Abstract
Background:
Lamotrigine is an anticonvulsant as well as a mood stabilizer. Apart from its established use in the treatment of epilepsy, there has been an expansion of its use in the treatment of mental disorders. Patients with epilepsy as well as those with mental disorders are at increased risk of deliberate drug overdoses. An evidence base for the safety profile of lamotrigine in overdose is an essential tool for prescribers. The objective of this study was to carry out a narrative synthesis of the existing evidence for the safety profile of lamotrigine in overdose.
Methods:
A systematic search was conducted of EMBASE (1974 to December 2015), MEDLINE (1946 to December 2015), PsycINFO (1806 to December 2015) and CINAHL (1981 to December 2015) databases. Studies were included in which there was a deliberate or accidental single drug overdose of lamotrigine, with its toxic effects described. Studies that did not involve an overdose were excluded. A narrative synthesis of the described toxic effects was carried out.
Results:
Out of 562 articles identified, 26 studies were included, mainly in the form of case reports and series. The most commonly described toxic effects of lamotrigine were on the central nervous system, specifically seizures, movement disorders and reduced consciousness. Other toxic effects included QTc interval and QRS complex prolongations, hypersensitivity reactions, serotonin syndrome as well as rhabdomyolysis possibly due to seizures and/or agitation. Deaths were recorded in two studies, with cardiovascular and neurological toxic effects described.
Conclusions:
Even though lamotrigine has been reported to be well tolerated, there is a risk of toxic effects which can be life threatening in overdose. This needs to be borne in mind when prescribing to patients at an increased risk of deliberate drug overdose.
Introduction
Lamotrigine [3,5-diamino-6-(2,3-dichlorophenyl)-1,2, 4-triazine] a phenyltriazine derivative inhibits high-voltage activated-calcium channels [Stefani et al. 1996] and voltage-sensitive sodium channels as well as reduces neuronal glutamate release [Biton, 2006]. Lamotrigine has effects on the serotonergic pathway with reuptake inhibition [Sagud et al. 2008], which may explain its antidepressant property.
In terms of pharmacokinetic properties, lamotrigine has 98% bioavailability, with peak concentrations (Cmax) reached within 1–3 hours after ingestion. It is 55% protein bound, with an apparent volume of distribution (Vd) reported to be 0.9–1.2 l/kg in healthy volunteers and 1.28–1.36 l/kg in patients who have epilepsy and are on concomitant therapy. It undergoes hepatic glucuronidation with a conversion to the inactive 2N-glucuronide metabolite, which is excreted via the kidneys. A half-life of 24–30 hours has been reported for lamotrigine when it is used as monotherapy, but with multiple dosing it can induce its own metabolism reducing its half-life by 25% [Splinter, 2005], possibly through a weak induction of some phase II enzymes (uridine diphosphate glucuronosyltransferases) [Benedetti, 2000].
The toxic effects of lamotrigine on the central nervous and cardiovascular systems can be explained via its action on voltage-gated sodium channels, which are responsible for the initiation and propagation of action potential in excitable cells such as nerve and muscle cells [Catterall et al. 2005]. By virtue of its serotonin reuptake inhibition property, there is a risk of serotonin syndrome especially when it is ingested along with other substances that may potentiate this effect [Kotwal and Cutrona, 2015]. As central nervous system features of serotonin syndrome include: an altered mental state which could range from agitation to coma; neuromuscular hyperactivity which can present with a variety of features including hyperreflexia, ataxia and nystagmus; and generalized tonic–clonic seizures (in severe cases) [Iqbal et al. 2012], the toxic effects of lamotrigine on the central nervous system may also occur through excess serotonin.
An anticonvulsant hypersensitivity syndrome, which consists of a triad of fever, skin eruption and internal organ involvement [Knowles et al. 1999], has been associated with lamotrigine, despite its having a different structure to the aromatic anticonvulsants (phenytoin, phenobarbitone and carbamazepine), which have been more commonly associated with the anticonvulsant hypersensitivity syndrome. The exact mechanism for the hypersensitivity syndrome associated with lamotrigine is not clear, but it has been reported even with normal doses (100 mg) of lamotrigine (although in this case, the patient was also receiving valproic acid 1000 mg/day as treatment) [Chang et al. 2006].
Lamotrigine has an established role as an anticonvulsant in the treatment of partial, primary generalized tonic–clonic seizures and generalized seizures of Lennox–Gestalt syndrome in patients aged 2 years or older [Goldenberg, 2010]. In the treatment of mental disorders, benefits of lamotrigine have been demonstrated in bipolar depression [Calabrese et al. 1999], as well as in the prophylaxis of rapid cycling bipolar disorder [Calabrese et al. 2000]. There is increasing evidence of its possible benefits in the control of affective instability and impulsivity [Reich et al. 2009] in patients with borderline personality disorder, as well as its benefits in managing aggression amongst this group of patients [Leiberich et al. 2008].
Lamotrigine is reported to be generally well tolerated, with commonly reported adverse effects being dizziness, somnolence, nausea, asthenia and headaches in 8–20% of patients [Sabers and Gram, 2000]. Rash as a side effect, has been reported to develop in about 12% of patients [Sabers and Gram, 2000], with the incidence of serious rashes in bipolar studies reported to be around 0.1% [Goldsmith et al. 2003]. Stevens–Johnson syndrome, a severe rash characterized by cutaneous erythema with blister formation and mucosal lesions as well as fever [Mockenhaupt, 2011], has been described to occur in 1/1000 lamotrigine treated adults and 1/100 treated children [Varghese et al. 2006]. The exact mechanism by which Stevens–Johnson syndrome occurs in some patients treated with lamotrigine is not clear. A molecular target analysis of drugs associated with Stevens–Johnson syndrome using a bio-informatics approach, reported the sodium channel alpha 2 (Nav1.2) as a target highly associated with Stevens–Johnson syndrome [Burkhart et al. 2015]. This voltage-gated channel has been found to be expressed in keratinocytes present in skin biopsied from patients with painful skin conditions such as complex regional pain syndrome type 1 [Zhao et al. 2008]. It may play a role in the development of Stevens–Johnson syndrome in patients treated with lamotrigine, in view of the action of lamotrigine on voltage-gated sodium channels.
Other relatively less-common adverse events, which were reported from 12 controlled bipolar clinical trials, include dry mouth (6.3%), diarrhoea (6.2%) and tremor (3.9%). Data from the bipolar clinical trials showed the serious adverse events of mania, hypomania and mixed mania occurred in 2.5% of 1256 patients treated with lamotrigine, although this was felt to be comparable to the level found in patients treated with placebo (2.2% of 1094 patients) and lithium (2.5% of 280 patients). A 0.5% incidence of these adverse events were felt to be related to lamotrigine in the studies [Seo et al. 2011].
Generally, mental disorders are associated with a relatively increased risk of suicidal behaviour, of which self-poisoning has been identified as a common method, accounting for 20.2% (N ≈ 1259) and 38.2% (N ≈ 2381) of female and male suicides, respectively, in the United Kingdom in the year 2013 [Office for National Statistics, 2015]. A risk of suicide has also been associated with epilepsy [Christensen et al. 2007], even in the absence of a mental disorder. Hence, patients prescribed lamotrigine may be at higher risk of deliberate drug overdose.
The evidence regarding toxic effects of lamotrigine in overdose is mainly in the form of specific case reports, most of which concentrate on a number of specific features related to the case in question. In view of the established and increasing use of lamotrigine in groups of patients who are at an increased risk of drug overdoses, prescribers are in need of evidence on its safety profile in overdose in assessing the risks and benefits of its use. Hence we carried out a review of the literature in order to synthesize the available evidence for the safety profile of lamotrigine in overdose.
Objectives
The objectives of this study were as follows:
to conduct a systematic literature search of the toxic effects of lamotrigine in overdose;
to carry out a narrative synthesis of the existing evidence.
Method
Search strategy and selection criteria
We searched the databases EMBASE (1974 to December 2015), MEDLINE (1946 to December 2015), PsycINFO (1806 to December 2015) and CINAHL (1981 to December 2015) for relevant studies in which the toxic effects of lamotrigine were described in accidental or deliberate overdose.
We defined lamotrigine overdose using any of the following criteria.
For patients who were prescribed lamotrigine: lamotrigine taken accidentally or deliberately in doses higher than what they were prescribed or intended to take by their prescriber.
For individuals not prescribed lamotrigine: lamotrigine taken accidentally or deliberately at doses which are above the British National Formulary (BNF) recommended (adult equivalent) maximum doses for monotherapy (400 mg daily for bipolar disorder and up to 500 mg daily for seizures).
We did not apply any age or language restrictions to our search. A manual search of the references of the obtained articles was also carried out for relevant studies.
We included studies in which there was a deliberate or accidental overdose of lamotrigine, with the toxic effects attributable to the drug described.
We excluded studies in which:
lamotrigine was taken at prescribed doses with features of toxicity described;
lamotrigine was taken at prescribed doses, but its toxicity was precipitated by other coprescribed medications;
there were coingestants along with lamotrigine.
Included studies were mainly case reports and case series and, hence, an assessment of the risk of bias was not possible. A preliminary search carried out by our team had revealed observational studies as the level of evidence available to address the study objectives, with case reports and case series the main evidence found, hence a narrative synthesis was planned given the lack of appropriate data to conduct a meta-analysis.
Results
The search revealed a total of 562 records, out of which 26 eligible articles were included in the narrative review. Figure 1 shows the study selection process. Of these 26 articles which represented 543 individual cases, 21 were case reports, 4 were case series and 1 study had a case control design. One of the case series [Lofton and Klein-Schwartz, 2004] utilized data from 493 single-substance exposures to lamotrigine reported to the American Association of Poison Control Centers toxic exposure surveillance system. Another case series [Moore et al. 2013] utilized data from nine patients admitted to an inpatient toxicology centre with lamotrigine only overdoses, whilst only one case in each of the other two case series was judged to have met the inclusion criteria and included.

Study selection process.
Table 1 shows the characteristics of the included studies.
Characteristics of included studies.
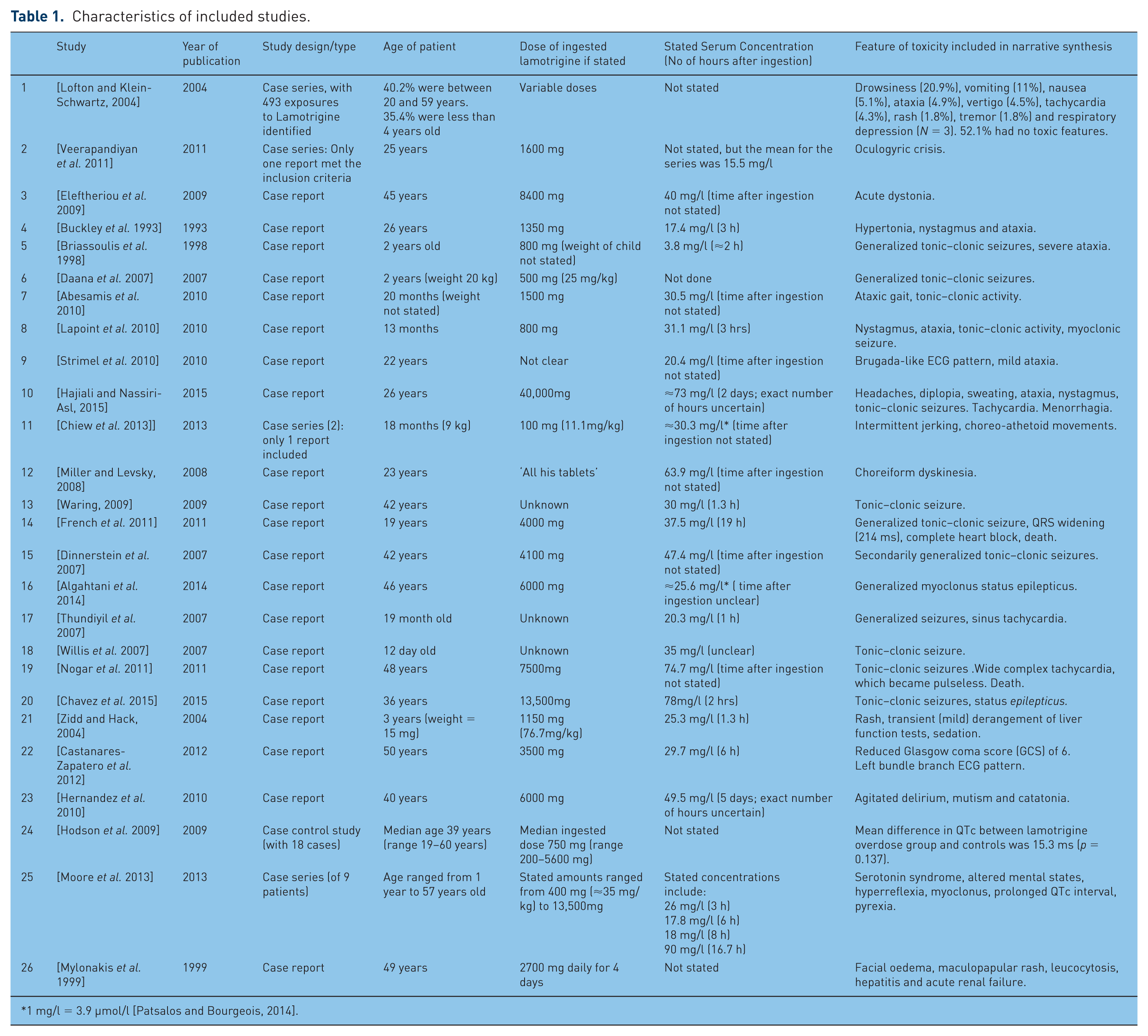
1 mg/l = 3.9 µmol/l [Patsalos and Bourgeois, 2014].
Frequency of toxic effects
The study by Lofton and Klein-Schwartz [Lofton and Klein-Schwartz, 2004] provided useful information on the frequencies of the observed toxic effects of lamotrigine in overdose. In this study of 493 patients, the frequencies of the following effects were mentioned: drowsiness/lethargy (20.9%); vomiting (11%); nausea (5.1%); ataxia (4.9%); dizziness/vertigo (4.5%); tachycardia (4.3%); confusion (2.2%); agitation (2.0%); rash (1.8%); slurred speech (1.8%); tremor (1.8%); nystagmus (1.6%); blurred vision (1.4%); and coma (1.2%). Seizure as a toxic effect was reported in eight people (1.6%), with three events of multiple seizures and two individuals with seizures of a status pattern. Respiratory depression was reported in three patients, whilst cardiac conduction disturbances were reported in two patients.
Described toxic effects of lamotrigine in deliberate overdose
Central nervous effects
In the studies included in this review, central nervous toxic effects appear to be the most commonly associated effect of lamotrigine in overdose. There were 249 individual reports of associated central nervous toxic effects in the form of 20 case reports and 4 case series.
Lamotrigine in overdose was associated with a wide range of central nervous system effects, such as: oculogyric crisis [Veerapandiyan et al. 2011], dystonia [Eleftheriou et al. 2009], nystagmus, ataxia [Buckley et al. 1993; Briassoulis et al. 1998; Daana et al. 2007; Abesamis et al. 2010; Lapoint et al. 2010; Strimel et al. 2010; Hajiali and Nassiri-Asl, 2015], slurred speech [Hajiali and Nassiri-Asl, 2015] and hypertonia [Buckley et al. 1993]. Abnormal movements with intermittent jerking, choreo-athetoid movements [Chiew et al. 2013] and choreiform dyskinesia [Miller and Levsky, 2008] have been described in case reports.
Paradoxically, lamotrigine which is an anticonvulsant has been associated with seizures in overdose [Waring, 2009; French et al. 2011]. The seizures associated with lamotrigine have occurred in patients without a documented prior history of seizures [Lapoint et al. 2010; French et al. 2011] and in patients with a history of seizures [Dinnerstein et al. 2007; Waring, 2009; Algahtani et al. 2014]. The seizures have been described as generalized [Thundiyil et al. 2007], with specific seizure types reported with single-drug overdoses of lamotrigine being: tonic–clonic seizures [Briassoulis et al. 1998; Daana et al. 2007; Dinnerstein et al. 2007; Willis et al. 2007; Waring, 2009; Abesamis et al. 2010; Lapoint et al. 2010; French et al. 2011; Nogar et al. 2011; Hajiali and Nassiri-Asl, 2015] and myoclonic seizures [Lapoint et al. 2010]. The tonic–clonic seizures were reported along with the clinical findings of clonus and hyperreflexia [Nogar et al. 2011] in a case, and status epilepticus developing in another case [Chavez et al. 2015]. The myoclonic seizures were described as a generalized myoclonic status epilepticus [Algahtani et al. 2014] in a report.
Reduced levels of consciousness (without an associated description of seizures) have been reported in single-drug overdoses of lamotrigine [Zidd and Hack, 2004; Castanares-Zapatero et al. 2012].
An encephalopathic picture which included an agitated delirium, mutism, nystagmus, upper motor neurone features as well as catatonia was described in a 40-year-old woman, with a previous history of glioblastoma multiforme and seizures, who took 30 × 200 mg tablets (6000 mg) of the extended-release (XR) preparation of lamotrigine [Hernandez et al. 2010].
Two patients in a case series from an inpatient toxicology centre [Moore et al. 2013] were reported to have met the Hunter criteria [Dunkley et al. 2003] for serotonin syndrome. One of these patients was a 1-year-old child with a serum lamotrigine level of 18 mg/l (taken at 8 hours), who had presented with agitation, diaphoresis and an inducible clonus. All nine patients in this case series were reported have had altered mental states with four patients having a depressed mental state, five patients having an agitated mental state and three patients having both depressed and agitated mental states. Five of the patients in the series had hyperreflexia and intermittent myoclonus.
Cardiac effects
There were 36 individual reports of associated cardiac toxic effects; these were in the form of 6 case reports, 2 case series and 1 case control study.
Widening of the QRS complexes in association with lamotrigine overdose, has been reported in a patient who had a normal heart rate of 88 beats/min, but with a left bundle branch block ECG pattern [Castanares-Zapatero et al. 2012]. A widening of the QRS complex of up to 210 ms has also being associated with a tachycardia, with outcomes of this being complete heart block [French et al. 2011] and a pulseless ventricular tachycardia [Nogar et al. 2011]. Sinus tachycardia as a nonspecific finding without a description of QRS complex widening, has also been reported [Thundiyil et al. 2007].
Chavez and colleagues [Chavez et al. 2015] described a male patient who developed a new onset right bundle branch block, with the prolongation of QRS interval (up to 128 ms) and QTc intervals (up to 458 ms). Much longer QTc prolongations (up to 586 ms) were described in a case series study [Moore et al. 2013]. Prolongation of QTc intervals as a toxic effect of lamotrigine in overdose was examined further in a case control study [Hodson et al. 2009]; even though the QTc interval was found to be longer in the lamotrigine overdose group compared with the control group, the difference between the groups was not statistically significant (p = 0.137). However, this was a small study with 18 cases.
A brugada-like ECG pattern has been described in an adult patient who had a serum lamotrigine level of 20.4 mg/l [Strimel et al. 2010].
Other described toxic effects
A range of other features of toxicity have been described in the course of a lamotrigine overdose, including nausea, vomiting, vertigo, lethargy, respiratory depression [Lofton and Klein-Schwartz, 2004]. Headaches, diplopia, dizziness, sweating and abdominal pain [Hajiali and Nassiri-Asl, 2015] were other described features which appeared to have preceded the onset of a tonic–clonic seizure in a patient.
Abnormal vital sign readings were stated in some of the reports, these included: tachypnea with recorded respiratory rates of up to 32 breaths per minute [Hajiali and Nassiri-Asl, 2015]; tachycardia with heart rates up to 131 beats/min [Nogar et al. 2011]; elevated blood pressure readings of up to 185/95 mmHg [Hajiali and Nassiri-Asl, 2015]; and pyrexia with a temperature above 38°C [Moore et al. 2013].
Rhabdomyolysis has been reported in the context of lamotrigine overdose, although this might have been the consequence of the associated seizure [French et al. 2011] or agitation [Vanholder et al. 2000].
Mylonakis and colleagues [Mylonakis et al. 1999] described a hypersensitivity-like syndrome with low-grade pyrexia, erythema and oedema involving the periorbital region, leucocytosis, elevated phosphokinase (7222 U/l), elevated alanine aminotransferase (76 U/l), elevated aspartate aminotransferase (154 U/l) and a raised creatinine (24 mg/l) in a 49-year-old man, who inadvertently received 4 daily doses of 2700 mg of lamotrigine.
Hajiali and Nassiri-Asl [Hajiali and Nassiri-Asl, 2015] described menorrhagia of a 2-day duration observed in a 26 year old who was reported to have taken up to 40 g of lamotrigine. The patient was reported to have had a normal gynaecological examination at the time. Her menstrual periods were reported to have been normal prior to this. Apart from the drop in the patient’s haemoglobin levels, investigations carried out in this patient were reported to be normal, although no specific mention was made of whether the investigations involved coagulation tests.
Rash, a common and important adverse reaction in normal doses, has also been described in two case reports of lamotrigine overdose. A flat, lacy, reticular blanching rash occurred along with a transient mild derangement of liver function tests in a 3 year old who accidentally ingested up to 1150 mg of lamotrigine [Zidd and Hack, 2004]. A diffuse, discrete confluent, branching red maculopapular rash without mucosal involvement was described in the context of lamotrigine overdose presenting with a hypersensitivity-like syndrome [Mylonakis et al. 1999].
Death
Death occurring in the course of lamotrigine overdose was well described in two separate case reports.
French and colleagues [French et al. 2011] described a case of a 19-year-old patient who took 4000 mg of lamotrigine. Events following the overdose had involved the development of seizures and a cardiac arrest. The patient subsequently developed a respiratory and metabolic acidosis as well as cardiac arrhythmias including complete heart block. He developed an acute renal failure, which was felt to be due to rhabdomyolysis with a recorded CK of over 23,000 U/l. A bacterial pneumonia infection (with Staphylococcus and Klebsiela) and a disseminated intravascular coagulation occurred prior to his death.
Nogar and colleagues [Nogar et al. 2011] described a 48-year-old man who ingested 7500 mg of lamotrigine, who developed a tonic–clonic seizure and subsequently developed a broad complex tachycardia which became pulseless, with a resulting anoxic brain injury and death 4 days later.
Both overdoses were solitary drug overdoses of lamotrigine.
Discussion
Lamotrigine taken in overdose is not always associated with toxic effects. In the case series of 493 patients [Lofton and Klein-Schwartz, 2004] who ingested lamotrigine as a single substance overdose, the majority (52.1%) did not experience any toxic effects, and there were no deaths. However, a study using a calculated Shannon information index to compare requirements for interventions in overdoses of newer anticonvulsants, reported lamotrigine as the most toxic in overdose by this definition [Wills et al. 2014], compared with gabapentin, levetiracetam, pregabalin, tiagabine, topiramate and oxcarbazepine.
This review reported single-agent overdoses of lamotrigine with doses of 100–40,000 mg and serum concentrations of 15.5–74.7 mg/l, have been associated with a wide variety of toxic effects on the central nervous and cardiovascular systems, as well as hypersensitivity-like syndrome. Figure 2 shows mechanisms for the toxic actions of lamotrigine on the body. Toxicity has been correlated with increasing serum lamotrigine levels in some [Froscher et al. 2002; Hirsch et al. 2004] but not all studies [Kilpatrick et al. 1996]. In view of the available evidence in this review for the toxic effects of lamotrigine being mainly case reports and case series, it was not possible to calculate a minimum dose or serum concentration associated with toxic effects.

Proposed mechanisms of the toxic effects of lamotrigine, in overdose, on the human body.
Treatment of lamotrigine toxicity is largely based on the observed clinical effects, and may involve the use of measures to prevent further absorption of the ingested lamotrigine such as use of activated charcoal and gastric lavage.
There has been a recommendation for the use of alkalinisation via sodium bicarbonate when cardiotoxicity due to sodium channel blockade is suspected [Castanares-Zapatero et al. 2012], which is similar to its suggested use in the management of tricyclic antidepressant toxicity [Newton, 2015]. In view of lamotrigine’s lipophilic properties, use of intravenous lipid emulsion has been suggested as a potential treatment strategy in cases of cardiac toxicity in the context of sodium channel blockade, if refractory to alkalinisation therapy [Castanares-Zapatero et al. 2012]. The exact mechanism by which lipid emulsion therapy works is not entirely clear. It has been suggested that lipid emulsion therapy may work by creating a ‘lipid sink’ [Weinberg et al. 1998] in the blood stream, which draws in a lipophilic substance thereby reducing the plasma aqueous concentration of the substance. It may also work by increasing the cardiac energy supply by improving fatty acid metabolism [Picard and Meek, 2006] or by activation of calcium channels [Castanares-Zapatero et al. 2012], thereby increasing the inotropic action [Chavez et al. 2015]. In a case report, there was a narrowing of a previously widened QRS complex and a normalisation of conduction interval disturbances, a few minutes after the initial bolus of lipid emulsion therapy was administered to a patient whose ECG had showed no response to sodium bicarbonate therapy [Castanares-Zapatero et al. 2012]. As the toxic effect of lamotrigine on the central nervous system can also be explained via sodium channel blockade [Catterall et al. 2005], the criteria for the use of lipid emulsion therapy would include life-threatening cardiac and/or neurologic toxicity refractory to conventional therapy.
Prompt and effective management of seizures is required, with parenteral benzodiazepines recommended as the first-line treatment for drug-induced seizures, and barbiturates or propofol recommended as second-line anticonvulsants. It is felt that propofol may have a synergistic effect when used with benzodiazepines or barbiturates [Chen et al. 2016]. Sedation with benzodiazepines may also play an important role in the management of features that may occur with a serotonin syndrome, such as: anxiety, agitation, increased muscular activity, tremors, hypertension, tachycardia and pyrexia [Dvir and Smallwood, 2008; Iqbal et al. 2012; Volpi-Abadie et al. 2013].
The strategies for managing lamotrigine overdose are summarized in Table 2.
General principles for the management of associated effects of lamotrigine overdose.

The role for haemodialysis in lamotrigine overdose has been considered. Using a 100 mg dose of lamotrigine in a study of the pharmacokinetics of lamotrigine in renal impairment, haemodialysis was reported to have shortened the elimination half-life from 59.6 ± 28.1 hours during the interdialysis period to 12.2 ± 6.4 hours during the dialysis period, with 17% of the drug extracted by haemodialysis [Fillastre et al. 1993]. Lu and colleagues [Lu et al. 2012] described a reduction in the serum concentration of lamotrigine from 61.2 to 7.2 mg/l over a 62-hour period using blood perfusion which involved haemodialysis and haemoperfusion. Taking into consideration its half-life and a potential to induce its own metabolism [Splinter, 2005], it was difficult to assess the role of haemodialysis in this instance. Further studies are required to clarify the role (if any) of haemodialysis in the management of lamotrigine overdose.
In conclusion, even though lamotrigine has been reported to be well tolerated at normal doses (and even in some overdoses), there is evidence of clinically important toxicity in overdose, with variable outcomes; this may be a complete recovery occurring in majority of the cases, but death has been reported. Its toxic profile must be considered when prescribing lamotrigine to patients who are potentially at an increased risk of taking overdoses. This should form part of the risk–benefit analysis particularly in the drug’s longer term and increasing use in psychiatric populations, when weighed against alternative mood stabilizers. There is a paucity of systematic observational studies for the toxic effects of lamotrigine in overdose, with only one case control study found and included in this study. Hence, frequencies of specific toxic effects could not be judged from this single study alone. There remains a need for more systematic evidence for the toxic effects of lamotrigine in overdose, in the form of case control studies as well as retrospective cohort studies, which can help confirm a direct association between the observed toxic effects and the overdose, including the consideration that some of the observed effects may well be indirect effects, with the triggering event being the overdose.
Limitations
Due to the nature of the review, only evidence in the form of observational studies (mainly case reports) was available. We sought to follow the principles of the PRISMA reporting guideline [Moher et al. 2009], but this was limited by the absence of quantitative studies; for instance, there was no risk of bias assessment we could conduct. Although we did not place a language restriction on our search criteria and on the included studies, we were not able to include one of the obtained articles in this review, due to our inability to translate its full text from Farsi to the English language.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
