Abstract
Serotonin syndrome is thought to arise from serotonin excess. In many cases, symptoms are mild and self-limiting. But serotonin syndrome can become life threatening, when neuromuscular hyperexcitability spins out of control. Uncontainable neuromuscular hyperexcitability may lead to cardiovascular complications, linked to extreme changes in blood pressure. Currently, there is little guidance on how to control blood pressure in hyperserotonergic states. We report a case with treatment-resistant arterial hypertension, followed by a clinical review (using systematic review principles and techniques) of the available evidence from case reports published between 2004 and 2016 to identify measures to control arterial hypertension associated with serotonin syndrome. We conclude that classic antihypertensives may not be effective for the treatment of severe hypertension associated with serotonin syndrome. Benzodiazepines may lower blood pressure. Patients with severe hypertension not responding to benzodiazepines may benefit from cyproheptadine, propofol or both. In severe cases, higher cyproheptadine doses than currently recommended may be necessary.
Keywords
Introduction
Awareness of serotonin syndrome (SS) as a severe form of serotonin toxicity has increased. Yet, it remains a diagnostic challenge in everyday practice. SS is thought to arise from 5HT1A and 5HT2 receptor stimulation, resulting in neuromuscular hyperexcitability. SS has been linked to a variety of drugs with direct or indirect serotonergic actions (Table 1).1–4 The risk of SS increases with the use of serotonergic agents in escalating doses, or combination of two or more serotonergic agents. The risk of SS also increases with the concomitant use of other agents reducing the metabolism of serotonergic agents, or hepatic or renal impairment.4,5 In many cases, symptoms are mild and self-limiting. But if not recognized in good time, neuromuscular hyperexcitability can spin out of control. Then, SS can become life threatening. Uncontainable neuromuscular hyperexcitability may ensue in cardiovascular complications. Such are linked to extreme changes in blood pressure (BP). Currently, there is little guidance on how to control BP in hyperserotonergic states. We report a case of SS with treatment-resistant hypertension as the lead symptom. We then present a clinical review of the literature to identify measures to control arterial hypertension associated with SS.
Agents associated with an increased risk of serotonin syndrome.

Also promotes serotonin release.
5HT, 5-hydroxytryptamine; LSD, lysergic acid diethylamide; MAOI, monoaminoxidase inhibitors; MDMA, 3,4-methylenedioxymethamphetamine; SNRI, serotonin–noradrenaline-reuptake inhibitor; SSRI, selective serotonin-reuptake inhibitor; TCA, tricyclic antidepressant.
Case report
A 73 year-old White woman was admitted to the emergency department with a 4-week history of cough and nausea and a 1-week history of dyspnoea. At 8 days prior to admission, she had been started on erythromycin because of pulmonary crackles suggestive of a chest infection. Feeling worse, she stopped erythromycin after 6 days.
She had a prior history of arterial hypertension and mild coronary heart disease treated with furosemide and acetylsalicylic acid. She was treated with a combination of 15 mg escitalopram and 225 mg venlafaxine for a chronic depression, which had not responded to monotherapy alone. Escitalopram had been increased to its current dose 3 months ago.
At admission, she presented fully orientated with pallor, tachypnoea (24 bpm) and bilateral crackles. There was no oedema. The initial BP was 130/60 mmHg, rising to 170/66 mmHg after 40 min. The temperature was 36.1°C and oxygen saturation 100% with 3 l O2 via nasal cannula. The patient’s previous electrocardiography (ECG) had been normal. Now, the ECG showed sinus rhythm (100 bpm) with episodes of fast atrial fibrillation at 160/min and short bursts of ventricular tachycardia, both self-limiting. The ECG showed deep anterolateral and inferior ST-depressions. The patient was immediately transferred for catheterization, which showed an old occlusion of the right cardiac artery. At 14 months prior to the current episode, creatinine was normal with 70 µmol/l. At 5 months prior to the current episode, the venlafaxine concentration was 698 nmol/l, well within the therapeutic range of 90–900 nmol/l. Now, the laboratory results showed a severe hyperkalaemia in the context of renal failure (for complete laboratory results, see Table A1).
The patient received calcium, sodium bicarbonate and glucose–insulin to treat the hyperkalaemia. She was transferred to the intensive care unit. At 10 min after the initiation of haemodialysis without ultrafiltration, the BP fell to 60/35 mmHg. The patient stabilized quickly but stayed anuric thereafter. The abdominal computed tomography (CT) was normal.
Further into the dialysis, the patient became increasingly agitated. Her BP rose to 240/110 mmHg. She developed atrial fibrillation, which responded to 4 mg intravenous (i.v.) metoprolol. However, the BP remained high. The patient then decreased in consciousness and scored 7/15 on the Glasgow Coma Scale (GCS). She developed muscular rigidity with hyperreflexia, inducible clonus and upgoing plantars. She also had mydriasis despite morphine administration and slow, pendular, horizontal (roving) eye movements. A cranial CT was normal. As her hypertension did not improve and the neurological abnormalities persisted without any apparent cause, we reconsidered our differential diagnosis. Re-review of the patient’s drug chart alerted us to the fact that she was treated with two serotonergic antidepressants and had recently been exposed to erythromycin, which might have interfered with the metabolism of her antidepressants. Neither prior to admission nor during her hospital stay had the patient received any other serotonergic agents, such as serotonergic opioids. Apart from erythromycin, the patient had not either received any other agent that could have interacted with her antidepressants pharmacologically.
As the symptoms fulfilled all three diagnostic criteria systems, SS was diagnosed. At this point, the patient had a constant BP around 220/85 not responding to any conventional treatment (Table 2) or benzodiazepines. Cyproheptadine was started. After administration of 12 mg, the BP began to decrease and finally fell to 150/65 after 4 h. At the same time, the patient improved neurologically and achieved 14/15 points on the GCS. The BP started to increase again 11 h later despite maintenance with 2 mg cyproheptadine every 2 h. However, the BP could now be contained with amlodipine and metoprolol. After 24 h, cyproheptadine was stopped.
Antihypertensive treatment in our case.

BP measurements in mmHg.
BP, blood pressure; i.v., intravenously; p.s., via nasogastric tube; p.o., orally.
Around 12 h later, after discontinuation of cyproheptadine, high BP and neurological symptoms recurred despite continued treatment with midazolam, amlodipine and metoprolol. Neither clonidine, labetalol, minoxidil, nor reinstitution of cyproheptadine in maintenance dose had effect on BP. Finally, propofol was initiated, leading to a rapid improvement of both BP and neuromuscular symptoms. After 10 h, propofol was discontinued. The patient was then able to swallow. BP was successfully contained with amlodipine, doxazosin, bisoprolol and diazepam. She also received ECG monitoring for a suspected type 2 myocardial infarction for another 48 h. However, 4 days after her initial presentation to the emergency department, she developed ventricular fibrillation unresponsive to resuscitation and asystole. The post-mortem examination revealed a myocardial infarction and a myeloma kidney. No post-mortem CYP genotyping was performed.
The Ethical Review Board in Umeå declared that according to the Swedish Ethical Review Act, this study of a deceased person was not in need of an ethical review board approval. The Ethical Review Board declared further that it did not see any research ethical problems with the case report. The husband of the deceased person had given his verbal informed consent to publication, which was documented in the deceased patient’s case notes at the time.
Literature review
We conducted a clinical review, using systematic review principles and techniques, of interventional and observational studies on the effect and outcome of BP-lowering treatment in SS.
Search strategy
We searched MEDLINE, ISI Web of Science: Science Citation Index Expanded, Cochrane Central Register of Controlled Trials CENTRAL, the Cochrane Library, CINAHL, TOXNET Toxline search and ClinicalTrials.gov. We used the following search terms: ‘serotonin syndrome’ OR ‘serotonin toxicity’ and ‘hypertension’ OR ‘hypertensive’. As this review did not identify any relevant studies, we proceeded to searching cases reporting on treatment of hypertension in SS. For this, we screened two databases that contained case reports, MEDLINE(R) and ISI Web of Science. We searched for articles published between 1 January 2004 and 31 December 2016 for articles containing the terms ‘serotonin syndrome’ or ‘serotonin toxicity’. We assessed all case presentations concerning patients over 18 years fulfilling the criteria for SS according to at least one of the three available classification systems (Sternbach, Radomski et al. or Hunter Serotonin Toxicity Criteria).6–8 We chose the year 2004 as a cut-off point, because by that time, all three classification systems were available (Table A2 and A3).
Inclusion criteria
We considered all cases meeting the definition of at least one of the three available diagnostic systems. In all cases, after differential diagnostic consideration, SS emerged as the most likely diagnosis. Two investigators independently double rated all cases regarding the three diagnostic systems. This method has been reported in detail elsewhere. 4 We screened these case reports for information regarding BP and antihypertensive treatment. We included all cases that reported severe hypertension, defined as a systolic BP of 175 mmHg or more, measured in an acute setting.
Exclusion criteria
We excluded all cases (a) not meeting any of the diagnostic criteria despite claiming a diagnosis of SS; (b) being aetiologically uncertain despite meeting the diagnostic criteria; (c) containing insufficient clinical information to rate; (d) being historical; or (e) implicating use of first-generation antipsychotics or concomitant neuroleptic malignant syndrome.
Data extraction
We abstracted all eligible cases into a new dataset, including general patient characteristics, onset, clinical course, mode of presentation, symptoms, diagnostic criteria, associated medications, treatment and outcome. All cases were summarised according to two criteria: (a) type of hypertensive treatment; and (b) type of response. Type of response was further stratified into (a) therapeutic response within less than a day (rapid); or (b) therapeutic response after 24 h or no response at all (slow or no).
Results
The systematic search of the literature yielded 403 articles with 493 potentially eligible cases. Of these, 119 were excluded so that 374 cases remained (Appendix 4). Of these, 49 cases reported severe arterial hypertension of at least 175 mmHg systolic and 22 commented on the antihypertensive treatment and outcome (Table 3).9–29
Treatment of high blood pressure (>175 mmHg) in serotonin syndrome as reported in the literature 2004–2016.

BP measurements in mmHg.
BP, blood pressure; i.v., intravenously; MAP, mean arterial pressure; n.s., not specified; p.o., orally; p.t., via gastric tube.
Sixteen patients received benzodiazepines. As monotherapy, this led to a relevant reduction of BP in three cases (cases 1, 5, 6). Combination therapy decreased BP in a further three cases (cases 2, 7, 9). ‘Classical’ antihypertensive agents included calcium antagonists, nitrates and beta blockers. Of these, only diltiazem, used in monotherapy, took effect. In the respective case (case 8), however, diltiazem, led to severe hypotension. Hydromorphone was effective in two cases (cases 10, 12). In case 10, hydromorphone was used in combination with fentanyl. This combination led to apnoea. Propofol was used in four cases. It was clearly effective in two cases (case 4, 13), and in another case (case 9), when used in combination with lorazepam. Cyproheptadine was used in seven cases. It proved effective as a single agent in two cases (case 11, 15) and as combination treatment in two further cases (case 7, 14) (Table 4).
Effect of antihypertensive agents, number of cases. #

Including our case.
Followed by severe arterial hypotension.
In combination with cyproheptadine.
Apnoea (combination hydromorphone with fentanyl).
Discussion
Severe SS is often the result of a chain of events leading to a catastrophic outcome. It is well known that SS can precipitate autonomic instability and BP changes. But clinicians may not be aware that SS may be associated with severe hypertension that resists treatment with conventional antihypertensive therapies.
Reconstructing the likely catastrophic chain of events: our case
In our case, we established ventricular fibrillation with cardiac arrest as the immediate cause of death, secondary to a type 2 myocardial infarction. This myocardial infarction was most likely an aftermath of an 8 h episode of severe hypertension in the context of a SS.
Until the event, this patient had successfully and uneventfully been maintained on the combination of two antidepressants over a period of 9 months.
Potential role of hepatic CYP inhibition
In our case, the SS may have been precipitated through addition of erythromycin, which is a CYP3A4 inhibitor.
Venlafaxine is metabolized by several hepatic microsomal enzymes. CYP2D6 metabolizes venlafaxine to O-desmethylvenlafaxine, an active metabolite. CYP2C19 and CYP3A4 facilitate N-demethylation (a) of venlafaxine to the inactive metabolite N-desmethylvenlafaxine and (b) of O-desmethylvenlafaxine to the clinically insignificant metabolite N,O-desmethylvenlafaxine.32,33 When adding erythromycin as a CYP3A4 inhibitor, the levels of the various agents will change. The net result will depend on patient’s CYP2D6/2C19 genotypes. However, most likely, inhibition of CYP3A4 will lead to higher levels of venlafaxine and O-desmethylvenlafaxine.
Escitalopram is metabolized to the active metabolite S-desmethylcitalopram by CYP2C19 and to a lesser degree by CYP2D6 and CYP3A4. S-desmethylcitalopram is further metabolized by CYP2D6 to S-didesmethylcitalopram, which is also pharmacologically active.33,34 Erythromycin can also increase levels of escitalopram. This will depend on CYP2C19 genotype. If CYP2C19 activity is reduced, CYP3A4 becomes important.
We have identified three further case reports of SS in the context of macrolide antibiotics in combination with selective serotonin-reuptake inhibitors (SSRIs). The first case concerned a combination of paroxetine and clarithromycin in a 36-year-old woman, 35 the second sertraline and erythromycin in a 12-year-old boy, 36 and the third case fluoxetine and clarithromycin in a 53-year-old man. 37
The individual risk of developing SS will partly depend on gene polymorphisms that increase serotonin sensitivity. In our case, the patient’s CYP2C19 and CYP2D6 genotypes would have been of great interest, but no post-mortem genotype testing was performed. Gene polymorphisms can vary considerably, for instance, according to ethnicity. They may affect pharmacokinetic factors, such as the CYP system. They may also affect pharmacodynamic mechanisms, such as serotonin signal transduction via the 5HT2 receptor. 38
We also considered whether the underlying infection could have changed the permeability of our patient’s blood–brain barrier. However, this is unlikely because the patient had a chest and not a central nervous system infection. We also considered the likelihood of arrhythmia caused by erythromycin. However, erythromycin has a short half-life and the patient had discontinued erythromycin 2 days prior to admission.
Potential role of renal impairment
In our case, renal failure has most likely also contributed to the development of SS; 92% of venlafaxine and its metabolites are renally eliminated. 32 Around 8–10% of escitalopram is excreted renally unchanged. 34 We do not know when the patient had developed multiple myeloma kidneys. As the decline of renal function was not detected, escitalopram and venlafaxine were not adjusted and may have increased in plasma concentration.
Hypertension as a consequence of serotonin toxicity
In therapeutic doses, venlafaxine, a serotonin and noradrenaline reuptake inhibitor (SNRI), raises BP very modestly. 39 Hypertensive crisis in overdose has been described. 40 In that case, which did not have features of SS, BP responded to labetalol, a beta blocker with antinoradrenergic properties. In our case, treatment with labetalol did not show any effect, whereas cyproheptadine led to dramatic improvement. This suggests that the arterial hypertension was a consequence of serotonin toxicity rather than a hyperadrenergic state.
Understanding hypertension in the context of serotonin toxicity
Neuromuscular symptoms and hypertension may not lie on a continuum, and only a minority of patients with SS develops hypertension. 7 It is unclear why serotonin toxicity can precipitate a hypertensive crisis. Serotonin plays a crucial role in cardiovascular regulation. Serotonin exerts effects on BP by a multitude of mechanisms. In the peripheral circulatory system, serotonin binds to 5-HT receptors in the blood vessels. Here, it causes predominantly direct arterial constriction. Under certain conditions, serotonin can even have vasodilatory effects. In the heart, serotonin has been shown to increase inotropy. It is also arrhythmogenic. Serotonin influences the sympathetic nervous system, both peripherally and centrally. Dependent on which receptor is activated, serotonin can either lower or raise BP and effects vary over time.41–43 In patients with renal insufficiency, free plasma serotonin accumulates and is not eliminated by dialysis. 44 Since serotonin does not cross the blood–brain barrier, central neurologic signs of SS cannot be explained by impaired renal excretion. However, the proportion of central and peripheral effects of serotonin on BP in our patient will remain unresolved.
One other setting that links serotonin to hypertension is pulmonary hypertension of the newborn (PPHN) associated with SSRI use in the second half of pregnancy. The mechanism behind this adverse SSRI effect remains unclear. Serotonin is a potent pulmonary vasoconstrictor, and the serotonin transporter is involved in the pulmonary artery smooth muscle proliferation.45,46 One study has shown that SSRI caused ductus arteriosus constriction and made vessels less sensitive to prostaglandin-induced vasodilatation. 47 Prostaglandin E2 agonists are potent vasodilators. Yet, to our knowledge, they have never been used in the treatment SS-associated hypertension.
Treating hypertension in the context of SS: lessons from our case report analysis
SS can occur in a variety of clinical settings. As each case is different, tailoring the treatment to the underlying pathophysiology is not easy. Hence, it comes as no surprise that experts do not agree. Boyer and Shannon have recommended nitroprusside and esmolol.1,5 Others have suggested propranolol or cyproheptadine. 7 All agents used in the treatment of SS are used off label.
Beta blocker and combined beta and alpha blocker (labetalol) had no effect on the BP in our case. Another case in our review also reported a lack of effect (case 18). One patient became even hyperdynamic after esmolol (case 12). In our patient, conventional antihypertensive agents only yielded a response effect after administration of the serotonin antagonist cyproheptadine and (later in the course) propofol. Only then, metoprolol, co-administered with amlodipine, and bisoprolol, co-administered with amlodipine and doxazosin, took effect.
Calcium antagonists (calcium-channel blockers, CCBs) interact with serotonin-induced vascular contraction via pharmacologic overlap between L-type calcium- and 5-HT receptors. 48 In one case, the nondihydropyridine-CCB diltiazem had fast effect on BP (case 8). This led to prolonged hypotension, necessitating norepinephrine. Treatment with nicardipine, a dihydropyridine-CCB, did not lower BP substantially in a second case (case 19). A third case (case 14) with autonomic instability was difficult to rate. In that case, the patient was hypotensive both before and after the treatment with nicardipine, given in combination with metoprolol. Nondihydropyridine-CCBs may be superior to dihydropyridine-CCBs in hyperserotonergic states. Possibly, nondihydropyridine-CCBs inhibit serotonin-induced vasoconstriction. 49 This may be a 5-HT2-receptor-mediated effect. 50 At higher serotonin doses, though, oral diltiazem lacked effect on radial arteries. 51
The use of central antihypertensive drugs in SS has not been previously described. The central alpha2-agonist clonidine affects serotoninergic neurons and decreases serotonin levels in the medulla oblongata. 52 But in our case, clonidine lacked effect on BP.
Nitroglycerine provides nitric oxide to induce vasodilatation via generation of cyclic guanosine monophosphate (GMP). This activates calcium-sensitive potassium channels in the cell membrane. Nitroglycerin proved effective in a case with hypertension (case 2), but ineffective in a second (case 21). A third case is difficult to rate since nitroglycerine was given very late in the clinical course at a lower BP (case 16). In our patient, we did not see any effect on BP despite escalating intravenous doses. Data on nitroglycerine remains limited, and no data are available on nitroprusside. As sublingual nitrate is widely available, it may be worth a try.
Benzodiazepines have direct vascular vasodilatory effects on both arteries and veins. It has been hypothesized that the reduction of peripheral vascular resistance is a consequence of decreased catecholamine levels. Binding to central gamma-amino butyric acid receptor would lead then to decreased adrenergic amine production. In addition, benzodiazepines can peripherally depress the baroreceptor reflex.53–56
Cyproheptadine is an antihistamine with anticholinergic and antiserotonergic properties. It has antagonistic effects on several serotonin receptors including 5-HT1A,D, 2A,B,C, 3, 6 and 7. 57 Cyproheptadine has been used to treat hyperserotonergic states in carcinoid syndrome. 58 Today, cyproheptadine is the antidote of choice for moderate SS. 59 It is only available in oral form and quickly resorbed. 60 Positron emission tomography (PET) scan studies in two healthy men show a blockade of 85% and 95% of 5-HT2 receptors in the prefrontal cortex, respectively, with 12 mg and 18 mg daily over 6 days. 61 In SS, an initial dose of 12 mg is recommended, followed by 2 mg every 2 h to a maximum of 32 mg in 24 h until symptoms settle. 62 This dose was insufficient in our case. Our patient showed a good initial response but relapsed after 8–11 h under maintenance with 2 mg every 2 h. While the effect faded, we were able to control the BP with CCB and beta blocker. Once cyproheptadine was stopped, this regimen did not work any longer.
In our review, we identified seven cases with hypertension treated with cyproheptadine (cases 7, 11, 14, 15, 17, 20, 22). Two cases describe a quick effect within hours (cases 7 and 11). Case 14 observed reversal of the autonomic instability. Case 17 did not see any improvement, using 8 mg cyproheptadine initially followed by three 4 mg doses. Equally, case 22 must be regarded as a treatment failure. Here, normalization of the high BP took 96 h. But this patient was continuously treated with fentanyl at the same time. Fentanyl may cause SS in its own right and may have maintained the hypertension that way.
Cyproheptadine has been used in higher doses in maintenance therapy outside the SS arena: in carcinoid syndrome, up to 48 mg daily has been used. 63 In adults, 0.5 mg/kg is considered the maximal daily dose for treatment of hypercortisolism or allergy. 60 Toxicity for single doses is believed to lie beyond 250 mg 64 or 3 mg/kg. 60 Thus, there is scope to give higher doses than 32 mg daily if the clinical course develops unsatisfactorily.
Propofol has been established as a possible treatment in SS, mostly due to its effect on the neuromuscular symptoms. Propofol inhibits 5HT3-receptors at supratherapeutic concentrations. 65 Direct effects of propofol on 5-HT1 or 5-HT2 receptors have not been studied. Propofol antagonizes serotonin-induced arterial contraction, 66 either as a direct effect on 5-HT receptors or via inhibitory effects on platelet aggregation. 67 Probably, propofol reduces systemic vascular resistance. 68 The antihypertensive effect of propofol is well known to anaesthetists.
In three of the four available cases, propofol was effective (cases 5, 9, 13). The case not responding to therapy was the one on continuous fentanyl (case 22). In our patient, we saw an immediate drop of BP, leading to normal BP after 5 h. If the antihypertensive effect of propofol was mediated through 5-HT receptors, propofol could retain its antihypertensive properties in hyperserotonergic states. This would make propofol the antihypertensive drug of choice in SS for patients in need of intensive care.
Preventing severe serotonin syndrome
Choosing and dosing serotonergic drugs
Ultimately, the risk of SS should be minimized whenever possible. Preventive measures include combining multiple serotonergic agents only if absolutely necessary and avoiding dose escalation. It is also important to remain alert of pharmacokinetic factors that could increase plasma concentrations, such drug–drug or gene–drug interactions.
In our case, an SNRI (venlafaxine) and an SSRI (escitalopram) were used in combination for a chronic depression, which had not responded to monotherapy alone. Here, the question arises, whether this combination of venlafaxine and escitalopram is an appropriate augmentation strategy. The medical records suggest that the patient was difficult to treat and had not responded to a combination of venlafaxine and mianserin. Presumably, because mianserin is structurally similar, mirtazapine augmentation was not attempted. It remains unclear why the combination of SSRI and SNRI was preferred over other augmentation strategies apart from mirtazapine. Pharmacologically, the combination of venlafaxine and escitalopram offers little advantage. Venlafaxine, like escitalopram, has predominantly serotonergic properties. 69 Thus, the therapeutic gain may be small. Yet, pharmacology does not always predict treatment response. 70 Gonul and colleagues reported four cases, in whom an SSRI brought additional effect to venlafaxine. 71 This combination carries a risk of SS. In a meta-analysis of 299 cases of SS published since 2004, 4 we found nine cases, in which venlafaxine and SSRIs had been combined or where a swap had occurred in close temporal relation. In seven of these cases, however, there were also other serotonergic agents present. Whereas, clinicians need to remain flexible in their approach to treatment-refractory combinations, 70 they need to inform patients of the risk of SS when combining two serotonergic agents. Moreover, they should alert patients to the fact that not only medicines used for depression, but also some medicines used for physical health problems, can increase risk of SS.
Patients treated with serotonergic drug combinations should be monitored regularly to pick up signs of serotonin toxicity early. Serotonin toxicity lies on a continuum from mild to severe. Symptoms are variable (Table A2). Mild cases may only present with restlessness (akathisia) with or without tremor. Severe cases may present with high temperatures and rigidity (hypertonicity). 5 Rigidity in its own right can mask other symptoms of neuromuscular hyperactivity, such as tremor and hyperreflexia. Even for severe cases, symptoms are variable and nonspecific. In our meta-analysis, we found that only 75% of SS cases requiring intensive care presented with fever, defined as a temperature >38°C (100.4°F). Only 18% of cases presented with hyperthermia, defined as a temperature >41.1°C (106.0°F). 4
Strengths of our review
To our knowledge, this the first review to use systematic review principles and techniques to assess SS-associated severe hypertension and its treatment. All included cases were validated by two reviewers to ensure all cases met at least one diagnostic criteria system. We identified and analysed all published cases, following techniques as outlined in the PRISMA guidelines for systematic reviews. By integrating our own case with all other cases identified from the literature we hope to maximize the utility of our review for clinicians working in emergency scenarios. Acute threat to life demands quick decisions that are not reversible.
Limitations of our review
Only little evidence is available regarding the management of severe hypertension in the context of SS. As we could not identify any trial or observational study, we had to resort to individual case reports. Such anecdotal observations can be used as a starting point to explore this clinical problem of potentially life-threatening dimensions. Ignoring case reports due to methodological concerns such as selection bias is not an option where clinical evidence is virtually absent and clinical guidance relies mainly on expert opinions. 4 Uncommon or emerging clinical phenomena rely on pattern recognition of cases. Applying systematic review techniques to the present analysis is a robust way to collate and analyse anecdotal, but important, clinical information.
Conclusion
Severe arterial hypertension due to SS is a life-threatening condition. SS occurs in a multitude of settings and disguises. The therapeutic approach has to take individual comorbidities into consideration. Some classic antihypertensives may not be effective. The vast majority of patients are initially treated with benzodiazepines targeted at neuromuscular symptoms.
Based on the limited evidence available, we conclude that patients with severe hypertension not responding on benzodiazepines may benefit from cyproheptadine with or without propofol with the shortest delay possible to prevent serious cardiovascular damage. Both substances may even improve the neuromuscular symptoms. Since propofol is given i.v., the dosage can be easily titrated. Current dosing recommendations for cyproheptadine may be too low in severe cases. Maintenance of antiserotoninergic treatment may be necessary for periods of several days.
Supplemental Material
Appendix_1_Diagnostic_criteria_for_Serotonin_Syndrome – Supplemental material for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques
Supplemental material, Appendix_1_Diagnostic_criteria_for_Serotonin_Syndrome for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques by Michael Ott, Julie K. Mannchen, Fariba Jamshidi and Ursula Werneke in Therapeutic Advances in Psychopharmacology
Supplemental Material
Appendix_2_Search_strategy-16Oct2018 – Supplemental material for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques
Supplemental material, Appendix_2_Search_strategy-16Oct2018 for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques by Michael Ott, Julie K. Mannchen, Fariba Jamshidi and Ursula Werneke in Therapeutic Advances in Psychopharmacology
Supplemental Material
Appendix_3_Laboratory_results – Supplemental material for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques
Supplemental material, Appendix_3_Laboratory_results for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques by Michael Ott, Julie K. Mannchen, Fariba Jamshidi and Ursula Werneke in Therapeutic Advances in Psychopharmacology
Supplemental Material
Appendix_4_Publications_included_FINAL – Supplemental material for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques
Supplemental material, Appendix_4_Publications_included_FINAL for Management of severe arterial hypertension associated with serotonin syndrome: a case report analysis based on systematic review techniques by Michael Ott, Julie K. Mannchen, Fariba Jamshidi and Ursula Werneke in Therapeutic Advances in Psychopharmacology
Footnotes
Appendix 4
Table A1.
Laboratory results.

|
|
|
|
|
|---|---|---|---|
| Na+ | mmol/l | 142 | 137–145 |
| K+ | mmol/l |
|
3.6–4.6 |
| Creatinine | μmol/l |
|
45–90 |
| Urea | mmol/l |
|
3.1–7.9 |
| Creatine kinase | μkat/l | 2.94 | <3.5 |
| Troponin (high sensitive) | nmol/l |
|
0–15 |
| Myoglobin | μg/l |
|
25–58 |
| Phosphate | mmol/l |
|
0.8–1.5 |
| CRP | mg/l |
|
<10 |
| Arterial blood gas analysis | |||
| pH |
|
7.35–7.45 | |
| pCO2 | kPa |
|
4.6–6.0 |
| Standard bicarbonate | mmol/l |
|
22–27 |
| Base excess | mmol/l |
|
±3 |
| Urine analysis | |||
| Protein | g/l |
|
<0.03 |
CRP, C-reactive protein.
Table A2:
Diagnostic criteria for Serotonin Syndrome.
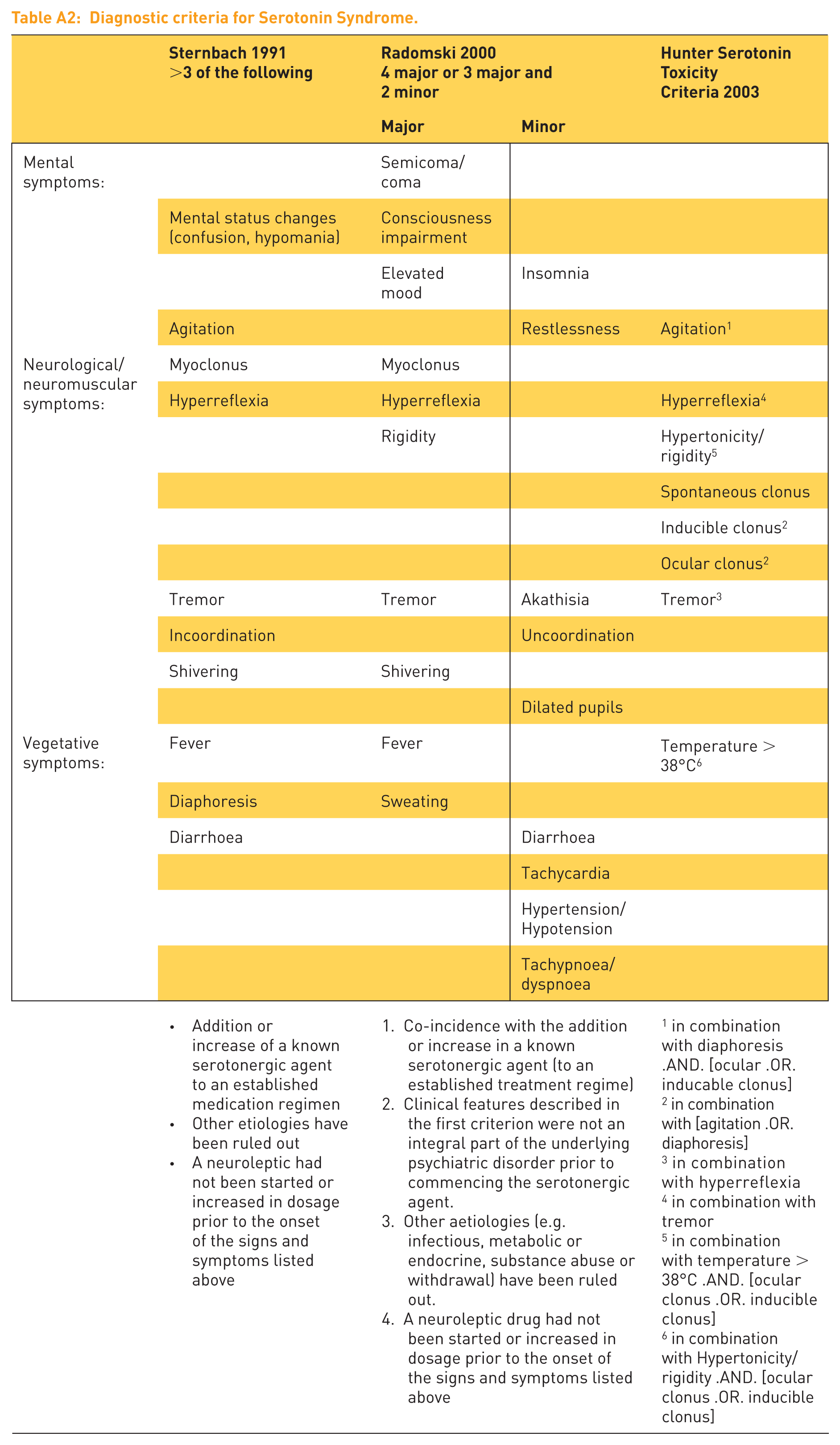
| Sternbach 1991 |
Radomski 2000 |
Hunter Serotonin Toxicity |
||
|---|---|---|---|---|
| Major | Minor | |||
| Mental symptoms: |
Semicoma/ |
|||
| Mental status changes |
Consciousness |
|||
| Elevated |
Insomnia | |||
| Agitation | Restlessness | Agitation1 | ||
| Neurological/neuromuscular |
Myoclonus | Myoclonus | ||
| Hyperreflexia | Hyperreflexia | Hyperreflexia4 | ||
| Rigidity | Hypertonicity/ rigidity5 | |||
| Spontaneous clonus | ||||
| Inducible clonus2 | ||||
| Ocular clonus2 | ||||
| Tremor | Tremor | Akathisia | Tremor3 | |
| Incoordination | Uncoordination | |||
| Shivering | Shivering | |||
| Dilated pupils | ||||
| Vegetative |
Fever | Fever | Temperature > 38°C6 | |
| Diaphoresis | Sweating | |||
| Diarrhoea | Diarrhoea | |||
| Tachycardia | ||||
| Hypertension/ |
||||
| Tachypnoea/ |
||||
| ● Addition or increase of a known serotonergic agent to an established medication regimen |
1. Co-incidence with the addition or increase in a known serotonergic agent (to an established treatment regime) |
1 in combination with diaphoresis .AND. [ocular .OR. inducable clonus] |
||
Table A3.
Search strategy and method for case report analysis.

| Objective | To identify pharmacological treatments to control arterial hypertension in the setting of serotonin syndrome |
| Eligibility criteria | Trials, observational studies or case reports in which participants fulfil the following criteria: Inclusion criteria: Patients (of any age) fulfilling one or more of the current three diagnostic criteria for serotonin syndrome [Sternbach criteria (SC), Radomski criteria (RC) or Hunter criteria (HC)]. Exclusion criteria: we excluded all cases: (1) not meeting any of the diagnostic criteria despite claiming a diagnosis of serotonin syndrome; (2) being aetiologically uncertain despite meeting the diagnostic criteria; (3) containing insufficient clinical information to rate; (4) being historical; or (5) implicating first-generation antipsychotics or concomitant neuroleptic malignant syndrome |
| Information sources (databases) | (1) MEDLINE(R) (2) ISI Web of Science: Science Citation Index Expanded (3) Cochrane Central Register of Controlled Trials CENTRAL, the Cochrane Library (4) CINAHL (5) TOXNET Toxline search (6) ClinicalTrials.gov (www.clinicaltrials.gov) |
| Search | Search was performed with the terms [‘serotonin syndrome’ OR ‘serotonin toxicity’] AND [‘hypertension’ OR ‘hypertensive’] Limits for case reports: from 2004, by which time the HC had been published and all three diagnostic criteria became available, until 31 December 2016 |
| Study selection | Inclusion and exclusion criteria as above |
| Data collection process | Systematic review: we identified no studies meeting the inclusion criteria Case report analysis: we abstracted all eligible cases into a new dataset, including general patient characteristics, onset, clinical course, mode of presentation, symptoms, diagnostic criteria, associated medications, treatment and outcome; two investigators (UW and FJ or UW and MO) independently double rated all cases regarding the HC, SC or RC. If the article fulfilled the inclusion criteria, two authors then reviewed the full text of each article to extract data on medication, clinical data, treatment and outcome; disagreements between authors’ ratings were resolved by consensus |
| Data items | Symptoms of serotonin syndrome: 20 symptoms appearing in any of the three diagnostic criteria sets |
| Blood pressure (BP) at presentation and highest/lowest BP reported. |
|
| Bias | As there were no trials or observational studies available, we relied on case reports only |
| Summary measures | Time to normalization of BP after given treatment |
| Synthesis of results | Descriptive |
Funding
This work was supported by a grant of the Norrbotten County Research & Development Fund, Sweden.
Conflict of interest statement
Ursula Werneke has received funding for educational activities (Masterclass Psychiatry Programme/EAPM 2016, Luleå, Sweden): Astra Zeneca, Janssen, Eli Lilly, Lundbeck, Novartis, Servier, Sunovion, Otsuka and Shire.
Michael Ott is a scientific advisory board member of Astra Zeneca Sweden.
Julie Mannchen and Fariba Jamshidi declare that there is no conflict of interest.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
