Abstract

Depot antipsychotic medication, often referred to as long-acting injections (LAIs), are an important treatment option for mental health problems such as schizophrenia and bipolar disorder.1,2 They were designed over 50 years ago to promote adherence to maintenance antipsychotic medication, but arguably remain stigmatized and underused. 3 Table 1 compares oral formulations with LAIs.
Summary of the pros and cons of LAIs versus oral medications.
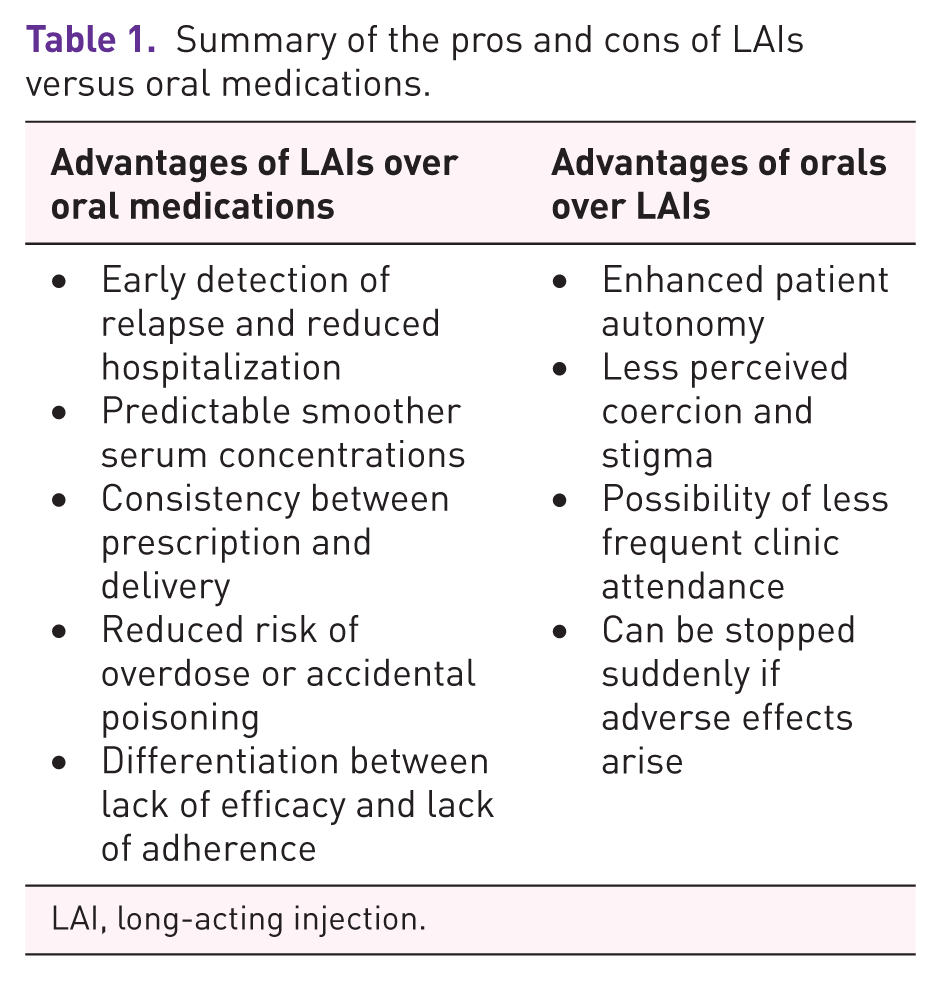
LAI, long-acting injection.
International guidelines recommend LAIs for both patients with demonstrated poor medication adherence and those who prefer an LAI.1,2 Both an independent long-term comparative trial by Subotnik et al. 4 and a systematic review by Taylor and Ng 5 have highlighted the value of earlier use of LAI preparations in psychosis, reporting decreased relapse rates through improved medication adherence, which reduces the risk of toxic relapse and possible subsequent rehospitalisation.4–6 This begs the question, would a cardiologist wait till the second or third heart attack before instituting the most effective treatment? 6
Antipsychotic medication development has stagnated over recent years, so the introduction of the first 3-monthly or quarterly LAI antipsychotic deserves review.
Paliperidone palmitate 3 monthly (PP3)
Paliperidone palmitate is a benzisoxazole derivative, and the 3-monthly variant (PP3; also known as INVEGA TRINZA®) is available in prefilled syringes that require vigorous shaking, unlike many other LAIs or depots. The transport substrate of PP3 has been modified to increase its elimination half-life, which allow dosing at 3-monthly intervals. PP3 is the first and only currently available quarterly injectable, and as such, represents a significant advance in the maintenance treatment of schizophrenia.
PP3 is available in four strengths (175 mg, 263 mg, 350 mg, and 525 mg), which are equivalent to the original monthly paliperidone LAI strengths of 50 mg, 75 mg, 100 mg, and 150 mg, respectively. Formulation of PP3 equivalent to a paliperidone monthly dose of 25 mg is not yet available. There is a 2-week window for administration of PP3.
Importantly, PP3 is only recommended after stability on monthly paliperidone for a minimum of 4 months, where the last two monthly doses are required to be stable prior to switching.
PP3 has an elimination half-life of approximately 85–95 days (when administered as a deltoid injection), and 118–139 days (when administered as a gluteal injection). PP3 would likely remain in the body for a period of 340–695 days, depending on injection site, dose, and individual patient pharmacokinetic factors.
The two approval trials for PP3 were both sponsored by the manufacturer. The first trial demonstrated efficacy compared with placebo for prevention of schizophrenia relapse, and involved 379 participants who each received a single dose of PP3, with 145 individuals then receiving placebo and 160 receiving at least one further dose of PP3. The second trial was a long-term double-blind non-inferiority study, which recruited 1429 participants. All of them were treated with monthly paliperidone palmitate before 512 people were then randomly assigned to continue monthly paliperidone, and 504 were randomly assigned to switch to PP3, with a 48-week continuation phase. Relapse rates over that period were not significantly different between PP3 (8%) and monthly paliperidone (9%) arms in this study. No other studies of PP3 are yet in the public domain.
Given that PP3 contains the same active molecule as monthly paliperidone, it has the same potential adverse effects,7,8 including prolactin elevation; weight gain; and, less frequently, sedation and extra-pyramidal symptoms. Key differences between PP3 and monthly paliperidone are the requirement for vigorous shaking of the vial for 15 seconds to ensure uniform dosing, as well as the increased volume of injection, with the highest PP3 dose (525 mg) being administered over a 2.6 ml injection. Despite the increase in volume of injection, the non-inferiority study established comparable tolerability between preparations, without significant increases in local injection site reactions. Dealing with emergent adverse effects with a 3-monthly preparation could be intrinsically challenging, although stabilization on the monthly preparation beforehand should reassure clinicians as to the patient’s ability to tolerate PP3.
Current experience
The authors’ experience (admittedly limited to 12 cases) of switching to PP3 has revealed that no patients have so far declined to switch, with all indicating a preference for the less frequent 3-monthly injections. Patients are reassured that the therapeutic molecule is the same, with a very similar efficacy and tolerability profile, it is simply a ‘slower release’ variant of paliperidone LAI. There have also been no significant new adverse effects compared with monthly paliperidone, with a global impression of similar local injection site reactions and pain.
One anticipated concern with it is that the decrease in frequency of injection may result in decreased patient contact and care. Although the intramuscular administration of PP3 is 3 monthly or ‘seasonal’, that does not mean the frequency of concomitant psychosocial interventions or monitoring has to diminish. Indeed, one perspective is that a slow-release injectable liberates time for other aspects of treatment, such as physical healthcare or cognitive remediation.
A related concern might be that PP3 facilitates transition away from expert mental health care providers to primary care, where the multidisciplinary holistic therapy may not be available. As always, clinical needs should be assessed on a case-by-case basis, and not be dictated by political or financial constraints.
Clinicians administering PP3 have at times commented that it is more difficult to be aware of when the next LAI dose is due, resulting in medication charts being checked repeatedly. PP3 does offer a 2-week window for administration, easing this burden and accommodating weekends and staff time off.
Looking ahead
With current evidence indicating treatment with antipsychotic medication for at least 18 months following a first episode of psychosis, 2 and studies demonstrating poor medication adherence in this specific population, 6 the prospect of a slower-release antipsychotic LAI is a step forward. Use of a 3-monthly depot early in the schizophrenia disease course would, however, represent a paradigm shift. Nevertheless, it is noted that 3 monthly (and longer-acting implantable) contraceptives are widely sought after by the general public.
Patient choice and autonomy need to be upheld, but patients also deserve access to appropriate evidence-based medication from the outset of their diagnosis, including information on the benefits of antipsychotic medication; the risks of early cessation or poor adherence with antipsychotic medication; and available preparations including oral and LAI antipsychotics. There is evidence that some psychiatrists themselves view LAIs or depot as stigmatizing or too coercive, and perhaps unconsciously filter out this potential therapeutic option. 9 This is despite a national study indicating the same antipsychotic molecule in LAI formulation reduced relapse rate by 64% compared with the identical oral molecule. 6
The unveiling of PP3 serves as a timely reminder that antipsychotic LAIs have a role across the disease pathway in schizophrenia. Given the evidence noted above, it could be argued that mental health clinicians should be justifying why they are not using clozapine or an LAI in established schizophrenia.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
MT has accepted fees and hospitality from Janssen, the manufacturer of PP3. HH has nil to declare.
