotic drugs, including aripiprazole, risperidone, trifluoperazine, clonazepam, amisulpride, and quetiapine. One patient was readmitted 4 months later with another attack of acute pancreatitis due to resuming olanzapine treatment. 10 Among the 12 patients with elevated blood sugar, five patients (41.7%) returned to normal levels after olanzapine was stopped, and four patients (33.3%) still needed hypoglycemic treatment. Nine patients (81.8%) with hypertriglyceridemia returned to normal blood sugar levels after olanzapine was stopped.
Relevance evaluation
We used the Naranjo scale to assess the possibility of olanzapine-induced acute pancreatitis in 22 patients. Among them, 20 patients were classified as probable cases, including 13 patients with a score of 7–8, six patients with a score of 6, and one patient with a score of 5. One patient with a score of 4 could be considered to have possible pancreatitis. The Naranjo Adverse Drug Reaction Probability Scale was not applicable in one case because the lethal outcome occurred only 2 h after admission.
Discussion
Acute pancreatitis is an inflammatory disorder of the pancreas that is associated with substantial morbidity and mortality. Gallstones and alcohol misuse, trauma, hypertriglyceridemia, hypercalcemia, and drugs are the main risk factors for acute pancreatitis. 17 Many drugs may induce pancreatitis through various pathophysiological mechanisms. 18 Olanzapine was the most commonly prescribed antipsychotic. In a survey in Qatar, 45.9% of patients were prescribed olanzapine. 19 In an Asian psychotropic prescription pattern (REAP) survey, 20.1% of prescriptions for psychosis included olanzapine. 20 Acute pancreatitis seems to be an uncommon complication of olanzapine, considering the frequency of olanzapine use and the rarity of reports of pancreatitis related to the drug. Underreporting may also, however, be an influencing factor.
Most of the patients we included did not have other causes or risk factors related to acute pancreatitis. The duration of olanzapine use varied from 6 days to 4 years after the beginning of olanzapine treatment. Abdominal pain is still the most common clinical manifestation of acute pancreatitis induced by olanzapine. Some patients will have asymptomatic pancreatitis. For patients with asymptomatic elevations of amylase and lipase levels, whether continued olanzapine treatment under given conditions will cause pancreatitis requires further research. Elevated serum amylase levels can reach 23 times the ULN and lipase can reach 30 times the ULN. Olanzapine-induced acute pancreatitis can manifest as mild pancreatitis, moderate pancreatitis, or severe pancreatitis and imaging manifestations are acute edematous pancreatitis, acute hemorrhagic pancreatitis, and acute necrotizing pancreatitis. Olanzapine can cause acute pancreatitis with hypertriglyceridemia and hyperglycemia and can also cause pancreatitis with normal triglyceride levels. Acute pancreatitis with normal triglycerides is mainly manifested as MAP, while acute pancreatitis with elevated triglycerides is more serious, manifesting as moderate to severe acute pancreatitis. Patients with a preexisting diagnosis of hypertriglyceridemia may further raise their triglyceride levels after the initiation of olanzapine therapy, thereby having a higher likelihood of pancreatitis. 21 Therefore, lipid screening should be performed before determining whether a patient is suitable for olanzapine treatment. Similarly, lipid levels monitoring during olanzapine treatment should also be performed.
The exact mechanism by which olanzapine causes acute pancreatitis is unclear. One of the current hypotheses is that olanzapine may indirectly induce pancreatitis through metabolic adverse reactions (especially weight gain, hyperlipidemia, and hyperglycemia).22–24 The second hypothesis is the direct effect of olanzapine because of its rapid onset. 25 The serum concentration of olanzapine may be linked to the success of treatment and the risk of adverse drug reaction. 26 The dosage form of olanzapine is an important factor that affects the serum concentration of olanzapine. 27 For example, ODTs, which have higher bioavailability and rapid dissolution rates, lead to earlier plasma concentrations compared with standard formulations. 25 This adverse drug reaction may also be related to the patient’s genetic variations. 25 The metabolism of olanzapine is affected by polymorphisms in the CYP1A2, CYP2D6, FMO3, and UGT1A4 coding genes, which affect the steady-state serum concentration of olanzapine. 28 In addition, prescribing multiple (two or more) drugs related to acute pancreatitis may have an additive effect. 29
The possibility of rapid clinical deterioration of drug-induced acute pancreatitis and the lack of unique features that distinguish pancreatitis caused by drugs or other etiologies stress the need to identify and discontinue offending agents as soon as possible. 30 After discontinuation of olanzapine and symptomatic treatment, almost all patients did not report long-term complications related to olanzapine-induced acute pancreatitis. The time it takes for serum amylase/lipase levels to return to normal levels varies from person to person, regardless of the degree of elevation. Hypertriglyceridemia gradually returned to normal levels in these patients. Blood sugar levels gradually returned to the normal range in some patients with elevated blood sugar, while other patients still needed to continue hypoglycemic therapy.
Conclusions
Clinicians should be aware of the rare adverse reaction of olanzapine-induced acute pancreatitis. Patients taking olanzapine should regularly monitor their abdominal condition, especially if they have one or more of the above risk factors for pancreatitis. When symptoms related to acute pancreatitis occur, the medication should be stopped immediately. In addition, to prevent the recurrence of pancreatitis after resuming olanzapine treatment, we recommend that patients who still need antipsychotic treatment switch to antipsychotics other than olanzapine.
Footnotes
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Natural Science Foundation of Hunan Province (No. 2020JJ5869).
