Abstract
Background:
Asenapine is a second-generation antipsychotic approved in Europe for treating moderate-to-severe manic episodes in adults affected by type I bipolar disorder (BD-I). We aimed to compare its efficacy in psychiatric inpatients with BD-I, with or without substance use disorder (SUD).
Methods:
We administered flexible asenapine doses ranging from 5–20 mg/day to 119 voluntarily hospitalized patients with Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM-IV-TR) BD-I diagnosis, with or without SUD. Patients were assessed with clinician-rated questionnaires [i.e. Brief Psychiatric Rating Scale (BPRS), Young Mania Rating Scale (YMRS), Hamilton Depression Rating Scale (HDRS), Hamilton Anxiety Rating Scale (HARS), and Global Assessment of Functioning (GAF)]. Assessments were carried out at baseline (T0, prior to treatment), and 3 (T1), 7 (T2), 15 (T3), and 30 days (T4) after starting treatment for all clinical scales and at T0 and T4 for the GAF.
Results:
Patients improved on all scales (p < 0.001) across all timepoints, as shown both by paired-sample comparisons and by applying a repeated-measures, generalized linear model (GLM). Patients without comorbid SUD showed greater reductions in BPRS scores at T2 and T3, greater reduction in YMRS scores at T3, and lower HARS scores at all timepoints. HDRS scores did not differ between the two groups at any timepoint. However, the reduction in HARS scores in the comorbid group was stronger than in the BD-I only group, albeit not significantly. Side effects were few and mild-to-moderate.
Conclusions:
The open-label design and the relatively short observation period may expose to both type I and type II statistical errors (false positive and false negatives). Asenapine showed effectiveness and safety in hospitalized BD-I patients. Its effect was stronger in patients without comorbid SUD.
Introduction
Bipolar disorder (BD) is a circular, chronic, progressive mood disorder characterized by major depressive episodes alternating with manic/hypomanic or mixed episodes [Judd et al. 2002; Leboyer and Kupfer, 2010]. BD with a history of a manic episode is type I (BD-I), whereas BD without manic episode history is type II [American Psychiatric Association, 1987, 2000]. Combined estimated prevalence of type I and type II BD ranges from 0.5–2% [Warren and Dubovsky, 2013]. The prevalence of BD rises to 4.4–6.4% when adding subthreshold/subsyndromal forms [Merikangas et al. 2011].
BD is one of the main causes of disability worldwide, being associated to important psychological and social consequences, long-lasting unemployment, and medical comorbidity [Zimmerman et al. 2010; Merikangas et al. 2011]. BD has a high mortality rate that can be partly attributed to suicide (BD patients have the highest suicide risk after first hospital contact [Nordentoft et al. 2011] and their lifetime suicide attempt rate is 10–15%), to accidents, to a reckless dysmetabolizing lifestyle, and to the negative consequences of substance abuse [Craig et al. 2006; Khalsa et al. 2008; Young et al. 2013]. Due to its recurrent nature, patients present post-onset symptoms for almost half of their lives; this clinical and epidemiological finding imposes effective [Judd et al. 2002; Collins et al. 2011] and safe treatment that may ensure compliance, since the lack of treatment adherence is the most common cause of relapse or recurrence [Gutiérrez-Rojas et al. 2010].
Many second generation antipsychotics (SGAs) have been approved for acute, subacute, and maintenance treatment of BD. According to the World Federation of Societies of Biological Psychiatry (WFSBP) [Grunze et al. 2010] and to the Canadian Network for Mood and Anxiety Treatments (CANMAT) guidelines [Grunze et al. 2013], SGAs are first choice treatment in managing manic episodes, either in monotherapy or in association with mood stabilizers; quetiapine and olanzapine have obtained fair results also in the control of depressive episodes [Grunze et al. 2010, 2013; Yatham et al. 2013].
Asenapine has been recently approved in Europe for the treatment of moderate-to-severe manic episodes in adults, whereas in the US and some other countries it is used in the acute treatment of schizophrenia. Asenapine blocks 5-HT2A, 5-HT2B, 5-HT2C, 5-HT6 and 5-HT7 serotonin receptors, dopamine D2, D3 and D4 receptors, and α1A, α2A and α2C adrenoceptors [Szegedi et al. 2011; Azorin et al. 2013; Young et al. 2013]. Its moderate affinity for histamine receptors and almost complete lack of affinity for muscarinic acetylcholine receptors may explain its low propensity for metabolic changes [Potkin, 2011]. Clinical studies of asenapine to date showed efficacy in acute manic and mixed episodes [Panagides et al. 2007; McIntyre et al. 2010a], possibly in controlling depressive symptoms [Szegedi et al. 2011], and in maintenance [McIntyre et al. 2010b]. However, currently available data are insufficient for drawing conclusions, hence the need for further studies.
While there is consensus as to the importance of dopamine receptor blockade in mediating the antipsychotic effects of antipsychotics, and also regarding the importance of accumbens dopaminergic transmission in the maintenance of addictive behaviour [di Chiara et al. 2004], there is no clear indication for the use of atypical antipsychotics in the treatment of substance use disorders (SUDs). A study found clozapine to reduce symptoms of substance dependence more than olanzapine, and this has been attributed to the faster dissociation of the former from the D2 receptor [Machielsen and de Haan, 2009]. Although asenapine shows not so fast a dissociation from D2 receptors, it nevertheless blocks the D3 and D4 dopamine receptors, that have been linked to addiction [di Ciano et al. 2014; le Foll et al. 2014]. So it is possible that this drug improves sideways both the addiction and the psychosis. If true, we may expect a differential response of people with BD-I and SUD comorbidity.
Our aim was to assess differences in the clinical response to asenapine between patients with BD-I patients alone and BD-I patients comorbid with SUD from a cohort of voluntarily hospitalized BD-I patients observed in a naturalistic setting.
Material and methods
Patients
The study has been conducted in adult patients (aged 18–60) with BD- I who were voluntarily admitted between May 2014 and March 2015 at the Neuropsychiatric Hospital Villa von Siebenthal, Genzano, Rome, Italy. We recruited consecutive patients with DSM-IV-TR [American Psychiatric Association, 2000] BD-I on asenapine; each patient received the minimal effective dose. Diagnosis was confirmed through the Structured Clinical Interview for DSM-IV-TR Axis I Disorders (SCID-I) [First et al. 2002]. Patients who were on mood stabilizers or benzodiazepines were allowed to keep this regimen unaltered during the course of the study. Excluded were patients with neurological disorders, and intellectual disability.
Assessment scales
Patients were assessed through clinician-rated clinical scales and a functioning scale at baseline (T0, start of the treatment), 3 days later (T1), 1 week later (T2), 15 days later (T3), and 1 month later (T4, endpoint).
To minimize errors, all evaluations for each patient were carried out by the same trained psychiatrist or psychologist (PM, CF, PZ, RP). Cohen’s κ for interrater reliability was 0.87 for all instruments used; p < 0.0001 (i.e. high). History and sociodemographic data were collected in a purposely constructed database, where annotation was made also for past drug treatment history and psychotropic drug use.
The following clinical scales were completed at T0, T1, T2, T3, and T4:
Hamilton Depression Rating Scale (HDRS) [Hamilton, 1960]: this is a scale sensitive to change that measures the severity of depressive symptoms. It contains an important proportion of somatic symptoms. It is a clinician-rated 21-item scale whose first 17 items are added to obtain the total score; single items are Likert, ranging 0–4 (8 items) or 0–2 (9 items); 0–7 is normal, 8–13 mild depression, 14–18 moderate depression, 19–22 severe depression and ⩾23 very severe depression. In antidepressant clinical trial research, a drop of at least 50% from baseline scores is considered as treatment response, while a score <8 is a remission. HDRS interviews were carried out by certified clinicians whose interrater reliability was 0.875 (Fleiss’ kappa).
Hamilton Anxiety Rating Scale (HARS) [Hamilton, 1959]: it is a scale that is sensitive to treatment-related change and measures the severity of anxiety symptoms. The scale consists of 14 items that are scored after interview on a five-point Likert scale, ranging from 0 = not present to 4 = severe. Total score may range from 0–56, with scores of 14–17 indicating mild anxiety, 18–24, moderate, and 25–30 severe anxiety.
24-item Brief Psychiatric Rating Scale (BPRS) [Lukoff et al. 1986; Roncone et al. 1999]: it is a scale of general psychopathology mainly focusing on psychotic symptoms. Initially developed as a 16-item [Overall and Gorham, 1962] and 18-item scale [Overall, 1974], has been later expanded to 24 items [Lukoff et al. 1986], but conserved its factor structure [Ventura et al. 2000]. Items 1–14 are rated on the basis of patient’s self-report and additionally, items 7, 12 and 13 are also rated according to observed behaviour. Items 15–24 are rated on the basis of observed behaviour and speech. All items range from 1 to 7 on a Likert scale, where 1 is symptom not present and 7 is extremely severe. The possible score range of the scale is 24–168; symptom severity rises with increasing score.
Young Mania Rating Scale (YMRS) [Young et al. 1978, 2008]: it is a scale assessing the severity of manic symptoms of BD. This is an 11-item scale whose items are rated differently (i.e. items 1–4, 7, and 10–11 receive scores of 0–4 each, while items 5–6 and 8–9 are scaled 0–8 with only even numbers possible). In this scale, a score ⩾39 is severe mania, while ⩽12 is no mania (remission), with scores between 13–38 representing mild-to-moderate mania. Response is considered an at least 50% drop from baseline values.
The Global Assessment of Functioning (GAF) [Endicott et al. 1976] has been completed at T0 and T4. For each of the two assessments, the last month from the interview was scored. This pseudo-continuous 1–100 scale is subdivided in ten 10-point content layers with higher scores indicating better psychosocio-occupational functioning.
In the BD with SUD group we also evaluated substance craving by using a Visual Analogue Scale (VAS); this is a 10 cm bar with no craving at one extreme and extreme craving at the other. The bar had no other indications and the participants were asked to place a sign anywhere along the bar, where they considered their craving to be appropriate for that day, combining both frequency of urge and intensity. The rating was made by applying a 10 cm-long millimetre ruler on the VAS.
All patients provided free, informed consent for all treatments and procedures to which they were subjected, for participating in the study, for their treatment of sensible data and publication of their data, as related to this study.
The study has been conducted in accordance with the Principles of Human Rights, as adopted by the World Medical Association at the 18th WMA General Assembly, Helsinki, Finland, June 1964 and subsequently amended by the 64th WMA General Assembly, Fortaleza, Brazil, October 2013. The study has received approval from the Villa von Siebenthal local ethical committee (approval number: Nr 3754/2014) prior to its initiation.
Statistical analysis
Mean differences of scores on assessment scales across the various time-points were compared through repeated-measures analysis of variance (ANOVA); covariance analysis (ANCOVA) has been used to adjust for possible confounders. For categorical data comparisons we used the Chi-square test. All continuous variables were reported as mean ± standard deviation (SD). For nonparametric statistics, data were expressed as median and percentiles. For all statistical analyses we used the Statistical Package for the Social Sciences version 19.0 software (IBM SPSS Statistics v. 19, ©2010 SPSS). To identify potential confounders we entered age and sex in a repeated-measures generalized linear model (GLM) as factors.
Results
Recruited were 119 consecutive patients with DSM-IV-TR [American Psychiatric Association, 2000] BD-I who were receiving asenapine. Patients received variable asenapine doses, ranging from 5–20 mg/day according to their clinical needs. Of the 119 patients included, 57 (47.9%) were male and 62 (52.1%) were female; the mean age of the sample was 43.3 years (SD = 10.92); its sex-wise socio-demographic characteristics are shown in Table 1. Table 2 shows distribution according to diagnosis. Table 3 reports sex-wise recreational psychotropic substance problem use (legal and illegal). There were significantly more index depressive episodes in the BD without SUD group than in the comorbid group (χ2 = 8.09; p < 0.05). There were no sex differences in the type of substance use in the BD with SUD group (Table 3). Table 4 shows the median scores on the clinical scales per time point in the two groups (i.e. BD without and with SUD). Both groups improved significantly on all scales at every time-point versus baseline. However, the BD without SUD group improved significantly more than the BD with SUD group at T2 and T3 on the BPRS, at T3 on the YMRS, while on the HARS, the BD with SUD group scored higher than the non-SUD group at each time point, including baseline. It should be noted that in the BD with SUD group the effect size of treatment on the anxiety scale was 0.64 (Cohen’s d = 1.68), while in the BD without SUD the effect size was less [i.e. 0.58 (Cohen’s d = 1.42)], although they may be both considered to represent strong effects.
Sociodemographic characteristics of the sample (n = 119).
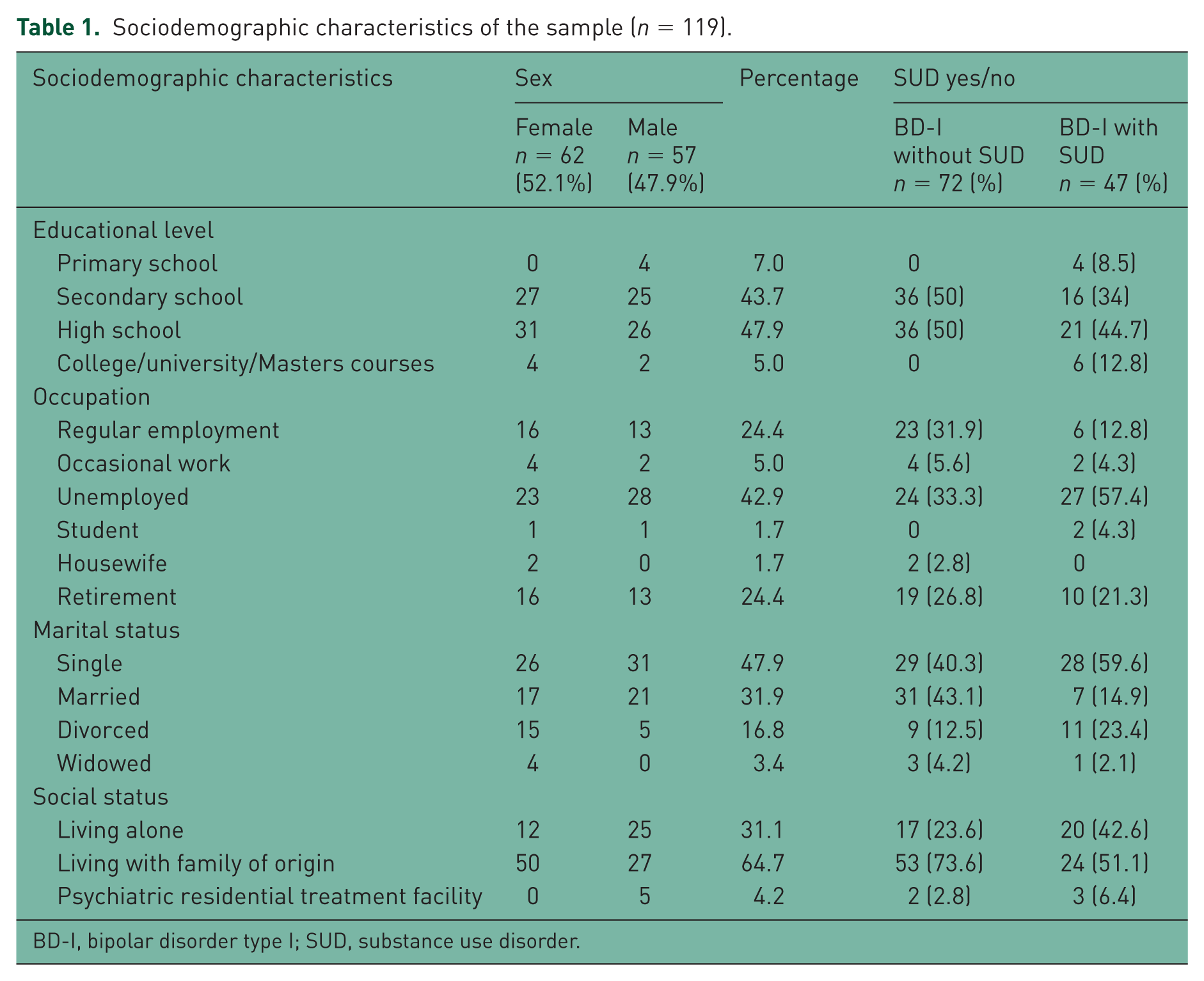
BD-I, bipolar disorder type I; SUD, substance use disorder.
Distribution of the sample according to DSM-IV-TR diagnosis.

BD, bipolar disorder; DSM-IV-TR, Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision; SUD, substance use disorder.
Chi-square test for comparing sex-wise psychotropic substance problem use in the sample.

Mean scores on psychopathology scales during the study in the bipolar subgroups with and without SUD. Intragroup comparisons show Wilcoxon rank-sum test’s Z and intergroup comparisons Mann–Whitney test’s U.

p < 0.05; **p < 0.01; ***p < 0.001. Significant differences in bold.
BD, bipolar disorder; BPRS, Brief Psychiatric Rating Scale; HARS, Hamilton Anxiety Rating Scale; HDRS, Hamilton Depression Rating Scale; SUD, substance use disorder; T0, baseline prior to treatment; T1, 3 days after treatment; T2, 7 days after treatment; T3, 15 days after treatment; T4, 30 days after treatment; YMRS, Young Mania Rating Scale.
Asenapine treatment was associated with a significant improvement on GAF scores from baseline to endpoint in the entire group (T0 mean = 46.23, SD = 6.13; T4 mean = 60.32, SD = 7.28; ANOVA F = 6.25; p < 0.0001). In the BD without SUD group, mean GAF increased from 46.57 (SD = 6.33) to 59.87 (SD = 7.64) (Student’s t = 8.72; p < 0.00001), while in the BD with SUD group, mean GAF increased from 45.70 (SD = 5.84) to 61.30 (SD = 7.92) (Student’s t = 7.4; p < 0.00001); means between the two groups were not significantly different at baseline or endpoint, with strong effect sizes obtained in both groups [i.e. 0.69 (Cohen’s d = 1.90)] in the BD without SUD group and 0.75 (Cohen’s d = 2.24) in the BD with SUD group.
Craving, as assessed through the VAS, showed no significant reductions from baseline (mean = 6.85, SD = 1.82) to T1 (mean = 6.43, SD = 2.03; T = 1.07, p = 0.29), but significant reductions at T2 (mean = 5.02, SD = 2.29; T = 4.29, p < 0.001), T3 (mean = 3.89, SD = 2.48; T = 6.60, p < 0.00001) and T4 (mean = 2.49, SD = 2.27; T = 10.27, p < 0.00001).
During the study period, 23 patients discontinued asenapine treatment for minor side effects, like excessive sedation (43.5%) and intolerance to sublingual anaesthesia (21.7%); 34.8% of dropout cases were attributed to lack of efficacy. No patient discontinued for onset of severe side effects.
Repeated-measures GLM analysis confirmed that the significant change obtained on psychopathology scales was not influenced by potential confounders, such as sex and age. The ANCOVA (Table 5) showed substance use to predict greater drops in HDRS and HARS scores from T0 to T4, longer hospital stay to predict greater HARS score drops from T0 to T4, married marital status to predict greater drops in BPRS scores, older age to predict improved global functioning, while higher educational status predicted a greater drop in manic symptoms from T0 to T4 only marginally.
Covariance analysis (ANCOVA).
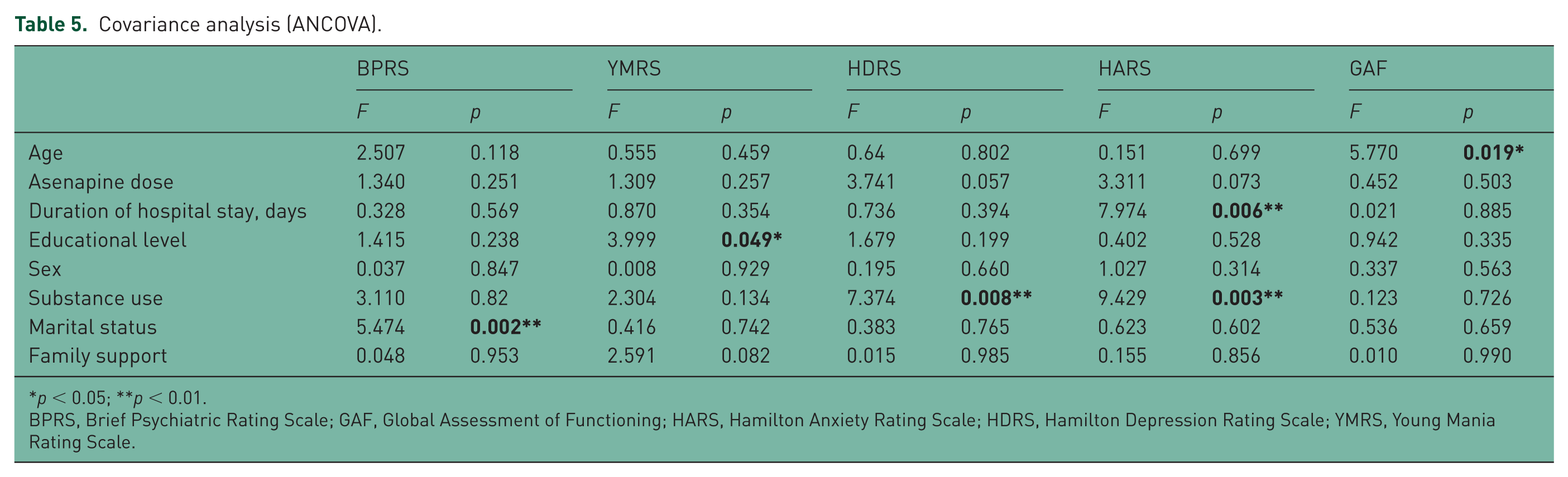
p < 0.05; **p < 0.01.
BPRS, Brief Psychiatric Rating Scale; GAF, Global Assessment of Functioning; HARS, Hamilton Anxiety Rating Scale; HDRS, Hamilton Depression Rating Scale; YMRS, Young Mania Rating Scale.
Discussion
In this naturalistic longitudinal study we observed 5–20 mg/day asenapine to progressively improve psychotic, manic, depressive, and anxiety symptoms and to increase global functioning in patients with BD-I. The general psychopathology and manic symptoms of patients without comorbid SUD responded better to asenapine, while their anxiety symptoms were milder than those of patients with comorbid SUD throughout the study period.
Asenapine received US Food and Drug Administration approval in 2009 for schizophrenia and the manic phase of BD [Traynor, 2009]. Various studies showed its efficacy and safety in BD. A 3-week double-blind study found it to be superior to placebo in acute mania and in mixed states [McIntyre et al. 2009a]. Patients who showed a positive response to this study were subsequently randomized to asenapine and olanzapine in a 9-week extension study which showed noninferiority for asenapine with respect to olanzapine, but also a lower incidence of weight gain with asenapine, compared with olanzapine [McIntyre et al. 2009b]. Later, the same team of investigators conducted a 3-week randomized, double-blind controlled trial and observed superiority for both asenapine and olanzapine versus placebo, but it was the latter, and not the former, that showed significantly greater response and remission rates with respect to placebo [McIntyre et al. 2010a]. This study confirmed the lower tendency of asenapine, compared with olanzapine, to induce weight gain. Finally, a study of asenapine versus olanzapine extending to 40 weeks has shown similar effectiveness and safety for both [McIntyre et al. 2010b]. However, reanalysing data for the effect on the Montgomery–Åsberg Depression Rating Scale (MADRS) [Montgomery and Asberg, 1979], asenapine was significantly superior to placebo in reducing MADRS scores after 3 weeks, while olanzapine was not [Szegedi et al. 2011]; this finding was subsequently confirmed by a French study [Azorin et al. 2013]. In these studies, the superior antimanic effect of both drugs with respect to placebo was already apparent by the second day of administration [Szegedi et al. 2013]. This has been confirmed by a further post hoc analysis [Cazorla et al. 2013]. A 52-week add-on study confirmed the positive antimanic effects of asenapine, as the association between asenapine and a mood stabilizer proved to be significantly better than the mood stabilizer alone [Szegedi et al. 2012]. Our naturalistic study also confirmed the antimanic and antidepressant efficacy of asenapine, alone or in association, and evidenced also a prominent anxiolytic effect.
The frequency of mixed states in BD populations is estimated to be between 6.7–40% [Hantouche et al. 2006; National Institute for Health and Care Excellence, 2014], varying according to the adopted diagnostic criteria [Ouanes et al. 2014]. A greater effect of asenapine has been hypothesized for mixed states compared with other antipsychotics, on the basis of the stronger reduction of depressive symptoms in BD patients with manic excitement [Young et al. 2013]. This became observable because in most studies patients were recruited regardless of whether they were in a manic phase only or were experiencing a mixed state [McIntyre, 2010]. While a study that analysed data from five clinical trials did not find a significant effect of asenapine on depressive symptoms [Gonzalez et al. 2011], other studies showed that with increasing depressive symptom severity in a patient, responsiveness of the manic episode to olanzapine, but not to asenapine, decreases [McIntyre et al. 2013]. Other studies showed a favourable cost–benefit profile for asenapine in mixed states [Caresano et al. 2014; Sawyer et al. 2014] and improved quality of life; the latter was more impaired in patients with mixed states compared with those with a nonmixed manic episode; the improved quality of life was more pronounced with asenapine, compared with olanzapine and placebo [Michalak et al. 2014].
Our data match those found in literature, as more than half of our sample had a mixed state and because depressive, anxious, manic, and psychotic symptoms improved at a similar pace (Tables 4 and 5). However, the lack of a control group precludes any speculation as to a possible specificity of the action of asenapine in the mixed state.
The safety profile of asenapine was acceptable, with a low incidence of adverse side effects. The dropout rate was <20%, thus favourably comparing with other antipsychotic trials [Song, 1997]; however, dropout rate does not appear to constitute a valid measure for assessing tolerability [Rowsell et al. 2001]. At any rate, we may speculate that the low incidence of side effects could have fostered treatment adherence. The most frequent side effects were sedation and sublingual anaesthesia intolerance. As predicted by asenapine’s receptor occupancy profile, no significant weight gain occurred in our sample.
Repeated-measures ANCOVA showed BD and substance use comorbidity to predict a greater response to asenapine. To our knowledge, this a previously unreported finding that needs replication. Should it find confirmation by double-blind studies, it could suggest a specific indication of asenapine in BD with prominent anxious-depressive symptoms and concomitant psychotropic substance use. There is great debate about how to treat BD substance use comorbidity, with some authors stressing the need for specific, targeted treatments [Pettinati et al. 2013]. However, the results of the ANCOVA are at odds with the finding of higher anxiety symptoms in the comorbid subsample and the better response of manic symptoms to asenapine in the non-SUD group.
Limitations
The lack of a control group and the relatively brief observation period constitute the major limitations of this study. Furthermore, patients with mixed states were overrepresented in our BD-I sample, hence our results may not extend to a prototypical BD-I population.
Conclusion
Asenapine administered as add-on or monotherapy showed efficacy and acceptable safety in the medium term in patients with BD-I. Drug treatment was associated with improvement in psychotic, manic, depressive, and anxious symptoms as well as in global functioning. Comorbidity with SUDs impacted negatively the course of anxious symptoms and the response of general psychopathology and manic symptoms, while not affecting depressive symptoms. Future studies using double-blind controlled designs will address whether this finding is not an artefact.
Footnotes
Acknowledgements
The authors wish to thank Dr Mimma Ariano, Dr Ales Casciaro, Dr Teresa Prioreschi, and Dr Susanna Rospo, Librarians of the Sant’Andrea Hospital, School of Medicine and Psychology, Sapienza University, Rome, for rendering precious bibliographical material accessible, as well as their Secretary Ms Lucilla Martinelli for her assistance during the writing of the manuscript.
Funding
All authors of this paper have no relevant affiliations or financial involvement with any organization or entity with a financial interest in, or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Conflict of interest statement
The authors declare that there is no conflict of interest.
