Abstract
Aims:
This cross-sectional pharmacoepidemiologic study examined the prevalence of polypharmacy and psychotropic polypharmacy among inpatients in a tertiary psychiatric hospital in Belgium.
Methods:
Current prescriptions of all inpatients suffering from mental disorders were extracted from the hospital Computerized Physician Order Entry. Two methods were used to examine definitive polypharmacy (defined as the concomitant use of at least five medicines): number of medicines per active component and per prescription. Psychotropic polypharmacy was defined as the concomitant use of at least two psychotropic medicines, based on the first counting, i.e., per active component.
Results:
In 292 included patients, the prevalence of definitive polypharmacy was 65.8%, with a mean number of 6.8 ± 4.2 medicines per patient. The most prevalent medicines were related to the central nervous system (55.7%), followed by medicines related to the gastro-intestinal (17.6%) and cardiovascular (9.4%) systems. A prevalence of psychotropic polypharmacy of 78.1% was observed, with a mean of 3.0 ± 1.7 psychotropic medicines per patient. Psychotropic polypharmacy was classified in same-class (71.5%), multi-class (82.5%), augmentation (20.6%), and adjuvant (35.5%) polypharmacy.
Conclusion:
These findings are consistent with previous reports of highly prevalent polypharmacy in patients with mental disorders. Although, in some cases, polypharmacy can be an important part of good clinical practice, the high prevalence of both polypharmacy and psychotropic polypharmacy emphasizes that attention must be paid to the potentially associated risks. Consensus on the definition and method of determination of polypharmacy is needed to support further research.
Introduction
In the general population there has been an increasing trend towards polypharmacy, commonly defined as the concomitant use of five or more medicines, over the last number of decades. 1 The prevalence of polypharmacy is, in some ways, unsurprising in people with psychiatric illness, considering the complex mental health presentation and the high prevalence of multimorbidity.1,2
Worldwide, trends in psychotropic polypharmacy, commonly defined as the concomitant use of two or more psychotropic medicines, are increasing.3–5 This finding is not in keeping with common guidelines in psychiatric care that tend to promote psychotropic monotherapy. 6 In patients experiencing a serious psychiatric disorder, such as schizophrenia and bipolar disorder, studies often demonstrate more complex psychotropic polypharmacy, defined as the concomitant use of four or more psychotropic medicines, with prevalence estimates of approximately one-third of patients.7,8 Causes for this discrepancy between theory and practice are many. A high comorbidity among people with psychiatric disorders contributes to psychotropic polypharmacy. 9 Moreover, it is assumed that lack of knowledge about the aetiology and the physiopathology of psychiatric disorders, as well as the limited pharmacological targets, the restricted efficiency, and the large differences in response are important contributors. 10 A study showing polypharmacy in patients with borderline personality disorder, 11 a disorder for which there are no evidence-based psychopharmacological treatment guidelines, highlights high rates of off-label use of psychotropic medicines. 12 Most evidence regarding psychotropic medicines is derived from clinical trials with relatively small samples of highly selected patients. Additionally, partial response to psychotropic medicines may contribute to the addition of such medicines. Furthermore, restraint towards deprescribing psychotropic medicines is seen even when the effects are limited or nonexistant. 13 Other, more socio-cultural explanations for psychotropic polypharmacy could be the expectations of patients, social pressure, and discrepancy between the subjective urge for treatment and the time needed for ideal therapy start up. 10
Strictly speaking, polypharmacy according to the numerical definition is not necessarily bad practice.1,14 Polypharmacy is, however, established as a health risk and is associated with increased mortality. 15 Previous research in Belgium reveals that patients leaving a psychiatric hospital take an average of 6.5 medicines having 3.1 possible drug–drug interactions. 10 Patients taking multiple medicines are at risk of drug-related problems, such as drug–drug interactions or side effects. 16 Additionally, polypharmacy has proven to cause more potentially inappropriate prescribing such as contra-indicated medicines, under or overdosing of medicines, combining medicines with opposite pharmacodynamics, etc. 17 The literature shows a high incidence of drug-related problems (15.8%) and potentially inappropriate prescribing (59%) in psychiatric patients.16,17 Furthermore, a complex medicine schedule with multiple medicines and different times of administration may reduce adherence.
In the assessment of polypharmacy, some have suggested that a purely numerical approach is insufficient, especially when it comes to drawing conclusions about the appropriateness of treatment. 18 There is, however, no consensus about another way of defining polypharmacy.
The first aim of this study was to develop a clear and comprehensive method to determine polypharmacy. It was hypothesized that the number of active components constituted the major factor in the determination of polypharmacy, particularly since the active components contributed to the greatest pharmacological risks in terms of drug–drug interactions and side effects. Furthermore, combination medicines – being medicines containing multiple active components – are increasingly prescribed, especially in cardiovascular pharmacotherapy. 19
This hypothesis was applied by combining two different counting methods in order to evaluate polypharmacy. The first was based on the number of different active components per patient; the second was a more classic numerical approach based on prescriptions per patient.
Using this two-method determination, the prevalence of polypharmacy in a tertiary psychiatric hospital was determined amongst full-time admitted patients of all age categories suffering from one or more mental disorders. Organ-targeted proportions of somatic medicines were examined in order to compare them with proportions in a general population.
Furthermore, psychotropic polypharmacy was determined by means of the counting method based on active components. Specific proportions of certain classes of psychotropic medicines were determined to evaluate the contribution of these different classes to polypharmacy. Subtypes of psychotropic polypharmacy were also determined. 5 It was assumed that largely psychotropic medicines largely contribute to polypharmacy within a psychiatric population.
The impact of variables such as age, gender, and length of stay (LOS) on the prevalence of polypharmacy were investigated.
A second aim of this study was to explore the reasons for polypharmacy and the proportion of rational polypharmacy. Although not exhaustive, hypotheses were set out to determine the reasons that may contribute to some part of the high prevalence in polypharmacy and psychotropic polypharmacy. Furthermore, the study investigated the proportion of rational polypharmacy in psychotropic polypharmacy. Rational polypharmacy was defined as polypharmacy based on clinically relevant data regarding the advantage of combinations.
Methods
Design
A descriptive, retrospective, cross-sectional study was performed in a tertiary psychiatric hospital, University Psychiatric Center Katholieke Universiteit (UPC KU) Leuven, in Belgium. The hospital has 440 places with 336 beds for adolescents, adults, and elderly psychiatric patients.
Population
All patients included were hospitalized full time. Patients in day-care or outpatients were excluded because prescriptions of these patients were not entered systematically into the hospital software. The population included young patients from the age of 12 years onwards to elderly patients. Patients were all diagnosed with a psychiatric disorder based on Diagnostic and Statistical Manual of Mental Disorders (DSM) IV criteria.
Data extraction
The dataset was extracted at one point in time from the hospital computerized physician order entry (CPOE). It was anonymized by the chief pharmacist. The dataset contained patient information including date of birth, gender, date of admission, and prescription information including ATC-code, start and stop date, moment of administration and current dose.
Data processing
To identify polypharmacy, the number of medicines per patient was determined. Medication was defined as “a medicine that can be found in the medicines list of the Belgian Center for Pharmacotherapeutic Information.” 20 Both acute and chronic medicines were included. Single administration prescriptions were excluded. PRN (as needed) prescribed medicines were included.
The number of medicines per patient was determined using two methods (Figure 1). The first method consisted of the addition of active components. Active components were defined as products that are active in themselves and not just synergistic or only protecting the active component from degradation. In this method, different doses of the same active component were counted as one medicine (e.g., quetiapine 25 mg + quetiapine 100 mg = one medicine), yet combination medicines were counted as more than one medicine (e.g. Deanxit® = melitracen + flupentixol = two medicines). The second method consisted of an addition of prescriptions, i.e., all the medicines with the same dosage. This more classic approach did not account for active components (e.g., quetiapine 25 mg + quetiapine 100 mg + quetiapine 100 mg = two medicines).

Determination of polypharmacy.
In the event of a discrepancy between method outcomes, priority was given to the second method. An exception was made when a discrepancy occurred in case of a combination medicine. In that case, active components of the combination drug were counted as different medicines, even though preference was given to the second counting method.
The outcome of the combined use of these two methods finally resulted in what will be further referred to as “definitive polypharmacy.”
In order to examine the contribution of somatic medicines, somatic prescriptions were determined by sorting prescriptions per ATC code. All prescriptions with ATC Codes N06A, N05A, N05AN01, N03A, and N05B/C were excluded in the determination of proportions of somatic medicines. Somatic prescriptions were sorted per ATC class.
Psychotropic polypharmacy was defined as the use of two or more psychotropic medicines. The first counting method, based on active components, was used in the determination of psychotropic polypharmacy. Afterwards, subtypes were identified (Figure 2). Same class polypharmacy is the simultaneous use of medicines of the same class, for instance two or more antipsychotics. Augmentation polypharmacy is the use of a subtherapeutic dose of one drug in combination with a drug in a therapeutic dose for the same indication. For this subtype, the indications were based on the summary of product characteristics (SmPC) of the BCFI (official compendium of medicines). Adjuvant polypharmacy is the use of a non-psychotropic drug to counter the side effects of a psychotropic drug. In this study, only constipation and Parkinsonism were withheld as side effects. The concomitant use of a laxative or an anticholinergic drug with psychotropic medicines was examined.
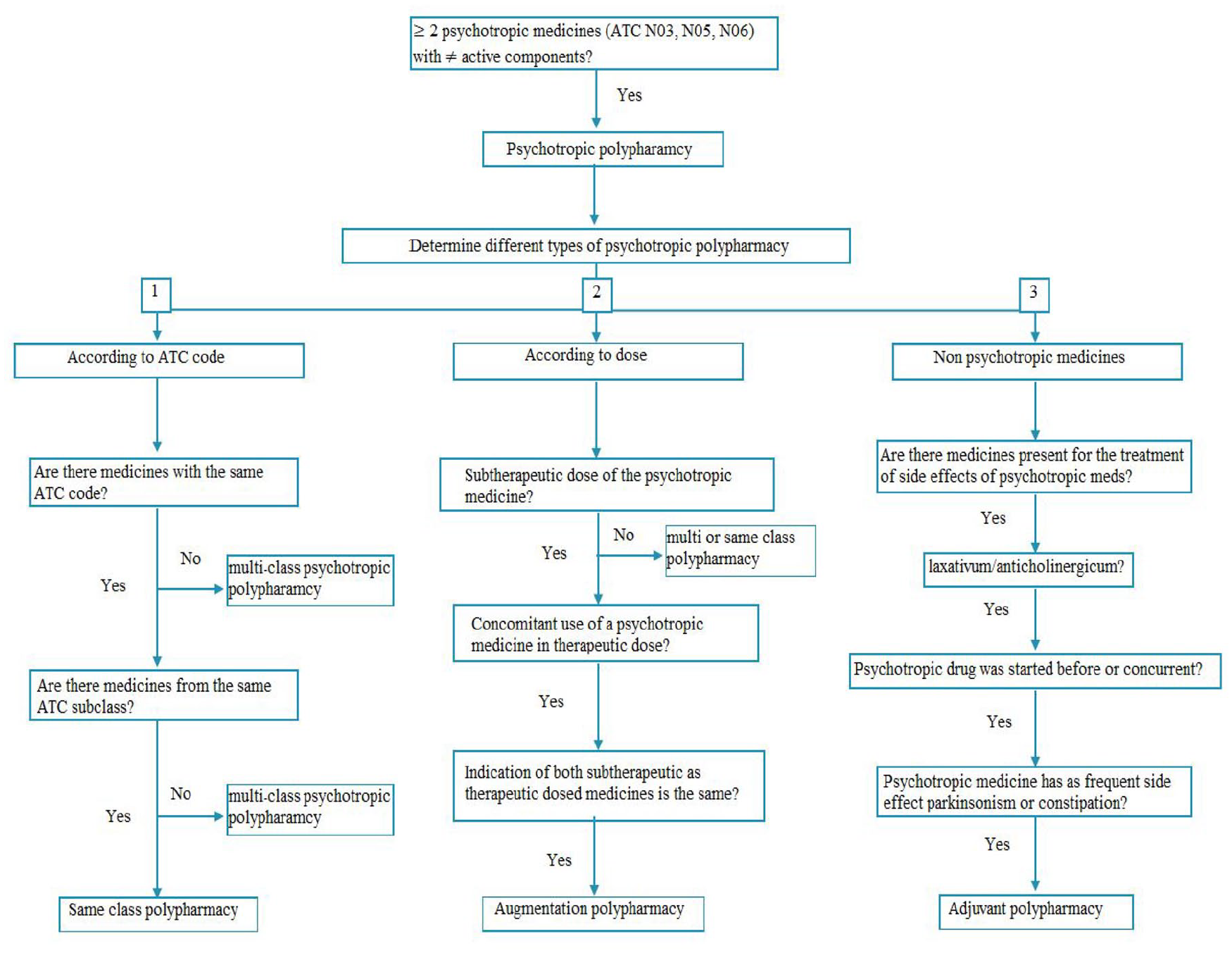
Determination of psychotropic polypharmacy.
Age and LOS were calculated for each patient to determine if these variables are potentially to related to polypharmacy.
Data analysis
Descriptive statistics were performed with the Statistical Package for the Social Sciences (SPSS, version 25).
Patient demographics were analyzed. Normal distribution of data was determined using the Shapiro–Wilk test. Normally distributed data were reported using mean and standard deviation (SD). Non-normally distributed data were reported using median and interquartile range (IQR). Three age categories were withheld: minors (0–18 years), adults (18–65 years), and elderly (+65 years).
Different proportions within polypharmacy were determined: the proportion of patients with polypharmacy according to the active component counting method in relation to the total number of patients included, the proportion of patients with polypharmacy according to the prescription counting method in relation to the total number of patients included, and, finally, the proportion of patients with definitive polypharmacy in relation to the total number of patients included.
Furthermore, proportions of somatic medicines based on ATC codes in relation to the total number of somatic medicines were determined.
Different proportions within psychotropic polypharmacy were determined. The proportion of patients with psychotropic polypharmacy was determined in relation to the total number of patients included as well as in relation to the patients with definitive polypharmacy. The proportion of patients treated with a certain class of psychotropic medicines sorted by ATC class, for example, antidepressants, versus the total number of patients with psychotropic polypharmacy was determined. Finally, the proportions of subtypes of psychotropic polypharmacy, for example, augmented polypharmacy, versus the total amount of polypharmacy were determined.
In antipsychotic polypharmacy, specific subdivisions were determined based on route of administration, per os (PO) or intramuscular (IM), and on class, typical or atypical.
In antidepressant polypharmacy, dosage of the first prescribed antidepressant was determined and compared with the maximum licensed dose (SmPC). Furthermore, augmentation combinations of antidepressants with lithium, benzodiazepines and antipsychotics were determined based on ATC class.
To evaluate the impact of variables on polypharmacy, a multinomial logistic regression analysis was conducted, the null hypothesis being that there was no influence of age, gender, and LOS on the prevalence of psychotropic and definitive polypharmacy.
Results
Sociodemographic and clinical data
A total of 292 patients were included, of whom 45.2% (n = 132) were male and 54.8% (n = 160) female. The mean age was 46.9 ± 21.4 years. The mean LOS at the time of data extraction was 180.2 ± 268.3 days (Table 1). The prevalence of main DSM IV diagnoses retrieved from the hospital computer software in the included patient sample is shown in Table 2. The most prevalent DSM IV diagnoses were schizophrenia and other psychotic disorders (n = 94), mood disorders (n = 72), and adjustment disorders (n = 34) (Table 2).
Sociodemographic data of all included patients (n = 292).

LOS, length of stay.
Prevalence of main DSM IV diagnoses in the included patients’ sample (n = 292).
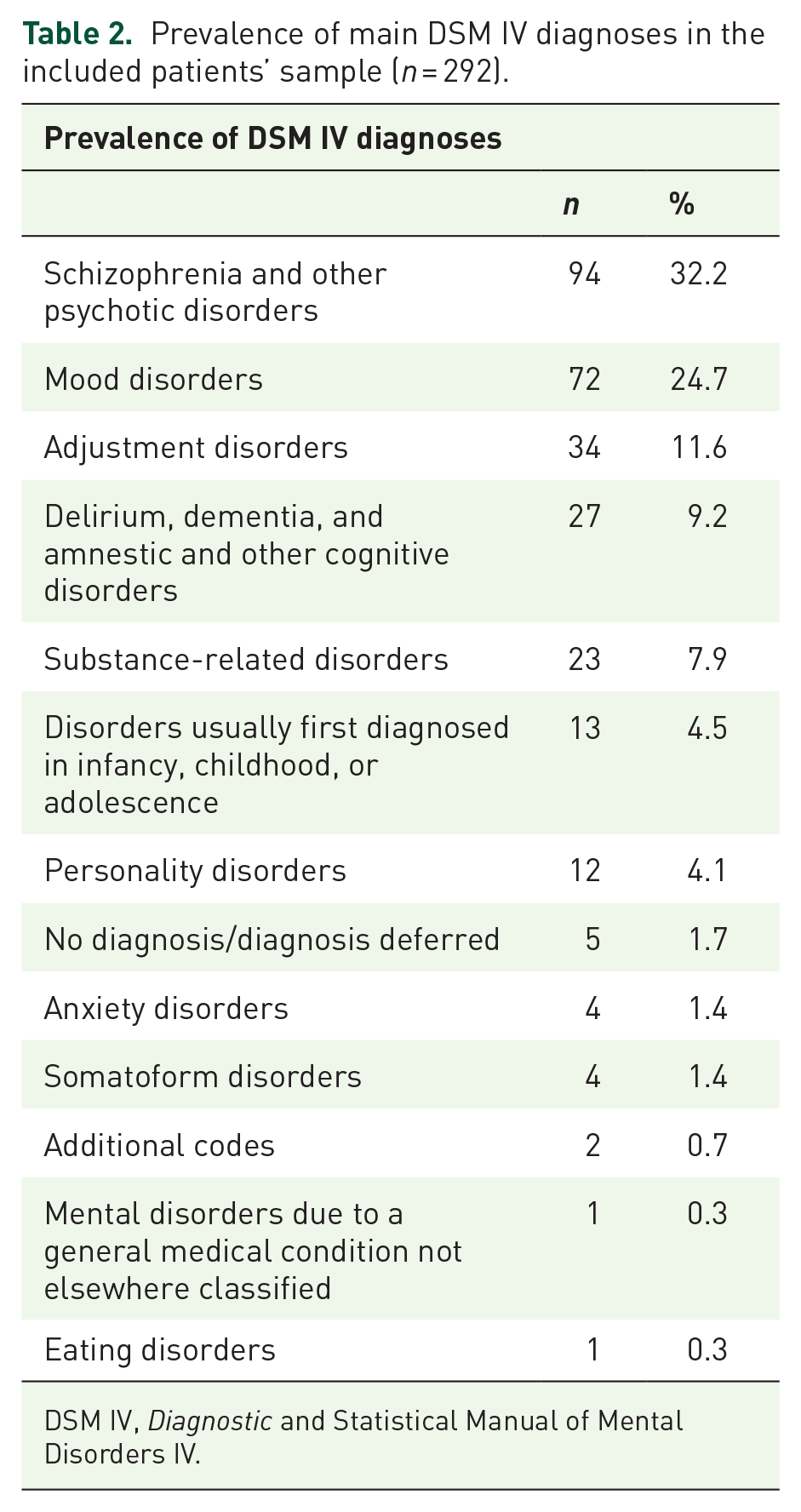
DSM IV, Diagnostic and Statistical Manual of Mental Disorders IV.
Prevalence of polypharmacy
Based on the first method, i.e., counting per active component, polypharmacy was found in 66.4% (n = 194) of the included patients. Based on the second method, i.e., counting per prescription, polypharmacy was found in 64.7% (n = 189) of the included patients. Definitive polypharmacy was found in 65.8% (n = 192) of the included patients, with a mean number of 6.8 ± 4.2 medicines per patient. The first method prevailed in 2 cases of the 292 when definitive polypharmacy was determined.
Proportions of somatic medicines
After applying the exclusion criteria, 2045 prescriptions were withheld (Figure 3). After sorting medicines per ATC class, a vast contribution of central nervous system medication was found, with a share of 55.7% (n = 1139) of all prescriptions. Within somatic medicines (n = 1060), 34.0% (n = 360) were gastro-intestinal medicines, 18.0% (n = 191) cardiovascular medicines, 7.2% (n = 76) circulatory system medicines, and 6.4% (n = 68) respiratory system medicines (Figure 4). Other ATC class medicines were far less prevalent.

Flowchart of inclusion of patients and prescriptions. Patients that were not admitted to hospital full time (daycare, polyclinic) were excluded.

The proportions of somatic medicine prescriptions of all included patients categorized per ATC code. In the ATC-N class (central nervous system medication) presented in this graph, the psychotropic N class medicines mentioned in Table 4 were excluded.
Prevalence of psychotropic polypharmacy
Psychotropic polypharmacy was found in 78.1% (n = 228) of all patients included, with a mean number of 3.0 ± 1.7 psychotropic medicines per patient. The distribution of psychotropic medicines within psychotropic polypharmacy is shown in Figure 5. The prevalence of psychotropic polypharmacy in the patients with definitive polypharmacy was 91.1% (n = 175) (see Table 3).
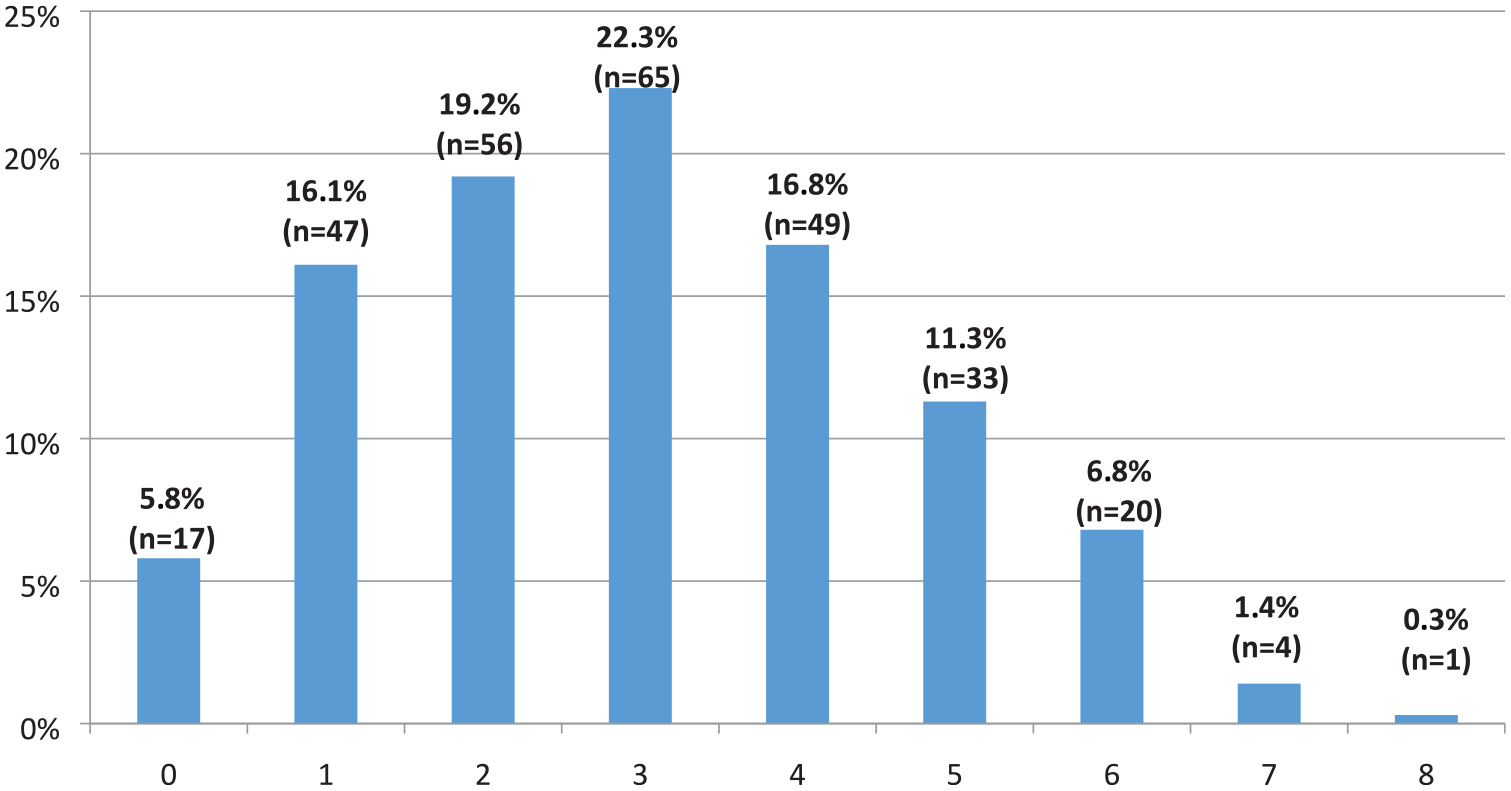
The number of psychotropic medicines prescribed (x-axis) in relation to the proportions of all patients included (N = 292) (y-axis).
Number of patients with/without definitive PP in relation to patients with/without psychotropic PP.

PP, polypharmacy.
Table 4 shows the proportions of patients on specific psychotropic medicines balanced against the total number of patients with psychotropic polypharmacy. Of the people with psychotropic polypharmacy, 66.2% (n = 151) were on at least one antidepressant (ATC: N06A), 81.6% (n = 186) received at least one antipsychotic drug (ATC: N05A minus lithium), 10.5% (n = 24) received lithium, 20.6% (n = 47) received at least one mood stabilizing antiepileptic (ATC: N03A), and 53.9% (n = 123) received at least one sedative/anxiolytic/hypnotic drug (ATC: N05B/C).
Number of psychotropic medications determined in patients ordered by class. The right column indicates the number of patients on the respective number of psychotropic medication as well as the proportion of these patients related the total number of patients with psychotropic PP (n = 228).

PP, polypharmacy.
Within the antipsychotic polypharmacy, the most prevalent combinations were the combination of multiple atypical antipsychotics PO (n = 41) and the combination of typical and atypical antipsychotics PO (n = 34). Combinations with intramuscular antipsychotics were far less prevalent (see Table 5).
Prevalence of combination of AP in AP PP, based on route of administration (PO/IM) and class (typical/atypical).

AP, antipsychotics; IM, intramuscular; PO, per os; PP, polypharmacy.
In augmentation strategies with antidepressants, lithium was associated in 4.8% (n = 11), benzodiazepines in 33.8% (n = 77), and antipsychotics in 51.8% (n = 118) of patients with psychotropic polypharmacy.
Within antidepressant polypharmacy, 68.8% of the longest prescribed antidepressants were underdosed, 24.7% were maximally dosed, and 6.5% were overdosed as compared with the licensed dose (see Table 6).
Dosing of longest prescribed antidepressants compared with the licensed dose (SmPC) within proportion of antidepressant PP (n = 77).

NDRI, norepinephrine and dopamine reuptake inhibitor; PP, polypharmacy; SmPC, summary of product characteristics; SNRI, serotonin–norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressants.
Specific proportions within psychotropic polypharmacy were 71.5% (n = 163) of same class polypharmacy, 82.5% (n = 188) of multi-class polypharmacy, 20.6% (n = 47) of augmented polypharmacy, and 35.5% (n = 81) of adjuvant polypharmacy.
Impact of age, gender, and LOS
The multinominal logistic regression analysis showed that older age was significantly associated with definitive polypharmacy [odds ratio (OR) 10.58 (95% Cl: 3.63–30.79)]. As for psychotropic polypharmacy, both adult age [OR 6.26 (95% Cl: 2.52–15.55)] and older age [OR 8.56 (95% Cl: 3.01–24.18)] were significantly associated.
Discussion
Though research on the prevalence and effects of polypharmacy in patients with psychiatric disorders is scarce, there are concerns about the high prevalence of polypharmacy and the possible negative effects on health outcomes. This study shows that 65.8% of hospitalized psychiatric inpatients have polypharmacy and 78.1% psychotropic polypharmacy.
Our first aim was to develop a clear method to determine polypharmacy. Throughout the literature, polypharmacy is defined most commonly as the concomitant use of five or more medicines. However, there is no clear consensus.18,21 This may result in difficulties in research, interpretation of literature, and, more importantly, in drawing conclusions regarding the impact of polypharmacy. In this study, a numerical approach to address polypharmacy is used, defined as the use of at least five medicines. The association between the number of medicines, i.e., a quantitative determination of polypharmacy, and the presence of inappropriate medication is strong. 21 In an effort to deliver a more comprehensive determination of polypharmacy, two different quantitative methods are suggested in this study. Apart from determining the number of prescriptions for each patient, the number of active components prescribed per patient is also determined. The major risks of polypharmacy, i.e., side effects and interactions, are attributed to the pharmacologically active components. It is hence argued that counting per active component in assessing polypharmacy yields a more relevant result in the risk assessment of drug–drug interactions and side effects, particularly since there is a trend towards combination medicines. The first method is combined with a second more classic approach of counting per prescription, because of its clinical relevance for the therapy adherence. The total number of medicines taken by patient is known to affect medication adherence. 22
The use of two different methods enabled us to compare the respective results. As expected, the prevalence of polypharmacy based on our first counting method, i.e., counting per active component, is higher as compared with the result of our second counting method, i.e., counting per prescription. It is true that the difference assessed between the two counting methods is small, with a difference in prevalence of merely 1.7% (n = 5). However, the difference resulting from counting per active component is valuable to keep in mind. The use of combination medicines involves a risk of underestimating the polypharmacy to which patients are exposed. When applied at a larger scale, this method might influence risk assessment when it comes to the determination of polypharmacy.
The prevalence of definitive polypharmacy in our population, based on the combined counting methods, is established at 65.4% (n = 192). To put this number in perspective, these numbers are compared with a large study that was conducted in the Netherlands. The study examined trends in polypharmacy, which they define as taking at least five medicines with a different ATC code, in a database of pharmacies with data of +600,000 adults over a time span of 15 years. They found the prevalence of polypharmacy in a general outpatient population to be 8.0% and 26.7% in the geriatric proportion. 23 The most common medicines contributing to this polypharmacy are medicines of the cardiovascular and the central nervous system. Our study shows, as expected, a significant higher prevalence of polypharmacy in a hospitalized psychiatric population than in a general outpatient population.
A prevalence of psychotropic polypharmacy of 78.1% (n = 228) is found in our study. This is in line with the prevalence of psychotropic polypharmacy of 76.1% found in a study of Weibel et al., 24 although their study involved only geriatric inpatients. In a general population, psychotropic medication significantly contributes to polypharmacy. 25 Our study confirms this finding, with 91.1% (n = 175) of all patients with definitive polypharmacy having psychotropic polypharmacy.
Reasons for polypharmacy
In our study, psychotropic polypharmacy has a large share in the prevalence of general polypharmacy. In psychopharmacology, reasons for polypharmacy can be multiple. Treating comorbidities, increasing efficacy of the primary treatment, achieving acute amelioration, intervening in an acute phase of an illness, and switching medication are some reasons described in literature that apply with certainty in a tertiary psychiatric hospital. 26 In our study, we were unable to systematically retrieve information about the reasons behind the high prevalence of polypharmacy. However, we addressed antipsychotic polypharmacy because of its high prevalence and little evidence. 27 In our study, 42.1% (n = 96) of patients with psychotropic polypharmacy were taking at least two antipsychotics. This is in line with the findings of Nguyen et al., who report a prevalence of antipsychotic polypharmacy in 39% of the general population. 28
The most common reasons for antipsychotic polypharmacy presented in recent literature are the combined use of antipsychotics with different routes of administration, enhancement of response, and treatment of a different symptom domain. 29 Findings of Shenoy et al. show that, in more than one-third of schizophrenic patients with antipsychotic polypharmacy, the combination of an oral antipsychotic with a depot is the reason for polypharmacy. 29 However, in our study, this applies to only 15.6% (n = 15) of patients with antipsychotic polypharmacy (Table 5). The highest proportion is the combination of oral antipsychotics, without clear evidence that this combination was temporary (without prescription stop data).
Another reason for polypharmacy could be the temporary combination of medicines, e.g., when switching medication or to counter initial side effects. This is the case in 6.7% (n = 13) of patients with definitive polypharmacy and 6.6% (n = 15) of patients with psychotropic polypharmacy. In those cases, a stop date is found in one or more scheduled prescriptions. In 16.3% (n = 37) of patients with psychotropic polypharmacy, some of the psychotropic medicines are prescribed PRN, often indicating a temporary drug combination.
Regarding somatic co-medication, particularly gastro-intestinal medicines appear to be relatively more prevalent in our study (see Figure 4) than in literature reports on the general population. 25 This may indicate a high prevalence of side effects inherent to psychotropic medicines, such as constipation with antipsychotics or nausea with antidepressants. Since certain side effects of psychotropic medicines eventually wear off, the somatic co-medication contributing to polypharmacy should be reconsidered after some time.
In the evaluation of demographic data, definitive polypharmacy is associated with old age (65+ years), whereas, for psychotropic polypharmacy both adult and old age are significantly associated. These findings further underline the considerable contribution of psychotropic medicines to the prevalence of polypharmacy.
Rational polypharmacy
Rational polypharmacy could be interpreted as polypharmacy based on clinically relevant data regarding the advantage of the combinations. 26
In general, different types of polypharmacy are assessed. Multi-class polypharmacy, augmentation polypharmacy and adjunctive polypharmacy might be considered as rational pharmacological strategies. In our study the respective prevalence was 82.5% (n = 188), 20.6% (n = 47), and 35.5% (n = 81). On the other hand, same-class polypharmacy, with a prevalence of 71.5% (n = 163), is often considered irrational. 5
In major depression disorder, for instance, the most commonly used combination strategies are said not be the strategies with the best controlled evidence in all circumstances. Lithium is a first line augmentation strategy. 30 In our study this combination has a prevalence of 4.8% (n = 11) within psychotropic polypharmacy. This is rather low when compared with the combination of antidepressants with benzodiazepines in 33.8% (n = 77) and antipsychotics in 66.2% (n = 151) of patients with psychotropic polypharmacy. Strictly speaking, these combinations may be considered as irrational polypharmacy.
Similarly, in antipsychotic treatment, antipsychotic polypharmacy with clozapine in patients with treatment resistant schizophrenia may be considered rational. 31 In our study this is found in 19.8% (n = 19) of patients with antipsychotic polypharmacy. This indicates that there is a large proportion of possibly irrational antipsychotic polypharmacy with potential adverse side effects and minimal added value.
An adequate trial of monotherapy is another important pillar of rational polypharmacy, especially when it comes to psychopharmacology. It is suggested that clinicians nowadays often tend to stick to the licensed dose of medicines. 26 Interestingly, our study shows that, in antidepressant polypharmacy, 68.8% (n = 53) of the longest prescribed antidepressants are underdosed, and only 6.5% (n = 5) are dosed above the licensed dose, which could indicate an inadequate trial of the original antidepressant therapy.
Furthermore off-label medicine use may contribute to psychotropic polypharmacy. In 55.8% (n = 43) of patients with antidepressant polypharmacy, trazodone – an antidepressant often used in treating insomnia rather than for its antidepressant properties – is associated. 32 The use of psychotropic medicines because of their side effects, most commonly their sedative properties, however, may be considered irrational. Remarkably, in 24.0% (n = 23) of patients with antipsychotic polypharmacy, prothipendyl – a low potent neuroleptic – is administered at night. Although it is structurally considered to be an antipsychotic, it is generally prescribed for its sedative side effects. Similarly, clotiapine in multiple low doses (10–20 mg) throughout the day is prescribed to 28.1% (n = 27) of patients. Clotiapine is often prescribed for its acute sedative properties in agitated patients. So, a significant part of psychotropic polypharmacy can be found in co-administration of psychotropic medicines used for their side effects.
Strengths
A first strength is that the data were collected through the hospital software CPOE. Hence, the counting methods are based on reliable data, issued by a solid registration system.
A second strength is that a considerable number of high-care psychiatric patients were included. This study offers a unique view on the pharmacological management of this population, and differentiates between definitive and psychotropic polypharmacy.
The third strength of this study is the development of a two-method determination of polypharmacy. Active components were being considered, contributing directly to the greatest risk in polypharmacy. The results of this method were set out against a more classic numerical approach of counting prescriptions of each patient to gain a more comprehensive view of polypharmacy.
Limitations
The most important limitation in the numerical approach of polypharmacy is that no statement can be made about the appropriateness of polypharmacy. When a patient suffers from multimorbidity and medication is managed well, it might be good clinical practice to prescribe more than five medicines. In this study, underlying comorbidities were not accounted for, and, consequently, only an absolute prevalence of polypharmacy was rendered.
Another limitation is that neither daily defined dose (DDD) nor any other measure of medication dose was considered. This means that, although a statement about the definitive polypharmacy of a patient at a specific moment in time can be made, conclusions about the appropriateness of the medication load are not possible.
Our study may over-report the prevalence of polypharmacy. Data were extracted at one specific point in time; hence, both chronic and acute prescriptions were included. The assessed polypharmacy may also be temporary; for instance, the combined use of antipsychotics in a switching period from one antipsychotic to another. It can be argued that this is particularly plausible in our hospital setting, where patients are admitted mainly because of a mental health crisis or relapse and a medication switch is often required. The high prevalence of psychotropic polypharmacy and the 60.0% prevalence of antipsychotic polypharmacy suggest, however, that it occurs more than is desirable. In addition, the inclusion of PRN medicines (16.4%) may add to this overestimation of psychotropic polypharmacy.
More specifically the indication for the use of anti-epileptic medication, whether for a mood disorder or epilepsy, was not retrieved. This could possibly lead to an overestimation of psychotropic polypharmacy.
The fact that this study was conducted in only one hospital is a limitation. Furthermore, one could argue that more complex pathologies may be found in a tertiary psychiatric hospital.
Conclusion
Psychiatric patients are often assumed to be at risk of polypharmacy. This may lead to a variety of challenges, including described increased mortality risk. 13 Our study indicates that both polypharmacy and psychotropic polypharmacy are highly prevalent in an inpatient psychiatric population, with a respective prevalence of 65.8% and 78.1%.
In the absence of homogeneity in the literature, a clear consensus on the definition of polypharmacy would be useful, or even necessary, for future work. In this study, a two-method numerical determination of polypharmacy was used, based respectively on active components and prescriptions per patient. A higher prevalence of polypharmacy was found using the first counting method based on active components. Although the difference with the second counting method was not significant, its relevance may increase in the light of a growing trend for combination medicines and should be considered when addressing polypharmacy.
This study underlines the high prevalence of psychotropic polypharmacy (78.1%) and the large contribution of psychotropic polypharmacy to general polypharmacy (91.1%). Although the sample includes a fully hospitalized tertiary psychiatric population (n = 292), which could indicate complex pathologies, our findings suggest that reasons for combination strategies and polypharmacy are not always rational. The low prevalence of stop dates in prescriptions, the low prevalence of evidence-based combinations, like antidepressant lithium augmentation or antipsychotic polypharmacy with clozapine within psychotropic polypharmacy, the large contribution of off-label use of psychotropic medicines in psychotropic polypharmacy, and often low dosage of the longest prescribed antidepressant in antidepressant polypharmacy, are some of the findings that suggest a high prevalence of irrational psychotropic polypharmacy. Considering the high prevalence of polypharmacy within a vulnerable population, rational strategies with adequate dosing and frequent reconsideration should be pursued.
Footnotes
Author contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Julie Boeyckens, Annelies Gilis, and Astrid Lammens. The first draft of the manuscript was written by Jeroen Govaerts, and all other authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Availability of data
Data and codes are available upon request
Conflict of interest statement
The authors declare that there is no conflict of interest.
Ethics approval
Approved by the Education-Support Committee (OBC) of the Biomedical Sciences Group KU Leuven, reference code: MP001342
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
