Abstract
With the exception of fluoxetine, all selective serotonin reuptake inhibitors (SSRIs) commonly cause hyperprolactinemia through presynaptic mechanisms indirectly via 5-hydroxytryptamine (5-HT)-mediated inhibition of tuberoinfundibular dopaminergic neurons. However, there is little insight regarding the mechanisms by which fluoxetine causes hyperprolactinemia via the postsynaptic pathway. In this text, analysis of five spontaneously reported clinical cases of hyperprolactinemia resulting in overt symptoms of amenorrhea with or without galactorrhea, were scrupulously analyzed after meticulously correlating relevant literature and an attempt was made to explore the putative postsynaptic pathway of fluoxetine inducing hyperprolactinemia. Hypothetically, serotonin regulates prolactin release either by increasing oxytocin (OT) level via direct stimulation of vasoactitive intestinal protein (VIP) or indirectly through stimulation of GABAergic neurons. The pharmacodynamic exception and pharmacokinetic aspect of fluoxetine are highlighted to address the regulation of prolactin release via serotonergic pathway, either directly through stimulation of prolactin releasing factors (PRFs) VIP and OT via 5-HT2A receptors predominantly on PVN (neurosecretory magnocellular cell) or through induction of 5-HT1A-mediated direct and indirect GABAergic actions. Prospective molecular and pharmacogenetic studies are warranted to visualize how fluoxetine regulate neuroendocrine system and cause adverse consequences, which in turn may explore new ways of approach of drug development by targeting the respective metabolic pathways to mitigate these adverse impacts.
Keywords
Clinical cases
Case one
A 35-year-old married woman consulted the psychiatry department in February 2010 for depression for the past 1.5 years which had become aggravated during the past 3 months. Using the Montgomery–Åsberg Depression Rate Scale (MADRS) [Montgomery and Åsberg, 1979] the patient scored 25 points and was diagnosed having depression and was prescribed fluoxetine 20 mg/day. After 21 days, the dose was increased to 40 mg/day along with alprazolam 0.5 mg/day for poor treatment response and persistent insomnia. After 2 weeks, she showed significant improvement although she experienced slight nausea and headache. During her subsequent follow up, the dose of alprazolam was reduced to 0.25 mg/day and tapered to complete cessation over the next week, and fluoxetine was continued with excellent therapeutic response.
However, in July 2011, she complained of amenorrhea for five consecutive cycles and her serum prolactin level was found to be 25 ng/ml (normal 0–20 ng/ml). The dose of fluoxetine was reduced to 20 mg/day, but amenorrhea was not resolved during the subsequent 3 months.
Case two
In March 2011, a 34-year-old married woman, presented with an aggravation of first episode of major depressive illness and was prescribed fluoxetine 20 mg/day for the first week and 40 mg/day for the following 3 weeks. During the first follow up visit after 21 days, total remission of symptoms was achieved and the prescribed pharmacotherapy was not changed.
However, in September 2011, she complained of irregular menstrual bleeding since June 2011, which further progressed to even complete cessation of her menstruation in August 2011. Her physical examination was unremarkable with slightly elevation of serum prolactin level to 42 ng/ml. She was advised to continue fluoxetine 40 mg/day and referred to gynecology department.
Case three
A 31-year-old unmarried woman presented in September 2010, with a 2-year history of severe and worsening obsessive compulsive disorder (OCD) as defined by the
Intriguingly, in July 2011, the patient complained of amenorrhea for 4 months with a 15-day history of a milky, nonhemorrhagic bilateral breast discharge while on 20 mg/day fluoxetine. Her serum prolactin level was found to be 122 ng/ml and the physical manifestation was highly suggestive of hyperprolactinemia associated with amenorrhea and galactorrhea. She was then advised to continue 20 mg/day fluoxetine, but amenorrhea and galactorrhea persisted with further elevation of prolactin level until August 2011.
Case four
A 33-year-old woman presented in June 2010 with symptoms of OCD according to the DSM-IV criteria [American Psychiatric Association, 1994] for 8 months and the features started after an interpersonal stressor that initially lasted for 2 months and had a waxing and waning course thereafter. She was prescribed fluoxetine 20 mg/day and at the end of the third week, the dose was increased to 40 mg/day. At the 12th week of treatment the symptoms of OCD decreased and it was decided to maintain her on 40 mg/day of fluoxetine.
In March 2011, during her scheduled follow up, she showed much improvement of her OCD associated symptoms, but reported absence of menstruation for four consecutive cycles. Her prolactin level at that time was found to be 93 ng/ml.
Case five
A 22-year-old young unmarried woman, with a newly diagnosed case of hypochondriasis, was initiated with fluoxetine 20 mg/day along with clonazepam 0.5 mg/day in November 2011.
In January 2012, she presented with a 3-day history of spontaneous bilateral nipple discharge associated with irregular menstruation cycles since December 2011. Her physical examination and vitals were found to be unremarkable for any features of hyperprolactinemia and her serum prolactin level was 138 ng/ml.
In all of the presented five cases, primary physical, biochemical examinations and negative pregnancy tests strongly suggest that their amenorrhea were temporally associated with fluoxetine trials. Further, in all cases the associated undesirable neuroendocrine clinical symptoms were accompanied by various degrees of elevated serum prolactin levels (Table 1).
Information regarding patients, treatment details including management strategy and duration of resolution with follow-up periods.
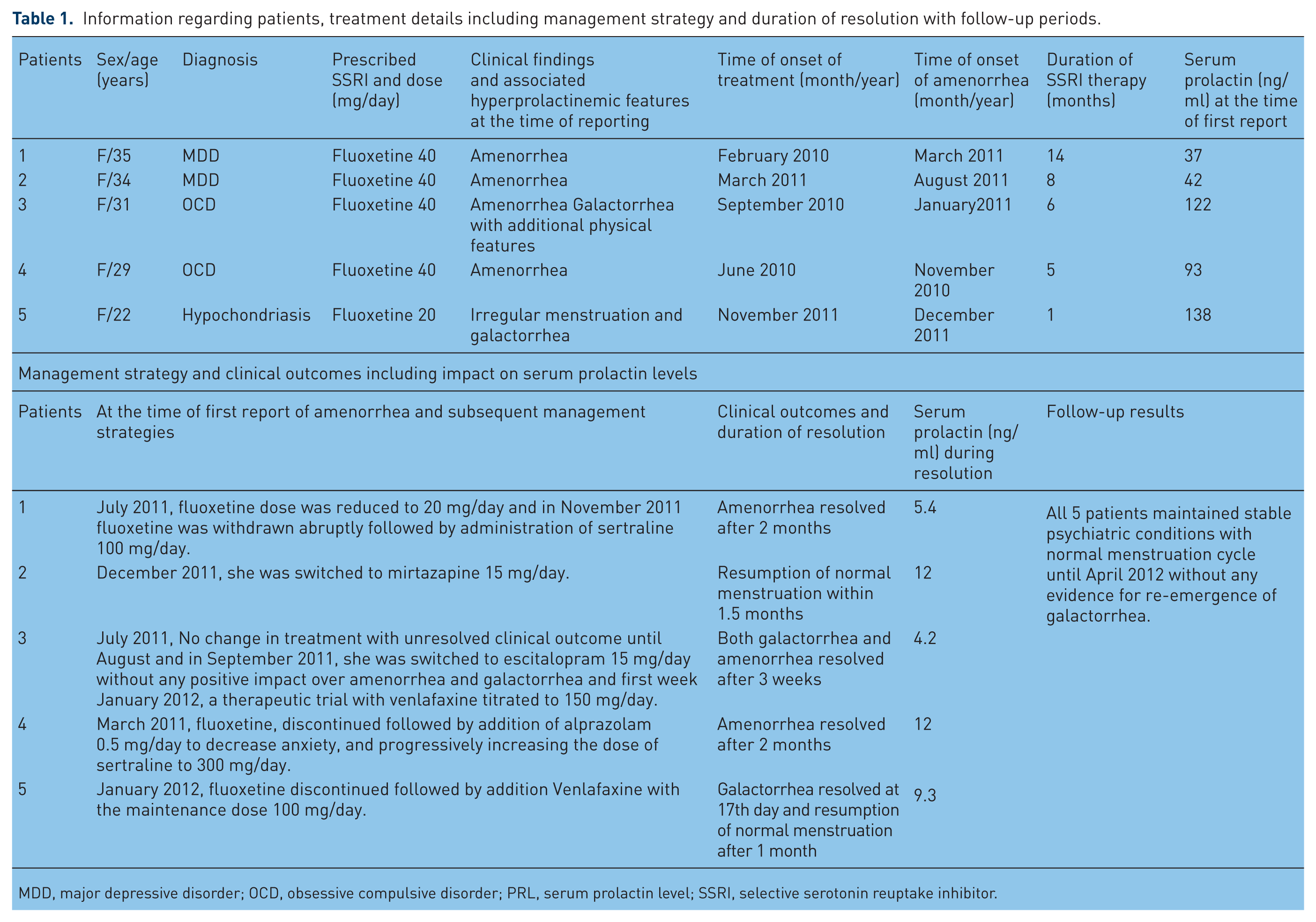
MDD, major depressive disorder; OCD, obsessive compulsive disorder; PRL, serum prolactin level; SSRI, selective serotonin reuptake inhibitor.
Analysis of the cases
A careful history and physical examination revealed that all of the patients had their regular menstrual cycle before these events of amenorrhea. There was no evidence of recent weight gain or loss and no history of eating disorder or excessive athletic activities. There was no previous contributing medical or family history of any other possible hereditary, traumatic, surgical, metabolic, infective, organic or pathologic diseases. None of the patients were smokers, alcoholic, or diabetic. Further meticulous clinical and physical examinations were negative for other psychiatric illnesses, surgeries, or substance abuse. The vital signs of all five patients were essentially within normal limits. The first four out of five patients were sexually active and were continuously on oral contraceptive pill (OCP). Hence, sudden withdrawal of oral contraceptives cannot be implicated as a likely cause of their amenorrhea. The fifth patient denied taking any OCPs. None of patients reported any hot flushes, severe headaches, or visual field disturbances.
Examination of the breasts of the first, second, and fourth patients revealed no secretions or tenderness. In the case of patients three and five, bilateral breast secretion could be expressed without any tenderness or dimpling. Skin examination of patients three and five showed mild papular acne on their faces and mild hair growth on their chins. There was evidence for mild painful pustular lesion on back of the fifth patient. There were no abnormalities in their routine blood chemistry, liver function tests, or renal function tests. Routine electrolyte and urine analysis were essentially within normal limits. The first, second, and fourth patients had mild elevated serum prolactin levels without any associated physical signs and symptoms. However, the third and fifth patients had substantially higher serum prolactin levels.
During systemic evaluation, preliminary exclusion of potential causes of secondary amenorrhea and hyperprolactinemia such as adenopathy, celiac disease, hypergonadism, polycystic ovary syndrome (PCOS), primary ovary insufficiency, Turner syndrome, Asherman’s syndrome, and insulin sensitivity studies were done by correlating their hormonal levels, past and present menarche histories and associated physical findings followed by expert opinions from respective fields. The pertinence of the above preliminary findings was further evidenced by unremarkable pelvic examinations, pelvic ultrasounds, magnetic resonance imagining (MRI) scans (focused on the brain and particularly the pituitary gland), hysterosalpingographies (HSG), and mammography tests. Based on the absence of atypical symptoms, a mutual clinical judgment ruled out the rare plausible etiologies of secondary amenorrhea, such as pheochromocytoma, systemic lupus erythematosus (SLE), rheumatoid arthritis, uterine and cervical tuberculosis, or any other related infections.
Regarding the question of drug interactions, there is no known evidence of either pharmacokinetic or pharmacodynamic interactions between clonazepam, alprazolam, and fluoxetine, rather most of the published literature demonstrated that cotherapy of fluoxetine and clonazepam or alprazolam resulted in superior efficacy than fluoxetine alone in various indications [Papakostas et al. 2010; Eric et al. 2003]. However, there is a dearth of literature correlating the impact of this cotherapy in hypochondriasis, even though this combination therapy is often used in regular clinical practice with satisfactory clinical outcomes.
In all of the above cases, several features pointed to fluoxetine as the cause of secondary amenorrhea. First, there was a temporal relationship between the drug administration and onset of secondary amenorrhea. Second, in all cases the secondary amenorrhea was associated with elevated serum prolactin levels and a conspicuous systematic evaluation of each individual patients could not explain any alternative potential causes for hyperprolactinemia and associated clinical consequences, including the menstrual abnormalities, amenorrhea and galactorrhea (particularly in cases three and five). Third, hyperprolactinemia and associated clinical consequences were resolved only after discontinuation of fluoxetine in the first four cases. Use of the Naranjo probability scale [Naranjo et al. 1981] indicated a highly probable relationship between secondary amenorrhea, namely hyperprolactinemia and fluoxetine in all five cases.
Final diagnosis and follow up
In all five cases, the single most important attributable factor was fluoxetine, owing to its strong temporal correlation with the onset of amenorrhea, hence, it was withdrawn (dechallenged) in all five patients and each one was managed on a case-by-case basis, with involvement of clinical pharmacologists in the decision-making process to determine the balance of risks and benefits for each individual patient category.
Case one
In July 2011, the dose of fluoxetine was reduced to 20 mg/day but amenorrhea was not resolved during the subsequent three cycles. Serum prolactin level was further elevated up to 59 ng/ml, without any evidence of additional physical features such as hyperprolactinemia. In November, 2011 fluoxetine was withdrawn abruptly followed by administration of sertraline 100 mg/day. After 2 months, in January 2012, menstruation resumed and serum prolactin level dropped to 5.4 ng/ml. As of April 2012, the patient continued on sertraline 100 mg without any evidence for aggravation of her depression-associated symptoms or reappearance of amenorrhea.
Case two
Until 17 December 2011, she was continued with fluoxetine 40 mg/day with unresolved amenorrhea and aggravated hyperprolactinemia, as her serum prolactin level was found to be elevated up to 83 ng/ml. Eventually, she was switched from fluoxetine to mirtazapine 15 mg/day. Menstrual abnormalities were resolved in February 2012 and serum prolactin level dropped down to 12 ng/ml. As of April 2012, she was psychiatrically stable without any manifestation of hyperprolact-inemia, while being maintained on mirtazapine 15 mg/day.
Case three
The patient was advised to continue 20 mg/day fluoxetine, but her amenorrhea and galactorrhea persisted without further elevation of prolactin level until August 2011. In September 2011, she was switched to escitalopram 15 mg/day without the resolution of any of hyperprolactinemia-associated symptoms until the first week of January 2012. A therapeutic trial with venlafaxine titrated to 150 mg/day brought a near-complete symptomatic remission of all associated hyperprolactinemic features with resumption of normal menstruation cycle within 3 weeks. On February, 2012 her serum prolactin level was found to be essentially within the lower limit of normal value (0–20 ng/ml). The patient remained psychiatrically stable without hyperprolactinemia from the time she was switched to venlafaxine until April 2012.
Case four
In March 2011, treatment was first modified by discontinuing fluoxetine and adding alprazolam 0.5 mg/day to decrease anxiety, and progressively increasing the dose of sertraline up to 200 mg/day. In May 2011, approximately 2 months after initiation of the sertraline treatment, her serum prolactin levels was normalized (12 ng/ml) and menstrual abnormalities were resolved. Until April 2012, the patient remained on the same therapeutic regimen and continued to be psychiatrically stable with absolutely regular menstrual cycles.
Case five
Fluoxetine was discontinued in January 2012, and it was decided to switch her over to venlafaxine 75 mg/daily in two divided doses, which was gradually increased to 100 mg/daily over the next 2 weeks with complete cessation galactorrhea at the 17th day after stopping fluoxetine. She resumed her normal menstruation after 1 month (February 2012) and her serum prolactin level returned to a normal level (9.3 ng/ml). At the 3-month follow-up visit in May 2012, the patient was well maintained on venlafaxine and there was no re-emergence of either galactorrhea or amenorrhea.
Essential review of the literature and discussion
A literature survey revealed that a correlation exists between weight loss and menstrual cessation, and between regain of weight and menstrual resumption [Mitan, 2004]. There are several published articles that depicted high prevalence of menstrual disturbances among women associated with bipolar disorder. Although the mechanism had not been ascertained, disruption of the hypothalamic–pituitary–adrenal (HPA) axis function similar to that seen in depression is likely [Rasgon et al. 2000; Rasgon et al. 2003]. However, there is a dearth of published scientific articles justifying the correlation between OCD and hypochondriasis with amenorrhea. As these two psychiatric conditions could also be associated with stress, both conditions might have a role in hypothalamic dysfunction.
It is notable that patients with high emotional stress or physical distress can have hyperprolactinemia and associated amenorrhea or menstrual irregularities related to hypothalamic dysfunctions [Kaplan and Manuck, 2004; Young and Korzun, 2002]. In a 3-year study of women aged 36–45 years [Harlow et al. 2003], those with a history of depression exhibited 1.2 times the rate of perimenopause as nondepressed women. Subjects with Hamilton Rating Scale for Depression [Hedlung and Vieweg, 1979] scores >8 at enrollment had twice the rate of perimenopause after 3 years compared with women without depression.
PCOS causes 20% secondary amenorrhea is a prevalent and frequently encountered endocrine disorder [Lobo and Carmina, 2000]. In a study, 16 of 32 women with PCOS had depression as diagnosed by Sub-fertility Center for Epidemiological Studies - Depression Rating Scale (scores >16) [Rasgon et al. 2000; Rasgon et al. 2003]. This suggests a high prevalence of depression among women with PCOS, but was limited by possible selection bias, no further diagnostic evaluation for depression, small sample size, and lack of an age-matched control group.
A case report describes high dose of alprazolam-induced amenorrhea and galactorrhea in a 35-year-old unmarried female psychiatric patient [Petrić et al. 2011]. In another clinical report, there was evidence of pharmacodynamic interactions between citalopram, alprazolam in tramadol-induced galactorrhea in a female patient [Bondolfi et al. 1997; Hall et al. 2003]. However, likelihood of either pharmacokinetic or pharmacodynamic interactions with alprazolam was easily eliminated as alprazolam was discontinued long before.
The advent of fluoxetine was the beginning of a new era of safe and effective treatment for patients with various psychological disorders [Wong et al. 1995; Rossi et al. 2004]. The most commonly reported side effects of fluoxetine include sexual dysfunction, headache and nausea, but, fortunately, only in a small minority of patients and such effects generally disappear after about 2 weeks, although, as with other antidepressants, sexual dysfunction can persist [Eli Lilly, 1995]. A comprehensive literature review deciphered fluoxetine is well tolerated and rarely associated with serious side effects. Endocrine and reproductive side effects of serotonergic antidepressants (particularly with fluoxetine) are infrequent and uncommon, galactorrhea and amenorrhea is rather rarely mentioned among SSRI-related adverse effects.
A MEDLINE search revealed two case reports of fluoxetine-induced galactorrhea. A 71-year-old woman taking estrogen replacement therapy developed galactorrhea after initiation of fluoxetine for depression and was found to have an elevated prolactin level. Fluoxetine was discontinued with complete resolution of galactorrhea and normalization of prolactin level [Peterson, 2001]. A 29-year old woman newly diagnosed to be suffering from generalized anxiety disorder according to the DSM-IV criteria developed unilateral galactorrhea 21 days after initiation of fluoxetine therapy with excellent resolution of anxiety-associated symptoms without any evidence for elevated serum prolactin level. Withdrawal of prolactin resulted in cessation of galactorrhoea [Canan et al. 2011].
A systematic search of the literature in English was performed in Embase and MEDLINE and references cited in all relevant trials were searched iteratively to identify any link between SSRI and neuroendocrine abnormalities such as hyperprolactinemia and its clinical consequences. It revealed a significant number of studies describing that all SSRIs (paroxetine, fluoxetine, fluvoxamine, citalopram, escitalopram, sertraline, etc.) are associated with prolactin abnormalities (hyperprolactinemia) and/or manifest galactorrhea, amenorrhea, and breast tenderness.
However, the number of published cases in the literature is limited. To date there are few published case reports describing nonpuerperal lactation associated with the use of SSRIs in women. In these reports, the patients were mostly premenopausal. The common features of all of the case reports were the onset of galactorrhea with or without significant elevated prolactin levels and, in a very few cases, associated amenorrhea resulting shortly after initiation of a SSRI. In all of the reports, symptoms promptly subsided with discontinuation of the SSRI drug. Most SSRIs were implicated in these reports [Bondolfi et al. 1997; Iancu et al. 1992; Bronzo and Stahl, 1993; Morrison et al. 2001; Arya and Taylor, 1995; Spigset and Mjorndal, 1997; Otero et al. 2002; Pablos et al. 2001; Lesaca, 1996; Jeffries et al. 1992; Davenport and Velamoor, 2002; Bonin et al. 1997; Gonzalez et al. 2000; Cowen and Sargent, 1997; Hall, 1994; Gulsun et al. 2006; Gulsun et al. 2007; Shim et al. 2009].
There have also been numerous uncontrolled studies implicating changes in prolactin levels with therapy with SSRIs. All reports showed varied degrees of basal prolactin elevations with SSRI treatments [Attenburrow et al. 2001; Amsterdam et al. 1997; Dulchin et al. 2001; Laine et al. 1997; Urban and Veldhuis, 1991]. The link between acute serotonin stimulation and prolactin release has long been established, but the clinical and pathological impact of chronic serotonin stimulation on prolactin release has only been investigated recently. In a systematic study, the incidence of mammoplasia in 59 women taking SSRIs or venlafaxine was highest with paroxetine compared with other antidepressants. Paroxetine-treated patients exhibited statistically significant elevations in prolactin levels, although all subjects on fluoxetine, sertraline, or venlafaxine showed nonsignificant elevations of their basal prolactins. Although significant effects were only seen with paroxetine, this might be related to the relatively greater number of patients on paroxetine, compared with other drugs fluoxetine, sertraline, and venlafaxine, resulting in inadequate power to detect changes in the latter groups [Amsterdam et al. 1997].
A putative mechanism and argument
The exact mechanisms by which SSRIs render hyperprolactinemia and cause several clinical consequences such as amenorrhea and galactorrhea (neuroendocrine effects) remain elusive. There is evidence that serotonin might stimulate prolactin release directly via postsynaptic 5-HT receptors in the hypothalamus [Nicholas et al. 1998], or indirectly via 5-HT-mediated inhibition of tuberoinfundibular dopaminergic neurons [Arya, 1994]. The serotoninergic neurons project from the dorsal raphe nucleus to the medial basal hypothalamus and exert their action via 5HT1A and 5HT2 receptors and paraventricular nucleus containing different populations of neurosecretory cells producing oxytocin, vasopressin, vasoactive intestinal peptide (VIP), thyrotropin-releasing hormone (TRH) and other neuropeptides [Emiliano and Fudge, 2004; Aizawa and Hinkle, 1985; Benker et al. 1990; Bjoro et al. 1990].
It is known that serotonin affects the prolactin level through the action of one or more of these prolactin-releasing factors (PRFs), among which the VIP pathway is the best studied. VIP acts both via hypothalamic afferents and direct paracrine and autocrine mechanisms, through lactotroph cell receptors binding, enhancing adenylate cyclase activity and increasing prolactin gene transcription. Oxytocin seems to participate in VIP-induced prolactin release and could act through the inhibition of the tuberoinfundibular dopamine pathway (TIDA) [Emiliano and Fudge, 2004; Ben-Jonathan, 1994; Wanke and Rorstad, 1990; McCann et al. 1984; Lightman and Young, 1987; Mogg and Samson, 1990; Samson et al. 1986, 1989]. However, there is little synaptic contact between serotonin fibers and dopaminergic cells. Hence, if direct inhibition of dopaminergic cells occurs, it is rather through this serotonin that volume transmission occurs in this region [Kiss and Halasz, 1986]. A wealth of scientific and clinical evidence also supports the concept that direct stimulation of GABAergic neurons in the vicinity of dopamine cells, the tuberoinfundibular–GABA (TI-GABA) system, is involved in modulating prolactin secretion in humans, possibly through serotoninergic stimulation of GABA interneurons via the 5HT1A membrane receptor, resulting in inhibition of TIDA cells and causing the tonic inhibition of prolactin release [Ondo and Dom, 1986; Wagner et al. 1994; Fuchs et al. 1984; Mirkes and Bethea, 2001] (Figure 1).

Putative pathways through which fluoxetine and norfluoxetine may stimulate prolactin releasing factors (PRF) such as oxytocin (OT) and vasoactive intestinal peptide (VIP) via 5HT2A receptor predominantly on PVN (neurosecretory magnocellular cell) via direct stimulation of GABA which in turn block the dopamine D2 receptor in the tuberoinfundibular region and block dopamine release [predominant prolactin inhibitory factor (PIF)] and by elevating postsynaptic-receptor-mediated serotonin release indirectly through TIDA and PVN cell stimulation. This ultimately leads to uninhibited action on PRF. Rest SSRIs mediate their action only through presynaptic elevation of serotonin level via serotonergic projection from dorsal raphe in resemblance fashion.
With the exception of fluoxetine, SSRIs generally have little interaction with postsynaptic 5-HT receptors and are mainly thought to work through presynaptic mechanism. In general, all SSRIs exert their therapeutic actions and their undesirable effects by increasing synaptic serotonin concentration, where re-uptake is blocked and serotonin release is disinhibited. Ultimately, increasing serotonin in desirable pathways and at targeted receptor subtypes leads to well-known therapeutic actions of all SSRIs and
The incidence and prevalence of hyperprolactinemia in patients taking SSRIs will be important to pursue in future controlled studies. Based on cumulative case reports, all SSRIs have the potential to cause elevation of basal prolactine. This observation was recently confirmed by the French Pharmacovigilance Database Study, an epidemiological study that investigated the rates of hyperprolactinemia induced by multiple prescription medications from 1985–2000 [Petit et al. 2003]. Of the total of 159 cases of drug induced hyperprolactinemia studied, 17% had been induced by SSRIs, which included sertraline [odds ratio (OR) 15.74], fluoxetine (OR 49), paroxetine (OR 8.10), fluvoxamine (OR 5.96), and citalopram (OR 3.62). Citalopram was the only SSRI not to reach any statistical significance. The available data indicate that SSRI-induced hyperprolactinemia is a class related effect [Petit et al. 2003].
If we change our notion here towards the management strategy of each individual patient as depicted in cases one and four, hyperprolactinemia and associated amenorrhea resolved within 2 months of withdrawal of fluoxetine and both the patients responded well to sertraline. In case three escitalopram was tried initially without any positive impact on the patient’s condition, rather it resulted in further elevation of prolactin. The resolution of hyperprolactinemia-associated symptoms was achieved after 3 weeks of escitalopram withdrawal and almost 5 months of fluoxetine discontinuation and the patient remained psychiatrically stable while being maintained on venlafaxine. In cases two, three, and five, normalization of serum prolactin level, resumption of normal menstruation and cessation of spontaneous milk discharge were achieved by chemically and pharmacologically different category drugs mirtazapine and venlafaxine. Resolution was also reported within a month of discontinuation of fluoxetine in patients two and five.
The important noticeable features of all these single case reports are the delayed onset time for hyperprolactinemia (0.5–1.0 months) and variable recovery time after fluoxetine withdrawal (between three weeks and two months). In cases two, three, and five, the final management strategy justifies the superiority of mirtazapine and venlafaxine over fluoxetine in respect to prolactin releasing pathway. However, in cases one and four, management was achieved by sertraline, another SSRI, without affecting recoveries from hyperprolactinemia. In patient three, escitalopram was tried initially without any benefit over fluoxetine with regards to alleviation of hyperprolactinemia and associated features; instead, escitalopram elevated prolactin level further.
These interesting observations raise certain important questions. First, whether SSRIs, with their own pharmacological individuality are of one class with different members? Second, what is the reason for prolonged onset time for symptoms to be appearing after fluoxetine administration and why these patients had delayed recovery after fluoxetine withdrawal? Third, does fluoxetine possess any special pharmacological property with regards to pharmacodynamic and pharmacokinetic aspects of individual patient, which might have contributed to these prolongations?
Although all of the SSRIs clearly share the same mechanism of actions, therapeutic profiles, and overall spectrum of side effects, individual patients often react very differently to one particular SSRI than the other. This might be the reason why cases one and four both responded well to sertraline, resulting in rectification of hyperprolactinemia attributed clinical consequences, whereas in case three, escitalopram failed to exert any therapeutic benefit over fluoxetine. The reality is that one or other individuals of the SSRIs has pharmacologic actions within one or two orders of magnitude of their potencies for serotonin reuptake inhibition over a wide variety of receptors and enzymes. Furthermore, no two SSRIs have identical secondary pharmacological characteristics. These actions can include norepinephrine reuptake blockade, dopamine reuptake blockade, serotonin agonist actions, muscarinic cholinergic antagonist actions, interaction with the sigma receptors, inhibition of enzyme nitric oxide synthetase, and inhibition of the cytochrome P450 enzymes 1A2, 2D6, and 3A4. Whether these secondary binding profiles can account for the differences in efficacy and tolerability in individual patients remain to be explored. To find the answers to the remaining questions we have focused on certain exceptions of fluoxetine pharmacodynamics. Fluoxetine imparts its serotonergic action by interacting with postsynaptic 5-HT receptors where as the rest of the members of this SSRI family are mainly thought to exert their therapeutic actions through presynaptic mechanisms. This specific exception could clearly be correlated with the fact that serotonin can stimulate prolactin release directly via postsynaptic 5-HT receptors in the hypothalamus [Nicholas et al. 1998; Stahl, 2000; Goodnick and Goldstein, 1998; Hyttel, 1984; Tatsumi et al. 1997; Dubovsky, 1994].
In one study, oral fluoxetine administration (5 mg/kg) for 21 days elevated the cerebrospinal fluid (CSF) GABA levels by approximately two-fold (
It is also pertinent here to examine the pharmacokinetics of fluoxetine. Fluoxetine is almost completely absorbed after oral administration, but its systemic availability is reduced because of extensive first-pass metabolism in the liver. Owing to its lipophilic character, it has a larger volume of distribution and accumulates in several tissues. Fluoxetine is extensively metabolized in the liver. The only identified biologically equipotent and active metabolite, norfluoxetine, is formed by demethylation of fluoxetine. The primary route of elimination is largely through oxidative metabolism and conjugation, but more than half of the metabolic end products are unknown. Evidence from several
Therefore, the concentration of the drug and its active metabolite in the blood continues to grow through the first few weeks of treatment, and their steady concentration in the blood is achieved only after 4 weeks [Perez et al. 2001; Brunswick et al. 2002]. Moreover, the brain concentration of fluoxetine and its metabolites keep on increasing through at least the first 5 weeks of treatment [Henry et al. 2005]. This means that the full benefits of the current dose received by a patient are not realized for at least a month after initiation. For example, in one 6-week study, the median time for achieving consistent response was 29 days [Perez et al. 2001]. Likewise, complete excretion of the drug may also take several weeks. During the first week after treatment discontinuation, the brain concentration of fluoxetine decreases by only 50% [Guze and Gitlin, 1994], the blood level of norfluoxetine after 4 weeks following treatment discontinuation is about 80% of the level registered by the end of the first treatment week, and norfluoxetine was still detectable in blood after 7 weeks after the discontinuation [Perez et al. 2001].
This extended half-life appears to protect against sporadic noncompliances [Guze and Gitlin, 1994] and against the occurrence of several withdrawal phenomenon of fluoxetine over other SSRIs. However, in the context of this discussion, the long half-life of fluoxetine and its desmethyl metabolite may account for such late onset hyperprolactinemia and resulted in prolonged recovery time after fluoxetine discontinuation in all of these patients. The prominence of clinical implications of inter-individual variability and the possibility of impact of genetic polymorphism cannot be ruled out in this context. However, we are not aware of any study conducted to date addressing these relevant issues. By considering all of these aspects of discussion an attempt was made to depict putative mechanism of increasing prolactin level by fluoxetine (Figure 1).
The exact insight of increased risk for neuroendocrine abnormalities is uncertain, but their prevalence must be correlated as the classic pathological manifestations of hyperprolactinemia are galactorrhea, amenorrhea, infertility, and decreased libido in women, and erectile dysfunction, hypogonadism, and infertility in males. The long-term clinical sequelae of hyperprolactinemia are obscure and can lead to deleterious chronic pathological conditions such as osteopenia both in men and women, and the possibility of increased risk of breast cancer in women. Association of prolactin levels with impaired fertility, decreased bone density, and breast cancer are yet to be established. The likelihood of developing these perilous neuroendocrinological complications should also be an important consideration as these unpredictable conditions might pose a major public negative health impact [Segal et al. 1979; Seppala, 1978; Gomez et al. 1977; Carter et al. 1978].
The growing number of individual case reports could be signifying a strong association of SSRIs with prolactin abnormalities. Therefore, knowledge of their effect on prolactin homeostasis is extremely important. As the large studies conducted in premenopausal women treated with SSRIs have not addressed the impact and prevalence of hyperprolactinemia and its further deleterious consequences, and our observations include only five patients, individual variation in the effect of SSRIs on the HPA axis cannot be ruled out. So, larger studies are needed to investigate the effect on prolactin regulation and dopamine regulation. Pharmacogenetic studies involving fluoxetine metabolism are essential. As cytochrome P450 isoforms involving fluoxetine metabolism exhibit wide genetic polymorphisms, accumulation of metabolites and consequent (endocrine) effects in a common population pool such as this might result from such genetic variations in fluoxetine metabolism.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The author declares that there are no conflicts of interest.
