Abstract
Background:
There is limited evidence on interventions to minimise weight gain at clozapine commencement. We compared the effect of adjunctive metformin versus placebo at clozapine initiation.
Methods:
People with schizophrenia commencing on clozapine were randomised to either metformin or placebo for 24 weeks. The primary outcome was difference in the change of body weight. Secondary outcomes included comparative rates of weight gain of more than 5%, overall weight gain/loss, and differences in metabolic and psychosis outcomes.
Results:
The study was closed prematurely in March 2020 due to COVID-19 restrictions. Ten participants were randomised to each of the metformin and placebo groups. Eight metformin group and five placebo group participants completed the trial and were included in the analysis. The study was insufficiently powered to detect difference between the metformin and placebo groups for the primary outcome of change in weight (0.09 kg vs 2.88 kg, p = 0.231). In terms of secondary outcomes, people in the metformin group were significantly less likely to gain >5% of their body weight (12.5% vs 80%, p = 0.015) and were more likely to lose weight (37.5% vs 0% p = 0.024) compared to placebo. There was no difference between the groups in terms of adverse drug reactions (ADRs).
Conclusion:
While limited by the forced premature closure of the trial due to COVID19, the findings from this randomised controlled trial are promising. Clozapine and metformin co-commencement may be a promising treatment to prevent clozapine-associated weight gain, especially given the low rates of ADRs associated with metformin. This supports the consideration of use of metformin to prevent weight gain in people initiated on clozapine; however, further studies are needed to confirm this finding.
Trial registration:
ACTRN12617001547336
Introduction
People with schizophrenia are three times more likely to die prematurely than the general population, 1 leading to a 16-year reduction in life span. 2 This mortality gap is largely driven by morbidities associated with metabolic disorders and cardiovascular disease. 2 The increased risk for cardiometabolic disease among people with schizophrenia is multifactorial, including genetic risks of impaired glucose regulation, 3 as well as lifestyle factors like poor diet, 4 lack of physical activity 5 and smoking. 4 However, a major contributing factor for glucose dysregulation is antipsychotic medications. 6
Up to 33% of people with schizophrenia will not respond to first- and second-line antipsychotic medications. 7 This cohort of people have treatment refractory schizophrenia (TRS). 8 Clozapine is the most effective medication for reducing positive symptoms, 9 hospitalisations, 10 and overall mortality among people with TRS. 11 However, clozapine is associated with the highest rates of weight gain, type-2 diabetes mellitus (T2DM), and metabolic syndrome of all antipsychotics.6,12,13
Lifestyle modification, though effective in reducing weight and improving cardiometabolic health, is hampered by low rates of uptake and long-term engagement among people with schizophrenia. 14 For example, people with schizophrenia frequently suffer from comorbid cognitive deficits which can create barriers to engaging in physical activity programmes or planning meals. 15 As such, there is a need for effective pharmacological agents to ameliorate weight gain among people on antipsychotic medications, particularly clozapine.
Clozapine-induced weight gain is multifactorial, with involvement of serotonergic, histaminic, muscarinic and dopaminergic pathways. 16 Variants in the LEP and HTR2C genes have been associated with greater weight gain with clozapine. 17 Pharmacological agents, such as metformin, can improve glucose homeostasis and lead to weight loss. 18 Metformin mediates weight loss in part through the peptide hormone growth differentiation factor 15 (GDF15). 19
A meta-analysis of randomised controlled trials (RCTs), including 478 participants with schizophrenia who were already taking clozapine and obese, showed that commencement of metformin led to a 3.12-kg greater loss in weight than placebo (95% confidence interval [CI]: 1.37–4.88 kg, p < 0.001). 20 In a retrospective naturalistic cohort study of patients routinely commenced on clozapine, patients who were taking metformin in the subsequent 12 months had a significantly lower increase in percentage body weight gain than patients who were not taking metformin (1.32% versus 5.95%, p = 0.031). 21
Preventing rather than ameliorating weight gain is important. Weight gain compromises treatment adherence which can lead to relapse in psychosis among people with TRS. 22 However, to date, there is limited evidence on interventions that can prevent weight gain at the time of clozapine commencement. There have been no RCTs that have explored the concomitant prescription of metformin at the time of clozapine initiation to attenuate weight gain.
In CoMET: a randomised controlled trial of co-commencement of metformin, we aimed to investigate the effectiveness of adjunctive metformin versus placebo in attenuating weight gain in people with schizophrenia newly commenced on clozapine.
Methods
This trial received ethics approval from the Metro South Human Research Ethics Committee (HREC/17/QPAH/538). It was registered on the Australia and New Zealand Clinical Trials Registry (ACTRN12617001547336). A pre-trial protocol was published. 23
Recruitment was undertaken at four Hospital and Health Service (HHS) Districts in South-East Queensland: Metro North HHS, Metro South HHS, West Moreton HHS, and Gold Coast HHS. Recruitment was from inpatient units, residential community care units, and community clinics. All patients participating in the study provided a written informed consent.
Participant inclusion criteria were people with schizophrenia or schizoaffective disorder who were either clozapine naïve or had not been on clozapine in the previous year, had capacity to consent, and who were being commenced on clozapine as part of routine care by their treating team. Participants were excluded from the study if they were underweight (body mass index (BMI) ⩽ 18 kg/m2), morbidly obese (BMI ⩾ 40 kg/m2), had a pre-existing diagnosis of T2DM, or were already taking metformin or any other weight-lowering medications. The full inclusion and exclusion criteria are detailed in Supplementary Appendix 1.
As clozapine is a highly monitored drug in Australia, clozapine recipients attend structured weekly medical appointments, with weekly biometric measurements and absolute neutrophil counts (ANC) for the first 18 weeks of clozapine treatment. After Week 18, the clozapine treatment protocol in Australia stipulates 4-weekly medical reviews and ANCs. Prior to clozapine commencement, pre-registration with a clozapine manufacturer patient -monitoring system is required. At the time of clozapine registration, potential participants for this study were approached by clinical teams regarding the CoMET study and referred to our clinical trials team for further information, assessment for eligibility and consent.
Participants who met eligibility requirements and provided consent were randomised to metformin (active treatment) or placebo in a 1:1 ratio using blocks of four via a computer-generated randomisation table provided by a statistician independent of the study and treating teams. The treating team, participants and the research team were blinded to allocation of intervention.
Allocation concealment was maintained using placebo tablets that were identical in packaging, appearance, colour, and taste to the metformin tablets. The number of placebo tablets dispensed were increased to match the titration of metformin. The study medications were packed in blinded sequentially numbered bottles by a pharmacist external to the study group. All other study assessments and procedures were identical between the two groups. As part of usual care in the public mental health clinics providing care to the trial participants, all patients are offered dietary and physical activity advice.
We based our metformin dose regime on clinical recommendations, starting at 500 mg daily and titrating up to 2000 mg daily over 3 weeks.24,25
Based on the primary outcome, change in body weight at 24 weeks, using repeated measures with two-way analysis of variance (ANOVA), we calculated a required total sample size of 86 participants. Using data from our meta-analysis of metformin for clozapine-associated obesity, 20 we estimated that to observe a minimal clinical difference in weight change of 3.12 kg, with an assumed standard deviation (SD) of 9.6 in both groups (from our meta-analysis), α = 0.05, and correlation between baseline and repeated measures of 0.7, we would require 34 participants per group to achieve 80% power. We assumed an attrition rate of 20% from baseline to follow-up.
Treatment protocol
Metformin group
Participants in the metformin group were provided with metformin extended release (XR) formulation to be taken nightly 24 weeks. Metformin XR 500 mg tablets were titrated, as tolerated, over 3 weeks period with one tablet daily (500 mg) for the first week, two tablets daily (1000 mg) for the second week and four tablets daily (2000 mg) for the remainder of the study.
Placebo group
Placebo group participants were provided with placebo tablets identical to the metformin XR tablets to be taken nightly over the 24-week trial. The dose was titrated, as tolerated, over a 3-week period with one tablet daily for the first week, two tablets daily for the second week and four tablets daily for the remainder of the study.
Adherence was monitored through the count of returned unused tablets at each visit.
Outcomes
Primary
The primary outcome was a difference in the change between baseline and endpoint body weight in kilogrammes (kg) at 24 weeks between the placebo and metformin groups.
Secondary
The secondary outcomes were differences between the placebo and metformin groups in terms of the following: the proportion of participants with weight gain of ⩾5%, overall weight gain or loss, and changes in metabolic outcomes. These included BMI, waist circumference, hip/waist ratio, heart rate, diastolic and systolic blood pressure (BP), vitamin B12, glycosylated haemoglobin (HbA1c), fasting glucose and insulin, homeostatic model assessment (HOMA), high-density lipoprotein (HDL) and low-density lipoprotein (LDL) cholesterol and triglycerides. Other outcomes were measures of hepatic fibrosis including the Non-Alcoholic Fatty Liver Disease (NAFLD) Fibrosis Score or Fibrosis-4 (FIB-4) Index.
We also compared the placebo and metformin groups in terms of changes in Positive and Negative Syndrome Scale (PANSS) and Global Assessment of Functioning (GAF) scores from baseline to endpoint, as well as adverse drug reactions (ADRs; SAFTEE – a systematic ADR checklist) 26 and dropouts.
Trial visits, assessments and outcome measures
Trial visits and assessments were identical in both groups. 23 Participants were seen by trial investigators weekly for the first 4 weeks and then every four weeks for the remainder of the study, with investigational product (either metformin or placebo) dispensed at every study visit. Anthropometric assessments and ADRs were recorded every study visit. Metabolic bloods, including fasting glucose, HDL and LDL cholesterol, triglycerides, HbA1c, Vitamin B12 and insulin (for the calculation of HOMA) were taken at baseline and Weeks 12 and 24. Oral Glucose Tolerance Test (OGTT) was conducted at baseline and Week 24. PANSS and GAF were assessed at baseline and at Weeks 12 and 24.
Height was recorded at the screening assessment. Weight was recorded to the nearest 0.1 kg using calibrated scales with participants wearing light clothing, no shoes and after emptying their bladder. Waist circumference was measured horizontally around the abdomen at a level halfway between the top of the iliac crest and the bottom of the ribs using a non-stretchable measuring tape to the nearest 0.5 cm. 27 Hip circumference was measured at the maximum circumference of the buttocks. 27 The hip/waist ratio was calculated as the ratio of hip circumference to waist circumference. Pulse and BP were recorded after the participant sat for 5 min. 28
The NAFLD Fibrosis Score was calculated using age, BMI, presence of diabetes, liver enzymes, albumin and platelets, with a low cut-off threshold of <–1.455 to exclude fibrosis. 29 The FIB-4 Index was calculated using age, liver enzymes and platelets, with a low cut-off threshold of <1.45 to exclude fibrosis. 30
Food craving was measured using the Food Craving Inventory, 31 assessed at baseline and endpoint.
Psychotic symptoms were measured using the PANSS, a validated 30-item investigator-rated measure. 32 The GAF, a validated investigator rated scale, measures symptom severity, psychological, social, and occupational functioning. 33
Data analysis
For the primary outcome (change in weight between baseline and endpoint), we undertook a repeated measures analysis using two-way analysis of variance over the nine data collection time points to assess both the within-subjects’ time and time by intervention effect. This included baseline weight as a co-variate. Repeated measures analysis was also used for all secondary outcomes with continuous variables, except food craving which was analysed by t-test. Where data violated the assumption of sphericity, Greenhouse–Geisser correction was used. Chi-square tests were used for categorical variables and t-tests for continuous variables. Differences between the baseline characteristics of the metformin and placebo groups were compared using t-tests or chi-square tests as appropriate. Number needed to treat was calculated from rates of participants with >5% weight gain between the metformin and placebo groups. Analysis was undertaken using SPSS (Version 27 for Mac).
Results
Study recruitment was commenced in March 2018 but shut down prematurely in March 2020 due to COVID-19 lockdown restrictions. Two hundred and five people were assessed for eligibility. Ninety-two people did not meet eligibility criteria (24 were already taking metformin and 68 did not meet other eligibility criteria), 16 were assessed to be deprived of capacity to provide informed consent, 7 had their clozapine ceased by the treating team prior to randomisation, and 70 declined to participate. Twenty participants were randomised, 10 to metformin and 10 to placebo. From the metformin group, one participant discontinued the trial because they declined to take the study medication, while the other requested to leave the trial so they could be commenced on metformin open label by their treating team. Five participants in the placebo arm discontinued the trial, one because they declined to take the study medication, two because of non-adherence with clozapine, and two because of a treating team decision to cease clozapine. Eight participants in the metformin arm and five in the placebo arm completed the trial and were included in the endpoint analysis. Of those who did not complete the trial, only one had data beyond the 4-week timepoint. The CONSORT Flow Diagram is provided as Figure 1. The CONSORT checklist is provided as Supplementary Appendix 2.

CONSORT 2010 flow diagram.
All participants were titrated to 2000 mg XR metformin daily (or four tablets placebo daily) over the first 3 weeks as per protocol without the need to slow titration or down titrate.
Baseline characteristics of the participants in the metformin and placebo groups are provided in Table 1. The participants in the placebo group had statistically significantly higher body weight, BMI, waist circumference, Hip/Waist ratio and fasting triglycerides than those in the metformin group. Other measures were not statistically significantly different. Mean clozapine dose and levels at Week 24 did not statistically significantly differ between the groups. Baseline characteristics of participants who completed the study are provided in Supplementary Table 2. These baseline characteristics were broadly similar to those of all participants; however, the difference in weight between the placebo and metformin groups was not statistically significant.
Baseline demographics and clinical measures.
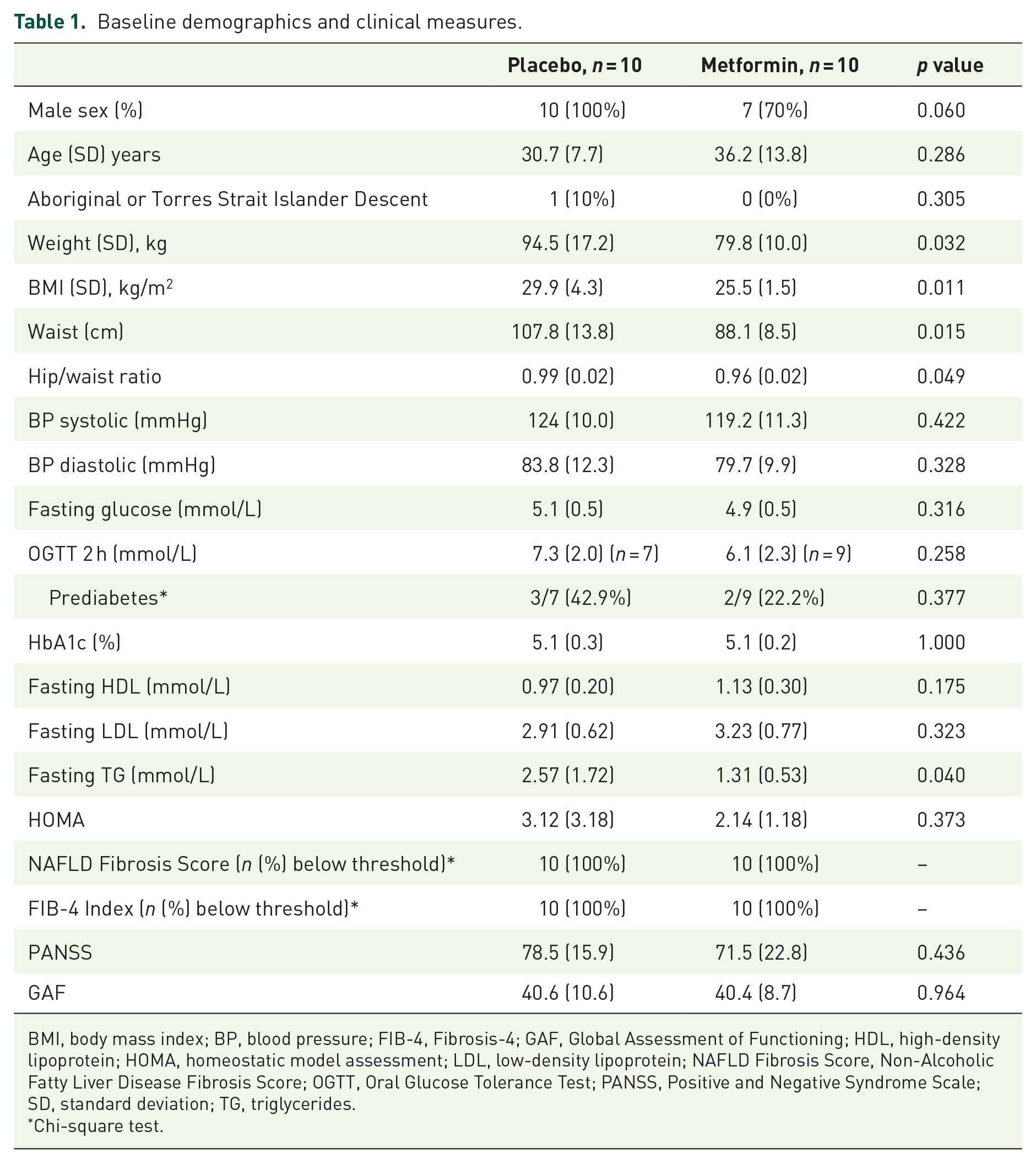
BMI, body mass index; BP, blood pressure; FIB-4, Fibrosis-4; GAF, Global Assessment of Functioning; HDL, high-density lipoprotein; HOMA, homeostatic model assessment; LDL, low-density lipoprotein; NAFLD Fibrosis Score, Non-Alcoholic Fatty Liver Disease Fibrosis Score; OGTT, Oral Glucose Tolerance Test; PANSS, Positive and Negative Syndrome Scale; SD, standard deviation; TG, triglycerides.
Chi-square test.
For the primary outcome, there was no statistically significant difference in the change in weight between the metformin and placebo groups on repeated measures analysis (0.09 kg vs 2.88 kg, p = 0.231). There was also no statistically significant difference between participants in the metformin and placebo groups for the secondary continuous outcomes, apart from HbA1c (0.24% lower in the metformin group, p = 0.047) and diastolic BP (0.26 mmHg lower in the metformin group, p = 0.047); however, neither of these results were a clinically meaningful change 34 (Table 2). Change in weight over time for each participant is provided in Figure 2.
Change in variables from baseline to Week 24 (per protocol analysis).
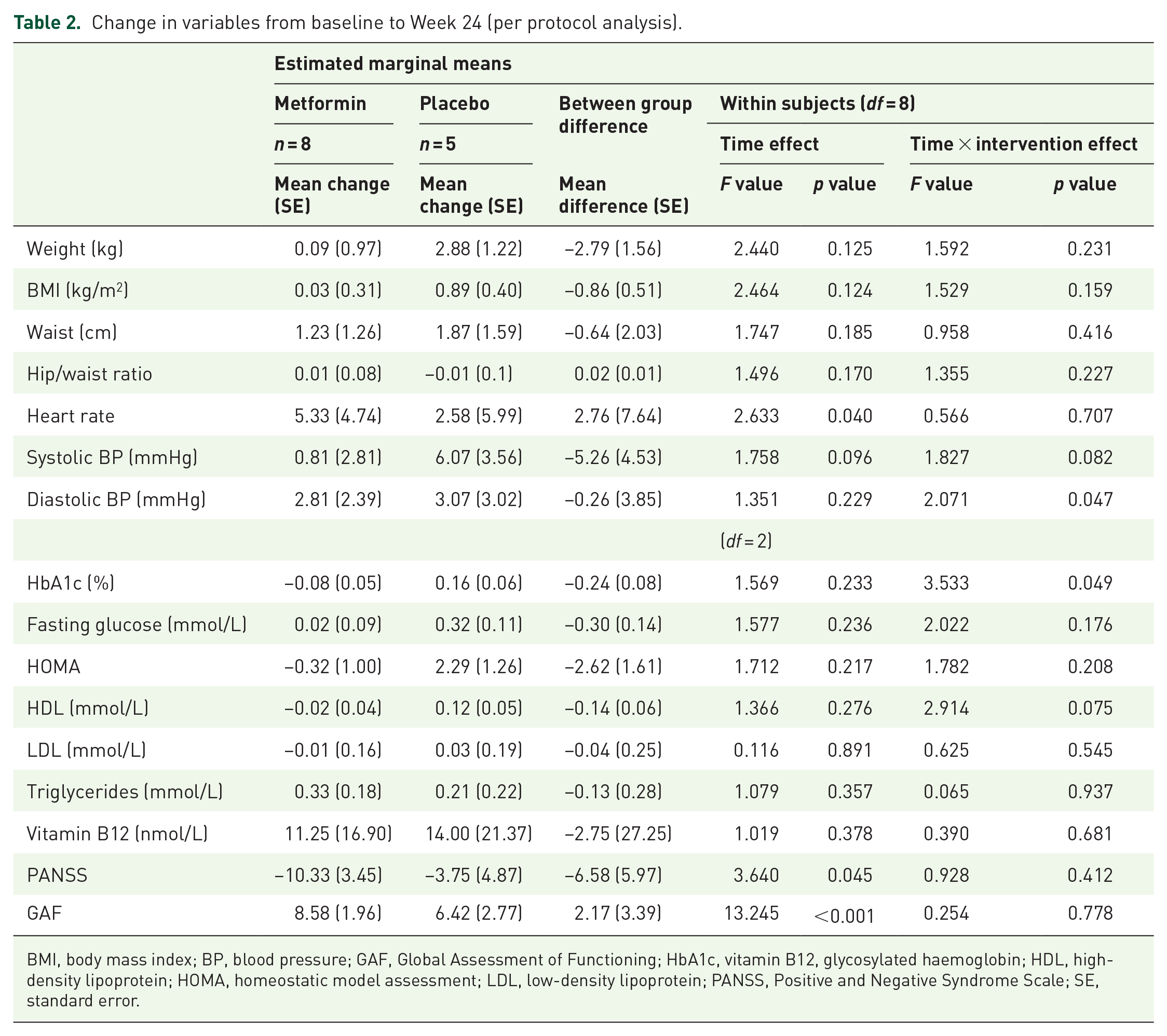
BMI, body mass index; BP, blood pressure; GAF, Global Assessment of Functioning; HbA1c, vitamin B12, glycosylated haemoglobin; HDL, high-density lipoprotein; HOMA, homeostatic model assessment; LDL, low-density lipoprotein; PANSS, Positive and Negative Syndrome Scale; SE, standard error.

Weight change throughout the study period.
Compared to participants in the placebo group, participants in the metformin group were significantly less likely to gain more than 5% of their body weight, (12.5% vs 80%, p = 0.015), were more likely to lose weight (37.5% vs 0%, p = 0.024; Table 3) and have a reduction in BMI (37.5% vs 0%, p = 0.024). The number needed to treat to prevent a >5% gain in body weight was 1.5.
Metabolic changes from baseline to 24-week endpoint.
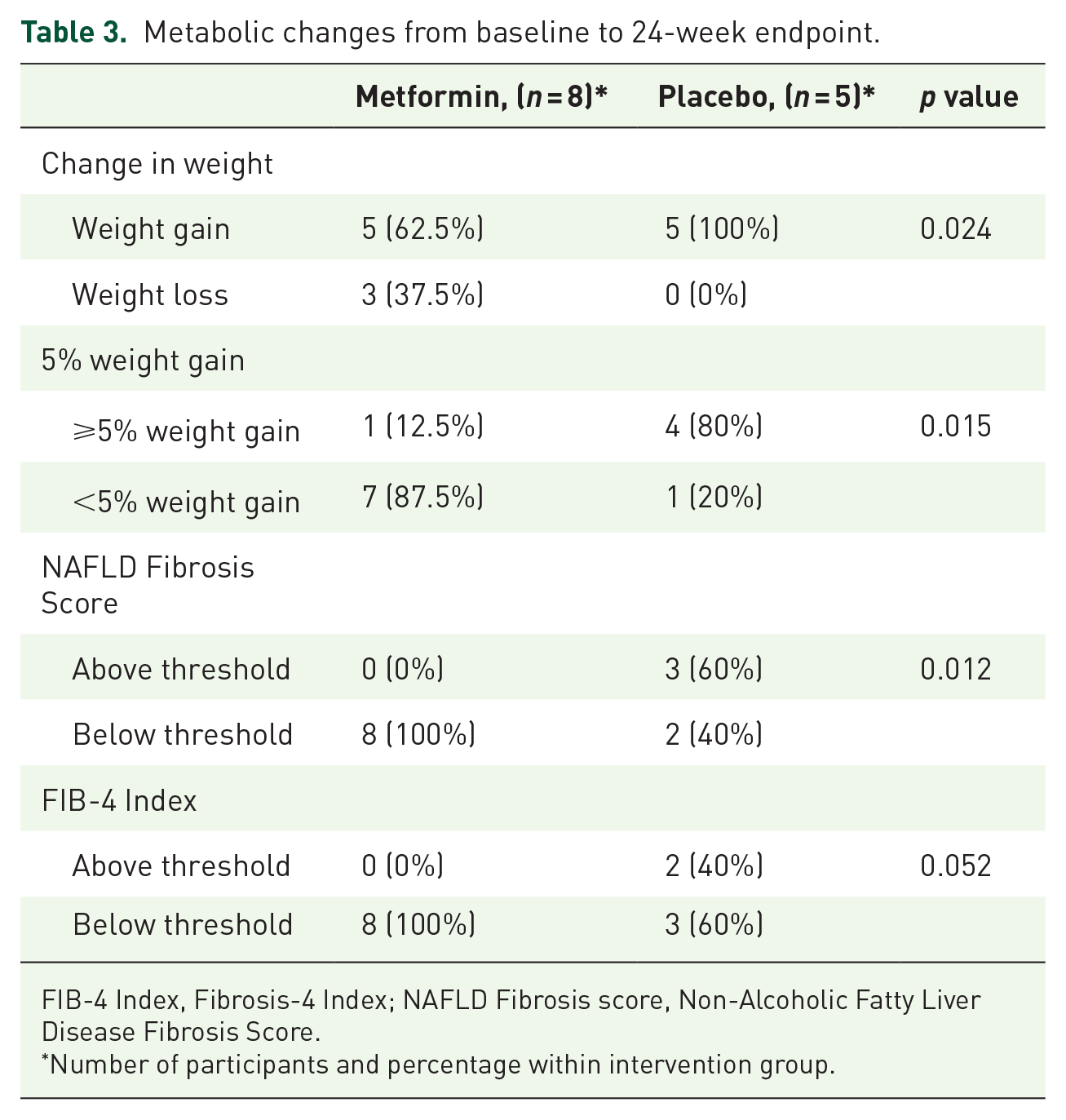
FIB-4 Index, Fibrosis-4 Index; NAFLD Fibrosis score, Non-Alcoholic Fatty Liver Disease Fibrosis Score.
Number of participants and percentage within intervention group.
Using the NAFLD Fibrosis Score threshold for excluding NAFLD, all metformin participants were below the threshold, compared to 40% of the placebo group (p = 0.012). Using the FIB-4 Index threshold for excluding NAFLD, the result failed to reach statistical significance (100% vs 60%, p = 0.052; Table 3). No participants had NAFLD Fibrosis Scores or FIB-4 Indices above the threshold for high risk of hepatic fibrosis.
There was no difference between the groups on change in the Food Craving Inventory total score from baseline to endpoint (placebo: –16.2, SD: 29.2 versus metformin: –6.3, SD: 24.0, p = 0.480).
There was no statistically significant difference between participants in the metformin and placebo groups for any of the ADRs (Supplementary Table 1). The most common ADR was tachycardia, in 50% of all participants in both groups. This is most likely to be secondary to commencement of clozapine than the intervention. No participants in either group had Vitamin B12 deficiency, an ADR associated with metformin previously reported in the literature. 35 No participants developed diabetes based on fasting glucose, HbA1c or OGTT.
Discussion
This study is the first double-blind RCT to examine the effectiveness of metformin on ameliorating weight gain among people commenced on clozapine. We were not able to achieve our recruitment targets due to early trial closure secondary to the COVID-19 global pandemic. Despite being underpowered for the primary outcome of change in weight between metformin and control, we did find that fewer people on metformin than placebo gained more than 5% body weight, a clinically significant change in body weight 36 and that significantly more people on clozapine than placebo lost weight. The number needed to treat to prevent a >5% body weight gain was 1.5. These results should be viewed with caution in the context of the small sample size and require further exploration in future adequately powered studies.
These findings are in keeping with a recent retrospective cohort study by our group. We previously found that in the year after commencement of clozapine, people who were commenced on metformin by their treating team had significantly less weight gain than those who were not commenced on metformin. 21 Two previous RCTs examined metformin co-commencement with olanzapine, an antipsychotic with a similar, albeit slightly less-adverse, metabolic profile to clozapine. One RCT demonstrated amelioration of weight gain, 37 while the other RCT had equivocal results. 38
Although change in weight is a proxy measure for future cardiometabolic disease, it remains an important issue for people with schizophrenia. Weight gain is the most distressing side effect reported to callers of mental health helplines 22 and is associated with poorer quality of life outcomes 39 and barriers to social engagement. 40 Weight gain may compromise adherence with treatment. 22 Greater body weight is associated with higher mortality risk, with all-cause mortality risks increasing for those who are overweight and obese.41,42 In addition, obesity has also been found to increase risk of accelerated brain ageing in schizophrenia. 43
We found statistically significantly greater reductions in the metformin group than the placebo group for HbA1c and diastolic BP, although these reductions were not clinically meaningful. Previous studies of hypoglycaemic medications among people on clozapine have shown reductions in HbA1c38,39 although it is possible that our findings may reflect a Type-I error associated with multiple analyses.
Our results on NAFLD warrant further inquiry. Obesity and NAFLDs are often comorbid. 44 The rapid weight gain that can occur with clozapine commencement increases the risks of developing NAFLDs. NAFLD increases the risk of T2DM, cardiovascular disease, liver disease and chronic kidney disease. 45 If metformin can reduce clozapine-associated weight gain, it may also reduce the development of NAFLDs. This finding needs further exploration in future studies.
Rates of ADRs were not different between the placebo and metformin groups reflecting findings from previous studies in already obese patients taking stable doses of clozapine. 20 Our findings add further evidence that metformin is well tolerated among people with schizophrenia.
Our study had several significant limitations. First, due to the COVID-19 pandemic, we were not able to achieve our recruitment target, and as such, we were underpowered to undertake analysis for the primary outcome. With the smaller sample size, there were baseline differences between the metformin and placebo groups in terms of weight, BMI and waist circumference. It is unclear how the greater baseline BMI of the placebo group would have influenced the weight trajectory of the participants. Participants with lower baseline BMI, as in the metformin group, may have had greater opportunity for weight gain, although people with higher baseline BMI, as in the placebo group, may have had greater innate risk of further weight gain. Of note, the two participants with the greatest weight gain (one on placebo and one on metformin) both were in the normal weight range at baseline. We do not have genetic data to confirm whether these participants had variants of the LEP and/or HTR2C genes. The differences in baseline weight and BMI between the groups meant we had to change our pre-published planned primary analysis outcome from difference between groups on endpoint weight to difference between groups on change in weight. Using repeated measures analysis with two-way analysis of variance yielded the same statistical significance irrespective of whether change or endpoint data are used. When there are baseline differences in weight between intervention and control groups, the magnitude of differences between intervention and control groups on endpoint weight can be more challenging to interpret than change in weight. We did not have data on prior antipsychotic and dose for the full 6 months before commencing clozapine, and as such, were unable to include this as co-variate in the analysis. Although the collection of the variables required for calculating NAFLD and FIB-4 Scores was described into our published study protocol, we overlooked specifically mentioning NAFLD and FIB-4.
Even prior to the premature closure of recruitment, we found that one of the challenges in recruitment was the high rate of use of metformin among people with schizophrenia, in the absence of T2DM. This may reflect influence of the ongoing research on metabolic disorders among people on antipsychotics undertaken by our group on the clinical services in which we are embedded.
In conclusion, while there are significant limitations related to the small sample size, our findings are consistent with previous findings from observational studies, suggesting that metformin co-commencement with clozapine may reduce risk of clinically significant weight gain, without significant risk of ADRs. The risks and benefits of metformin should be discussed with patients and their carers for consideration of co-commencement at time of clozapine initiation. Further studies are required to confirm this result prior to a formal recommendation for routine co-commencement of metformin with clozapine.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253211045248 – Supplemental material for CoMET: a randomised controlled trial of co-commencement of metformin versus placebo as an adjunctive treatment to attenuate weight gain in patients with schizophrenia newly commenced on clozapine
Supplemental material, sj-docx-1-tpp-10.1177_20451253211045248 for CoMET: a randomised controlled trial of co-commencement of metformin versus placebo as an adjunctive treatment to attenuate weight gain in patients with schizophrenia newly commenced on clozapine by Dan Siskind, Anthony W. Russell, Shuichi Suetani, Dylan Flaws, Steve Kisely, Vikas Moudgil, Korinne Northwood, Gail Robinson, James G. Scott, Terry Stedman, Nicola Warren, Karl Winckel, Peter Cosgrove and Andrea Baker in Therapeutic Advances in Psychopharmacology
Footnotes
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: D.S. and this clinical trial were supported in part by an NHMRC Early Career Fellowship (grant no. GNT1111136 (2016–2019)). This trial was also supported in part by the 2018 Society for Mental Health Research Early Career Researcher Project Grant Scheme. D.S. is supported in part by an NHMRC Emerging Leadership Fellowship (grant no. 1194635 (2021–2025)). S.S. is on Seqirus advisory board.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
