Abstract
Background:
Even when patients experience remission with antidepressants, many continue to report anger attacks and excessive irritability despite continued treatment. Iloperidone antagonizes 5-HT-2a, D2, and alpha-1 receptors, which can have anti-aggressive effects. We examined iloperidone’s safety and efficacy as an augmentation agent in outpatients with partially remitted major depressive disorder (MDD) with residual symptoms of anger and irritability.
Methods:
A total of 13 outpatients with partially remitted MDD [currently treated with selective serotonin reuptake inhibitors (SSRIs)] received four weeks of iloperidone or placebo, followed by one week of washout. Patients were then crossed over to the other treatment arm for 4 weeks. Treatment arms were randomized and double blind; and two sites were used for the study. Analyses compared treatment response using the Symptom Questionnaire (SQ) Anger/Hostility Subscale as the primary outcome measure.
Results:
There was no significant differential effect of iloperidone × weeks on the SQ Anger/Hostility Subscore over the course of the study, compared with placebo × weeks, regardless of administration order (p = 0.77).
Conclusions:
Iloperidone did not significantly outperform placebo on measures of anger or irritability in patients with partially remitted MDD and residual anger/irritability.
Introduction
Major Depressive Disorder (MDD) is a common and highly disabling disorder [Smith, 2014]. Despite the availability of various treatment modalities, it has been estimated that the rates of remission in efficacy trials with the selective serotonin reuptake inhibitor (SSRI)-type antidepressant are only between 30% and 40% [Gaynes et al. 2009, Thase et al. 2010]. Up to 46% of depressed patients show only partial or no response after a single antidepressant trial [Fava et al. 1996], with irritability as a common residual symptom. Interestingly [after bipolar disorders were excluded in the National Comorbidity Survey Replication (a national US adult household survey)], irritability during depressive episodes was reported by half of respondents with a lifetime history of DSM–IV MDD. Furthermore, the Diagnostic and Statistical Manual of Mental Disorders, 5th Edition (DSM–5) classifies irritability as a diagnostic symptom of MDD in children and adolescents (but not adults) [American Psychiatric Association, 2013]. Irritability in MDD was associated with early age of onset, lifetime persistence, comorbidity with anxiety and impulse-control disorders, fatigue and self-reproach during episodes, and elevated rates of disability [Fava et al. 2010].
Similarly, anger attacks are sudden intense spells of anger that are accompanied by irritability and symptoms of autonomic activation (such as tachycardia, flushing, sweating, and tightness of the chest). Anger attacks are reported by 30–40% of depressed patients, and disappear in up to 71% of those treated with antidepressant drugs (such as fluoxetine, sertraline, or imipramine) [Fava et al. 1999]. The presence of anger attacks in MDD has been associated with higher depression measures scores, as well as decreased serotonergic neurotransmission (as suggested by a blunted prolactin response to fenfluramine compared with MDD alone) [Fava et al. 2000], and decreased striatal dopamine D1 binding [as measured using positron emission tomography (PET)] [Dougherty et al. 2006]. A substantial proportion of patients who respond to SSRIs or serotonin norepinephrine reuptake inhibitors (SNRIs) continue to report anger attacks and excessive irritability despite continued treatment [Fava et al. 1999]. Several augmentation strategies are commonly used in clinical practice to address these residual symptoms (e.g. atypical antipsychotics), but without any published evidence of clinical efficacy on anger and irritability to date.
Iloperidone is an antipsychotic drug that has been approved by the US Food and Drug Administration (FDA) for the acute treatment of schizophrenia in adults in 2009 [Marino et al. 2010]; thus far, there have been no published studies on iloperidone’s efficacy among depressive disorder patients. However, iloperidone is an antagonist of serotonin 5-HT-2a receptors and dopamine D2 receptors [Citrome, 2009], and these pharmacological actions have been extensively shown to have anti-aggressive effects in specific clinical populations with pathological aggression (e.g. aggressive behaviors related to dementia, brain injury, mental retardation, and personality disorders) [Fava, 1997]. In addition, iloperidone is an norepinephrine alpha-1 adrenoreceptor antagonist; compared with the other atypical antipsychotics, it has the highest receptor affinity [Richelson et al. 2000]. Data suggest that the agitation and aggression experienced among patients with Alzheimer’s disease may be mediated with antipsychotics via alpha-1 receptor blockade [Wang et al. 2009]. A recent study in 3580 subjects with schizophrenia has shown a very robust effect of iloperidone on both the hostility and depression factors of the Positive and Negative Syndrome Scale (PANSS) [Citrome et al. 2011]. Thus, this study aimed to explore iloperidone’s safety and efficacy as an augmentation agent in outpatients with MDD (partially remitted) with residual symptoms of anger and irritability.
Methods and materials
Overall study design
This nine-week randomized, double blind, placebo-controlled, crossover study was conducted at the Depression Clinical and Research Program (DCRP) at Massachusetts General Hospital (MGH) and at the University of Alabama. MGH served as the coordinating site and the overall principal investigator of the study was Maurizio Fava. All patients underwent telephone and face-to-face screening to assess eligibility. All study patients met DSM–IV diagnostic criteria of MDD in partial remission (see below), with residual anger and irritability at screening. Eligible patients completed one visit per week for a total of nine study visits. Following the conclusion of the study, patients returned for two follow-up visits.
The study design was selected given the half-lives of iloperidone and its active metabolites, P88 and P95 (18, 26, and 23 hours, respectively). These half-lives would allow for the 1 week of washout in the crossover design.
Medication treatment arms
Eligible patients were randomly assigned by the research pharmacy, in a ratio of 1:1, to one of the following two treatment groups: (1) Group 1 received iloperidone at doses of 1–8 mg, orally, once nightly for the first 4 weeks, followed by one washout week of single (patient)-blinded placebo and then a further 4 weeks of placebo; (2) Group 2 received placebo for the first 4 weeks, followed by one washout week of single-blinded placebo and then iloperidone at doses of 1–8 mg, orally, once nightly for a further 4 weeks. Phase I was defined as the first set of four weeks of receiving iloperidone (versus placebo; weeks 1 to 4); phase II was defined as the second set of four weeks of receiving iloperidone (versus placebo; weeks 5 to 9).
For both treatment arms, the dosing regimen was fixed, such that at the baseline visit (week 0, or day 0), patients began a gradual titration from 1 mg of iloperidone (or placebo) up to 6 mg during the first week of treatment. This dosing regimen of gradual titration was chosen to optimize the tolerance of the medication and to decrease the risks of adverse events. At the end of the first week (week 1, or day 7), depending on drug tolerability and efficacy, the study physician had the options of: (1) increasing the dose to 8 mg; (2) decreasing the dose to 4 mg; or (3) remaining at 6 mg. The study drug was dispensed in blister packs that included a 1-week supply; two additional capsules were provided to account for scheduling variability.
Study patients
Between August 2013 and November 2014, a total of 34 adult patients (18–65 years of age) completed an in-person psychiatric evaluation and screen to assess eligibility. All were screened via telephone, prior to the screening visit. At the screening visit, informed consent was discussed (i.e. study aims, risks, benefits, and potential adverse events) and obtained prior to entering the study, in accordance to the Declaration of Helsinki and approved by the Institutional Review Board (IRB) at each institution.
Of the 34 screened patients, 20 were entered into the study. These were randomized and entered into one of the 2 groups. A total of 13 patients completed the study as scheduled. Of the randomized patients that did not complete the entire trial as scheduled: one patient never crossed over from the first phase and was given iloperidone throughout the entire study, one patient had a pharmacy medication administration error during the first week, and one patient who was scheduled to receive placebo in phase I was mistakenly given iloperidone during week 1 and then placebo in the following 3 weeks. Those patients were therefore removed from the primary analysis. The remaining five randomized patients were withdrawn or discontinued from the study for the following reasons: one patient was discontinued by the study staff secondary to nonadherance; two withdrew due to adverse events; one was discontinued by the study staff due to an alcohol relapse; and one was lost to follow up.
Inclusion and exclusion criteria
All patients met criteria for current MDD in partial remission, based on the Structured Clinical Interview for DSM IV–Axis I disorders (SCID I/P) [First et al. 1997], as administered by a study clinician and confirmed by clinical interview. Other inclusion criteria were: (1) a total score (at screening and baseline) greater than 8 on both the 17-item Hamilton Depression Rating Scale (HDRS17; indicating partial response) [Hamilton, 1960], and the Anger/Hostility Scale of the Symptom Questionnaire [Kellner, 1987]; and (2) current treatment with an SSRI, SNRI, or bupropion [a norepinephrine-dopamine reuptake inhibitor (NDRI)] at a stable dose (⩾4 weeks) and duration (⩾3 months) according to the MGH Antidepressant Treatment History Questionnaire (ATRQ) [Chandler et al. 2010]. All other antidepressants, antipsychotics, and augmentation agents were exclusionary, though benzodiazepines and hypnotics were permitted if taken at stable doses.
Exclusion criteria included: (1) history of psychosis or bipolar affective disorder; (2) substance abuse/dependence within the past 3 months; (3) antisocial personality disorder; (4) dementia; (5) current risk of suicide or homicide; (6) pregnant or breastfeeding women; (7) unstable cardiovascular illness, including electrocardiogram (ECG) abnormalities (i.e. QTc interval >450 ms for males or >470 ms for females), cardiac arrhythmia (any), with or without symptomatic uncontrolled hyper- or hypotension; (8) history of epilepsy, progressive neurologic disease, head trauma, or neuroleptic malignant syndrome history; (9) clinically significant gastrointestinal, hepatic, or kidney disease that could potentially impair metabolism and excretion of the study medication; (10) history of priapism that required surgery; or (11) history of electroconvulsive therapy.
Study assessments
After passing the initial phone screen, patients were scheduled to come in to the clinic for a full screening visit. At the screening visit, demographic information, relevant medical history, psychiatric and medication history, physical exam, height, weight, vital signs, urine pregnancy and drug tests, the ATRQ, and SCID I/P were completed by a study clinician. Routine fasting laboratory tests (hematology, chemistry, urinalysis) were completed at screening, visit 6 (week 4), and visit 11 (week 9); an EKG was performed at screening, visit 3 (week 1), and visit 8 (week 6).
At all weekly study visits, depression severity was assessed with the 28-item Hamilton Depression Rating Scale (HDRS28) [Hamilton, 1960]. The Clinical Global Impression rating scales for Improvement (CGI–I) and Severity (CGI–S) [Guy, 1976] were utilized to assess the overall change in depressive symptoms compared with baseline, and illness severity at the time of assessment, respectively. The Symptom Questionnaire (SQ) [Kellner, 1987], a self-rated measure of emotional states, was administered at each visit. The SQ includes four symptom subscales (depression, anxiety, anger/hostility, and somatic) and four wellbeing subscales (contentment, relaxed, friendly, and somatic well-being).
Patients also completed the self-rated MGH Cognitive and Physical Functioning Questionnaire (CPFQ) an assessment of cognitive and physical functioning [Fava et al. 2006, 2009] and the self-rated Concise Health Risk Tracking – Patient-Rated Module (CHRT), a measurement of hopelessness and suicidality [Trivedi et al. 2011]. The Anger Attacks Questionnaire (AAQ), and the Consumptive Habits Form were completed at the screening, visit 1 (week 0), visit 7 (week 5), and visit 11 (week 9). Concomitant medications and adverse reactions were assessed weekly using frequency of reported events.
Statistical methods
The primary outcome measure for this study was the difference in scores on the Anger/Hostility Scale of the SQ during iloperidone treatment, compared with placebo. Secondary outcome measures were the change in HDRS, depression subscale of the SQ, anxiety subscale of the SQ, and CPFQ. Adverse events were compared across placebo versus iloperidone groups using frequencies. General linear models were used to compare placebo with iloperidone treatment group responses on measures of anger, anxiety, depression, and quality of life. Antidepressant response was defined as a greater than or equal to 50% decrease in the HDRS17 score from baseline; antidepressant remission was defined as less than 7 on the HDRS17 [Nierenberg et al. 2001]. A standard intention-to-treat/last-observation-carried forward analysis approach was used. All tests were two-tailed, α = 0.05.
Results
Demographics
Of the 13 patients that completed the study 3 (23%) were male and 10 (77%) were female. The average age of the sample was 46.1 ± 9.7 years old. A total of 11 patients (84.6%) were Caucasian and 2 (15.4%) were African–American. Highest level of education was as follows: one (7.7%) did not graduate high school, one (7.7%) graduated high school (or equivalent), four (30.8%) completed some college, one (7.7%) graduated from a two-year college, two (15.4%) graduated from a four-year college, one (7.7%) completed some professional/graduate school work, and three (23.1%) were professional/graduate school graduates. Regarding marital status, data were obtained from 11 of the 13 randomized patients: three (23.1%) were never married, four (30.8%) were married or living with someone, one (7.7%) was separated, and three (23.1%) were divorced.
Of the 13 randomized patients, 9 (69.2%) were maintained on an SSRI, two (15.4%) were on an SNRI, one (7.7%) was on bupropion at the time of randomization, and one (7.7%) patient was on both an SSRI and an SNRI.
Randomization
Of the 13 patients who completed the study, 5 were randomized to the iloperidone–placebo sequence (Group 1), and 8 were randomized to the placebo–iloperidone sequence (Group 2).
Primary outcome measure
Primary outcome data are summarized in Table 1. There was no significant differential effect of iloperidone × weeks on the SQ Anger/Hostility Scale over the course of the study, compared with placebo × weeks, regardless of administration order (Figure 1) (p = 0.77). A significant drug × weeks × order effect was found (p = 0.002), in other words, regardless of whether patients received placebo or iloperidone first, the first treatment they received produced a greater decrease in scores over the weeks compared with the following treatment – suggestive of a significant carry-over effect.
Adverse events frequency table.
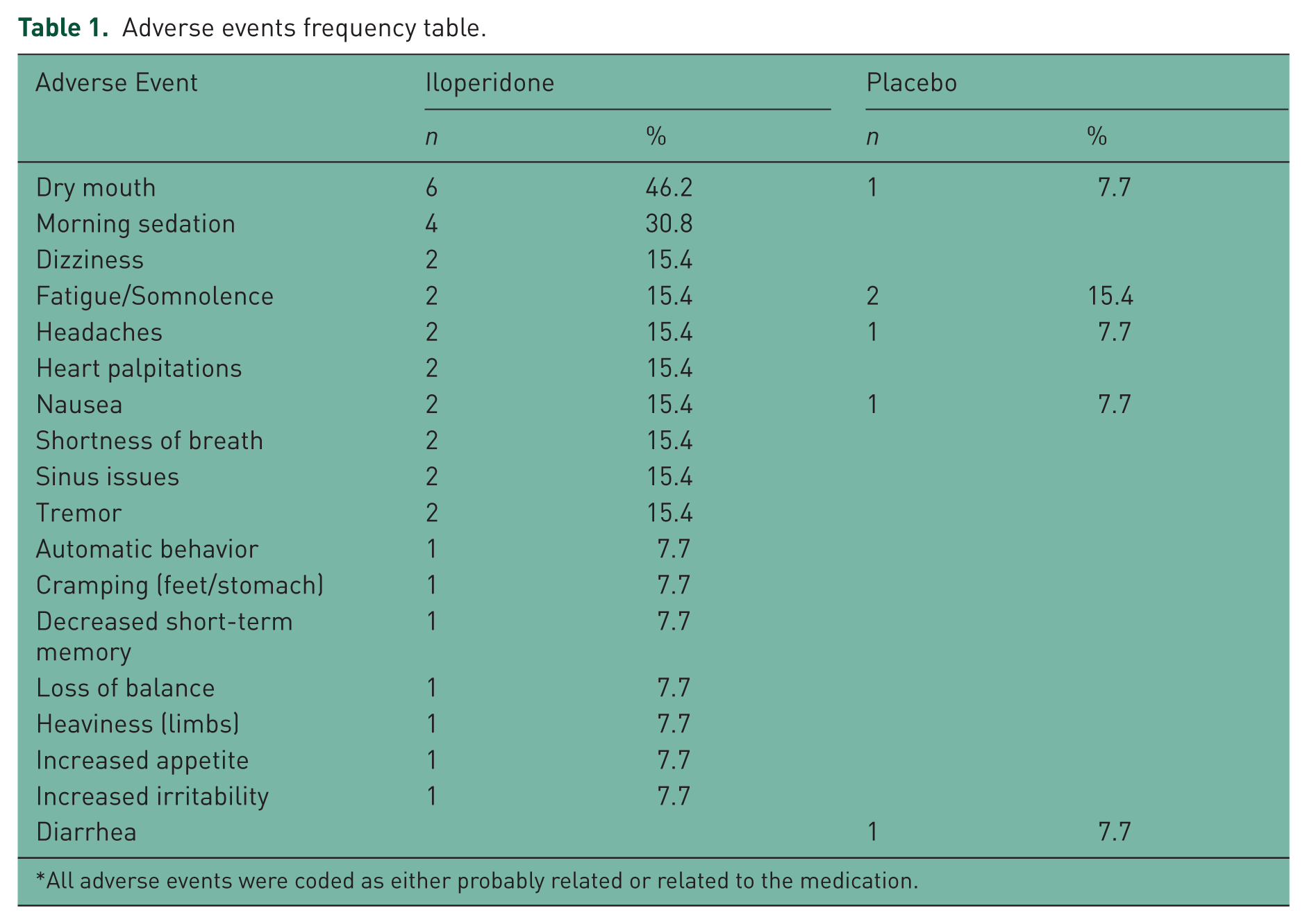
All adverse events were coded as either probably related or related to the medication.

SQ Anger Score change over weeks, iloperidone versus placebo.
Secondary outcome measures
With regard to secondary outcome measures, iloperidone led to no significant changes compared with placebo over the course of the study on the HDRS (p = 0.82). For HDRS, there was a significant effect of weeks (p < 0.01), meaning that scores decreased over time, regardless of whether iloperidone or placebo were given. There was also a significant effect of drug × order (p = 0.08), meaning that the efficacy of drug depended on which order they were administered; whichever was administered first was the most effective. With regards to overall depression response and remission rates, two (40%) out of the five patients randomized to Group 1 (iloperidone–placebo sequence) responded and one (20%) remitted. Four (50%) of the eight patients randomized to Group 2 (placebo–iloperidone sequence) responded and three (37.5%) remitted.
Similar to the HDRS findings, there was a significant drug × weeks × order effect on the SQ anxiety subscale total score (p = 0.03), but insignificant drug × weeks effect (p = 0.77). For the SQ depression subscale total score, there was a significant drug × weeks × order effects (p = 0.04), but no significant drug × weeks effect (p = 0.85). For the CPFQ total score, there was a significant drug × order effect (p = 0.06), but an insignificant drug × weeks effect (p = 0.75).
With respect to tolerability, Table 2 shows the spontaneously reported adverse events (AEs) while on drug or placebo. Dry mouth, morning sedation, dizziness, fatigue/somnolence, headaches, heart palpitations, nausea, shortness of breath, sinus issues, and tremor were the most commonly reported AEs on drug, occurring in at least 10% of the treated patients; all but one of the these AEs (fatigue/somnolence) were more frequently reported than on placebo. Of the two patients that withdrew from the study after randomization, one patient discontinued due to palpitations and sedation, and the other patient discontinued due to several side effects (dry mouth, sinus pressure, decreased short-term memory, increased urinary frequency, headache, fatigue, and increased irritability.
Summary of mean change of rating scale scores, iloperidone versus placebo.

Abbreviations: BL, baseline; SD, standard deviation; SQ, Symptom Questionnaire; HDRS, Hamilton Depression Rating Scale; CPFQ, Massachusetts General Hospital Cognitive and Physical Functioning Questionnaire.
Discussion
Contrary to our expectation, this pilot crossover study did not detect a significant advantage of iloperidone over placebo in the treatment of residual anger, irritability, anxiety, and depression in patients with incomplete response to antidepressant therapy in both the completer (n = 13) and in our samples. These findings do not support the hypothesis that iloperidone’s pharmacological action – especially on 5-HT-2a receptors and norepinephrine alpha-1 adrenoreceptors – would be efficacious as antidepressant augmentation among outpatients with MDD in partial remission who are experiencing residual symptoms of anger and irritability. This is also in contrast to the findings of a recent study in 3580 patients with schizophrenia, showing a very robust effect of iloperidone on both the hostility and depression factors of the PANSS [Citrome et al. 2011].
One possible explanation for the discrepancies between our findings and our hypotheses may be that iloperidone is not successful at treating residual symptoms in patients with depression (as opposed to those with a primary psychotic disorder). Indeed, psychosis (current or lifetime) was an exclusionary criterion for this current study of unipolar depressed patients. Perhaps the significant antihostility effects of iloperidone that were observed in the psychotic disorders sample [Citrome et al. 2011] actually required the presence of psychotic symptoms in order for iloperidone to take effect in treating residual symptoms of hostility. Indeed, the dissociative effects of ketamine have been hypothesized to mediate, and perhaps be necessary for, the antidepressant effects observed in patients with treatment-resistant depression [Luckenbaugh et al. 2014]. Similarly, perhaps the symptoms that define psychotic disorders are necessary for iloperidone’s antihostility properties in patients with schizophrenia.
The main limitation of our study is the very small sample size. We cannot rule out the possibility that significant benefits could have been detected in a larger group of depressed patients. Further, larger studies are needed to clarify this. The other significant issue of this study relates to the carry-over effect, which was significant and may have limited our ability to detect a therapeutic effect of iloperidone. Regardless of whether patients received iloperidone or placebo during the first four weeks of the study, their symptoms improved. Therefore, it made it difficult to analyze the second half of the study for meaningful effects. Furthermore, the placebo response rate (50%) was numerically greater than the iloperidone response rate (40%) in the primary analysis, though this is likely a function of ordering effects, since more patients received placebo first.
In addition, patients on iloperidone reported more adverse events, including dry mouth, morning sedation, dizziness, headaches, heart palpitations, nausea, shortness of breath, sinus issues, and tremor. The only symptom reported more often in the placebo group was fatigue/somnolence. Given iloperidone’s mechanism of action and known side effects, the adverse events in our sample were not surprising. Regardless, iloperidone clearly had more side effects compared with placebo, though no serious adverse events were reported.
In conclusion, iloperidone did not significantly outperform placebo on measures of anger, irritability, anxiety, and depression in patients with partially remitted MDD and residual anger. In addition, patients on iloperidone reported more adverse events. Results from this study do not support the use of iloperidone augmentation for residual symptoms of depression, anxiety, and anger in partially remitted patients with depression and residual anger. Larger studies are necessary to confirm or refute this finding.
Footnotes
Funding
This research was funded by Novartis Pharmaceuticals. The unique ClinicalTrials.gov Identifier for this study is NCT01464229.
