Abstract
There is still much to learn about the predictors of therapeutic response in psychiatry, but progress is gradually being made and precision psychiatry is an exciting and emerging subspeciality in this field. This is critically important in the treatment of refractory psychotic disorders, where clozapine is the only evidence-based treatment but only about half the patients experience an adequate response. In this case report, we explore the possible biological mechanisms underlying treatment failure and discuss possible ways of improving clinical outcomes. Further work is required to fully understand why some patients fail to respond to the most effective treatment in refractory schizophrenia. Therapeutic drug monitoring together with early pharmacogenetic testing may offer a path for some patients with refractory psychotic symptoms unresponsive to clozapine treatment.
Background
About a third of patients with schizophrenia are refractory to conventional antipsychotic treatments.1,2 For these patients, clozapine is the only evidence-based and licensed treatment. There is now some understanding of the neurobiological basis of treatment resistance, 3 but efforts to understand why clozapine is uniquely effective still proves elusive. Although there are possible candidates, no consistent predictors of clozapine response have been identified. Investigation of various clinical, demographic, neuroimaging and genetic predictors of clozapine response have yielded inconclusive results.4,5 There is some evidence that clozapine plasma levels greater than 350 µg/L predict response, but contradictory data exist.6,7 Several guidelines advocate clozapine therapeutic drug monitoring because of the significant inter-individual variation that exists in clozapine metabolism and pharmacokinetics. 8 Genetic and environmental factors contribute to this variation, which may ultimately determine the clinical outcome in patients with refractory psychotic disorders.
Case presentation
The patient is a 22-yea-old Black British male diagnosed with treatment-resistant schizoaffective disorder. The onset of his illness was at age 16 years, just before his General Certificate of Secondary Education examinations. His first psychiatric admission, in the same year, was to the adolescent unit where he was treated with risperidone up to 4 mg daily without response. He was then switched to olanzapine, to which he showed some response but experienced weight gain. He was then trialled on lurasidone, on which he was subsequently discharged. He experienced further psychiatric admissions during which various other antipsychotics, including haloperidol and the long-acting injection, paliperidone, were tried, without much success. Owing to the prominent affective symptoms, lithium was added 2 years later, with some success. At 4 years after his onset of illness, following unsuccessful treatments with multiple oral and depot antipsychotics and another psychiatric admission, clozapine was initiated due to the unrelenting nature of the psychotic symptoms characterised by thought disorder, bizarre religious delusions, paranoid delusions, and delusions of reference and auditory hallucinations. Based on this, our patient met the consensus criteria for treatment resistance. 9
The clozapine dose was gradually increased until a dose of 850 mg daily was reached. There was little change in clozapine levels between his dose at 500 mg and that at 850 mg daily, averaging 240 µg/L. He was eventually discharged to the home treatment team, with intensive input and daily reviews at home. At discharge, he was prescribed clozapine 850 mg daily and lithium 1200 mg daily (plasma level of 0.7 mmol/L). Although there was some improvement in his symptoms at discharge, 2 months later, he was referred to the Treatment Review & Assistance Team (TREAT) 10 service because of persistent paranoid delusions, thought disorder and disinhibited behaviour. The TREAT service at the South London and Maudsley NHS Foundation Trust provides specialist treatments for patients with ongoing or difficult to treat symptoms and investigates the causes of non-response. All investigations, including magnetic resonance imaging and autoantibodies, were within normal limits. It was suggested that clozapine levels should be optimised to between 350 µg/L and 500 µg/L, 11 although how this was to be achieved was not stated.
The following year, there was a major deterioration in his mental health, resulting in another hospital admission. He was eventually transferred to our Psychiatric Intensive Care Unit (PICU) as he became increasingly sexually disinhibited, thought disordered, verbally and physically aggressive, and unmanageable on an open ward. At this point, the clozapine dose was 850 mg daily. During the admission, clozapine was increased further to 950 mg (50 mg above the maximum British National Formulary permitted dose) due to persistently low clozapine levels. The graph of clozapine dose and plasma levels is presented in Figure 1. Questions about compliance were raised and he was put on enhanced nursing observations that confirmed that he was compliant with treatment. Meanwhile, clozapine levels remained largely unchanged at an average of 250 µg/L despite dose increments. He was not on any coprescribed medication known to affect clozapine metabolism; he was also a non-smoker.

Clozapine dose and plasma levels.
For several months during the admission, the patient remained profoundly psychotic as evidenced by hallucinatory and bizarre behaviour, running in the corridors naked, dancing in front of the camera and speaking to the closed-circuit television. Clinical assessment at 6 months of admission showed a Positive and Negative Syndrome Scale (PANSS) score of 109. Transfer out of PICU was impossible because of the nature and degree of his psychotic symptoms. Owing to the persistently low clozapine levels and lack of clinical improvement, the team decided to request a genetic test.
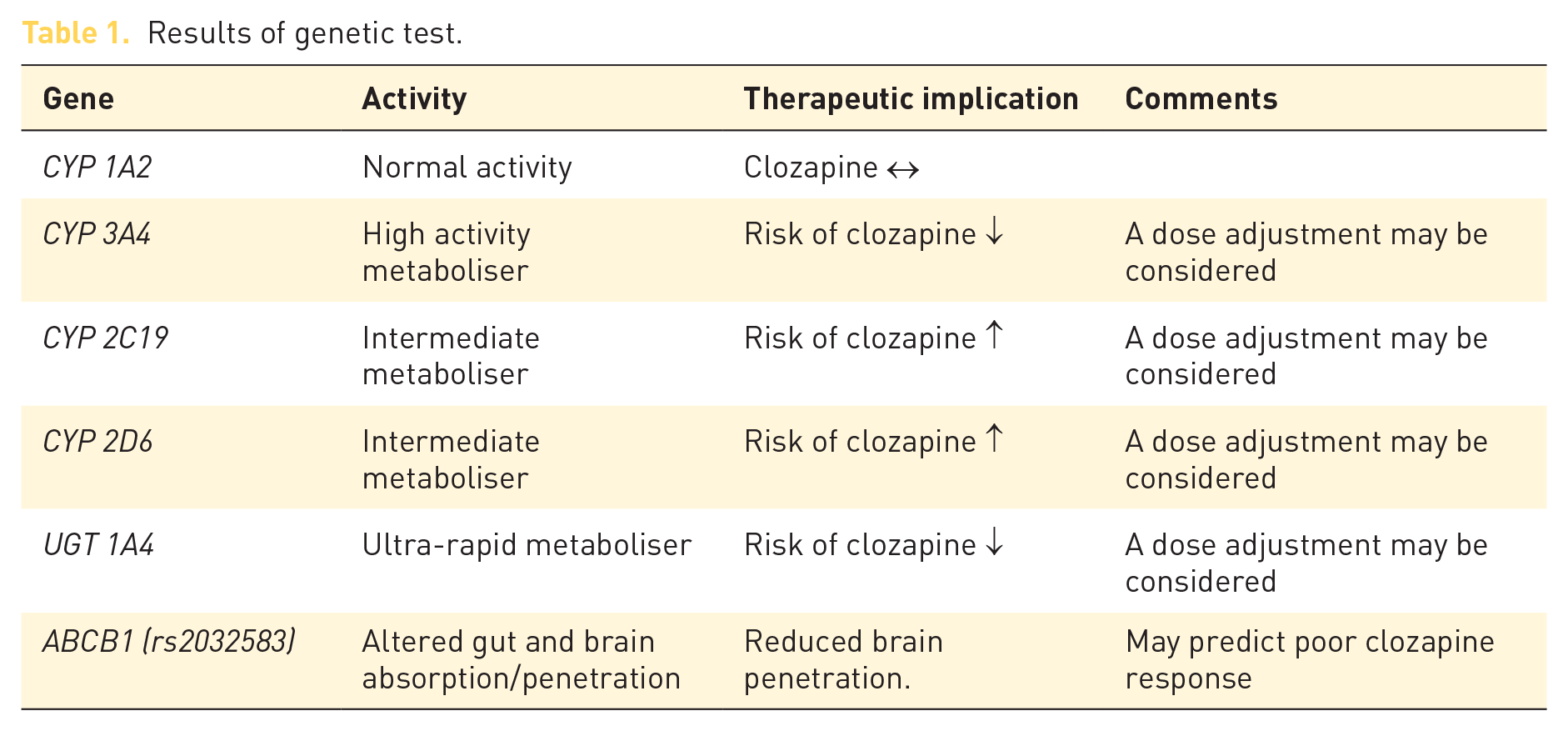
A genetic test was performed by Genomind® Professional PGx™, the summary results of which are displayed in Table 1 below.
Results of genetic test.
Intervention
Based on the results of the pharmacogenetic profile, we decided to give an empirical trial of fluvoxamine, a potent inhibitor of CYP 450 1A2 and 2C19, but with some activity on other CYP 450 enzymes. Fluvoxamine was initiated and kept at 25 mg daily throughout the period. The baseline clozapine level was 250 µg/L at a daily dose of 950 mg daily. The clozapine level was determined 12-h post-dose three times weekly using the Mycare™ point-of-care testing for clozapine. 12
Assessments
At the end of 1 week, clozapine levels had risen by 80% to an average of 450 µg/L and remained stable over the next 4 weeks (see Figure 1). There was no evidence of increased adverse effects as assessed using the Glasgow antipsychotic side-effects scale for clozapine. 13 Clinical assessment at 8 weeks showed significant improvement in psychotic symptoms and previously described behaviours substantially reduced with a 33% reduction in a PANSS score to 73, corresponding to an improvement from severely ill to moderately ill or approximating to a 2-point change in Clinical Global Impression scores.
Discussion
The superior efficacy of clozapine in treatment refractory schizophrenia (TRS) is unquestionable.14–16 However, even with clozapine, only about half of those with TRS achieve a satisfactory response.17,18 Hence, clozapine augmentation with a second antipsychotic is routine clinical practice, despite limited evidence.19–21 Data indicate that patients with TRS have normal dopamine function but elevated glutamate levels;3,22 however, neither the precise neurobiological mechanisms that underlie the superior efficacy of clozapine in patients with TRS nor definitive determinants of response to clozapine have been clearly identified.
Among the various possible determinants of clozapine treatment response is clozapine plasma levels, which are roughly related to dose, but there is significant inter-individual (up to 45-fold) variation in plasma levels on the same dose. 23 About 50% of this variation in plasma levels can be explained by the dose, patient’s age, sex, body weight, smoking status and the clozapine metabolic ratio. 24 Our patient is a non-smoker. Evidence from the literature suggests that clozapine plasma levels greater than 350 µg/L are associated with clinical response.6,8,25 Therefore, in our patient, treatment failure could be attributed, at least in part, to inadequate clozapine plasma levels.
Clozapine is metabolised primarily by CYP1A2 and 3A4, and to some extent by CYP2C19 and 2D6. However, CYP 1A2 appears to be the main route of clozapine metabolism.26,27 This is evidenced by the well-documented and clinically significant interaction and occasional toxicity observed in reported interactions between clozapine and CYP 1A2 inhibitors such as fluvoxamine.28–30 Induction of CYP 1A2 by smoking reduces clozapine levels and a significant increase leading to toxicity has been seen when people stop smoking.31,32 The effect and clinical consequences of inhibition of CYP 2D6, 3A4 and 2C19 on clozapine kinetics seem to be relatively modest.33–35
The pharmacogenetic profile of our patient showed normal activity at CYP 1A2, indicating no effect on clozapine metabolism and intermediate activity at CYP 2D6 and 2C19 that may, potentially, cause an increase in clozapine plasma levels. Perhaps of some relevance is the CYP 3A4 activity, where the patient is reported to show high activity metaboliser status that could potentially reduce clozapine levels. Although our patient had normal CYP 1A2 activity, we chose to use fluvoxamine because of its broad-spectrum activity on CYP 450 enzymes that could, potentially, affect clozapine metabolism. 36 Approximately 70% of the variation in clozapine clearance is due to CYP 1A2 activity; 37 hence, theoretically, up to a 10-fold increase in clozapine levels can be observed with fluvoxamine addition. 28 In our patient, unsurprisingly, we observed only a modest 80% increase in clozapine levels because of normal activity at CYP 1A2. It should be noted, however, that we used only 25 mg fluvoxamine and the effect is dose-dependent.
Of note is that our patient is an ultra-rapid metaboliser at UGT 1A4, which may also explain, in part, the observed low clozapine levels, although this is hypothesised to have a greater effect on norclozapine rather than clozapine levels. UDP-glucuronosyltransferase (UGT) enzymes comprise a family of proteins expressed in the liver that catalyse the glucuronidation of various drugs, including clozapine.38,39 Although it has been recognised, not much has been published on the role of and clinical significance of UGT enzymes in clozapine metabolism.40–42 Interestingly, many foods and medicinal plants contain ursolic acid and oleanolic acid, both of which can inhibit UGT1A4 activity and, therefore, have the potential to affect clozapine metabolism. 43
ABCB1 is a transmembrane transporter that is expressed in excretory organs such as the kidneys and liver, in intestinal mucosa and on the blood–brain barrier.44,45 In a study of patients with TRS, the presence of a specific genotype of ABCB1 was associated with therapeutic response. 46 Indeed, in a systematic review of gene polymorphisms potentially related to the pharmacokinetics of clozapine, the only gene polymorphism that has been found to be associated with clozapine plasma concentration and the therapeutic response was the ABCB1 gene. 47 In our patient, ABCB1 (rs2032583) is present. The potential contribution of this genetic variant on either the clozapine levels or the therapeutic response in our patient is unclear.
There is no doubt that the clozapine response is determined, to some extent, by genetic variability involved in its pharmacokinetic and pharmacodynamic characteristics. Some serotonin genes, i.e. HTR2A rs6314, rs6314 and HTR3A rs1062613, may be associated with the response. 48 The role of other genetic determinants in clozapine response, such as the nuclear factor 1 B-type, 49 is an exciting and developing area, but a full review is beyond the scope of this case report. Further, recent research suggests that polymorphisms at CYP 450 2C19 influence clozapine treatment response. 50
Several case reports and clinical studies where fluvoxamine has been used to increase clozapine levels have been published. These have been systematically reviewed. 37 This case is unique because the genetic profile of our patient demonstrates normal CYP 1A2 activity, resulting only in a modest rise in clozapine levels. It further shows that it is not clozapine plasma levels per se that makes the difference, but the amount of drug that enters the brain.
The possibility of other factors contributing to clozapine non-response in our patient cannot be ruled out. His illness started at a relatively young age and the age of onset of illness has been suggested as a predictor of response. 51 A more recent meta-analysis, however, did not find an association between the age of onset of illness and the therapeutic response to clozapine but, rather, a younger age may be associated with a better clinical response. 52 Delay in initiating clozapine is associated with poorer outcomes.53–55 It could be argued that there was some delay in clozapine initiation in our patient.
The prognosis for our patient is positive as his mental state continues to improve. He has a keen interest in mathematics and the sciences, engages well with occupational therapy and psychology and, most importantly, enjoys the support and encouragement of his family. Finally, although the role of pharmacogenetic testing was significant in our case, it must be borne in mind that the availability of testing in psychiatric clinics is still limited. We have used a commercially available test that may not provide a complete pharmacogenetic profile. These, however, should not preclude the use of fluvoxamine as an adjunct to optimise clozapine treatment in patients with low clozapine plasma levels.
Conclusion
Clozapine is the only evidence-based treatment in refractory schizophrenia. Unfortunately, almost half the patients fail to respond adequately. In addition, clozapine levels can be difficult to predict but optimising plasma levels can contribute to improving chances of successful clinical outcomes. The use of point-of-care finger prick testing for rapid therapeutic drug monitoring and pharmacogenetic testing may turn some clozapine non-responders into responders. It is our earnest expectation that personalised precision psychiatry is not a distant dream.
Footnotes
Acknowledgements
We thank our patient and his family, from whom we obtained permission, to share this learning. We thank our team and colleagues for their support in the care of the patient.
Authors’ contributions
EW compiled the initial draft. GR and KV made substantial contributions in revising the manuscript critically. DT supplied important intellectual content. KV was the lead clinician and EW, GR, MC, CA and ME were all directly involved in the treatment of the patient. All authors have read and approved the final manuscript.
Consent for publication
We obtained signed informed consent from the patient and his family for the publication of this report.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
