Abstract
Case reports and pharmacologic theory suggest that some antidepressants can interfere with the hepatic metabolism of metoprolol by cytochrome P450 2D6 (CYP2D6), potentially increasing the risk of bradycardia. The objective of this study was to characterize the clinical consequences of this potential drug interaction at the population level. We conducted a population-based, nested case–control study of Ontario residents 66 years of age or older receiving metoprolol. Cases hospitalized for bradycardia were compared with matched controls (4:1) to explore the odds ratio for initiation of antidepressants that inhibit CYP2D6 (fluoxetine and paroxetine) and those that do not inhibit CYP2D6 (fluvoxamine, citalopram, venlafaxine, and sertraline) 30 days before hospitalization. From April 1997 to March 2009, we identified 332,254 older patients continuously receiving metoprolol, of whom 8232 (2.5%) were treated in hospital for bradycardia. The adjusted odds ratio for exposure to fluoxetine or paroxetine compared with other antidepressants 30 days prior to hospitalization for bradycardia was 0.76 (95% confidence interval 0.42–1.37). Among older patients receiving metoprolol, the initiation of antidepressants that inhibit CYP2D6 was not associated with a significant increase in the risk of bradycardia compared with antidepressants that do not inhibit CYP2D6.
Introduction
Antidepressants are commonly prescribed for the treatment of depression and anxiety disorders. Since their introduction, selective serotonin reuptake inhibitors (SSRIs) such as fluoxetine, sertraline, and citalopram, along with other antidepressants, such as venlafaxine, have become very popular, with prescription sales doubling in the United States between 1996 and 2005 [Olfson and Marcus, 2009] and now far exceeding those of heterocyclic antidepressants [Mamdani et al. 2000]. Reasons for the increase include their relative safety in overdose compared with heterocyclic antidepressants and the perception of an improved safety profile. Although newer SSRIs may be better tolerated than the older heterocyclic antidepressants, they may still cause adverse effects in isolation [Murphy et al. 2008; Thomas and Ellingrod, 2009], and can interact with other medications by inhibiting various isoenzymes of the cytochrome P450 (CYP) enzyme system [Greenblatt et al. 1998; Kelly et al. 2010; Spina et al. 2003].
Depression is especially common in patients with cardiovascular disease [Schleifer et al. 1989], and metoprolol is commonly prescribed to patients with ischemic heart disease. Since the initial finding that depression is associated with increased mortality after acute myocardial infarction [Frasure-Smith et al. 1995], the association between depression and cardiovascular disease has also assumed a high profile [Bush et al. 2005], and has been the subject of a large, randomized controlled trial showing the safety of sertraline in patients with cardiovascular disease [Glassman et al. 2002]. Indeed, increased awareness of the implications of depression in cardiovascular illness has likely increased the rate of antidepressant prescriptions in patients with coronary disease.
Metoprolol toxicity manifests primarily as conduction disturbances, generally including bradycardia. Metoprolol is metabolized primarily by CYP2D6, a highly polymorphic enzyme inactive in roughly 7% of white people. People exhibiting this ‘poor metabolizer’ phenotype display more metoprolol-related adverse events [Wuttke et al. 2002]. This is relevant because fluoxetine and paroxetine are potent inhibitors of CYP2D6 [Alfaro et al. 2000; Lam et al. 2002]. Indeed, the initiation of paroxetine in patients receiving metoprolol could provoke metoprolol-related bradycardia, as has been described in case reports [Goryachkina et al. 2008; Onalan et al. 2008]. Metoprolol is frequently prescribed with fluoxetine or paroxetine [Molden et al. 2005] and
We speculated that, among older patients receiving metoprolol, the initiation of antidepressants that inhibit CYP2D6 would be associated with an increased risk of bradycardia-related hospital visits. To test this hypothesis, we conducted a nested case–control study examining the risk of bradycardia in older patients taking metoprolol who recently commenced an antidepressant.
Methods
Setting and design
We conducted a nested case–control analysis of multiple linked healthcare databases spanning 13 years (1 April 1997 to 31 March 2009) in Ontario, Canada. Ontario is Canada’s most populous province with a registered population of 13,069,200 in 2009, of whom 1,787,900 were 65 years of age or older. Ontario’s older residents have universal access to hospital care, physician services, and prescription drug coverage. This research was approved by the Sunnybrook Health Sciences Centre Ethics Review Board.
Data sources
We identified prescriptions for metoprolol and antidepressants from the Ontario Drug Benefit Program, which records prescription medication dispensed to Ontario residents 65 years of age and older. Admissions for bradycardia were identified from the Canadian Institute for Health Information (CIHI) Discharge Abstract Database, which contains a detailed record of all hospital admissions in Ontario. Emergency department (ED) visits were obtained from the CIHI National Ambulatory Care Reporting System, which contains detailed information for all ED visits in Ontario. The Ontario Registered Persons Database contains basic demographic information for each Ontario resident. These databases are routinely linked to study drug safety, including the clinical consequences of drug–drug interactions [Fischer et al. 2010; Juurlink et al. 2009; Kelly et al. 2010; Kurdyak et al. 2005]. All analyses were conducted anonymously using an encrypted version of the individual health card number.
Observation period
We studied older patients whose prescription records allowed us to define a period of continuous metoprolol use. For each patient, observation in the cohort began with the first metoprolol prescription following their 66th birthday. The observation period ended with a hospital visit for bradycardia, death, discontinuation of metoprolol, addition of another beta blocker, or the end of the study period, whichever came first. To ensure adherence to metoprolol, we employed an algorithm in which continuous metoprolol use was defined by refills of the drug at intervals not exceeding 1.5 times the days’ supply of the preceding prescription, as done previously [Fischer et al. 2010; Juurlink et al. 2009; Kelly et al. 2010; Kurdyak et al. 2005]. We did not study patients during their first year of eligibility for prescription drug coverage (age 65 years) to avoid incomplete medication records.
Identification of cases and controls
Within the cohort of patients receiving metoprolol, we defined case patients as those who had undergone a hospital visit for bradycardia. A hospital visit was defined as an ED visit with bradycardia as the main diagnosis or hospitalization with bradycardia as a preadmission diagnosis. We used the International Classification of Diseases, 9th revision (ICD-9) codes 426.0, 426.1 and 427.8, and 10th revision (ICD-10) codes R001, I440–I443. The date of hospital admission or ED visit, whichever occurred first, served as the index date for all analyses. Only the first hospital visit was considered for patients with more than one hospitalization or ED visit for bradycardia. We selected up to four controls for each case from the cohort of patients continuously receiving metoprolol, matched for age (within 1 year of birth date) and sex. The selected controls were assigned the same index date as their matched cases.
Exposure to antidepressants
We grouped antidepressants into categories based on the predicted likelihood of interaction with metoprolol, based on CYP2D6 inhibition and
Any potential interaction is most likely to manifest shortly after the institution of SSRI therapy in patients otherwise stabilized on metoprolol. To restrict our analysis to new users of antidepressants, we required that cases and controls have exposure to only one antidepressant in the 30 days prior to the index date and no antidepressant in the preceding 6 months. By design, therefore, we defined exposure as new use of a single antidepressant within 30 days of the index date.
Statistical analysis
Baseline characteristics were calculated for case patients and their matched controls, and were compared using standardized differences. Standardized differences of <0.1 are not generally meaningful [Mamdani et al. 2005]. We used conditional logistic regression to estimate the odds ratio (OR) and 95% confidence interval (CI) for the association between bradycardia and new antidepressant exposure. In the primary analysis, non-CYP2D6-inhibiting antidepressants (fluvoxamine, citalopram, venlafaxine, and sertraline) comprised the reference group.
We used a multivariable conditional logistic regression model to adjust for potential confounders. Potential confounders included socioeconomic status (income quintiles) at cohort entry date, prior hospitalizations for bradycardia in the year prior to the index event, number of drugs prescribed in the past year [Schneeweiss et al. 2001], use of other CYP2D6-inhibiting drugs in the past 90 days, and use of negative chronotropic drugs (verapamil, diltiazem, and digoxin) in the past 90 days. Although bupropion is also a CYP2D6-inhibiting antidepressant [Kotlyar et al. 2005], we included it as a covariate with other CYP2D6 inhibitors rather than as an exposure antidepressant
Results
We identified 332,254 people who were treated with metoprolol, with a total of 630,600 person-years of continuous metoprolol treatment. Of these, 53.2% were women. The mean age of entry into the cohort was 74.8 years (SD ± 6.4) for men and 77.2 years (SD ± 7.2) for women. Use of antidepressants was common; overall, 13,013 people (3.9%) had been prescribed either fluoxetine or paroxetine (CYP2D6-inhibiting antidepressants) and 33,897 (10.2%) had been prescribed the non-CYP2D6-inhibiting antidepressants (fluvoxamine, citalopram, venlafaxine, and sertraline) at some time during metoprolol therapy.
Within the cohort of older patients receiving metoprolol, we identified 8232 cases hospitalized with bradycardia. Of these, 99 were newly treated with a study antidepressant in the 30 days preceding the index date. Within this group, 23 (23.2%) had received either fluoxetine or paroxetine (strong inhibitors of CYP2D6) and 76 (76.8%) had received one of fluvoxamine, citalopram, venlafaxine, or sertraline (Table 1).
Characteristics of cases and controls.

Variables with cell sizes ≤5 were not included because of the need to protect confidentiality of patients.
CYP2D6, cytochrome P450 2D6; IQR, interquartile range; SD, standard deviation.
Table 2 outlines the univariate and multivariate analyses for the risk of bradycardia following exposure to CYP2D6-inhibiting antidepressants relative to noninhibiting antidepressants. The univariate OR estimate of relative risk of bradycardia was 0.78 (95% CI 0.47–1.31;
Results from logistic regression of bradycardia outcome.*
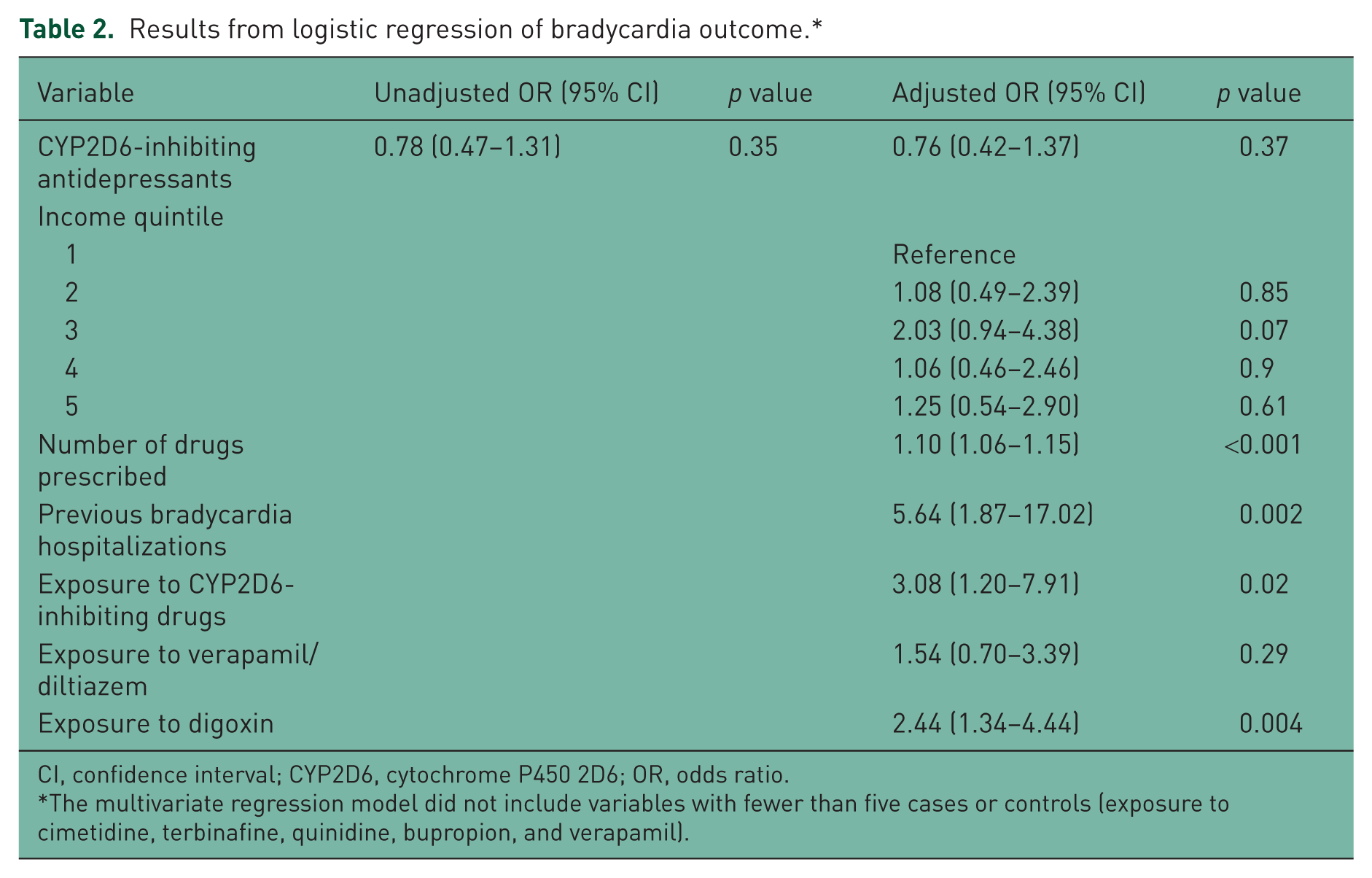
CI, confidence interval; CYP2D6, cytochrome P450 2D6; OR, odds ratio.
The multivariate regression model did not include variables with fewer than five cases or controls (exposure to cimetidine, terbinafine, quinidine, bupropion, and verapamil).
Discussion
Using population-based health services research methods, we found that in a large cohort of older patients receiving metoprolol, the addition of a CYP2D6-inhibiting antidepressant did not appreciably increase the risk of hospitalization for bradycardia compared with a non-CYP2D6-inhibiting antidepressant. To our knowledge, this is the first study using population health data to assess objective clinical consequences of the potential drug–drug interaction between metoprolol and antidepressants that inhibit CYP2D6. The absence of an elevated risk of bradycardia with fluoxetine and paroxetine is unexpected given the
The importance of our findings is highlighted by the high frequency of concomitant treatment with SSRI antidepressants and metoprolol in our sample. Overall, 14% of patients used any antidepressant, and 3.9% were exposed to one that inhibits CYP2D6. Concern about adverse events from drug–drug interactions is appropriate given evidence of an interaction [Alfaro et al. 2000; Belpaire et al. 1998; Hemeryck et al. 2001; Walley et al. 1993; Yoon et al. 2000] and the likelihood for exposure. The high antidepressant prescription rate is similar to other studies describing increasing use of antidepressants [Olfson and Marcus, 2009; Raymond et al. 2007]. However, surveys of physicians who prescribe in a general psychiatry outpatient setting suggest that the likelihood of drug interactions is not a major factor in selection of an antidepressant [Zimmerman et al. 2004].
Several limitations of our study merit emphasis. We used administrative data to identify exposures and outcomes and do not have access to serum drug levels, direct measures of heart rate, drug dose and other variable. This is important because an increased metoprolol level is a more proximate measure of the potential interaction between antidepressants and metoprolol. However, bradycardia is a clinically meaningful outcome measurable using administrative data. Some patients with bradycardia may not have sought medical attention or may have died in the prehospital setting. In addition, our results are derived from older patients; the generalizability of our findings to younger patients is unknown. However, most metoprolol users are likely to be represented by our population-based sample. We categorized antidepressants according to CYP2D6 inhibition based on
Some authors have discouraged the use of fluoxetine and paroxetine in patients already receiving metoprolol on the basis of
Footnotes
This study was supported by a grant from the Ontario Ministry of Health and Long-Term Care (MOHLTC) Drug Innovation Fund and the Institute for Clinical Evaluative Sciences (ICES), a non-profit research institute sponsored by the Ontario MOHLTC. Dr Paul Kurdyak is supported by a Canadian Institutes of Health Research (CIHR) New Investigator Award. We thank Brogan Inc., Ottawa for use of their Drug Product and Therapeutic Class Database.
Muhammad M. Mamdani has carried out consultancy work for Hoffman LA Roche Advisory Boards, GSK, Pfizer, Novartis, EI Lilly, Novo Nordisk, Astra Zeneca, and Bristol Myers Squid. The other authors have no conflict of interests to declare.
