Abstract
Objective
We investigated the clinical efficacy and safety of switching to risperidone long-acting injection (RLAI) in older patients with schizophrenia receiving oral risperidone.
Methods
The subjects were 48 inpatients who had been diagnosed with schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition. Their clinical symptoms were assessed using the Positive and Negative Syndrome Scale and the Clinical Global Impression – Severity of Illness scale, and their safety was assessed using the Drug-Induced Extrapyramidal Symptoms Scale (DIEPSS), body weight, body mass index, and blood biochemistry tests.
Results
No significant differences in clinical symptom improvement efficacy were seen between the group switched to RLAI and the control group. The mean changes from baseline on the DIEPSS total score and prolactin level were significantly greater in the older group switched to RLAI than in the control group. Furthermore, in older patients, RLAI allowed the dosage of the concomitant medication to be significantly reduced compared with the control group.
Conclusion
The results of this study suggest that switching older patients from oral risperidone to RLAI may result in superior efficacy and safety, and may also make it possible to reduce the dosage of the concomitant medication.
Introduction
Older patients generally have reduced liver and kidney function, are more susceptible to adverse drug reactions, and are more likely to experience a reduction in their activities of daily living (ADL) and in their quality of life (QOL) as a result of drug-induced adverse drug reactions. In older patients with schizophrenia, moreover, a decreased capacity for reality testing combined with a lack of insight make such patients more likely to lose their medication or make mistakes when taking their medication, resulting in severely inadequate treatment adherence.
Therefore, when using drug therapy in older patients with schizophrenia, it is important to ensure that the patients take their medication to prevent adverse drug reactions as much as possible, and to keep the dosing regimen uncomplicated.
Against this background, risperidone long-acting injection (RLAI) has been reported to yield improvement in clinical symptoms and extrapyramidal symptom rating scale scores in patients switched to RLAI from oral first-generation antipsychotics, first-generation long-acting injectable formulations, or oral risperidone [Chue et al. 2005; Kissling et al. 2007; Lasser et al. 2004; Schmauss et al. 2007]. However, in Japan, there are virtually no reports of RLAI being administered to older patients with schizophrenia to study its efficacy and safety.
In this study, we investigated the clinical efficacy and safety of switching to RLAI in older patients with schizophrenia receiving oral risperidone. We also investigated whether or not there were any differences in efficacy or safety compared with a group of younger patients who were switched to RLAI.
Methods
Subjects
The subjects were 48 patients who were being treated on an inpatient basis at the psychiatry departments of Tanzawa Hospital or Seimo Hospital and had been diagnosed with schizophrenia according to the Diagnostic and Statistical Manual of Mental Disorders, fourth edition (DSM-IV). Patients with chronic schizophrenia with persistent symptoms receiving oral risperidone monotherapy were enrolled into this study. Inclusion criteria were patients with schizophrenia according to the diagnostic criteria of the DSM-IV who had been treated with a stable dose of oral risperidone monotherapy for at least 6 months. There were no exclusion criteria. Study subjects were switched to RLAI from oral risperidone and were stratified into an older group of 18 patients, 60 years of age or older, and a group of 13 patients younger than 60 years of age. In addition, a group of 17 older patients was established as a control group who continued receiving oral risperidone. The patients were receiving oral risperidone monotherapy before they switched to RLAI. The results were the same as for the control group.
All the subjects who participated in the study were inpatients whose treatment compliance had been confirmed by a nurse and was thus assured. They were required to be symptomatically stable, as judged by the treating psychiatrist, to be able to complete all the clinical measures.
The study was an open-label, flexible-dose, naturalistic observational trial of patients with schizophrenia receiving oral risperidone but who required a change in their medication because of persistent symptoms or troublesome side effects. Patients had high scores on the Positive and Negative Syndrome Scale (PANSS), even though they were considered stable. However, these patients could not be considered refractory to antipsychotics.
Switching method
Subjects were switched to RLAI from their previous therapeutic medications as follows. They were given an initial dose of RLAI 25 mg in addition to their previous therapeutic medications, and received gluteal injections at 2-week intervals, alternating from the left to the right side. After 4 weeks, by which point the blood concentration had started to rise, doses of previous therapeutic medications were reduced so that the subjects would be receiving total doses equivalent to those of their previous therapeutic medications. After 8 weeks, the RLAI dose was increased as necessary to optimize the dose, and all subjects were receiving RLAI monotherapy. It was therefore possible to investigate the intrinsic effect of RLAI on cognitive function. Following RLAI optimal dose adjustment, wherever possible, the doses of any concomitant medications, including anti-Parkinson medications, were reduced.
When switching to RLAI, the calculation table of Inagaki and Inada was used as a guideline for calculating antipsychotic equivalents [Inagaki and Inada, 2010], and daily doses were calculated in terms of risperidone equivalents.
Only patients who had received a full explanation of the purpose and methods of the study and had provided voluntary informed written consent to participate were enrolled. Patient confidentiality was afforded all due consideration, as were ethical considerations.
Assessment methods
Clinical assessments were performed at baseline and at 24 weeks by the psychiatrist who was providing the therapy. There were no reliability tests for those who applied the PANSS [Kay et al. 1987], the Clinical Global Impression – Severity of Illness scale (CGI-S) [Guy and Rockville, 1976], and the Drug-Induced Extrapyramidal Symptoms Scale (DIEPSS) [Inada, 1996; Inada et al. 1996]. However, assessor training was provided to ensure a certain degree of reliability. The PANSS and the CGI-S were used to investigate efficacy. Meanwhile, the DIEPSS, body weight, body mass index (BMI), and blood biochemistry tests (total cholesterol, triglycerides, prolactin) were used to investigate safety. Injection site reactions were also assessed.
Statistical analysis
The Wilcoxon signed rank sum test was used to analyze efficacy and safety before and after RLAI switching. This test was also used to analyze differences between the older group and the younger and control groups in terms of efficacy or safety. The significance level was set at p < 0.05.
Results
No significant differences were seen between the older group switched to RLAI and the control group in the baseline PANSS total score, the baseline CGI-S score, the baseline DIEPSS total score, the baseline body weight, BMI, any of the blood biochemistry tests, the mean daily dose of the previous treatment drug, the mean duration of illness, or the mean age of the patients (Table 1).
Subject characteristics.

Data are presented as mean ± SD.
CGI-S, Clinical Global Impression – Severity of Illness scale; DIEPSS, Drug-Induced Extrapyramidal Symptoms Scale; PANSS, Positive and Negative Syndrome Scale.
The PANSS total score and the PANSS subscales decreased significantly from baseline in both the older and younger groups switched to RLAI, but no significant differences were seen between the two groups (Table 2). In addition, no significant differences in clinical symptom improvement efficacy were seen between the older group switched to RLAI and the control group. The CGI-S score decreased significantly from baseline in the older and younger groups switched to RLAI, but no significant differences were seen between the two groups (Table 2). However, the mean change from baseline in the CGI-S score was significantly greater in the older group switched to RLAI than in the control group. The DIEPSS total score decreased significantly from baseline in both the older and younger groups switched to RLAI, but no significant difference was seen between the two groups (Table 2). However, the mean change from baseline in the DIEPSS total score was significantly greater in the older group switched to RLAI than in the control group.
Efficacy and safety.

Data are presented as mean (SD).
p < 0.005 versus baseline, **p < 0.05 versus baseline.
BMI, body mass index; CGI-S, Clinical Global Impression – Severity of Illness scale; DIEPSS, Drug-Induced Extrapyramidal Symptoms Scale; PANSS, Positive and Negative Syndrome Scale.
The mean changes from baseline in body weight and BMI were small in all groups (Table 2). The total cholesterol and triglyceride levels decreased from baseline in both the older and younger groups switched to RLAI, but no significant differences were seen between the two groups (Table 2). In addition, the mean changes from baseline in the total cholesterol and triglyceride levels were substantial in the older group switched to RLAI and the control group, yet no significant difference was found between the two groups. The mean prolactin level (mg/ml) decreased significantly from baseline in both the older and younger groups switched to RLAI, but no significant difference was seen between the two groups (Table 2). However, the mean changes from baseline in the prolactin level were significantly greater in the older group switched to RLAI than in the control group.
The incidence of adverse events associated with injection site reactions was 22.6% (7 of 31); all of these adverse events were injection site pain; no redness, swelling, or induration was observed. Furthermore, all instances of injection site pain were mild in terms of severity and, in each case, the pain emerged at the time of the first or second RLAI administration, and subsequently resolved. Furthermore, in this study, no serious adverse events such as suicide attempt, neuroleptic malignant syndrome, or tardive dyskinesia occurred.
The mean risperidone equivalent dose and the mean biperiden equivalent dose were a significant decrease from baseline in both the older and younger groups switched to RLAI, but no significant differences were seen between the two groups (Table 3). However, the mean change from baseline in the risperidone equivalent dose and the biperiden equivalent dose was significantly lower in the older group switched to RLAI than in the control group. The mean diazepam equivalent dose was a significant decrease from baseline in both the older and younger groups switched to RLAI, but no significant difference was seen between the two groups (Table 3). However, the mean change from baseline in the diazepam equivalent dose was significantly lower in the older group switched to RLAI than in the control group. No significant difference was seen in the mean change from baseline in the mean doses of sennoside and magnesium oxide between the older and younger groups switched to RLAI. However, the mean change from baseline in the dose of sennoside was significantly lower in the older group switched to RLAI than in the control group.
Change of risperidone equivalent dose and concomitant medications.
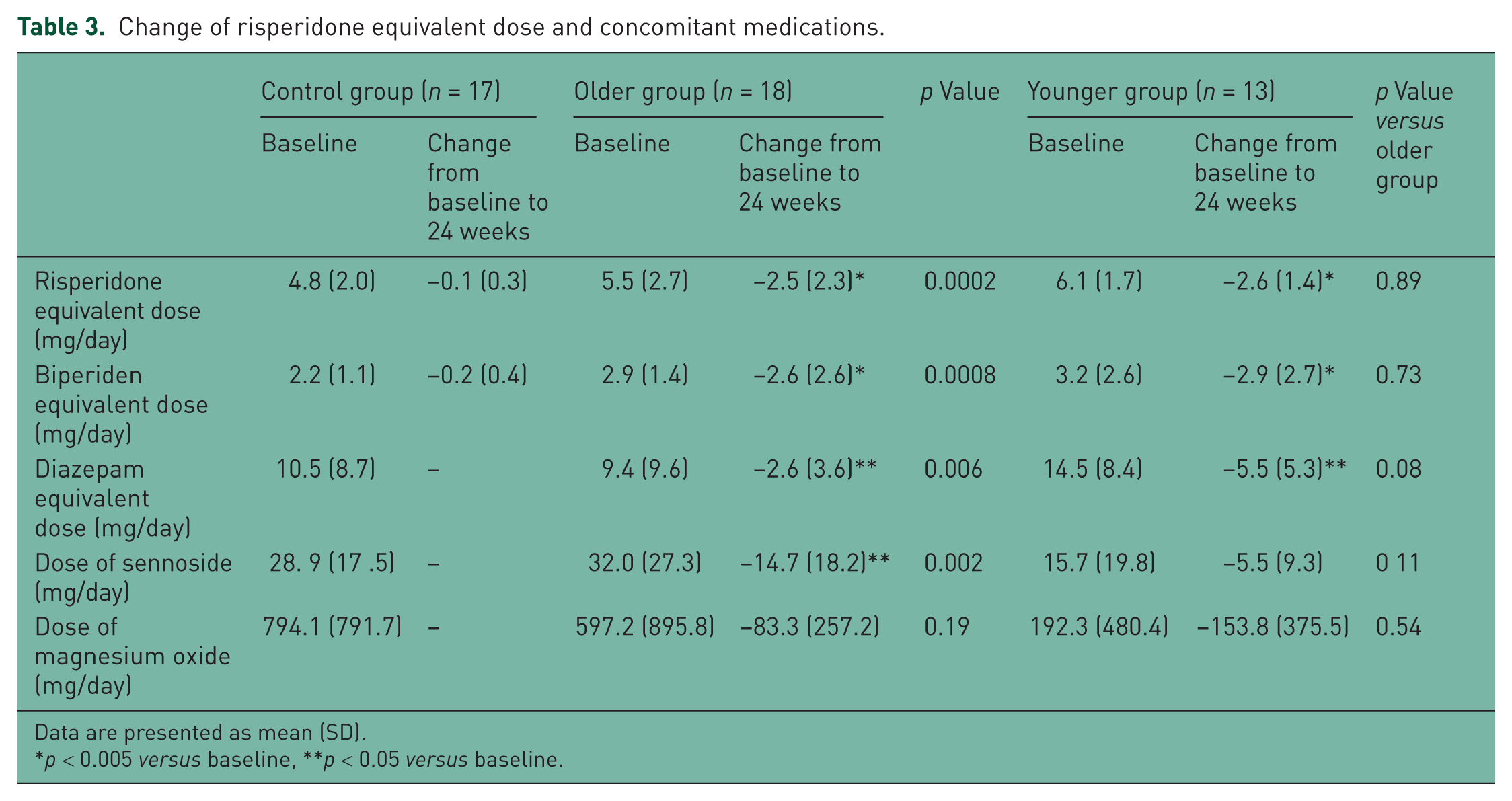
Data are presented as mean (SD).
p < 0.005 versus baseline, **p < 0.05 versus baseline.
Discussion
No differences were seen in efficacy in the improvement of clinical symptoms between inpatients with schizophrenia switched to RLAI for 24 weeks and those who continued to receive oral risperidone (control group). The results of this study suggest that switching from oral risperidone to RLAI resulted in similar clinical efficacy in both older and younger patients. Our findings are therefore consistent with the results of other clinical studies conducted to date [Kamijima et al. 2009; Kane et al. 2003; Lasser et al. 2004]. However, one previous study suggested that RLAI resulted in significantly lower serum concentrations of risperidone plus 9-OH risperidone than oral risperidone [Nesvag et al. 2006]. Furthermore, this may be a rather poor indication of the antipsychotic efficacy of risperidone. Although it is not known why the results of the present study differ from those of the previous study, one possibility is that the results may have been influenced by older patients with lower average body weight and racial differences. In the present study all patients initiated on treatment with RLAI continued for 24 weeks. However, in a previous study a small proportion of patients initiated on treatment with RLAI continued for 3 years [Taylor et al. 2009a] and the median number of days in hospital increased significantly in the 3 years after RLAI initiation [Taylor et al. 2009b]. Although it is not known why the results of the present study differ from the results of the previous study, one possibility is that they may have been influenced by the shorter study duration and symptomatically stable inpatients. The study results also suggest that switching from oral risperidone to RLAI prevents the emergence of drug-induced extrapyramidal symptoms, which is normally one of the risk factors for reduced ADL in older patients. This finding was consistent in both the older and younger groups compared with the control group who continued on oral risperidone. These findings are consistent with those of previous research.
Patients with schizophrenia are frequently obese, and over time, weight gain increases the risk of cardiovascular lesions, and is a factor that can contribute to a worse vital prognosis. This issue has therefore become of particular concern [Haupt, 2006; Newcomer and Haupt, 2006]. Our findings showed that switching from oral risperidone to RLAI, either in older or younger patients, resulted in virtually no change in body weight. In addition, BMI remained below 23, the level for normal body weight, suggesting that the risk of weight gain with RLAI switching is not high. These results are also consistent with those of previous research [Chue et al. 2005; Lasser et al. 2004; Newcomer and Haupt, 2006]. Switching from oral risperidone to RLAI resulted in similar (though not significant) reductions in total cholesterol, which is a risk factor for cardiovascular disease in both older and younger patients. Switching from oral risperidone to RLAI also resulted in a reduction (though not significant) in neutral lipids, which are an independent risk factor for coronary artery disease, in older patients compared with the control group who continued on oral risperidone. The results of this study therefore show that in older and younger patients switching from oral risperidone to RLAI resulted in a smaller effect on the lipid-metabolizing system than in the control group that was continued on oral risperidone. These findings are also consistent with those of previous research [Lasser et al. 2004; Tschoner et al. 2009].
Hyperprolactinaemia is a factor that can result in sexual dysfunction, one of the primary causes of reduced QOL [Baggaley, 2008], and risperidone-induced hyperprolactinaemia is dose dependent [Dossenbach et al. 2006]. The results of this study suggest that, in older patients, switching from oral risperidone to RLAI, with the same reduction in the risperidone equivalent dose as in younger patients, may result in a significant reduction in prolactin levels compared with the control group who continued on oral risperidone.
In this study, we converted the doses of antipsychotic medications before and after RLAI switching to risperidone equivalents to investigate changes in the risperidone equivalent dose. The results show that the older patients who were switched realized a significant decrease compared with the control group, although no significant difference was found compared with the younger patients who were switched to RLAI. Since older patients generally have reduced liver and kidney function and are thus more susceptible to adverse drug reactions, every effort must be made to reduce the dosing levels that are used in older patients. RLAI switching was also confirmed to result in a reduction in the dose in an overseas clinical study [Schmauss et al. 2007].
The results of this study suggest that, in older patients, switching from oral risperidone to RLAI, with the same reduction in the risperidone equivalent dose as in younger patients, may alleviate adverse drug reactions, even if only slightly compared with the control group who continued on oral risperidone. These results are consistent with those of previous research.
In addition, in this study, we also used biperiden equivalent doses and diazepam equivalent doses for the anti-Parkinson drugs, anxiolytics and hypnotics before and after RLAI switching to investigate the changes in each of these equivalent doses. Particularly in older patients, biperiden and diazepam are known to impair cognitive function, and older patients receiving these drugs must be closely observed for signs of delirium. The results of this study showed that switching older patients from oral risperidone to RLAI prevents extrapyramidal symptoms, which are risk factors for reduced ADL, compared with the control group who continued on oral risperidone. This suggests that it may be possible to reduce the equivalent doses of biperiden and diazepam, which result in cognitive impairment, in the same manner as in younger patients. Furthermore, the reason the diazepam equivalent dose was significantly lower in the younger group than in the older group may have been due to the difference in the dose at baseline.
Limitations
This study had a relatively small sample size, was a short-term study (24 weeks), and was an open-label, not a double-blind, study, so the possibility that bias was introduced to the results cannot be ruled out. Consequently, there are limits to the conclusions that can be drawn from this study. A double-blind, randomized, controlled study in older subjects may be necessary in the future to clarify the efficacy and safety of RLAI.
Conclusion
The results of this study suggest that switching older patients from oral risperidone to RLAI may result in superior efficacy and safety, and may also make it possible to reduce the dosage of concomitant medications.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
