Abstract
Background and aims:
A growing body of research shows that race contributes to disparities in mental health services utilization and influences the clinical diagnostic process. To our knowledge, no studies on current practice in the Unites States have documented whether these disparities impact the prescription of antipsychotic medications across individual patients based on race. Consequently, this study aims to describe the prescribing patterns of antipsychotic medications in the inpatient setting based on patients’ race, and to explore appropriateness of therapy based on Food and Drug Administration labeling and avoidance of inappropriate polypharmacy.
Methods:
Single-centered, retrospective, chart review of 398 psychiatric patients in the inpatient setting and who had a psychiatric diagnosis that warranted a prescription for an antipsychotic medication at the time of discharge. Frequencies were computed to describe differences in demographic variables (race, health insurance type, age, and gender), medical conditions (diagnosis, commodities, hospitalization status, antipsychotic medications, etc.), and screening tests (lipid panel, hemoglobin, urine and illicit drug use). Logistic regression, analysis of variance, and hypothesis tests were used to analyze the data.
Results:
Significant differences were not found in total chlorpromazine equivalent dose equivalencies by race or insurance. However, patients of involuntary admission status, past medication trials, a diagnosis of schizophrenia or bipolar disorder, and who lacked family support had higher total daily doses of antipsychotics upon discharge. Inappropriate therapy was significantly related to differences in increasing age and a diagnosis of insomnia.
Conclusion:
This single-centered study described patterns of antipsychotic prescribing based on race in an inpatient psychiatry facility. Future studies, using larger and more diverse sample populations, are recommended to elucidate the role that patients’ race, admission status, and family support play in the dose and appropriateness of antipsychotics prescribed for mental health care.
Background
In a 2015 survey performed in Louisiana, USA, 4.5% of adults had a diagnosable mental disorder resulting in serious functional impairment, though only 40.6% of those received mental healthcare services (inpatient treatment/counseling, outpatient treatment/counseling or prescription medication for problems with emotions, nerves, or mental health). 1 Although rates of any mental illnesses, excluding substance abuse (alcohol and illicit drugs) and developmental disorders (attention deficit hyperactivity disorder, autism, conduct disorder, etc.), are similar in African-Americans and the general population, disparities exist regarding mental health care services utilization across different levels of care. 2 In the 2012 National Surveys on Drug Use and Health, adult respondents were asked about any treatment or counseling services received for any problem related to emotions or mental health, except substance use disorders. Results showed that compared with non-Hispanic Whites, African-Americans self-reported half the rates of mental health service use (16.6% and 8.6%, respectively), while Whites reported greater use of outpatient services (7.8%) and prescription psychiatric medications (14.4%) than African-Americans (4.7% and 6.5%, respectively). On the other hand, African-Americans reported higher use of inpatient services (1.4%) than Whites (0.7%).3,4 Also, despite presenting with similar symptoms to their White counterparts, African -Americans are three-fold more likely to be diagnosed with schizophrenia or a psychotic disorder, and less likely than Whites to be diagnosed with mood disorders, such as bipolar disorder, even after adjusting for selected patient and hospital characteristics. 5 As a result, African-Americans are being exposed more frequently while on the inpatient setting to antipsychotic medications used to treat schizophrenia and other psychotic disorders.
Previous literature has shown that, compared with non-Hispanic Whites, African-Americans are more commonly prescribed long-acting injectable formulations of antipsychotics in both the inpatient and the outpatient setting, as well as being prescribed older agents in the outpatient setting.6–8 In terms of antipsychotic dosing and polypharmacy, previous studies around the turn of the century in the US have illustrated that antipsychotic exposures were higher in African-American populations, and the drivers of these exposures were first generation, depot antipsychotics.9–11 Of note, prescribing patterns have shifted since this period to a focus on second generation antipsychotics. Since then, several UK studies with prescribing patterns more similar to current practice reported a lack of difference in dose, type, polypharmacy, and costs of antipsychotics. The authors of these reports suggested that this lack of difference could be attributable to a more equitable healthcare system in the UK, a diverse multicultural mental health workforce, 12 and changing attitudes to racism.13–15 Because increased exposure to antipsychotic therapy can increase the risk of unwanted side effects, evidence in dose and adherence to treatment is needed. The limited available evidence suggests no benefit from high doses of antipsychotic medication, and treatment-related side effects are likely to be increased. 16 Similarly, previous guidelines have consistently recommended against the use of multiple antipsychotics except when the patient has failed multiple antipsychotics, including clozapine, as monotherapy,17–20 though recent literature has suggested polypharmacy may lower rehospitalization rates. 19 Nonetheless, the risks of harmful side effects such as movement disorders or metabolic concerns with increased antipsychotic exposure remain even when controlling by dose or by number of antipsychotic medications.21–23
Consequently, this study aims to describe the prescribing patterns of antipsychotic medications in an inpatient setting based on differences in race with a focus on medication dose, adherence to Food and Drug Administration (FDA) labeling, and avoidance of inappropriate polypharmacy. The primary objective of this study was to describe the prescribing patterns of antipsychotic medications for patients in the sample population and find out whether there are significant differences by racial/ethnical backgrounds. A secondary objective was to explore appropriateness of therapy based on FDA labeling, and avoidance of inappropriate polypharmacy in order to target potential mental health care disparities related to antipsychotic medication exposure.
Methods
This study was a single-centered, retrospective, chart review conducted at an academic hospital, the University Medical Center at New Orleans (UMCNO), in the inpatient behavioral health setting. Institutional Review Board (IRB) approval was acquired by Xavier University of Louisiana (IRB #685) and confirmed by the UMCNO Research Regulatory and Compliance Committee. Information was collected on a total of 398 randomly selected adult patients, of any gender, who were hospitalized during the study period and who had a psychiatric diagnosis that warranted a prescription for an antipsychotic medication at the time of discharge. The review was conducted from 1 October 2015 to 31 December 2017. Patients were excluded if (1) they were not prescribed a scheduled dose of an oral formulation of an antipsychotic at the time of discharge and/or were not administered a long-acting injectable formulation of an antipsychotic just prior to discharge; (2) they became medically destabilized or transferred to another unit of the hospital during the study timeline; (3) were hospitalized on other units of the hospital; or (4) did not have a psychiatric diagnosis at discharge. A sampling frame collected from electronic medical records was generated, with a total of 4071 patient encounters identified as meeting criteria. A final sample of 398 cases in total were selected at random through the utilization of the random number generator function in Excel.
Demographic data such as race, ethnicity, age, gender, weight, height, diagnosis, health insurance, and legal status were collected on each patient. Differences in antipsychotic drug, dose, type, number, and duration of treatment were characterized, and all dosing were standardized to “chlorpromazine equivalents” (CPZEs). Chlorpromazine was the prototypical first-generation antipsychotic, and it is used as the standard for classifying antipsychotic dosing equivalency.24,25 Data collected about adherence to FDA-guidance included indication, contraindications, dosing range, inappropriate use of multiple antipsychotics, and conversion of oral to long-acting injectable formulations (if applicable). Appropriateness of medications was categorized by the following criteria: (1) antipsychotics prescribed for an FDA-approved diagnosis, (2) dosing ranges following FDA-approved guidelines, (3) polypharmacy use only in treatment-refractory disease, (4) avoidance of use when documented contraindications to therapy existed, (5) avoidance of use with verified allergies to a particular medication. FDA labeling was defined by indication, dosing range, and contraindications listed in the package insert. Treatment-refractory disease was defined as two or more documented antipsychotic trial failures based on lack of efficacy, and inappropriate polypharmacy was defined as multiple antipsychotics prescribed concomitantly at discharge, with no documentation of a history of clozapine use or a short-term cross-taper from one antipsychotic to another.
Data was analyzed using the R statistical language, and the Holm and Bonferroni multiple comparison corrections were used.
Total CPZE (T-CPZE) dose was modeled using analysis of variance. The following explanatory variables were considered for the model: race, ethnicity, psychiatric diagnosis, comorbidities (such as intellectual disorder, conduct disorder, personality disorder, substance use disorder, and insomnia), age, weight, height, pregnancy, insurance, admission legal status (voluntary or involuntary), social support (defined as either family/friends or other home support), total past antipsychotic trials. The analysis used square root of T-CPZE as the response variable after preliminary tests showed that a variance stabilizing transformation was needed.
Whether the medication therapy followed FDA labeling and avoided inappropriate polypharmacy for the patient was modeled using logistic regression and the same explanatory variables as T-CPZE were considered for the model.
Results
Charts were reviewed for a total of 398 patients meeting the inclusion criteria. The majority of patients (Table 1) were African-American (66%) and non-Hispanic (91%); had a diagnosis of schizophrenia (50%); were involuntary hospitalized (51%); and had three or more past antipsychotic trials (38%).
Demographics of patients in the sample (N = 398).

Asians, Hispanics, other groups, or no response.
Totals include total past trials, not patient cases.
EPS, extrapyramidal symptoms; THC, tetrahydrocannabinol; BZD, benzodiazepines.
Total of psychiatric medication doses (CPZEs)
Although significant differences were not found in T-CPZE dose equivalencies by race or insurance, significant differences were found in T-CPZE values by legal hospitalization status, total past antipsychotic trials, psychiatric diagnosis, and family support (Table 2).
Mean values of total chlorpromazine equivalent dosing by different patient characteristics (N = 398).
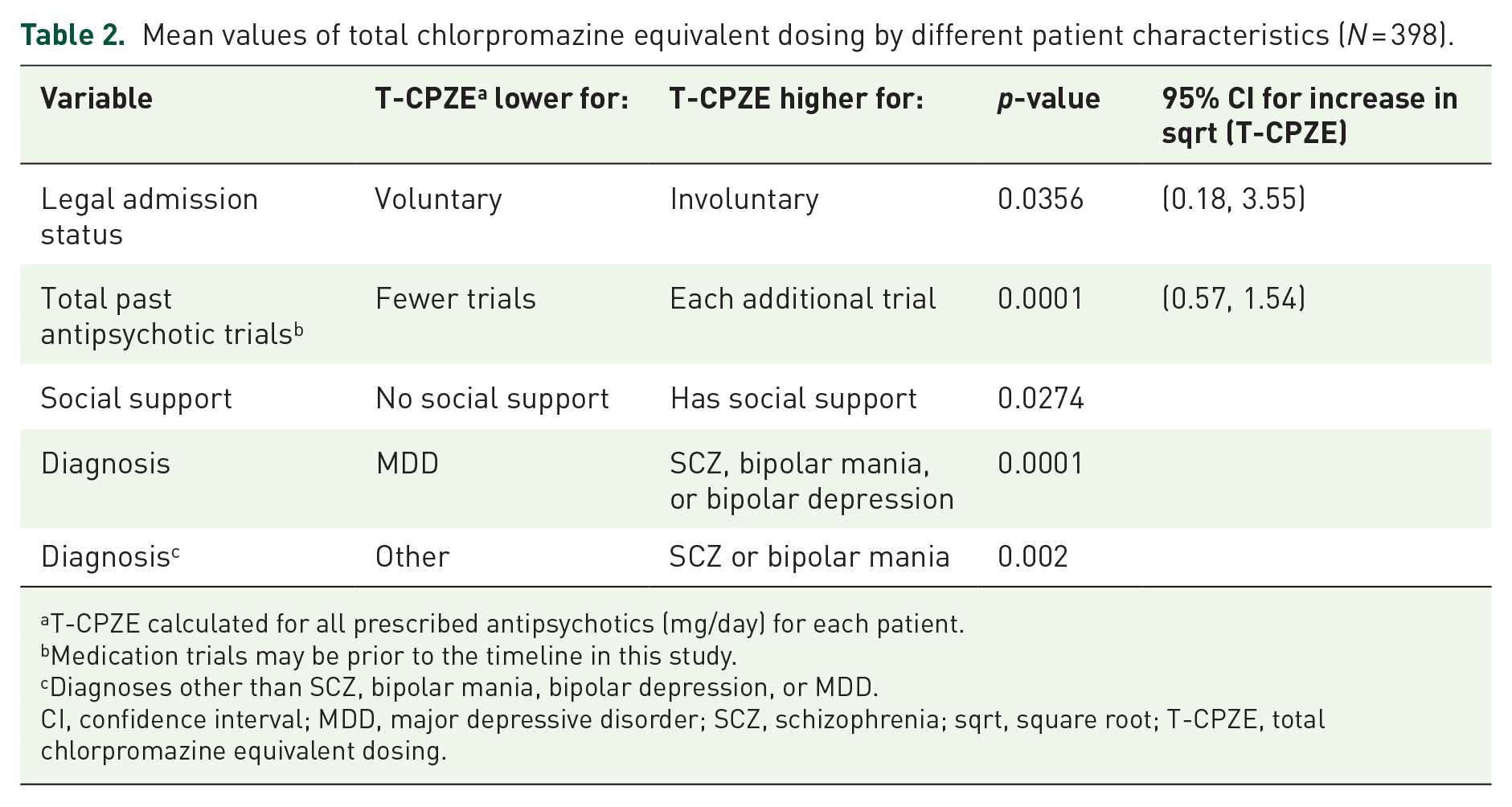
T-CPZE calculated for all prescribed antipsychotics (mg/day) for each patient.
Medication trials may be prior to the timeline in this study.
Diagnoses other than SCZ, bipolar mania, bipolar depression, or MDD.
CI, confidence interval; MDD, major depressive disorder; SCZ, schizophrenia; sqrt, square root; T-CPZE, total chlorpromazine equivalent dosing.
Specifically, patients who were forced to receive treatment for their mental illnesses without their consent (involuntary patients) had significantly higher mean T-CPZE values than those who voluntarily consented to receive treatment.
There was a significant increase in the T-CPZE related to each additional previous antipsychotic exposure. That is, patients who had been exposed to antipsychotic medications, often more frequently for the same mental health conditions in the past, had significantly elevated T-CPZE during the investigation period.
Psychiatric diagnosis also had a highly significant impact on T-CPZE, where patients with a major depressive disorder (MDD) diagnosis had a significantly lower T-CPZE than patients with a schizophrenia (SCZ) spectrum, bipolar mania, or bipolar depression diagnosis (p < 0.001). Also, patients with an ‘other diagnosis’ (such as schizoaffective disorder, substance induced mood or psychotic disorder) had a significantly lower T-CPZE than patients with a SCZ spectrum or bipolar mania diagnosis (p < 0.002).
Patients with reported social support had significantly lower T-CPZE values than patients without social support. Of note, African-American patients were more likely to lack social support than those of other races (p = 0.005).
Additionally, patients who declined to state their race had a marginally lower T-CPZE than those patients who self-identified as African-Americans (p = 0.063).
Last, women had significantly lower T-CPZE than men (p = 0.04). However, the effect was only marginally significant (p = 0.07) once other variables (legal status, ethnicity, psychiatric diagnosis, total past antipsychotic trials, and social support) were considered.
Appropriateness of therapy: following FDA-labeling and avoiding inappropriate polypharmacy
For all conditions together, patients with comorbid insomnia (p = 0.0281) and patients of older age (p = 0.0418) were significantly less likely to be given the appropriate medications than their counterparts (Table 3).
Estimated odds ratios, along with 95% confidence intervals.

Discussion
This single-centered retrospective study reviewed a random sample of 398 patients to describe the prescribing patterns of antipsychotics based on race and other patient characteristics. Findings of this study showed that regardless of race, patients of involuntary admission status, past medication trials, a diagnosis of schizophrenia or bipolar disorder, and lack of family support had higher total daily doses of antipsychotics upon discharge from the hospital. In this sample, there was no significant difference found in the T-CPZE dose based on race, though it is important to note that African-American patients were more likely to lack social support than White patients (p = 0.005). Exploring the role of race and social support on antipsychotic dose could elucidate this finding further.
A higher rate of antipsychotic exposure was found across all races in patients treated specifically for a diagnosis of schizophrenia. The diagnostic disparity found in this and previous studies5,26 could lend to a higher risk of antipsychotic exposure in the African-American population in particular. It could be hypothesized that an initial racial diagnostic bias towards African-Americans leads to long-term downstream effects on antipsychotic dose exposure where each inpatient antipsychotic exposure leads to a significant increase in antipsychotic dose in subsequent hospitalizations. A diagnostic bias could lead to improper treatment with an antipsychotic, 27 with risks increasing at each subsequent hospitalization as each hospitalization is associated with a higher dose of antipsychotic. It is important to note that the diagnosis of schizophrenia and other psychiatric disorders are clinical in nature; however, the validity of diagnosis was not assessed in this study, and future studies would be merited on this topic.
Involuntary admission status was also associated with a higher T-CPZE compared with voluntarily admitted counterparts. Although there is not a clear explanation of this difference, acuity of illness as well as aggressive treatment for rapid stabilization of symptoms could lead to higher dosing in this population. Though there was no difference based on race in terms of admission legal status, the majority of the patients with involuntary admission were African-Americans. Although other factors, not included in this study, may explain higher doses for involuntary admission, previous studies have shown that African-American populations receive more inpatient mental health services.3,4 This could be due to lack of availability of resources, access, stigma, or mistrust and may result in less utilization of outpatient resources. It is possible that though there may not be a significant difference in antipsychotic dose exposure between individual patients based on race in the inpatient setting, African-American populations could be exposed disproportionately to higher doses of antipsychotics due to higher rates of inpatient admissions. 28
In terms of appropriateness of therapy, patients with co-morbid insomnia and patients of older age were more likely to receive medications that did not follow strict FDA-labeling. Appropriateness was defined by deviations from FDA-labeling in terms of dose, indication, or contraindication.
Patients with insomnia were found less likely to be given appropriate medications based on FDA-labeling. This is an interesting find as an insomnia can be a symptom or harbinger of other psychiatric disorders. It can also be comorbid with other psychiatric disorders, thereby adding to the medical burden and increasing the risk of psychiatric relapse. 29 Because it is possible that patients could have been treated with an antipsychotic for the sole indication of insomnia or as part of a complex regimen for acute psychiatric illness, the role of antipsychotic therapy in the treatment involving insomnia should be explored in greater detail in future studies. Though none of the currently marketed antipsychotics have an FDA-labeling for insomnia, this modality is often used in clinical practice despite other alternatives. Caution should be taken when treating insomnia with an antipsychotic due to the risk of side effects, particularly in older patients, as those in our sample.
Older patients were also less likely to be given appropriate medications than younger populations. It is possible that older patients had a more treatment-refractory course, higher chronicity, and thus required more medication to treat symptoms appropriately.
A final note in terms of prescribing patterns seen in this analysis in patients of all races. There were two areas of note regarding current guidelines: a lack of documentation of metabolic panels in patients on antipsychotic therapy, and an underutilization of clozapine. First, metabolic panels are recommended at baseline, then initially every 3 months following antipsychotic therapy, 23 though over 80% of patients in this study did not have a hemoglobin A1c or lipid panel documented in their chart from the current admission. The second area of note regards the possible underutilization of clozapine, consistent with previous reports.30–32 Although a total of 70 patients were discharged with two or more antipsychotics (Table 1), only 23 of these patients (32.9%) had a past trial of clozapine. Data on which patients qualify for clozapine falls outside of the scope of this report, though it is likely that a subset of patients on dual antipsychotic therapy would have been proper candidates for this medication.
Limitations of this study include limited sample size documented through chart review from one hospital. Future studies should focus on expanding the sample size and spreading the population across multiple hospitals in order to become more generalizable as the description in this study could be a factor of the largely African-American, lower socioeconomic status, public health insurance population. Data was also not always clearly documented in patient charts, which may have affected some of the results. As this study was descriptive in nature, it may be more hypothesis-generating and follow-up studies are merited.
Though this study was unique in looking at total daily dose at discharge based on CPZEs, as well as appropriateness of therapy defined by FDA-labeling and avoidance of unnecessary polypharmacy, there are some potential limitations here as well. While CPZEs are the recognized means of standardizing antipsychotic dosing, there could be some variation between patients as blood plasma levels of antipsychotics do not necessarily correlate to efficacy, 33 and there is some debate in the literature over how to optimally calculate dose equivalence between antipsychotics.25,34 Also, while treatment is recommended to follow FDA-indication, this does not necessarily correlate with real-world practice as it can be difficult to ascertain a proper diagnosis during the acute episode of illness, and antipsychotics are often used in the acute treatment of comorbid symptoms, including insomnia. This could have led to mislabeling the practice as “inappropriate use of medication.”
Conclusion
This study described patterns of antipsychotic prescribing in patients of one hospital comprising a largely African-American population receiving public health insurance. Although no significant differences were found by race in terms of dosing, higher doses were found significantly in patients with involuntary admission status, increasing number of past medication trials, a diagnosis of schizophrenia or bipolar disorder, and lack of family support. Although higher doses of T-CPZE in patients with higher number of past medication trials or those with diagnosis of schizophrenia or bipolar disorder are explained by differences in treatment goals (antipsychotic drugs are used mainly as adjunctive treatment in MDD), differences for patients who are receiving treatment for legal requirements or those who do not have family support may be explained by other factors not considered in this study. Insomnia and age also played a role in prescribing patterns following FDA recommendations and current guidelines. While race did not play a large role in antipsychotic prescribing patterns, this descriptive study offers insight into patient characteristics that are associated with increased or possibly unnecessary exposure to antipsychotic therapy. Future studies in expanded populations are highly recommended to fully elucidate the role of race, as well as other patient characteristics, in the dose and appropriateness of antipsychotic therapy on the inpatient setting of behavioral health facilities.
Footnotes
Acknowledgements
Ethan Gipson, PharmD Candidate, Xavier University of Louisiana, Kayla Williams, PharmD Candidate, Xavier University of Louisiana, Donald Dang, PharmD Candidate, Xavier University of Louisiana.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
