Abstract
Dopamine-receptor blocking agent-associated akathisia (DRBA-A) is an adverse effect that can significantly limit the use of these important medications for the treatment of a variety of psychiatric diseases, yet there is no unifying theory regarding its pathophysiology. This knowledge gap limits clinicians’ ability to effectively manage DRBA-A and mitigate negative outcomes in an already vulnerable patient population. Based on a review of the current literature on the subject, it is hypothesized that dopaminergic and noradrenergic signaling is perturbed in DRBA-A. Accordingly, it is proposed that the optimal agent to manage this extrapyramidal symptom should increase dopamine signaling in the affected areas of the brain and counteract compensatory noradrenergic signaling via antagonism of adrenergic or serotonergic receptors.
Keywords
Introduction
In their 1977 publication, psychiatrists Robert Belmaker and David Wald described their experience after receiving a single dose of 5 mg intravenous haloperidol. They reported experiencing a myriad of debilitating symptoms, including anxiety and “profound inner restlessness” that rendered them unable to work for more than 36 hours. 1 This anecdote clearly demonstrates the consequences of the common yet often underappreciated side effect of dopamine receptor blocking agents (DRBAs) known as akathisia.
Akathisia is a psychomotor syndrome involving subjective feelings of agitation and dysphoria, accompanied by repetitive and/or purposeless movement and symptoms of cognitive dysfunction such as selective attention deficit, perceptual disorder, and impaired coping responses.2–4 Akathisia may develop at different time points during treatment with DRBAs. Acute akathisia has a relatively rapid onset and short duration, and is associated with intense dysphoria. 5 Chronic akathisia persists beyond 6 months and is generally associated with less severe feelings of restlessness than during the acute phase. 5 This discussion will focus on acute and chronic akathisia as opposed to tardive akathisia, which is more closely related to tardive dyskinesia and may develop via a different mechanism. 5
Akathisia can be detected using the Barnes Akathisia-Rating Scale (BARS), 6 yet it is still frequently overlooked or misdiagnosed.5,7–9 Symptoms can develop soon after DRBA administration, and may continue despite intervention. Patients often experience the effects of DRBA-associated akathisia (DRBA-A) within days to weeks of taking the medication, and lingering symptoms can persist even after a dose reduction. 10 The effects of DRBA-A can greatly impact daily functioning and wellbeing. Akathisia severity was found to be negatively correlated with quality of life in patients with schizophrenia, 11 and those with DRBA-A demonstrated significantly lower quality of life than those who regularly took DRBAs without experiencing akathisia. 12 Other significant consequences of DRBA-A include treatment non-adherence, 13 worsening psychiatric symptoms,14–16 and suicidality.17–20 Though the rates of DRBA-A are high, 21 data on morbidity and mortality is scarce.
Discussion
Current understanding of pathophysiology
DRBAs block dopamine signaling via dopamine D2 receptors. In specific areas of the brain, this dopamine receptor antagonism confers a therapeutic effect, but in the striatum it can trigger extrapyramidal symptoms (EPS) such as akathisia. 22 D2 receptor occupancy is believed to be paramount in understanding the development of DRBA-related EPS, which also includes dystonia, parkinsonism, and tardive dyskinesia.23–26 It has been observed that when > 80% of these receptors are occupied by DRBAs, this risk of EPS substantially increases.27–29 Along these lines, genotyping results among patients with schizophrenia have revealed an increased rate of DRBA-A with the use of second generation agents in those possessing a genetic variation associated with reduced striatal dopamine D2 receptor density, 30 which would allow for this occupancy threshold to be surpassed more readily. Other patient-specific risk factors for DRBA-A include young age, male gender, and concomitant substance use, while medication-related factors such as the use of high potency DRBAs, rapid DRBA dose escalation, and DRBA polytherapy can also increase the risk of akathisia (Table 1).
Description of different types of EPS and other movement disorders that can be associated with the use of DRBA.

This table was compiled using information from resources referenced throughout the manuscript. These references are cited in the column header. Prevalence ranges are based on the lowest and highest observed prevalence reported in the literature.
BID, twice daily; DRBAs, dopamine receptor blocking agents; EPS, extrapyramidal symptoms; IM, intramuscular injection; PO, by mouth.
It is generally believed that the first generation of DRBA medications developed in this class (e.g., chlorpromazine, haloperidol, perphenazine) confer a greater risk of EPS than second-generation agents.23,26,31,50 First-generation DRBAs are high potency striatal D2 receptor antagonists, whereas second-generation agents bind more loosely and can be rapidly displaced from D 2 by intrinsic dopamine, thereby ameliorating movement symptoms. 51 Nigrostriatal dopamine D2 receptor affinity and occupancy, as well as upregulation of the D2 receptor, have been correlated with the severity of EPS. 52 In vivo neuroimaging studies support the association between EPS and dopamine D2/3 receptor binding in the substantia nigra. 53
Second-generation DRBAs also block serotonin 5-HT2A receptors, which normally inhibit dopaminergic neurotransmission, thereby resulting in dopaminergic upregulation in the nigrostriatal region. 54 The ratio of 5-HT2A:D2 antagonism is believed to be another dynamic influencing a DRBA’s tendency to produce EPS, 36 as greater 5-HT2A receptor antagonism can mitigate the effects of EPS caused by dopamine blockade in the ventral striatum.55–58 The relative “EPS advantage” of second-generation agents is touted as producing multiple clinical benefits,59,60 but movement side effects can still occur in second-generation agents despite lower D2 receptor occupancy.5,24,34,61,62
The risk of akathisia, for instance, may be comparable or even greater in second-generation agents. 52 Akathisia does not correspond well with other types of EPS in terms of symptom severity and treatments, suggesting that there are other important mechanisms underlying its development. 63 For example, DRBA-A may result from an intrinsic homeostatic response to the depletion of dopaminergic activity in the ventral striatum portion of the brain. 64 The nucleus accumbens is an area specifically implicated in the pathophysiology of akathisia due to its importance for reward and movement. It is theorized that compensatory overstimulation of this region may result in “senseless” behaviors and feelings of dysphoria observed with akathisia. 65
Incidence rates of DRBA-A vary among different agents in the second generation class 66 (Table 2). For example, quetiapine is often considered to be “akathisia-sparing”, while aripiprazole has been associated with higher risks of DRBA-A development.10,67 Despite being structurally related to aripiprazole 68 and demonstrating a similar magnitude of D2 receptor antagonism, 69 brexpiprazole is believed to have lower rates of DRBA-A.70,71 This may be due to brexpiprazole possessing greater binding affinity for the 5-HT2A receptor than aripiprazole. 69
Rates of akathisia for second-generation DRBAs.
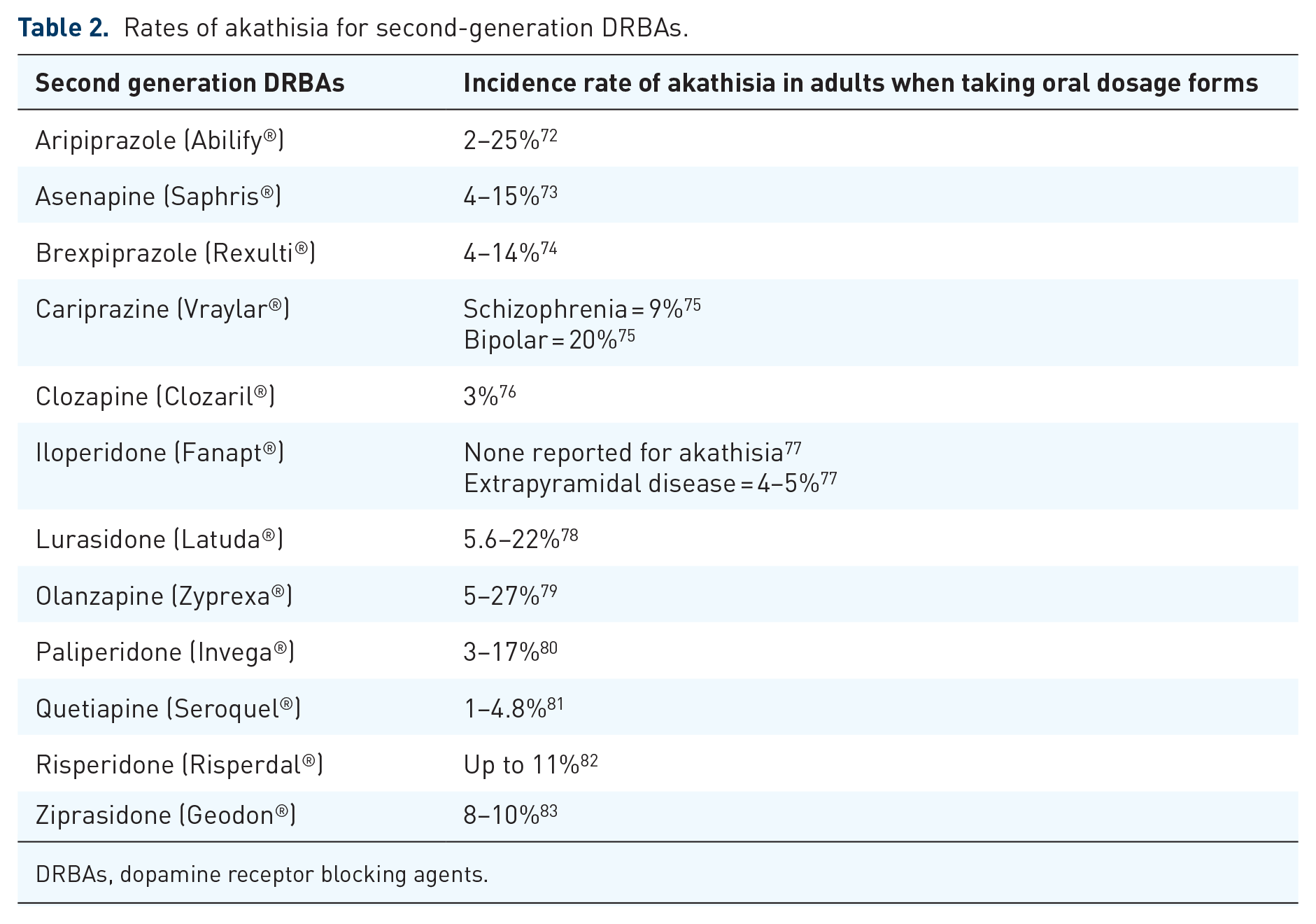
DRBAs, dopamine receptor blocking agents.
Indeed, greater 5-HT2A compared with D2 receptor antagonism appears to negatively correlate with DRBA-A risk. 84 Aripiprazole has serotonin 5-HT2A blocking effects that are less potent than D2, while quetiapine, which is associated with lower rates of DRBA-A, binds 5-HT2A to a greater extent than D2. 54 This preferential binding to 5-HT2A over D2 is most striking with clozapine (Table 3). Although DRBA-A from clozapine appears to be a rare clinical occurrence, there have been case reports of patients who both developed DRBA-A from clozapine and experienced a reduction of DRBA-A symptoms from clozapine. 85–87 Serotonin receptor activity has also been linked to risk of DRBA-A caused by first-generation agents. Patients with schizophrenia and reduced 5-HT1B receptor density were found to develop more frequent and severe akathisia when taking haloperidol compared with those with normal 5-HT1B receptor density. 88
Comparison of dopamine D2 receptor binding affinity to serotonin 5-HT2A receptor binding affinity for selected DRBAs.
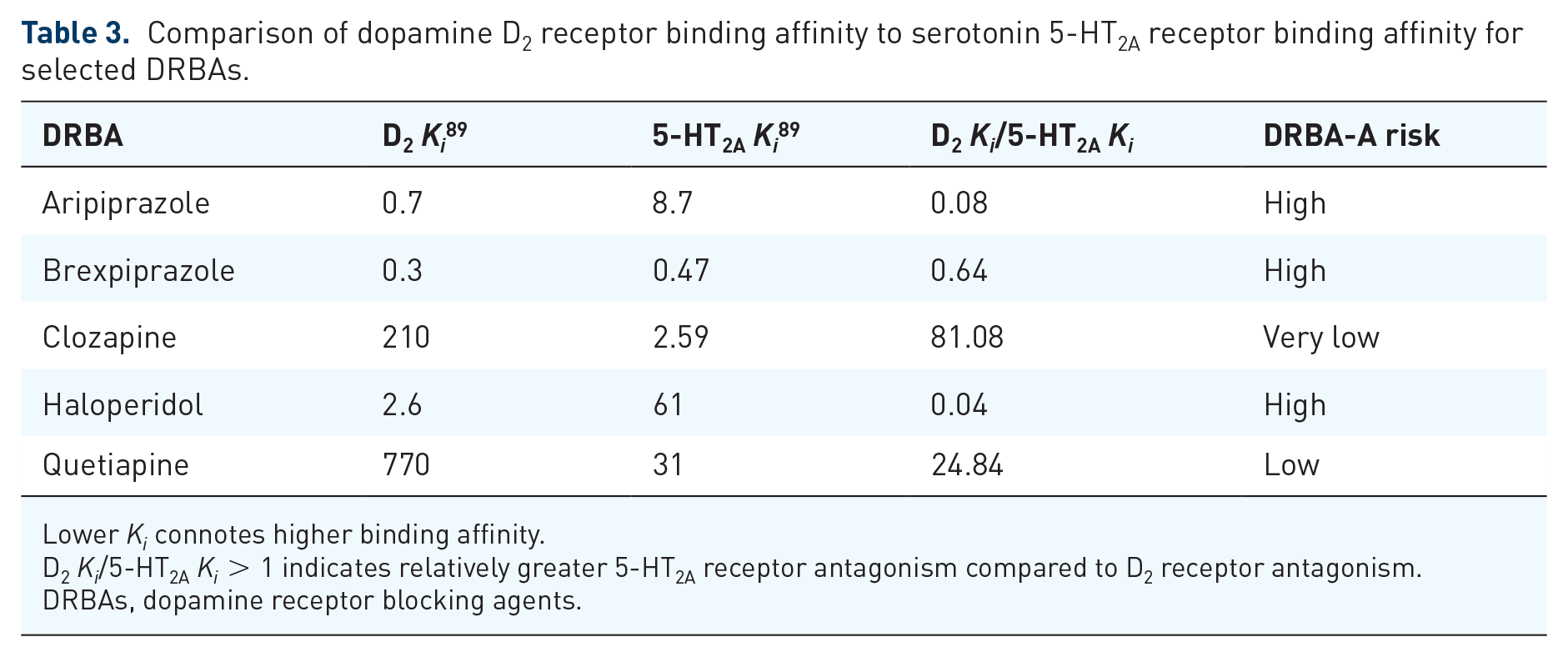
Lower Ki connotes higher binding affinity.
D2 Ki/5-HT2A Ki > 1 indicates relatively greater 5-HT2A receptor antagonism compared to D2 receptor antagonism.
DRBAs, dopamine receptor blocking agents.
Treatment mechanisms
Strategies for managing DRBA-A include psychosocial and pharmacological interventions. 5 Expert consensus guidelines recommend lowering the dosage of the DRBA, switching to an antipsychotic carrying lower akathisia risk (such as quetiapine), or initiating a short-term adjunctive medication as initial treatment options for DRBA-A.90–92 If it is safe to do so, decreasing the dose by 50% or completely discontinuing the DRBA may be required to relieve symptoms of akathisia. 31 If medications are used to treat DRBA-A, response rates for the commonly used agents propranolol and mirtazapine have been found to be 30% and 43.3%, respectively. 93
In general, support for the use of many DRBA-A treatments is based on anecdotal and empirical findings, often using data derived from studies with small sample sizes. Evidence for the use of the beta-blocking medication propranolol in treating DRBA-A dates back to the 1980s, making it one of the most well-studied treatments. Doses of 60–120 mg/day have been demonstrated to be effective and well-tolerated for reducing DRBA-A symptoms.94–97 The hypothetical mechanism of action, shared by the alpha-2 agonist clonidine, 98 is suppression of compensatory noradrenergic signaling that may trigger psychomotor activation associated with akathisia. D2 stimulation in the locus ceruleus normally inhibits norepinephrine outflow, 99 so it follows that reduced dopamine signaling caused by DRBAs subsequently increases norepinephrine signaling in the midbrain as part of a feedback response. 65 Noradrenergic antagonists such as propranolol and clonidine likely act by suppressing this excessive sympathetic response. Pramipexole, a dopamine agonist, is currently being studied to treat akathisia and other types of extrapyramidal symptoms related to DRBA therapy. 100 It acts not only to directly restore dopamine signaling in the ventral striatal region, but also to suppress noradrenergic overstimulation. These agents should be used with caution, however, as dopamine agonism carries with it the risk of exacerbating psychotic symptoms. 32
Recent literature has called into question the predominance of propranolol in the treatment of DRBA-A. 101 Comparative studies have shown cyproheptadine, zolmitriptan, and vitamin B6 produce similar levels of DRBA-A symptom reduction when compared with a range of doses (40–120 mg/day) of propranolol.102–104 Serotonin 5-HT2A receptor antagonists, such as mirtazapine,105–110 trazodone, 111 fluvoxamine, 112 zolmitriptan, 103 mianserin, 113 and cyproheptadine, 102 represent another class of treatments for DRBA-A. 114 Blocking this receptor likely plays a role in reducing symptoms by increasing downstream dopamine signaling in areas of the brain affected by DRBAs. 65 Though selective serotonin reuptake inhibitors (SSRIs) generally increase serotonergic neurotransmission and can precipitate akathisia, fluvoxamine possesses weak dopamine reuptake inhibition properties that have been shown to offset the resultant dopaminergic decrease.112,115 Mirtazapine, mianserin, and cyproheptadine also share antihistamine effects (Table 4), which may produce sedation and thereby ameliorate symptoms of akathisia.
Net neurotransmitter effects of compounds that have demonstrated efficacy in the treatment of dopamine-receptor blocking agent-associated akathisia (DRBA-A).

Sources: PubChem, Micromedex, Lundbeck Institute, Stahl’s Prescriber Guides, & StatPearls.
Anticholinergic medications are commonly used to treat other forms of extrapyramidal symptoms related to DRBA therapy, such as dystonia and parkinsonism, and may be useful when akathisia is present in combination with these types of EPS. 123 The purported mechanism of anticholinergics in the treatment of DRBA-A is restoration of dopamine signaling in areas of the brain where it is depleted by DRBAs. 65 Specifically, D2 receptors located on cholinergic interneurons in the basal ganglia, which normally inhibit acetylcholine release, 124 can activate the extrapyramidal pathway when blocked. This pathophysiologic theory is supported by the reduced rates of EPS seen with clozapine, which has greater intrinsic anti-muscarinic anticholinergic activity than other DRBAs.24,51,125 Excessive cholinergic outflow can be counteracted with the administration of agents such as benztropine, 126 diphenhydramine, 127 and trihexyphenidyl. 128 Unfortunately, the use of anticholinergics is limited by adverse effects 92 such as those related to cardiovascular, gastrointestinal, and cognitive dysfunction.
Combination therapy with propranolol and a benzodiazepine such as diazepam can also be effective in reducing DRBA-A symptoms. 129 Benzodiazepines are believed to counteract gamma-aminobutyric acid (GABA) inhibition caused by DRBAs. 101 One proposed mechanism of DRBA-A is via downregulation of GABA signaling in the pallidus through the blockade of D2 receptors. 123 5-HT2A receptor antagonism may also decrease GABA signaling in the prefrontal cortex, 130 which would offer an explanation as to why akathisia is still observed at high rates with second-generation DRBAs. In addition to correcting GABA hypofunctioning, benzodiazepines may also act via GABAergic depression of the central nervous system to reduce physical and psychological symptoms of agitation associated with DRBA-A.
Short (1–2 week) courses of clonazepam
131
and lorazepam
132
have been associated with a reduction in DRBA-A symptoms. Intravenous diazepam can rapidly relieve DRBA-A symptoms,
133
making it an acceptable option in the acute setting. However, the adverse effect profile, risk of overdose, and abuse potential of benzodiazepines make them far less attractive options for routine chronic and/or preventative treatment of DRBA-A.
134
Gabapentin enacarbil is another pharmacotherapy option that acts by potentiating GABA signaling and is associated with relatively fewer risks compared with benzodiazepines. Treatment with gabapentin enacarbil has been shown to significantly decrease the severity of DRBA-A within 2 weeks at doses similar to those recommended for restless leg syndrome.
135
Amantadine is an antagonist of the N-methyl-
Other treatments for DRBA-A have various mechanisms of action. Preladenant works by antagonism the adenosine receptor and has been studied to treat DRBA-A. 136 Reduced iron levels were found to correlate with DRBA-A development in patients with schizophrenia, 137 and the administration of IV iron produces a reduction in DRBA-A symptoms. 138 Heavy smoking has been associated with fewer instances of DRBA-A among patients with schizophrenia when compared with light smokers. 139 Accordingly, the administration of nicotine patches has been found to significantly reduce DRBA-A symptoms in non-smoking inpatients. 140 Vitamin B6 significantly reduces subjective symptoms of restlessness, distress, and global symptoms of DRBA-A compared with placebo. 141 Vitamin B6 is important for the synthesis of dopamine, serotonin, and GABA. 101 This treatment may also act outside of the neurotransmitter system to reduce symptoms via free radical scavenging. 142 Another antioxidant that may be useful for the treatment of DRBA-A is N-acetyl cysteine, which significantly decreases akathisia when used adjunctively in patients with schizophrenia taking DRBAs. 143
Conclusion
DRBA-A is a prevalent and potentially serious adverse effect of medications that are used widely in the field of psychiatry. It is crucial that DRBA-A is screened for in all patients prescribed these agents throughout all time points in their treatment. An accurate description of possible symptoms, including both common and uncommon presentations, must be provided to patients at the initiation of therapy so that they are aware of what to possibly expect. Any new motor or psychiatric symptoms resembling anxiety should include DRBA-A as a differential diagnosis.
The prevailing theory holds that dopamine hypofunctioning in the ventral striatal region of the brain is implicated in the pathophysiology of EPS such as akathisia, though this is likely not the only significant mechanism as rates of DRBA-A remain high in second generation DRBAs despite their comparatively lower dopamine D2 blocking potency than first generation agents. Figure 1 attempts to visualize neural circuits implicated in DRBA-A and illustrate their interactions in the pathophysiology of this drug-induced phenomenon.

Proposed neural circuitry underlying second generation DRBA-A.
While dopamine neurotransmission via the nigrostriatal pathway is believed to be important for other types of EPS, akathisia may more heavily involve alterations in the mesolimbic pathway. A deficiency in dopamine signaling via the former results in movement dysfunction, while effects on the latter can be linked to goal-directed behavior. Interruption in dopamine outflow through the ventral striatum may underlie agitation and obsessive-compulsive types of thoughts and behaviors unique to akathisia, such as purposeless and/or repetitive movements.
Noradrenergic signaling is likely also important in the pathogenesis of DRBA-A, and is already central to the treatment paradigm. It has been established that dopamine blockade by DRBAs can result in norepinephrine overactivity, 144 and thus it is reasonable to correlate this sympathetic response with the physical and psychological manifestation of akathisia. Consequently, it is key that any treatment for DRBA-A involve suppression of this system, by way of either alpha-1 receptor antagonism, alpha-2 agonism, or beta antagonism.
The development of new treatments for DRBA-A is relevant as traditional treatments are not always effective. 92 Based on the mechanisms of currently available therapies for DRBA-A, an effective agent should increase dopamine signaling in the affected areas of the brain (either via anticholinergic, serotonin receptor antagonism, or direct dopamine receptor stimulatory activity) and counteract compensatory noradrenergic signaling via one of the pathways mentioned previously. When balancing the two effects, a higher priority should be placed on noradrenergic inhibition than dopamine stimulation in order to avoid exacerbating psychotic symptoms. Therapies with significant GABAergic, anticholinergic, and/or antihistamine effects may have less impact on psychiatric symptoms (both beneficial and detrimental) than those with dopaminergic effects, but should generally be avoided due side effect burden. Ultimately, the management of DRBA-A should be a collaborative effort between patient and provider, and the benefit versus risk should be carefully weighed for any medications used.

