Abstract
Objectives:
Olanzapine is a well established treatment for schizophrenia. The olanzapine pamoate depot (long-acting injectable) formulation improves compliance and clinical trials have shown it to be effective. However, there are no previously published reports evaluating olanzapine depot in violent patients with schizophrenia in the community. We evaluated the clinical efficacy of olanzapine depot, its effect on violence, hospitalization and incarceration in community patients with schizophrenia and prior history of serious violence.
Methods:
This was a retrospective service evaluation in a community forensic psychiatry service where patients had schizophrenia spectrum disorder and a significant history of violence. Treatment resistance, substance use disorders and antisocial personality disorder were common. Nine deidentified patient records were audited for 12 months pre and 12 months post olanzapine depot initiation to identify any clinical changes, breaches of (legislated) psychiatric treatment orders, hospital admission days, days incarcerated and emergency presentations.
Results:
Community forensic psychiatric patients treated with olanzapine depot showed an improvement in psychotic symptoms (p = 0.008) with overall decreases in violence, supported by reductions in hospitalization days (p = 0.018) and days incarcerated (p = 0.043). Several patients had reduced psychiatric treatment order breaches and emergency presentations.
Conclusions:
Community forensic psychiatric patients with schizophrenia responded to olanzapine depot with decreased violence and reduced hostility. A depot antipsychotic medication that reduces violence and improves engagement has significant implications for greater effective community management of forensic patients with schizophrenia.
Introduction
Olanzapine is a well established and effective treatment for schizophrenia [Taylor et al. 2015], available in oral and depot formulations, the latter as olanzapine pamoate. Advantages of depot antipsychotic formulations over oral formulations include improved compliance, with earlier alerts to the treating team if patients do not attend for their depot, thus reducing relapse rates with absolute risk reductions of 10% and a number needed to treat of 10 to prevent relapse [Leucht et al. 2011]. Various therapeutic guidelines recommend depot antipsychotic medication be considered in patients in whom noncompliance is present or suspected [Royal Australian and New Zealand College of Psychiatrists, 2005; Kuipers et al. 2014]. Most depot antipsychotic medications are typical antipsychotics, with disadvantages including increased extrapyramidal side effects and tardive dyskinesia compared with atypical antipsychotics [Taylor et al. 2015]. Additionally typical antipsychotics are less effective for negative symptoms of schizophrenia [Falkai et al. 2005].
Olanzapine pamoate is one of a few atypical depot antipsychotics available and is not associated with an elevated risk of extrapyramidal side effects [Taylor et al. 2015]. However olanzapine depot is expensive compared with other antipsychotics and, like oral olanzapine, is associated with metabolic syndrome, lipid profile abnormalities, blood glucose elevation and weight gain [Citrome, 2009]. Additionally olanzapine depot is associated with postinjection syndrome (PIS), where olanzapine pamoate is absorbed directly into the circulation resulting in sedation or delirium [Citrome, 2009]. PIS is rare, occurring in 0.07% of olanzapine depot injections, with no known fatalities, although some patients have required intubation [Detke et al. 2010]. The risk of PIS does not reduce with future injections, thus patients always need monitoring for 2–3 h postinjection with half-hourly visual checks [Luedecke et al. 2015]. This has ramifications for services, as patients need adequate facilities and clinical staff available to observe for the timeframe. Ironically, noncompliant patients with poor insight, who may need olanzapine depot most, may not tolerate the observation period [Devadason, 2010; Baruch et al. 2014].
Violence and aggressive behaviour occur more frequently (nearly fourfold) in patients with schizophrenia compared with people with no psychiatric illness [Falkai et al. 2005; Fazel et al. 2009]. Clozapine is highly recommended for treatment-resistant schizophrenia and is effective in reducing hostility, aggressive behaviour and violent crime in patients with schizophrenia [Frogley et al. 2012; Fazel et al. 2014]. Some evidence suggests that clozapine’s antiaggressive effect is more marked in patients with treatment-resistant schizophrenia. Additionally these antiaggressive effects appear to be specific, being to an extent greater than clozapine’s antipsychotic and sedative effects [Frogley et al. 2012]. Despite the benefits of clozapine in violent patients with schizophrenia, clozapine can be difficult to maintain in community settings due to the lack of any depot or injectable formulation [Kasinathan and Mastroianni, 2007] and the need for regular blood tests to monitor for potential neutropenia, tests which patients may refuse.
Olanzapine is similar in molecular structure and pharmacological receptor profile to clozapine [Conley et al. 2003]. However, the evidence for olanzapine decreasing violence in schizophrenia is varied. Some trials showed no advantage for olanzapine in reducing violence relative to other antipsychotics [Volavka et al. 2004; Swanson et al. 2008]. However, other studies showed olanzapine was superior to risperidone [Swanson et al. 2004b] and typical antipsychotics in reducing violence [Swanson et al. 2004a; Bitter et al. 2005].
Only one study has evaluated olanzapine depot in violent male patients with schizophrenia [Baruch et al. 2014]. All eight patients were admitted to a high-security hospital and treated with olanzapine depot. Six had improvement in symptoms and decreased violence. Four patients had less seclusion hours.
To our knowledge, there were no published reports evaluating olanzapine depot in violent patients with schizophrenia in a community setting. The present study aimed to describe the clinical efficacy of olanzapine depot, its effect on violence, hospitalization and incarceration in community patients with schizophrenia and prior serious violence.
Methods
The Australian Capital Territory (ACT) community forensic psychiatry service is located in Canberra, Australia. The service treats mentally disordered persons with a history of prior serious violence that pose an elevated risk of violence in the community. The predominant diagnosis is schizophrenia, often with complex presentations, and a majority are treatment resistant. Noncompliance with oral medication in the community is major problem for this population.
Relevant clinical governance and the ACT Health Human Research Ethics Committee’s Low Risk Sub-Committee approved this retrospective service evaluation on 20 April 2016, with methodology loosely based on previous work by Baruch and colleagues [Baruch et al. 2014]. All patients treated with olanzapine depot and clinically managed by the ACT community forensic psychiatry service were included. Anonymized data were retrospectively collected from the patients’ electronic mental health clinical record. Outcome measures were collected for 12 months prior to and 12 months following olanzapine depot initiation.
Patient records were retrospectively examined to gain an impression of each patient’s clinical state, scored on the Brief Psychiatric Rating Scale (BPRS) [Overall and Gorham, 1962], the primary outcome measure. Secondary outcome measures included the Health of the Nation Outcome Scales (HoNOS) [Wing et al. 1998], the Behavior and Symptom Identification Scale (BASIS-32) [Eisen et al. 1986], Life Skills Profile (LSP) [Rosen et al. 1989], psychiatric hospital admission days, days incarcerated in prison, psychiatric treatment order breach warnings and enacted breaches, emergency presentations and body weight.
Analytic approach
Statistical analyses were undertaken with SPSS Version 23.0 (IBM, Chicago, IL, USA). Pre and post olanzapine depot outcome measures were paired for each patient. Given the nonparametric nature of the quantitative paired data, Wilcoxon signed ranks tests were applicable.
Results
Nine male patients were treated with olanzapine depot for at least 12 months each and clinically managed by the ACT community forensic psychiatry service. Mean patient age was 34 with a standard deviation of 9.8 years (range 23–55). Two patients identified as Aboriginal. All patients suffered chronic illness for several years. Seven had psychiatric diagnoses of schizophrenia and two had schizoaffective disorder as per the Diagnostic and Statistical Manual of Mental Disorders, fifth edition [American Psychiatric Association, 2013]. Six patients met criteria for treatment resistance [International Psychopharmacology Algorithm Project, 2006] prior to olanzapine depot.
Six patients met criteria for comorbid antisocial personality disorder and eight patients had active cannabis or amphetamine use disorders. All patients had a significant violent offending history, with index offences ranging from aggravated assault, grievous bodily harm, arson with intent to cause grievous bodily harm and attempted murder. In seven patients there was a direct connection between active psychotic symptoms and the index offence. Prior to olanzapine depot initiation, eight patients were on depot antipsychotics (six received paliperidone, one risperidone, one flupenthixol and one had fluphenazine and haloperidol combined) and one patient was on clozapine though becoming noncompliant.
Primary and secondary outcome measures are displayed in Figures 1–10, with statistical significance according to Wilcoxon signed ranks tests presented in Table 1. Patients overall showed significant clinical improvement after 12 months on olanzapine depot, with a mean BPRS reduction from 58 to 37 (Z = −2.67, p = 0.008). Eight patients were much improved and one was minimally improved. There were no significant changes in HoNOS, BASIS-32 or LSP. Hospital admission days were significantly reduced (Z = −2.37, p = 0.018), as were days incarcerated in prison (Z = −2.023, p = 0.043). Psychiatric treatment order breach warnings, enacted breaches and emergency presentations were reduced overall, however this did not reach statistical significance (see Table 1).

Brief Psychiatric Rating Scale (BPRS).

Health of the Nation Outcome Scales (HoNOS).

Behavior and Symptom Identification Scale (BASIS-32).

Life Skills Profile (LSP).

Psychiatric hospital inpatient days.

Days incarcerated in prison.

Psychiatric treatment order breach warnings.
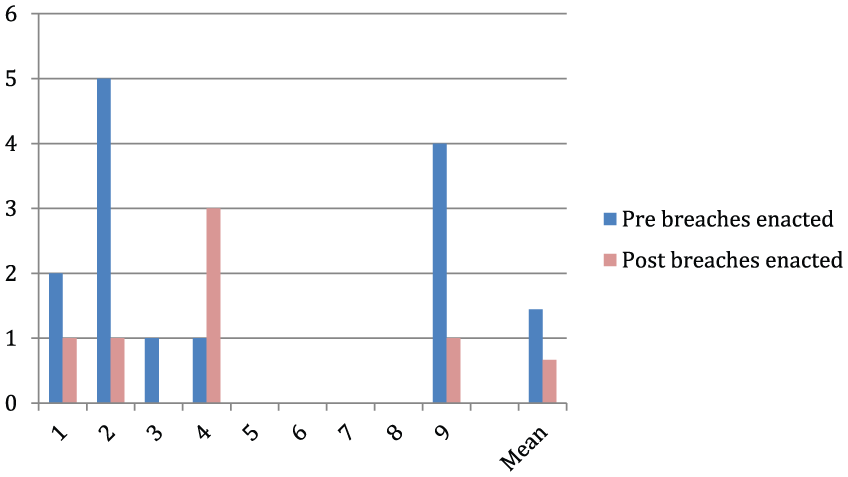
Enacted psychiatric treatment order breaches.

Psychiatric emergency presentations.

Body weight (kg).
Wilcoxon signed ranks test statistics.
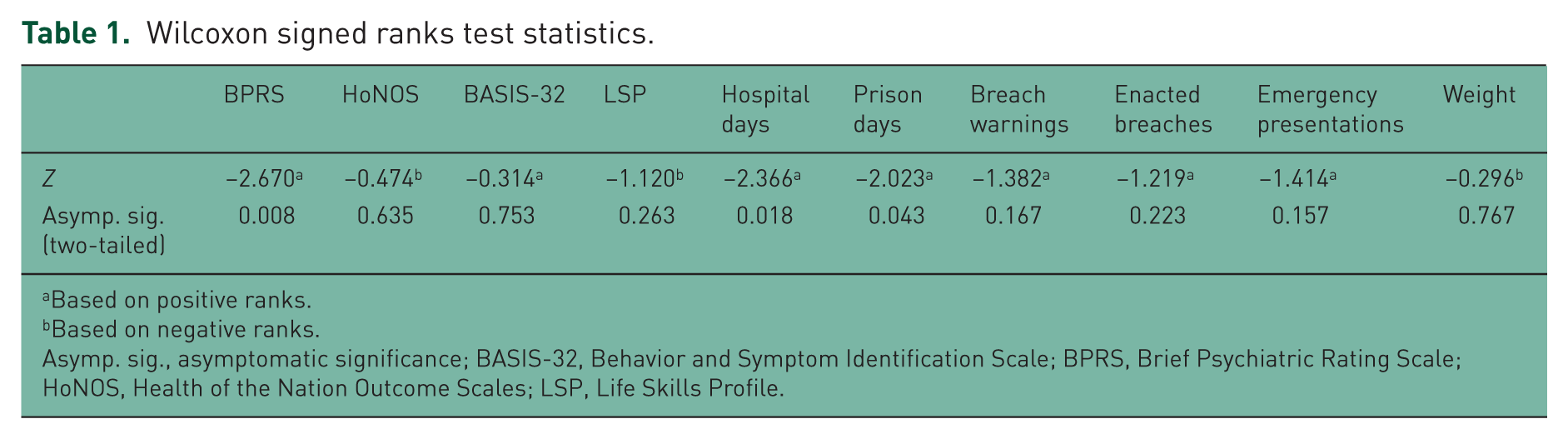
Based on positive ranks.
Based on negative ranks.
Asymp. sig., asymptomatic significance; BASIS-32, Behavior and Symptom Identification Scale; BPRS, Brief Psychiatric Rating Scale; HoNOS, Health of the Nation Outcome Scales; LSP, Life Skills Profile.
Metabolic monitoring was limited due to frequent patient refusal. Overall there was no significant increase in body weight. Five patients lost weight, four gained weight and the largest individual gain was 21 kg. One patient had insulin-dependent type 2 diabetes previously from clozapine; his blood sugars normalized on olanzapine depot and he no longer required insulin. Other patients (who agreed to monitoring) had no significant rise in blood glucose. There were three incidents of PIS, two in the same patient. The development of PIS did not necessitate cessation of olanzapine depot or changes to treatment regimes. No patient had side effects necessitating cessation of olanzapine depot.
Postinjection monitoring occurred as recommended by Australian regulatory authorities, with monitoring for alertness every 30 min for 2 h after each injection. Our clinicians endeavoured to engage patients in therapeutic activities and interventions during the monitoring period, such as psychiatric appointments, clinical psychology sessions, case manager reviews and social worker assistance for housing or disability support pension applications.
Discussion
We report our experience of olanzapine depot in a community setting with patients with schizophrenia spectrum disorder and significant past violence. All nine patients treated with olanzapine depot showed clinical improvement in psychotic symptoms, with lessened inpatient admission days and reduced prison days during 12 months on olanzapine depot.
Though a small sample, to our knowledge, our study is the first to demonstrate the utility of olanzapine depot as an effective community treatment for patients with schizophrenia and significant past violence. It adds to a very limited literature on the use of olanzapine depot in forensic psychiatric populations, with only one previous study that demonstrated benefits with olanzapine depot in a small inpatient sample in a UK high-security hospital [Baruch et al. 2014]. Wider evidence for the efficacy of olanzapine depot is promising, with studies showing equivalent effectiveness to oral olanzapine for acute schizophrenia [Lauriello et al. 2008] and maintenance treatment [Kane et al. 2010], though the cost effectiveness of olanzapine depot compared with other atypical and typical depots is mixed [Achilla and McCrone, 2013].
Our study was notable in that all patients on olanzapine depot for 12 months had clinically significant improvement in psychotic symptoms, which translated to real-world benefits of reduced violence and hostility, verified by significantly reduced psychiatric admission days and reduced days incarcerated in prison. In the majority of our sample, index offences were causally related to active psychotic symptoms. Additionally two-thirds were treatment resistant before olanzapine depot. As Baruch and colleagues [Baruch et al. 2014] reasoned, it is likely that the violence reduction from olanzapine depot was mediated by amelioration of psychotic symptoms, however we cannot exclude a specific antiaggressive effect with olanzapine depot, similar to clozapine [Baruch et al. 2014].
The study’s strengths included a quantitative paired 12-month comparison prior to and following olanzapine depot initiation. Olanzapine depot was maintained for 12 months throughout the entire sample. There were several limitations: a small sample size, retrospective analysis, no female patients and no control group, thus psychiatric management by a dedicated community forensic team, nursing care and psychological therapies could not be excluded as confounders for the improvements measured. However, research evaluating pharmacological treatments for forensic patients is extremely difficult. Patients enrolled in randomized clinical trials are not representative of forensic patients with substantial violence risk who are difficult to manage [Volavka et al. 2004]. This study adds to a barren research field.
Possible antiaggressive effects of olanzapine depot are of immense interest in forensic psychiatry. Treatment noncompliance involving forensic patients in the community poses high levels of violence risk, thus oral antipsychotics are not always a viable option, despite even clozapine’s antiaggressive effects [Baruch et al. 2014]. Importantly, both forensic psychiatric treatment adherence and medication response have been shown to ameliorate future violence risk [Douglas et al. 2013]. Our study demonstrates that for community forensic patients, olanzapine depot addresses both these clinical risk aspects, with promising indications of violence reduction. This has substantial implications for the improved management of forensic patients in the community.
Footnotes
Acknowledgements
Thanks are due to the ACT community forensic mental health team for their commitment and dedication to improving patient care.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest statement
Dr Kasinathan has received honoraria for speaking at educational meetings sponsored by AstraZeneca, Eli Lilly and Janssen. The authors declare no conflict of interest in preparing this article.
