Abstract
Despite their widespread use, long-acting injectable (LAI) antipsychotics (APs) are often regarded with some negativity because of the assumption of punishment, control and insufficient evolution towards psychosocial development of patients. However, LAI APs have proved effective in schizophrenia and other severe psychotic disorders because they assure stable blood levels, leading to a reduction of the risk of relapse. Therapeutic opportunities have also arisen after introduction of newer, second-generation LAI APs in recent years. Newer LAI APs are more readily dosed optimally, may be better tolerated and are better suited to integrated rehabilitation programmes. This review outlines the older and newer LAI APs available for the treatment of schizophrenia, with considerations of past and present pharmacological and therapeutic issues. Traditional, evidence-based approaches to systematic reviews and randomized clinical trials are of limited utility in this area so this paper’s blending of experimental trials with observational research is particularly appropriate and effective.
Keywords
Historical background of long-acting injectable antipsychotics
Soon after the introduction of antipsychotics (APs) in the 1950s, poor adherence to oral formulations was found to be a critical issue. This led to the development in 1966 of the first long-acting injectable (LAI) AP fluphenazine enanthate, and fluphenazine decanoate some 18 months later, to reduce the incidence of side effects of the former [Johnson, 2009]. Haloperidol decanoate became available in Europe in 1981 and in the USA in 1986 [Knudsen, 1985]. Clinical trial results showed a dramatic reduction in the morbidity of schizophrenia [Gottfries and Green, 1974; Johnson, 2009]. However, the concept of LAIs for schizophrenia was not initially received warmly by the medical profession for fears of increased side effects, lack of efficacy, and the fact this was seen as an attempt by psychiatrists to impose a treatment upon patients without due regard to their feelings or rights [Johnson, 2009] as well as the potential for medicolegal problems [Glazer and Kane, 1992]. However, with subsequent surveys and trials suggesting their benefits [Rifkin et al. 1977; Schooler et al. 1980; Hogarty et al. 1979], LAIs became more widely adopted.
The introduction of the oral second-generation APs (SGAs) brought claims of better tolerance and less severe side effects, even though their use may be hindered by metabolic syndrome [Meyer and Stahl, 2009]. They also have potential to prevent or reverse accelerated frontotemporal cortical grey matter decline, and to provide a greater degree of neuroprotection than first generation APs (FGAs) [Keefe et al. 2004; Robinson et al. 2006]. The introduction of SGAs led to a decline in the use of LAI FGAs [Patel et al. 2003; Patel and David, 2005]. However, it soon became clear that atypical characteristics did not bring better adherence rates with oral SGAs. The recent introduction of LAI SGAs allows psychiatrists once again to prescribe LAIs without losing any of the potential advantages of the SGAs by using this form of delivery [Johnson, 2009].
Bioavailability and dosing of LAI APs
While oral APs are converted to active and inactive metabolites and only a relatively small portion reaches the brain [Jann et al. 1985], LAIs bypass the initial deactivating process by avoiding first-pass metabolism in the liver [Dencker, 1984; Marder et al. 1989]. In this way, a higher proportion of the drug is available centrally [McEvoy, 2006], which arguably can allow the use of the lowest effective dose [Gerlach, 1995]. As a rule, calculation of total doses for oral versus LAI APs over time will usually show lower doses for the LAIs. That said, appropriate prescribing and dosing of LAIs is complicated by their long half-lives, delayed release (up to 28 days in the case of risperidone) and risk of postinjection delirium/sedation syndrome (olanzapine pamoate) [Novakovic et al. 2013]. Lack of clear dose–response data lead clinicians to approximate dosing in their clinical practice; even where fixed-dose studies have established ‘dose-response’ curves, there is debate regarding the clinical value of doses at the higher and lower end of the dose range [Taylor, 2009]. The pharmacokinetic and clinical characteristics of most commonly used LAI APs are described in Table 1.
Pharmacokinetic and clinical features of most commonly used first-generation LAI antipsychotics and new second-generation LAI antipsychotics.
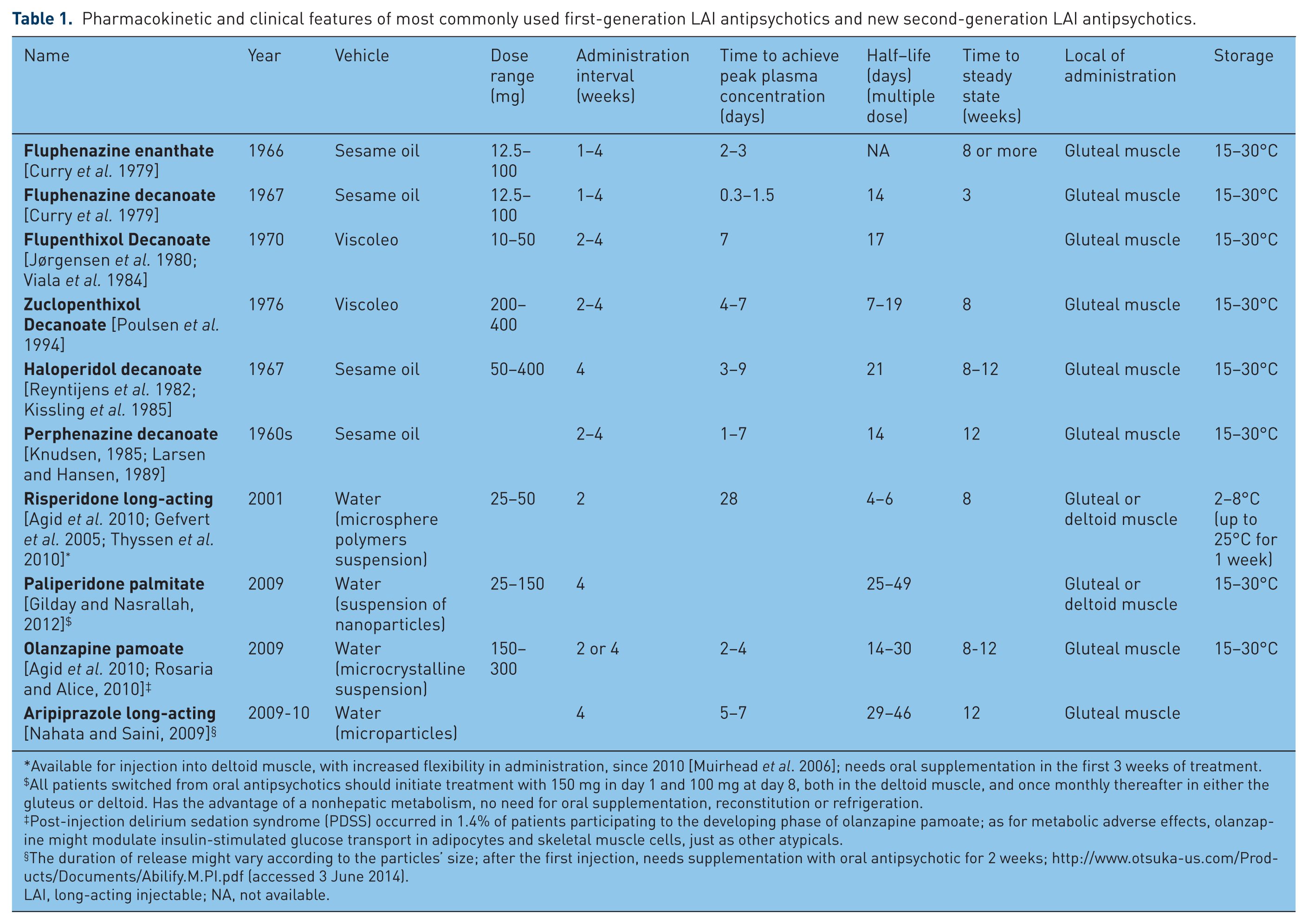
Available for injection into deltoid muscle, with increased flexibility in administration, since 2010 [Muirhead et al. 2006]; needs oral supplementation in the first 3 weeks of treatment.
All patients switched from oral antipsychotics should initiate treatment with 150 mg in day 1 and 100 mg at day 8, both in the deltoid muscle, and once monthly thereafter in either the gluteus or deltoid. Has the advantage of a nonhepatic metabolism, no need for oral supplementation, reconstitution or refrigeration.
Post-injection delirium sedation syndrome (PDSS) occurred in 1.4% of patients participating to the developing phase of olanzapine pamoate; as for metabolic adverse effects, olanzapine might modulate insulin-stimulated glucose transport in adipocytes and skeletal muscle cells, just as other atypicals.
The duration of release might vary according to the particles’ size; after the first injection, needs supplementation with oral antipsychotic for 2 weeks; http://www.otsuka-us.com/Products/Documents/Abilify.M.PI.pdf (accessed 3 June 2014).
LAI, long-acting injectable; NA, not available.
Attitudes of clinicians and patients to LAI APs
So far, few reports have addressed this question and the mixed quality of surveys makes generalizations problematic. A negative attitude amongst clinicians regarding LAI APs is common [Heres et al. 2006; Patel et al. 2010; Jaeger and Rossler, 2010], especially when considering recent onset patients [Heres et al. 2006; Kirschner et al. 2013]. Recently, a survey of 891 European psychiatrists and nurses revealed that 96% preferred LAI medications to oral treatment for patients with chronic schizophrenia, whereas only 40% preferred them for first-episode patients [Geerts et al. 2013]. The overestimated compliance by psychiatrists also limits their use [Heres et al. 2008; Samalin et al. 2013].
Regarding patients themselves, the majority have a negative impression of LAIs and perceive them as more coercive [Patel et al. 2010; Jaeger and Rossler, 2010], and patients are thought to be more negative towards LAIs than psychiatrists and relatives [Perkins, 2002; Patel et al. 2010].
However, patients are often not fully informed about LAIs by their psychiatrist [Patel et al. 2010; Potkin et al. 2013], and treatment decisions are usually made without patient or caregiver input, with LAIs not being discussed in about 50% of patients taking oral AP [Potkin et al. 2013]. However, when offered, more LAI-naïve patients express neutral (47%) or favourable (16%), rather than unfavourable (37%) responses [Potkin et al. 2013]. Moreover, patients who have tried LAIs prefer this treatment over oral APs [Patel and David, 2005; Heres et al. 2007; Patel et al. 2009; Waddell and Taylor, 2009; Caroli et al. 2011], referring that they ‘feel better’, have a more ‘normal life’ and find injections ‘easier to remember’ [Olfson et al. 1999; Svedberg et al. 2003; Caroli et al. 2011]. Regarding convenience, both patients and staff show a positive attitude and satisfaction towards LAIs [Walburn et al. 2001; Waddell and Taylor, 2009].
In subjects with a first episode of psychosis (FEP), psychiatrists frequently presume that patients will not accept LAI APs [Kirschner et al. 2013] and few patients are offered this treatment choice [Kirschner et al. 2013]. Although some FEP patients perceive LAI as more coercive and stigmatizing [Patel et al. 2010], others have a favourable perception, just like their relatives and primary caregivers [De la Gándara et al. 2009].
Finally, psychiatrists’ prescription of LAI APs may also be influenced by other factors; a study by Heres and colleagues [Heres et al. 2008] and a replicated French study from Samalin and colleagues [Samalin et al. 2013] found two clusters of factors preferentially taken into account by psychiatrists for the prescription of a depot form. Cluster I corresponded to patients with a past history of relapse and poor compliance with oral forms; however, there was a group of patients (cluster II), which corresponded to patients with a high level of insight and a high level of therapeutic alliance, factors not traditionally associated with the use of LAI APs. Another interesting point was the preferential use of LAI APs for patients with behavioural disorders to curb impulsiveness and aggressiveness or dangerousness [Llorca et al. 2013].
In short, preference for LAIs is mainly dependent on both the psychiatrists’ and the patients’ experience with such compounds. Educating staff, patients and families can help address prejudice and stigma towards LAI APs, increase familiarity and ultimately increase preference for this type of medication [Waddell and Taylor, 2009].
These attitudes determine, at least in part, the prevalence of use of LAIs. There is a lack of robust data on the frequency of use of LAI APs in schizophrenia patients [Adams et al. 2001], making it difficult to detail trends in use [Kissling, 1991]. LAI use varies greatly between different regions of the world and over the years [Kissling, 1991; Rothbard et al. 2003; Patel and David, 2005].
Overall, the use of LAIs diminished dramatically after the introduction of oral SGAs in the 1990s [Patel et al. 2003; Patel and David, 2005], and nowadays the proportion of patients prescribed LAI ranges from 10 to 50%
Potential advantages and disadvantages of LAI APs
The pros and cons associated with the use of LAI formulations are summarized in Table 2.
Summary of potential advantages and disadvantages of long-acting injectable antipsychotics as compared to oral antipsychotics.

LAI APs offer a number of advantages compared with oral medication, including not having to remember to take drugs daily, reducing the risk of unintentional or deliberate overdose, and transparency of adherence [Gerlach, 1995; Remington and Adams, 1995], allowing healthcare professionals to be alerted and to intervene appropriately if patients fail to take their medication [NICE, 2009]. When patients stop medication, plasma levels decrease more slowly than with oral formulations, giving time for the healthcare professionals to intervene at an early stage and lowering the probability of rebound symptoms or of a relapse occurring as rapidly. Moreover, if a patient suffers a relapse, despite receiving regular LAI treatment, it is then clear that compliance is not the reason [Remington and Adams, 1995].
Other benefits include more consistent bioavailability [Remington and Adams, 1995] and more predictable correlation between dosage and plasma levels [Rocca et al. 2013], reduced peak-trough plasma levels [McEvoy, 2006], improved patient outcomes [Peuskens et al. 2010a; Kaplan et al. 2013], improved patient and physician satisfaction [Peuskens et al. 2010b], lower relapse rates [Emsley et al. 2008a, 2008b; Gaebel et al. 2010] than oral therapy and more regular contact with the mental healthcare team [Pandarakalam, 2003].
The main disadvantage of LAIs relates to the slow dose titration and the long time required to achieve steady state levels [Agid et al. 2010]. This disadvantage is most evident in acutely ill individuals, in whom there would be a need for rapid dose titration within days of initiating treatment. In addition, during this time oral AP supplementation may be necessary, adding to the complexity of the titration process. It is also more difficult to make sensible dose adjustments because attainment of steady state plasma levels may take more than 2 months after a dose change. For these reasons, the initiation of LAIs has generally been confined to those periods when a patient is at least partially stabilized on their existing treatment. With some newer LAIs, such as paliperidone palmitate, rapid therapeutic levels can be attained by means of a ‘loading dose regimen’, allowing for its use in patients with moderate symptoms, and bringing about a rapid onset of action with no need for oral supplementation [Gilday and Nasrallah, 2012].
Severe side effects, such as tardive dystonia, dyskinesia and neuroleptic malignant syndrome (NMS) [Glazer, 1984; Kane et al. 1998], and postinjection syndrome with olanzapine [Novakovic et al. 2013], may occur; nevertheless, there are rare and there seems to be no increased risk for EPS liability [Glazer, 1984; Marder et al. 1984; Glazer and Kane, 1992; Adams et al. 2001], tardive dyskinesia [Adams et al. 2001], or NMS in LAI treatment.
Pain at the injection site can occur, and leakage of small amounts of the drug into the subcutaneous tissue and/or the skin may as well cause irritation and lesions. The lack of LAI formulations for all oral APs may also limit the use of some LAIs in clinical practice [Heres et al. 2008; Samalin et al. 2013]. Finally, risperidone LAI (RLAI) needs refrigeration, which may be cumbersome.
The importance of ensuring treatment continuity in schizophrenia
The relapsing, lifelong course of schizophrenia, along with the potentially progressive nature of the disease, justifies the continuity of treatment in the majority of patients. Today, the presence of structural and functional abnormalities in the brain of these patients has been well documented [Agarwal et al. 2010; Brugger et al. 2011; Nickl-Jockschat et al. 2011]. Although the interpretation of these data is often challenging and much remains to be understood, the observed brain abnormalities seem to progress with illness [Agarwal et al. 2010; Nickl-Jockschat et al. 2011].
Studies in animal models suggest that chronic exposure to APs may contribute to decreases in brain tissue volume associated with the disease [Ho et al. 2011], and a significant correlation between greater duration and intensity of AP treatment and reduced brain tissue volume has also been reported in patients [Ho et al. 2011]. However, a study carried out in recently diagnosed patients showed that prolonged treatment with the SGA RLAI was associated with stable white matter volume in comparison with a decrease in volume observed in patients treated with the oral formulation of the same molecule [Bartzokis et al. 2012]. These data suggest that, by modifying adherence, RLAI may differentially impact myelination and account for the better long-term outcomes compared with oral risperidone. Thus, the risk–benefit balance of long-term treatment needs to be further assessed.
Finally, a factor leading to inadequate and/or premature treatment discontinuation is poor adherence to AP medication, and inadequate adherence to treatment is one of the major issues in the management of patients with schizophrenia.
The consequences of (in)adequate adherence to AP treatment
The definition of medication adherence or compliance varies in the literature but has recently been defined as ≥80% of medication taken over 12 months and/or <1 week missed medications (over 3 months) [Velligan et al. 2009]. Studies have shown nonadherence rates to oral medication in schizophrenia to be as high as 50% [Glazer, 2007]. Moreover, adherence behaviour changes and fluctuates over time in a dynamic fashion and should be considered as a core aspect of the illness [Weiden, 2007].
Poor adherence is a relevant risk factor for relapse in both chronic [Valenstein et al. 2002; Leucht and Heres, 2006] and recent-onset patients [Morken et al. 2008], and most hospital readmissions are because of some degree of nonadherence, even if is often unclear whether nonadherence is the cause or the consequence of the relapse [Kane and Garcia-Ribera, 2009].
Among pharmacologic interventions aimed to enhance adherence, expert consensus and recent guidelines have proposed switching to a LAI when lack of insight, comorbid substance use, persistent symptoms, logistic problems, lack of routines or lack of family/social support makes adherence to oral APs problematic [NICE, 2009; Kane and Garcia-Ribera, 2009; Velligan et al. 2010; Koola et al. 2012].
Better outcomes, as evidenced by an improvement in clinical symptoms and functioning, a reduction in the use of concomitant medication, the rate of hospitalization and the duration of hospital stay, have been shown to correlate with better adherence [Olivares et al. 2009], and adherent patients have significantly lower hospital costs than nonadherent patients [Gilmer et al. 2004; Dilla et al. 2013].
Better outcomes among adherent patients may be a result of the continuous availability of medication, but it may also be easier for patients with good functioning levels and better controlled symptoms to take their medication on a daily basis [Olivares et al. 2009]. Thus, adherence is a key factor for treatment continuity and, at the same time, uninterrupted treatment administration appears to favour good adherence [Altamura et al. 2012].
Forensic implications of LAI APs
Psychosis has been considered an important risk factor for violence, but recently the UK Prisoner Cohort Study revealed that schizophrenia was associated with violence only in the absence of treatment (odds ratio = 3.76) [Keers et al. 2014]. Untreated schizophrenia was associated with the emergence of persecutory delusions at follow-up, which were associated with violence; therefore, maintaining psychiatric treatment after release can substantially reduce violent recidivism among prisoners with schizophrenia [Keers et al. 2014].
LAI APs have been recommended in involuntary admission to hospital, with the goal of increasing treatment compliance among first episode schizophrenia (FES) patients [Stip et al. 2011], and of avoiding future deterioration [Vaughan et al. 2000; Muirhead et al. 2006]. Even if it appears paradoxical, this coercive step does not seem to alter the therapeutic alliance [Trudeau et al. 1999].
In this context, naturalistic studies comparing oral to LAI APs for schizophrenia patients on involuntary outpatient treatment have revealed better medication adherence, fewer crisis referrals, and fewer relapses in the LAI group [Vaughan et al. 2000; Swartz et al. 2001; Muirhead et al. 2006; Swartz et al. 2001], and that LAIs may be an important tool to improve outcomes in these specific clinical population [Zhornitsky and Stip, 2012].
There are no data to suggest that a physician incurs greater liability with one route of AP administration than with another [Glazer and Kane, 1992]. The essential medicolegal issue is the extent to which physicians meet the standard of care by which liability is measured, rather than whether oral, short-term injectable, or a LAI was prescribed [Glazer and Kane, 1992]. For medicolegal purposes, a meaningful interaction between the physician and the patient can be documented by including brief quotes from the patient and/or family. It is difficult to discuss the benefits and risks of AP treatment with patients whose condition frequently involves neurocognitive deficits/dysfunction [Brissos et al. 2011; Ekerholm et al. 2012]. Nevertheless, physicians should always discuss treatment options and inform patients that LAI treatment is among the best treatment approaches [Glazer and Kane, 1992].
Guidelines and recommendations for the treatment of schizophrenia with LAI APs
The first guidelines published in 1998, already recommended that LAI APs should be considered for ‘any patients with schizophrenia for whom long-term treatment is indicated’ [Kane et al. 1998]. Several guidelines recommend LAI AP medication only for patients with recurrent relapses related to partial or full nonadherence, or patients with persistent positive symptoms [Lehman et al. 2004; Canadian Psychiatric Association, 2005], although more recent guidelines have introduced subtle changes. For example, the Texas Medication Algorithm Project [Moore et al. 2007] recommends that the clinicians consider LAI APs in patients who are inadequately adherent ‘at any stage’. Similarly, guidance from the National Institute for Clinical Excellence in the UK states that clinicians should consider offering LAI APs to patients who would ‘prefer such treatment after an acute episode and where avoiding covert nonadherence to AP medication is a clinical priority’ within the treatment plan [NICE, 2009].
In fact, several authors propose that LAI APs should not be restricted to patients with adherence problems, but instead should be more widely prescribed [Altamura et al. 2012], and systematically offered to all patients through shared decision-making [Llorca et al. 2013].
Kane and Garcia-Ribera further suggested that ‘any patient for whom long-term treatment is indicated should be considered a candidate for long-acting injections’, and that even if patients refuse this option, it would be helpful to discuss it further to better understand the potential advantages [Kane and Garcia-Ribera, 2009].
Regarding FES, current guidelines have a conservative position, but recent evidence suggests that these perhaps need to be updated [Parellada et al. 2012]. In fact, given the importance of continuous treatment in the early phases of schizophrenia, LAI APs may also be a treatment option [Kim et al. 2012; Emsley et al. 2013], and in the context of a shared decision-making approach [Parellada et al. 2012; Kirschner et al. 2013], it could reduce the negative image and stigmatization attached to depots [Kirschner et al. 2013].
Long-acting injectable APs in the early phase of schizophrenia
Treatment of FES is particularly important to improve long-term outcomes, as most clinical and psychosocial deterioration with cognitive decline and progressive structural changes in brain volume [Agarwal et al. 2010; Brugger et al. 2011; Nickl-Jockschat et al. 2011] occur within the first 5 years from the disease onset [Rocca et al. 2013; Kim et al. 2013].
In this initial phase, pharmacological intervention favourably affect symptomatic control and functional outcomes [Schooler, 2003; McGorry et al. 2007; Lindenmayer et al. 2009; Viala et al. 2012]; thus the primary goal of treatment during this period is to prevent a subsequent relapse and to restore sociooccupational functioning to the premorbid level [Kim et al. 2013]. However, poor medication adherence is particularly common in FES [Coldham et al. 2002], and is among the leading reasons for relapse in FES according to a recent meta-analysis [Alvarez-Jiménez et al. 2008]. This is due to several factors; many individuals in the early stages do not accept the illness itself or its severity, and there can even be a false sense of treatment being unnecessary or an unwanted imposition [Kane and Garcia-Ribera, 2009]. This leads to relapse rates in FES over 70% as early as 1 year after diagnosis [Emsley et al. 2012]. The cohort study by Tiihonen and colleagues in 2588 FES patients found that fewer than 50% of patients in the Finnish healthcare system continued treatment for the first 2 months after their initial hospitalization. Interestingly, in this study, route of administration affected relapse; LAIs had a 64% lower relapse rate than the equivalent oral medication [Tiihonen et al. 2011].
Despite this, in many countries fewer than 10% of psychiatrists offer patients LAIs after a FES [Knudsen, 1985]. However, some authors propose that LAI APs are perhaps most suited to patients in the early stage of illness, before disease progression associated with poor adherence occurs [Emsley et al. 2013], and since the advantages outweigh potential adverse effects [Rocca et al. 2013; Taylor and Ng, 2013]. The best rationale for using LAI APs in FES comes from the fact that frequent relapses occur during the first years of the illness [Alvarez-Jiménez et al. 2008] and there is evidence for decreased rate of relapse with LAI medication compared with oral APs in FES [Tiihonen et al. 2011; Kim et al. 2008].
Taylor and Ng carried out a systematic review to find out whether LAI APs should be used in early schizophrenia. The authors identified 10 studies: 2 cohort studies, 3 (RCTs) and 5 open studies. In spite of the limited number of studies, especially RCTs, they concluded that LAIs may be useful in the treatment of FES. The use of LAIs in FEPs may be more effective than oral medication in controlling symptoms and relapse [Taylor and Ng, 2013]. Tolerability of APs is a particular concern in FES. Usually, medication-naïve individuals are acutely sensitive to APs in terms of responsiveness as well as side effects [Kelly et al. 2005; Alvarez-Jiménez et al. 2008; Francey et al. 2010] including extrapyramidal symptoms (EPS) [Chatterjee et al. 1995] and weight gain [Strassnig et al. 2007]. However, it is generally admitted that LAIs have a more acceptable side effect profile in comparison with their oral counterparts due to lower variation in peak and trough levels, and any concerns over debilitating side effects may be due to dosing errors [Taylor, 2009].
Regarding which LAI is more suitable for FES, there is an absence of long-term RCTs comparing LAIs with oral medication regarding efficacy, tolerability, relapse prevention and global outcomes [Parellada et al. 2012], and most evidence concerns RLAI. We also need studies examining patients’ preferences, acceptability and attitudes toward LAIs in early phases of the illness, as well as data about nonadherence rates of LAI SGAs in early phases of schizophrenia. There is also a lack of cost-effectiveness studies comparing LAIs with oral AP treatments specifically focusing on FES.
The question of whether effective early intervention positively influences long-term outcome also needs to be more effectively addressed [Parellada et al. 2012]. We need to know whether we are able to alter disease trajectory to clinical and neurological deterioration that mainly occurs within the first 3–5 years following the onset of the illness, e.g. the ‘critical period’ [Birchwood et al. 1998].
A positive answer for benefits on disease progression would provide support to an emerging literature regarding the neuroprotective effects of APs, especially SGAs [Bartzokis et al. 2011; Gassó et al. 2010].
In summary, psychiatrists may start considering the option of LAI APs, especially SGAs, to more patients with first-episode or recent-onset schizophrenia in a shared decision-making approach [Parellada et al. 2012]. In this sense, current clinical guidelines regarding LAI AP use in FES may be too much conservative and need perhaps to be updated [Parellada et al. 2012]. Nevertheless, studies with high methodology quality are needed to confirm already existing results.
AP discontinuation after FEP: the role of LAI formulations
Considerable debate exists surrounding the need to maintain or discontinue AP medication after the FES [Emsley et al. 2013]. There are 10 RCT studies addressing discontinuation in cases of FEP, but the results are contradictory [Hogarty and Ulrich, 1977, 1998; Kane et al. 1982; Crow et al. 1986; McReadie et al. 1989; Gitlin et al. 2001; Wunderick et al. 2007; Chen et al. 2010; Boonstra et al. 2011; Gaebel et al. 2002, 2011] (Table 3). The rate of relapse in AP and placebo interventions showed heterogeneous results. Some authors [Kane et al. 1982; McReadie et al. 1989; Gaebel et al. 2002, 2011] reported lower rates of relapse for the AP intervention (0–6%); on the other hand, Crow and colleagues found that 46% of the patients on active medication had a psychotic relapse within 2 years [Crow et al. 1986], whereas Chen and colleagues reported similar rates, but in 1 year [Chen et al. 2010]. Boonstra and colleagues reported a higher rate of relapse on placebo intervention in 2 years (82%) [Boonstra et al. 2011], and a similar rate in 1 year (79%) was described by Chen and colleagues [Chen et al. 2010]. On the other hand, Gitlin and colleagues [Gitlin et al. 2001] and Gaebel and colleagues [Gaebel et al. 2010a, 2010b] reported low rates of relapse in conjunction with placebo intervention (13% in 6 months and 19% in 1 year, respectively). Overall, studies suggest that maintenance treatment is more effective than discontinuation, and only 1 of the 10 studies reports beneficial effects for the discontinuation intervention [Gaebel et al. 2002].
Randomized clinical trials evaluating the rate of relapse comparing discontinuation versus maintenance antipsychotic medication after a FEP.

p < 0.05.
FEP, first episode of psychosis; LAI, long-acting injectable.
To the best of our knowledge, there are no RCTs evaluating the efficacy of discontinuation with LAIs. In the studies evaluating this, the percentage of patients on LAIs varied greatly and the percentage of those relapsing after discontinuation of a depot was not reported [Hogarty et al. 1977; Kane et al. 1982; Crow et al. 1986; McReadie et al. 1989; Gitlin et al. 2011; Boonstra et al. 211]. Interestingly, all the studies but one [Crow et al. 1986] showed lower rates of relapse with the AP intervention.
The question of whether the onset of psychosis associated with AP withdrawal is a relapse of the disorder or else a new phenomenon caused by the withdrawal of APs (‘supersensitivity psychosis’) is an important matter [Moncrieff, 2006]. If the latter were the case, then the choice of the drug could prove significant. Chouinard and Jones proposed that APs with a short half-life would be at a greater risk of developing ‘supersensitivity psychosis’ [Chouinard and Jones, 1980]. The relation between onset and physiological withdrawal is clearer in drugs with a short half-life, especially clozapine [Verghese et al. 1996]. The rate of relapse after discontinuation is reduced after gradual discontinuation compared with abrupt discontinuation of APs [Moncrieff, 2006]. In a meta-analysis, Viguera and colleagues found that abrupt withdrawal was associated with 65% of relapse in six months as opposed to only 32% with gradual withdrawal [Viguera et al. 1997].
Bearing in mind the factors associated with psychosis relapse after discontinuation, there is some evidence in favor of using LAIs in FEP. Most FEP subjects will experience discontinuation 1 or 2 years after their index episode. The option of LAIs may minimize both the factor that increases the risk of supersensitivity and the risk of nonadherence, which is the major cause of relapse.
Supersensitivity psychosis is related with an upregulation of the dopamine D2 receptor density in the brain, as a consequence of long-term treatment with APs. Clozapine has been proposed as the treatment for supersensitivity psychosis [Kane et al. 1988] and RLAI has also been proposed as an effective treatment [Gaebel et al. 2002]. RLAI may prevent the fluctuation of dopamine D2 receptor occupancy over time associated with the relapse of psychosis after discontinuation [Kane et al. 2003; Eerdenkens et al. 2004]. In summary, RLAI may be considered as a first-line treatment in FEP, while RLAI may reduce the risk of relapse in two scenarios or strategies, both continuation and AP withdrawal.
Long-acting injectable versus oral AP formulations
The first studies that compared LAIs with oral AP treatment were carried out on inpatients during the 1960s and 1970s, following the introduction of LAIs. A meta-review of controlled clinical studies comparing oral versus LAI FGAs, including more than 800 patients, found no differences in relapse rates, tolerability and anticholinergic use, whereas clinical improvement was significantly more likely with LAIs [Adams et al. 2001]. However, these studies suffered from a number of methodological limitations, namely being carried out for a short period of time, and in inpatient settings where oral adherence may not be problematic. In fact, in a subsequent review, apart from one study, relapse was significantly higher for patients medicated with oral APs than depots [Schooler, 2003].
Later studies threw a wider net, addressing a broader range of outcome measures and employing different methodological approaches. In fact, studies of patients initiated or switched from oral to LAI APs reported significant improvements not only in schizophrenia symptom control, but also in quality of life, satisfaction and functioning [Kaplan et al. 2013].
The advantage of LAI versus oral APs in improving adherence has been demonstrated by Leucht and colleagues, who conducted a systematic review and meta-analysis on long-term RCTs on schizophrenia outpatients [Leucht et al. 2011]. The results also indicated a lower risk of relapse (10% and 30% relative and absolute risks, respectively) and dropout for inefficacy.
Kishimoto and colleagues performed the largest meta-analysis of 21 RCTs comparing LAI versus oral APs’ efficacy for relapse prevention in schizophrenia [Kishimoto et al. 2014]. Surprisingly, and in contrast to previous meta-analysis, they found that pooled LAIs were not superior to orals in all of the examined relapse-related outcomes; the only exception was fluphenazine-LAI, which showed significant superiority over oral APs in several relapse-related outcomes.
Regarding LAI SGAs, a recent meta-analysis showed greater efficacy versus placebo, but no significant differences compared with oral APs [Fusar-Poli et al. 2013]. Most RCTs comparing RLAI and oral APs have shown a superiority of RLAI in relation to adherence, clinical improvement, reduction in EPS and prolactin levels [Chue et al. 2005a; Bai et al. 2007] and reduction of relapses and hospitalizations [Olivares et al. 2001], but not all [MacFadden et al. 2010]. Regarding olanzapine, there seems to be similar efficacy between the oral and long-acting formulations [Kane et al. 2010]. At the time of writing, there was no comparative data regarding aripiprazol LAI.
Negative results of RCTs might have been driven by methodological questions, since the comparative effectiveness of AP formulations is sensitive to research designs [Kirson et al. 2013; Kane et al. 2013a]. Of note, most of the studies were too short in length to evaluate relapse prevention adequately. However, patients consenting to clinical trials of LAIs may not be representative of those prescribed LAIs in real-world settings [Haddad et al. 2009], since they might over-represent patients with better engagement with healthcare providers, better adherence to treatment, lower illness severity and better cognitive capabilities to understand complex issues. In fact, patients who do not reliably take their oral medications may be less likely to volunteer for RCTs, particularly if a treatment arm includes a LAI [McEvoy, 2006; Davis, 2010]. It is also important to recognize that participation in a controlled trial alters the ecology of treatment delivery and experience. Patients in RCTs are likely to receive more and different types of attention than those in routine care, from measures of adherence to reminders to attend clinical/research assessment sessions, or to the provision of free medication [Correll et al. 2011; Kane et al. 2013a]. In addition, more frequent monitoring during a trial enables psychiatrists to change dosages according to the symptoms and to provide supportive psychotherapy. It is difficult to determine what role these factors might have in altering patterns of medication-taking in contrast to routine care and to what extent they might, therefore, diminish the potential advantages of LAIs in RCTs [Offord et al. 2013; Kishimoto et al. 2014].
Since a major advantage of LAIs is overcoming partial/nonadherence, the differences between oral and LAI formulations are better evaluated in naturalistic settings, and especially in long-term studies [Schooler, 2003]. Therefore, observational, effectiveness studies provide more interesting information for clinicians and health authorities, as they include representative samples of patients, use pragmatic variables, reproduce routine treatment conditions, and are done in real-world clinical settings [Stahl, 2001; March et al. 2005; Kane et al. 2013a].
Several observational studies have compared LAI FGAs with first- and second-generation oral APs, but results have been inconclusive. Two prospective studies comparing LAI FGAs with various oral FGAs reported the former were associated with lowered risk of rehospitalization and longer time to discontinuation [Tiihonen et al. 2006; Zhu et al. 2008]. In contrast, two prospective studies comparing LAI FGAs with various oral SGAs found the oral SGAs to be associated with lower risk of readmission and higher odds ratio of achieving remission compared with LAIs [Conley et al. 2003; Haro et al. 2006, 2007]. In the same vein, the electronic records of patients medicated with LAI SGAs were compared with oral APs over a 30-month period revealed that LAI SGAs were associated with significantly lower mean number of rehospitalizations, emergency room visits and fewer hospitalization days compared with oral APs [Lafeuille et al. 2013].
Mirror-image studies evaluate the effect before versus after initiation of LAI treatment, with patients serving as their own controls. Mirror-image studies published in the early 1970s provided the first clinical evidence favouring LAIs versus oral APs in terms of frequency and duration of hospitalizations [Denham and Adamson, 1971; Gottfries and Green, 1974; Marriot and Hiep, 1976; Devito et al. 1978]. Later such studies performed during the 1980s confirmed relapse rates are significantly higher with oral APs versus LAIs [Johnson et al. 1983]. More recently, a large cohort observational study in France with 1859 patients followed for up to 12 months found a 34% reduction in the risk of hospitalization in patients treated with RLAI compared with patients receiving other treatments, including depot formulations [Grimaldi-Bensouda et al. 2012]. A 2-year, prospective, observational study also showed that, compared with oral APs, RLAI was associated with better treatment retention, greater improvement in clinical symptoms and functioning, and greater reduction in both number and length of hospital stays [Young and Taylor, 2006; Olivares et al. 2009; Kane et al. 2013b].
A recent retrospective study involving more than 3600 patients showed that those who initiate LAIs versus oral APs have greater reductions in the number of hospitalizations for any cause and for schizophrenia relapse compared with before treatment began [Offord et al. 2013]; this was found even though patients who initiated LAIs had a much greater disease severity compared with patients who began oral treatment.
Nevertheless, observational studies also have important limitations; the fact that patients are not randomized may bias the LAI group, including a greater proportion of patients with low adherence (i.e. treatment failures by previous history and comorbidity) that are more likely to receive a LAI [Olivares et al. 2011]. Moreover, these studies can be confounded by independent events, such as a reduction of hospital beds [Davis et al. 1994], and some authors have highlighted that this methodological strategy has an ‘inherent bias towards improvement’ [Haddad et al. 2009]. Finally, the few studies that report tolerability data [Adams et al. 2001; Marchiaro et al. 2005; Haro et al. 2006] did not use direct measures (i.e. rating scales) but clinical observations regarding anticholinergic use. Moreover, since relapse rates vary as a function of stage of illness, this too must be factored into the study design [Agid et al. 2010].
At present, no head-to-head studies of oral APs and olanzapine pamoate (other than with oral olanzapine), paliperidone palmitate, or aripiprazol long-acting are available. In face of methodological limitations, it is risky to extrapolate findings on other variables such as cost-effectiveness or adherence, and more studies are needed comparing RLAI, paliperidone LAI and aripiprazol LAI with oral APs to be able to make conclusions regarding their specific differences in clinical outcomes. In the next section, studies comparing LAI FGAs and LAI SGAs are summarized.
First- versus second-generation LAI APs
Data regarding differences between LAI FGAs and SGAs are much sparser. LAI FGAs have a ludicrously wide range of licensed doses and are often given in too high a dose; for haloperidol decanoate optimally effective doses appear to be around 50–100 mg per 4 weeks [Taylor, 2005]. Although use of higher doses is difficult to support given data available, slow rise to steady state may explain this common practice [Taylor, 2005].
Switching studies regard mainly RLAI and have shown that patients with mild, residual symptomatology treated with conventional depots experience significant improvement in psychiatric and movement disorder symptoms [Lasser et al. 2004; Turner et al. 2004; Marinis et al. 2007; Lai et al. 2009]. Of note, in real world settings, RLAI has been shown to significantly reduce hospitalizations rates as compared with conventional depots [Grimaldi-Bensouda et al. 2012].
Patients treated with RLAI have also been shown to be much more satisfied with themselves, their health and sleep than those on haloperidol depot [Marinis et al. 2007; Mihajlović et al. 2011], and have also reported better quality of life [Marinis et al. 2007].
More recently, in a 24-week open-label trial, Suzuki and Gen have shown that switching from haloperidol decanoate to RLAI may improve cognitive function including memory, executive function, motor processing function, and attention [Suzuki and Gen, 2012].
Covell and colleagues found no significant differences in psychopathology, hospitalizations, sexual side effects, new-onset tardive dyskinesia or EPS, and in time to treatment discontinuation in a naturalistic study of patients with schizophrenia or schizoaffective disorder who were taking haloperidol decanoate (n = 40) or fluphenazine decanoate (n = 22), and were randomly assigned to stay on current LAI FGA or switch to RLAI [Covell et al. 2012]. However, when the 6-month naturalistic follow-up period was included, time to treatment discontinuation was significantly shorter for individuals assigned to switch than for individuals assigned to stay (10% of stayers discontinued versus 31% of switchers; p = 0.01). On the other hand, patients who switch to RLAI from conventional depots may experience significantly more weight gain and in prolactin levels [Lai et al. 2009; Covell et al. 2012].
As far as we know, there are no data comparing the SGAs olanzapine pamoate, paliperidone palmitate and aripiprazol LAI with conventional FGA depots. An ongoing study will compare paliperidone palmitate to aripiprazol LAI [Clinicaltrials.gov identifier: NCT01795547].
Cost-effectiveness of LAI AP treatment
Schizophrenia accounts for 1.5–3% of national healthcare expenditures [Knapp et al. 2004], and to reduce the burden of illness and ensure efficient resource use, cost-effective treatment is essential [Achilla and McCrone, 2013]. In that sense, treatment options that improve compliance and reduce side effects, relapse rates and rehospitalizations are of paramount importance; LAI APs have potential advantages over daily medication in improving these factors [Fleischacker et al. 2003; Almond et al. 2004; Haycox, 2005; Keith, 2009]. However, high acquisition and administration costs may limit their more widespread utilization [Moore et al. 1998; Oh et al. 2001; Chue et al. 2005b]. On the other hand, even though drug costs of LAI APs are significantly higher than oral APs, schizophrenia patients initiating the former incur less healthcare cost in comparison with those initializing the latter [Lin et al. 2013].
Achilla and McCrone have reviewed the available evidence on the cost effectiveness of LAI APs [Achilla and McCrone, 2013]; the majority of studies are on RLAI and demonstrated that it was the dominant strategy in terms of cost-effectiveness relative to oral or other LAI drugs. RLAI was dominated by olanzapine in either oral or LAI formulation only in a Slovenian and a US study. Furthermore, in two UK studies, use of RLAI increased the hospitalization days and overall healthcare costs, relative to other atypical or typical LAI APs [Achilla and McCrone, 2013]. This was not found in a study of 250 patients 1 year after initiation of RLAI; mean days spent in hospital increased following initial administration of RLAI, with increase in direct healthcare costs [Young and Taylor, 2006]. The authors identified limitations to this study, including lack of a parallel control group and its short duration.
Therefore, RLAI is likely to be a cost-effective, first-line strategy for managing schizophrenia compared with other oral or depot formulations, irrespective of country-specific differences [Achilla and McCrone, 2013]. However, inconsistencies in study methodologies and in the reporting of study findings suggest caution needs to be applied in interpreting these findings.
Although indirect costs constitute a substantial proportion of the costs associated with schizophrenia, pharmacoeconomic studies have considered only the direct costs of health services, probably due to the limited availability of data and the difficulties associated with measuring them accurately. Thus, the adoption of a full societal perspective has been almost impossible. Finally, most economic evaluation studies employed decision analytical models rather than trial-based or observational designs.
A recent meta-analysis showed that costly LAI SGAs offer little advantage in reducing the psychotic symptoms, with no superior effect in comparison with oral administration [Fusar-Poli et al. 2013]. However, using clinical trial data to evaluate whether LAI APs provide a benefit for adherence or clinical outcomes over oral APs is difficult since patients involved in clinical trials are monitored to a much greater extent and are more likely to adhere to treatment [Offord et al. 2013], and hence lack external validity.
In addition to the likelihood of having a direct impact on hospital costs of care for schizophrenia patients, the marked reduction in the incidence of relapses and hospital length of stay will likely allow for patients using LAI APs to function better socially, maintain a job with less absenteeism and abuse substances to a lesser degree, all of which could potentially contribute towards a large reduction in the indirect costs of schizophrenia, which in the US in 2002 were estimated at $32.4 billion [Wu et al. 2005]. Earlier introduction of LAI APs may provide a considerable benefit to patient outcome and potentially reduce the burden on healthcare resources [Offord et al. 2013].
Finally, increase in medication adherence with the use of LAI APs may eventually induce a reduction in the pharmaceutical costs of schizophrenia treatment by a decrease in hospital stays that compensate their higher costs, especially SGAs [Niaz and Haddad, 2010]. Treatment with LAIs may be also more cost-effective than oral medication, and may reduce the suicide risk and the greater propensity to violence observed at least in a subset of persons with psychotic illnesses and comorbid substance/alcohol use disorders [Ravasio et al. 2009; Reichart and Kissling, 2013].
Unmet needs and prospect vistas
There is a need to increase the availability of additional LAI SGAs and to develop more reliable methods of AP delivery. Given the failure of the long-term oral treatments and bearing in mind that relapse can lead to serious consequences from all perspectives (biological and psychosocial), the future of schizophrenia pharmacotherapy will hopefully evolve to include better long-term delivery systems to more effectively address the high risk of relapse due to nonadherence in all phases of the illness. These should include not only longer extended release injectable formulations, but also intranasal formulations [Currier and Walsh, 2013], transdermal patches, subcutaneous implants of APs, and other long-acting devices like AP pumps [Rabin et al. 2008; Wang et al. 2012], since one of the primary reasons why patients reject LAIs is the fear of needles [Kim et al. 2012].
A radical change in attitude among clinicians and patients is required to reconsider LAI APs from a new perspective: no longer medications of last resort, but rather a potential first step to ensure treatment continuity and maximize clinical remission [Altamura et al. 2012]. Achieving and maintaining clinical remission is important [Andreasen et al. 2005], since patients in remission show significantly better social functioning and quality of life [Brissos et al. 2011].
A leading new issue is the utilization of LAIs in FES to prevent cognitive decline induced by demyelinization [Niaz and Haddad, 2010; Bartzokis et al. 2011; Olivares et al. 2011]. This approach implies increasing mental health staff relations with patients to overcome the ‘last resort treatment’ image of LAI APs [Knudsen, 1985; Glazer, 2007; Davis, 2010; Olivares et al. 2011], since treating with LAI APs as early as possible, ideally from the first episode, can reduce relapse, number and duration of rehospitalization and cognitive symptoms, and improve the quality of life and functional outcomes [Viala et al. 2012]. Clearly, head-to-head comparison studies between two or more LAIs are critically needed to clarify the role of each compound in the long-term management of psychotic disorders.
For these reasons, the obvious advantages of these agents with regards to the problem of noncompliance, refractoriness and relapse prevention, need not be lost because of unfounded fears if not prejudices [Glazer and Kane, 1992].
Footnotes
Acknowledgements
We thank Dr Nuno Cunha for his helpful advice regarding pharmacokinetics and pharmacodynamics in previous versions of the manuscript. M.R.-V. received grant Estudio NONSTOP PI11/02334.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
S.B. was Medical Affairs Manager at Janssen Pharmaceutical from April 2010 to August 2013. Since then, she has served as consultant/advisor for Janssen on a sporadic basis. M.R.-V. has received grants and served as advisor or Continuing Medical Education (CME) speaker for the following entities: Bristol-Myers-Squibb; Janssen; Juste; Otsuka; Spanish Ministry of Science and Innovation; Consejería de Salud de la Junta de Andalucía; Plan Nacional de Drogas; and Fundación Alicia Koplowitz. D.T. has received research funding from and undertaken paid consultancy with Janssen, Servier, Otsuka and AstraZeneca. V.B.-M. has received grants and served as consultant, advisor or CME speaker during the last three years for the following entities: Almirall; Angelini; AstraZeneca; Bristol-Myers-Squibb; Janssen; Juste; Lundbeck; Otsuka; Spanish Ministry of Science and Innovation (CIBERSAM); and Fundación Alicia Koplowitz.
