Abstract
People with antisocial personality disorder (ASPD-nonPPD) are described as insensitive to others and as relentlessly pursuing their goals. A severe form of antisociality is observed in psychopathic personality disorder (ASPD-PPD). In the spectrum of emotional reactivity, people with ASPD-nonPPD present more emotional dysregulation, whereas people with ASPD-PPD exhibit a reduced or nonexistent emotional response. To personally engage people with ASPD-nonPPD and ASPD-PPD, we used emotionally charged autobiographical stimuli, specifically their self-defining memories (SDMs). As these participants exhibit high control over voluntary responses, we measured neurophysiological indicators (heart rate variability (HRV) and electrodermal activity (EDA)). In the resting task and the SDM task, people with ASPD-PPD had significantly higher HRV, suggesting higher emotion regulation abilities. Conversely, the EDA of people with ASPD-nonPPD and ASPD-PPD reflects less activation during the SDM task than when resting. We suggest that people with ASPD-PPD are more adaptive to stimuli that provide less emotional activation. Furthermore, the correlation analysis results suggested that the higher people with ASPD-PPD score on Factor 1 of the Psychopathy Checklist-Revised (PCL-R), the less emotional activation they exhibit. This low activation (EDA) associated with good emotion regulation abilities (HRV) is thought to be the signature of psychopathy.
Keywords
Introduction
People with antisocial personality disorder without psychopathic personality disorder (ASPD-nonPPD) are characterized by difficulties in maintaining social norms and by their manipulation, intimidation, and deception toward others. Being indifferent to the feelings, needs, and suffering of others is one of the key criteria for this diagnosis (DSM-5; American Psychiatric Association, 2013). Indeed, people with this type of personality disorder have an impaired moral conscience and are motivated solely by their own desires when making decisions without any consideration of the negative effects that their actions can have on others (Martin et al., 2019; Swann et al., 2009). People with ASPD-nonPPD are described as insensitive to others and as relentlessly pursuing their goals, resorting to aggression if needed. They exhibit low levels of remorse and empathy. They may be aware of the wrongness of their actions, but rationalize their behaviour (Viding, 2019). Thus, these people are often widely associated with violent delinquency (Shepherd et al., 2018; Velotti et al., 2018) and remain difficult to manage even in secure environments due to their irritability and aggression (Black et al., 2010; Martin et al., 2019). The ability to correctly assess nonverbal emotional information (facial expressions) contributes to the development of the interpersonal skills that are essential to prosocial adjustment (Decety et al., 2016; Dols & Russell, 2017; Scherer, 2005; Schönenberg et al., 2013); however, people with ASPD-nonPPD face difficulties, particularly in recognizing the specific emotions of fear, (Dawel et al., 2012; Marsh & Blair, 2008; Robinson et al., 2012) and anger (Schönenberg et al., 2013). They underestimate the emotion of fear, which can be explained by dysfunction of the specific neuronal structures underlying the processing of expressions of fear, especially the amygdala (Marsh & Blair, 2008). Regarding anger, these people can only adequately detect this emotion at high intensity. When confronted with ambiguous facial stimuli (e.g. fear and anger both present at 50% intensity), they overestimate the emotion of anger, which is consistent with a hostile attribution bias perspective and can provoke or exacerbate aggressive behaviours (Sato et al., 2009; Schönenberg & Jusyte, 2014). These findings mainly suggest that in the emotion-decoding paradigm, people with ASPD-nonPPD exhibit specific issues with anger processing. People who exhibit an early and chronic anger pattern may be at risk for antisocial characteristics when they reach adulthood if they lack good cognitive control skills (Hawes et al., 2016) and adaptive emotion regulation (Taylor & Workman, 2023).
Psychopathic personality disorder (ASPD-PPD) represents a severe form of antisociality. Indeed, in the DSM-5 (2013), psychopathy is considered in a subset of ASPD cases, and involves a ‘pattern of disregard and violation of the rights of others’ (APA, p. 645). Psychopathy is therefore a specifier of antisociality when there is an absence of fear and anxiety and excessive attention seeking (Taylor & Workman, 2023). Among forensic inpatients, the prevalence of ASPD-PPD is 7.3% assessed with the Psychopathy Checklist-Revised (PCL-R) cut-off score of 30 (Hare, 1991) and 26.6% with a cut-off score of 25, as suggested for use in European countries (Cooke & Michie, 1999; Pham et al., 2019). The PCL-R is a multi-dimensional assessment tool composed of two factors: the interpersonal (factor 1) and social deviance factors (factor 2); each of these factors has 4 facets. Thus, we consider ASPD-PPD a complex clinical construct that involves a combination of interpersonal, behavioural, and affective characteristics, including egocentrism and manipulation (facet 1); a lack of empathy and remorse or guilt, and insensitivity (facet 2); irresponsibility and impulsivity (facet 3); and an antisocial trajectory (facet 4) (Hare, 2003). More specifically, some empirical studies have suggested that ASPD-PPD does not inherently involve an absence of emotions (Derefinko, 2015; Hoppenbrouwers et al., 2016; Kosson et al., 2015; Pham et al., 2021). This incongruency of results could be related to the type of task used (e.g. static and dynamic stimuli), the sample type (e.g. correctional offenders, forensic patients), the statistical methods (e.g. means comparison, signal detection theory), the experimental design (e.g. consideration of attention, language, anxiety), and the selection criteria (e.g. unselected samples vs. extreme manifestations of ASPD-PPD) (Brook & Kosson, 2013; Faith et al., 2022; Hicks & Drislane, 2018; Mokros et al., 2015; Newman et al., 2010). Using functional neuroimaging, Birbaumer et al. (2005) showed that physiological, cortical, and subcortical fear deficits in people with PPD are linked to PCL-R factor 1. Notably, this factor is associated with a decrease in emotional reactivity to fearful stimuli. Moreover, people with ASPD-PPD may exhibit hostile attribution bias, dependent on the attention that these people direct toward the person expressing anger (Faith et al., 2022). Moreover, some of the emotional capacities in which people with ASPD-PPD have been considered deficient are, in fact, dynamic: they are present in certain contexts and absent as a result of specific manipulations of participants’ attention, cognitive load, or motivation (Nentjes et al., 2022). Shane and Groat (2018) suggested that people with ASPD-PPD may lack the motivation, rather than the ability, to respond to emotion-evoking stimuli.
Interestingly, the literature has displayed discrepancies between clinical and neurophysiological results.
In clinical studies, people with ASPD-PPD are described as lacking empathy but pretending to care about others and hiding their insensitive behaviours (i.e. callous-unemotional traits) (Martens, 2014). Regarding the important distinctions between people with ASPD-nonPPD and ASPD-PPD, antisociality lies at the opposite end of the spectrum of emotional reactivity in comparison to psychopathy (Yildirim & Derksen, 2013). Indeed, people with ASPD-nonPPD struggle with emotion regulation (i.e. emotion dysregulation, the inability to control emotions such as anger and frustration), but people with ASPD-PPD exhibit a reduced or nonexistent emotional response (i.e. emotional hyposensitivity) (Taylor & Workman, 2023).
In the present study, to engage people with ASPD-nonPPD and people with ASPD-PPD and to increase their interest in the task, we used emotionally charged, personal stimuli that drew on their autobiographical memories and, more specifically, their self-defining memories (SDMs). SDMs refer to specific autobiographical memories that are characterized by their vividness, intense emotional content, and frequent recall (Singer et al., 2013). These memories play a significant role in shaping one’s identity and help people to maintain a sense of coherence, particularly during challenging transitions or upheavals (Conway et al., 2004). Compared to other autobiographical memories, SDMs have higher subjective importance and evoke stronger emotional responses (Singer, 1992). Consequently, SDMs serve as a suitable avenue for exploring the expression of spontaneous emotions, providing an ecologically valid and personalized framework for analysing the emotions experienced by people with ASPD-PPD or ASPD-nonPPD.
Moreover, to the extent that facial, vocal, and postural behaviours or expressions are classically controlled in people with ASPD-nonPPD or with ASPD-PPD, we specifically chose to measure automatic neurophysiological responses that preclude the voluntary control bias of emotional expressions. The neurophysiological responses of people with ASPD-nonPPD or ASPD-PPD have been well studied in recent years. The meta-analysis by de Looff (2022) reviewed the neurovegetative responses observed in people with these two types of personality.
Regarding the parasympathetic nervous system, which is involved in homeostatic balance and the regulation of emotional experience or behavioural guidance under intense emotional arousal (Flórez et al., 2017), studies have shown that people with psychopathic traits are characterized by more maladaptive forms of emotion regulation than people without psychopathic traits. Among neurophysiological indicators, heart rate variability (HRV) is best suited to measure emotion regulation (Laborde et al., 2017; Mather & Thayer, 2018; Porges, 2009). Among people with ASPD-nonPPD, a low resting heart rate (HR) may reflect the absence of fear, which could predispose some people to commit antisocial behaviour in spite of potential dangers and could also prevent fear conditioning in early childhood regarding socializing punishments (Raine, 2002). Moreover, high tonic HRV is linked to low anxiety (Thayer, et al., 1996) as well as a high capacity for self-regulation and high flexibility (Thayer & Lane, 2000).
Regarding neurophysiological measures, incarcerated offenders with ASPD-PPD (based on a PCL-R total score >30) had lower HRV indices than incarcerated offenders with ASPD-nonPPD (Flórez et al., 2017) during an emotional implicit association test (IAT). In addition, the authors did not observe any differences between incarcerated offenders with ASPD-nonPPD and those with ASPD-PPD (based on PCL-R total score >25) or during the resting state. However, heterogenous results have been obtained during cognitive tasks. Indeed, interpersonal facet (facet 1) was positively associated with HRV at rest or during cognitive tasks such as the working memory test and continuous performance test (Hansen, et al., 2007).
Regarding the sympathetic nervous system, there has been a great deal of research in recent years on electrodermal activity (EDA), corresponds to activation, in personality disorders. Some studies have noted a correlation among brain activity, alertness, personality disorders and electrodermal activity. Offenders with ASPD-nonPPD and ASPD-PPD typically have reduced tonic and phasic electrodermal activity (Farrell, 2001). In addition, low autonomic nervous system activity is a biomarker of aggressive and antisocial behaviour (Baker et al., 2013). People with ASPD-nonPPD and people with ASPD-PPD exhibit low levels of skin conductance both at rest (Lorber, 2004) and during a task (Broom, 2012; Lorber, 2004).
It should also be noted that the aforementioned studies were conducted in correctional facilities, so it is necessary to conduct studies in forensic psychiatric settings. Studies have shown that the profiles of forensic patients differ in many respects, such as their psychiatric history. Indeed, 72.50% of forensic patients have received treatment for psychiatric illness, whereas 94% of people incarcerated have never received psychiatric treatment (Arin & Mengchuay, 2022).
In this study, we sought to examine neurophysiological indicators in forensic inpatients with ASPD-nonPPD and ASPD-PPD during the recall of personal emotional memories. To our knowledge, these physiological responses have never been measured under conditions of high personal involvement, which can elicit something that most closely resembles a natural, spontaneous response in terms of activation (EDA) and emotion regulation (HRV). The main objective of our study is to provide an objective measure of the emotional processing (both activation and emotion regulation) of personal memories and to refine knowledge about emotional functioning in people with ASPD-nonPPD and people with ASPD-PPD.
Referring to the neurophysiological literature, we developed the following hypotheses: a) Based on the findings of Flórez et al. (2017) and Broom (2012), during the resting task, people with ASPD-nonPPD and people with ASPD-PPD will exhibit the same emotion regulation abilities (reflected by HRV) and low activation (reflected by EDA). b) Based on the findings of Flórez et al. (2017) and Broom (2012), during the SDM task, people with ASPD-PPD will exhibit lower emotion regulation abilities (reflected by lower HRV) than people with ASPD-nonPPD and equivalent levels of activation (reflected by EDA).
Methods
Participants
For our experimental sample, we recruited male French-speaking forensic inpatients from the Secure Psychiatric Hospital of the Regional Psychiatric Centre ‘Les Marronniers’ in Tournai (Belgium). All patients are hospitalized according to the law of May 5, 2014, on the internment of persons that allows mentally disturbed offenders and criminals to benefit from a security measure that is intended both to protect society and to ensure that the interned person receives the care required by his or her condition with to the ultimate goal of reintegration into society (Loi relative à l’internement des personnes, 2014). The patients whom we recruited for this study were initially chosen by the referring psychologist. The ward psychiatrist team and the care team also gave a favourable opinion for each patient whom we approached.
The criteria we set for inclusion in our study were as follows: native French speakers; no intellectual impairment that might interfere with comprehending instructions; no use of alpha-blockers and/or beta-blockers; no neurological or medical problems that might impair memory abilities; and a sufficiently stabilized mental state to participate in psychological interviews and understand self-report questionnaires.
Clinical description of the three samples.

NB: ASPD-nonPPD: antisocial personality disorder without psychopathic personality disorder; ASPD-PPD: antisocial personality disorder with psychopathic personality disorder; FC: forensic control; PCL-R: psychopathic checklist-revised; n and χ2 refer to Section II according to the DSM-5; M (SD) and Kruskal‒Wallis H values refer to the PCL-R assessment. Significant results are highlighted in bold.
To determine the power of the current sample, we carried out a post hoc calculation and obtained a power of .84 for our total sample of 46 participants.
Compliance with ethical standards
The ethics committee of Les Marronniers Hospital in Tournai, Belgium, gave us approval to conduct this study (reference DV/VJ/PB/2020). The procedure was also conducted in accordance with the 1964 Declaration of Helsinki and its subsequent amendments or comparable ethical standards. All participants were recruited on a voluntary basis. We met with and recruited the participants one at a time. Before beginning the experimental procedure, the investigator again explained the purpose and instructions of the study to the participants to ensure that they understood the procedure. They then signed two consent forms; the investigator kept one of these and the participants kept the other, in which their personal identification code was indicated. With their identification code, they could access their personal information and request its destruction. No one requested that their personal data be destroyed.
Experimental procedure
The participants were placed in a testing room inside the forensic hospital. In this room, the participants were seated facing the evaluator. Before starting the experimental task, the participants were asked to complete an information sheet including their age, weight, height, time since awakening, smoking and cannabis use, education level, professional status, laterality, history of neurological disorders, and current medications. The time since awakening reflects the state of alertness sufficient to participate in the research protocol and allowed us to check the participant’s cognitive capacity before the experiment.
The resting task consisted of the presentation of a series of neutral emotional images (IAPS; Lang et al., 1997). After 2 minutes of observing this series, participants were asked to recall the images as accurately as possible, while their neurophysiological indicators (HRV and EDA) were recorded.
Then, the investigator proposed the SDM recall task to the participants. For the participants, the experimental task consisted of recalling five important and meaningful autobiographical memories. The participants were free to take the necessary time to recall the important memories that they wished to recount. After each SDM task, the participant was debriefed and allowed to relax back to their physiological baseline. After narrating all five memories, the participant was offered a moment of exchange and debriefing.
Measures
During the SDM narration, the physiological signals were recorded by a Biopac MP150 system. For electrocardiogram (ECG) recording, two pre-gelled EL503 electrodes were placed on the participants’ wrists. To analyse the activation of the parasympathetic system and the emotion regulation processes, we used the HRV calculated a posteriori by filtering the signal over the .15–.40 Hz frequency band (Laborde et al., 2017; Sztajzel, 2004). SDMs with a minimum duration of 1 minute were selected for statistical analysis of heart rate variability. We used the HF-normalized unit (HF-nu) as a measure of high frequencies (HF-nu = HF/(tot_power-VLF) (very low frequency) and the HF-log indicator, the natural-log transformed HF-HRV value, as a measure of high frequency power. In addition to these frequency-domain measures, we used a measure of the time domain, the root mean square of successive differences (RMSSD), which reflects vagal tone and is relatively free of respiratory influences.
For sympathetic system activity, the electrodermal response was measured at a room temperature of approximately 21–23 degrees Celsius. Two electrodes were placed on the distal phalanx of the index and middle fingers of the nondominant hand. A GEL 101 electrolyte mixture was applied inside the electrodes and on the distal phalanges. The EDA corresponds to the variation in secretion from eccrine glands, which are innervated exclusively by the sympathetic nervous system (Braithwaite et al., 2013) and is a measure of activation. The skin conductance response (SCR) corresponds to an increase with a minimum amplitude criterion of .05 μS over the tonic level of skin conductance, driven by the impulse of the sympathetic nervous system on the eccrine glands. We used the mean phasic driver indicator as the average of the phasic component on the electrodermal signal.
Data analysis
We performed the analyses using the Statistical Product and Service Solutions (SPSS) program, version 25.0 (IBM Corp, Armonk, NY, USA). We assessed the normality of the data distribution with the Shapiro-Wilk test. After examining the three possible data transformations (log(X), √X, 1/X) (Field, 2013), we opted to use more conservative nonparametric rank-based statistics for comparisons. The chi-square (χ2) test and Fisher’s exact test statistic were used to assess the relationships between groups and categorical variables such as types of mental disorders (DSM-5, 2013).
To test for intra-individual variability in the repetition of SDMs, we used the Friedman test. We observed that the fifth SDM significantly differed from the other SDMs, probably due to reduced attention. As each SDM data point is independent, and there was no intra-individual variability among each SDM, we retained the first four SDMs for each participant. Thus, all group comparisons were performed on massed data, considering each of the 4 recalled memories a different measure. Thus, we collected 184 observations among our three groups. Only 168 of these observations were analysable based on the quality of the recordings.
Effect size comparisons during the resting task among our participant groups (ASPD-nonPPD/ASPD-PPD/FC) and community people (CP) were possible using data from the literature (HF-nu and RMSSD: Dantas et al., 2018; HF-log: Niveatha et al., 2022; mean phasic driver: Imtiaz et al., 2020). Cohen’s d criterion was used: .20 = small, .50 = medium, .80 = large (Cohen, 2013). Moreover, intergroup comparisons were performed using the Kruskal‒Wallis H test (when comparing three groups) and the Mann‒Whitney U test (when comparing two groups) on the PCL-R total score and factors and neurophysiological responses (HF-nu, HF-log, RMSSD and mean phasic driver) for the resting and SDM tasks. We reported effect sizes (r = z/√n) only for pairwise comparisons (Field, 2013). Cohen’s r criterion was used: .10 = small, .30 = medium, .50 = large (Cohen, 2013).
Then, we calculated a massed delta values corresponding to the difference in neurophysiological indicators between the SDM task and the resting task rather than the difference in means between the two conditions. This calculation reflected the degree of arousal in the SDM task. These analyses were performed on 46 participants.
In order to reduce the risk of type I error, Dunn–Bonferroni correction was applied to the group comparisons. We opted for selective comparisons with alpha levels of .05/3 = .017 for 3-group comparisons and .05/2 = .025 for 2-group comparisons (Field, 2013).
In addition, we performed Pearson correlations between the PCL-R total score, factors 1 and 2 and the indicator responses (HF-nu, HF-log, RMSSD, and mean phasic driver). Finally, with regard to the moderate correlations between facets 1 and 2 (r = .48) as well as facets 3 and 4 (r = .52), partial correlations were performed between these facets and neurophysiological responses. Cohen’s r criterion was used: .10 = small, .30 = medium, .50 = large (Cohen, 2013).
Results
Comparisons of physiological responses
Resting task
Comparisons of HF-nu, HF-log, RMSSD, and mean phasic driver responses for the three samples during the resting and SDM tasks.
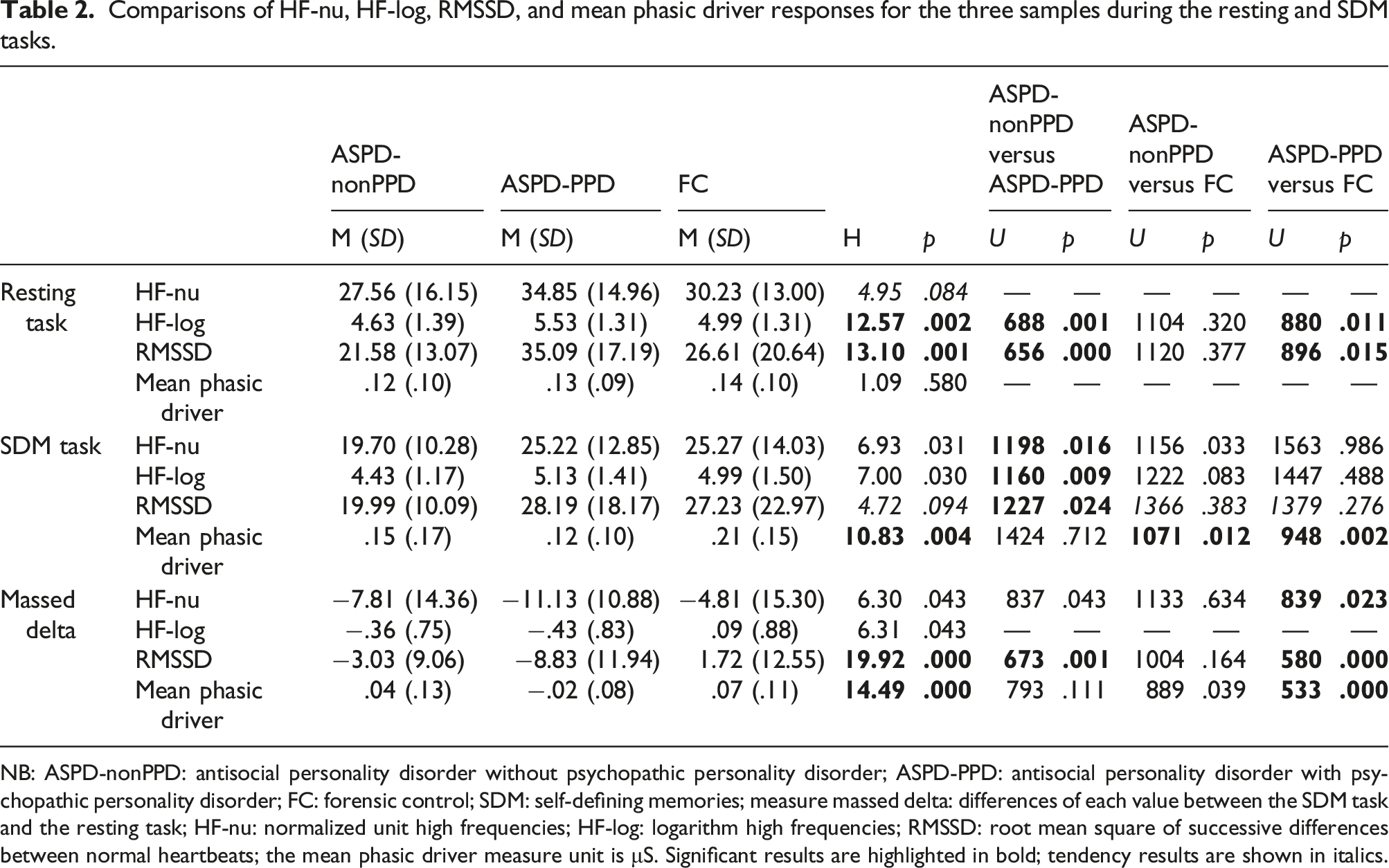
NB: ASPD-nonPPD: antisocial personality disorder without psychopathic personality disorder; ASPD-PPD: antisocial personality disorder with psychopathic personality disorder; FC: forensic control; SDM: self-defining memories; measure massed delta: differences of each value between the SDM task and the resting task; HF-nu: normalized unit high frequencies; HF-log: logarithm high frequencies; RMSSD: root mean square of successive differences between normal heartbeats; the mean phasic driver measure unit is μS. Significant results are highlighted in bold; tendency results are shown in italics.
However, when compared with the literature (CP), our participants had lower HF-nu values with a large Cohen’s d effect size (ASPD-nonPPD/CP = .87) or a medium Cohen’s d effect size (ASPD-PPD/CP = .47); FC/CP = .77). They had higher HF-log values than CP with a large Cohen’s d effect size (ASPD-nonPPD/CP = 2.91; ASPD-PPD/CP = 3.38; and FC/CP = 3.48). Only the ASPD-PPD group had higher RMSSD values than CP, with a moderate Cohen’s d effect size (ASPD-PPD/CP = .31). The ASPD-nonPPD group had lower RMSSD values than CP with a moderate Cohen’s d effect size (ASPD/CP = .59), and FCs, with a small Cohen’s d effect size (FC/CP = .19). On the other hand, they had higher mean phasic driver responses than the GP, with a large Cohen’s d effect size, (ASPD-nonPPD/CP = 1.07; ASPD-PPD/CP = 1.30; and FC/CP = 1.32).
SDM task
During the resting task (Figure 1), the HF-log responses showed a median value of 4.79 for people with ASPD-nonPPD, 6.06 for people with ASPD-PPD, and 4.75 for FCs. During the SDM task, the HF-log responses showed a median value of 4.65 for people with ASPD-nonPPD, 4.85 for people with ASPD-PPD and 4.80 for FCs. HF-log responses during the resting and SDM tasks.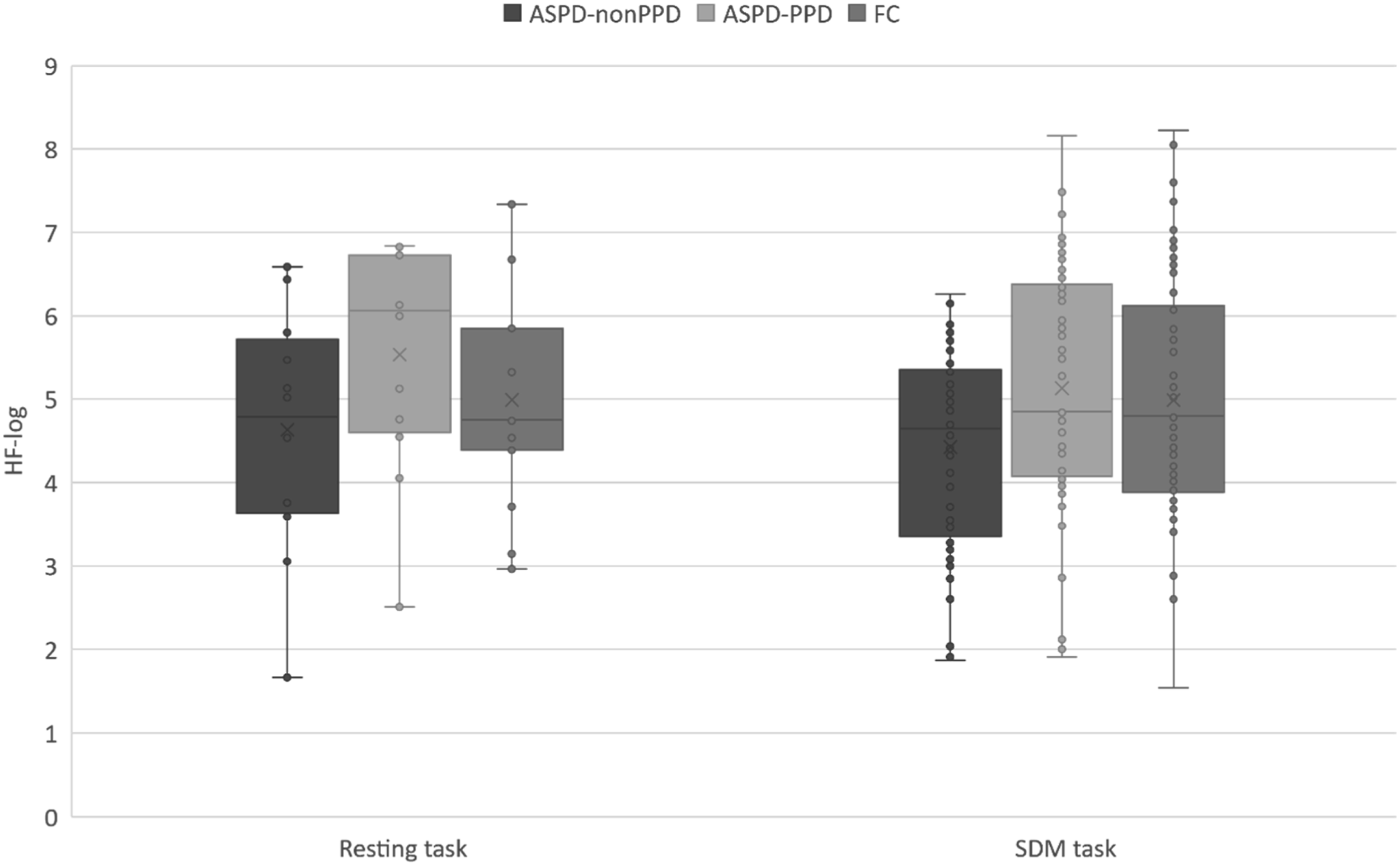
People with ASPD-PPD also had significantly higher HF-nu (r = .36) and HF-log values (r = .62) than people with ASPD-nonPPD (Table 2). In addition, FCs also had significantly higher HF-nu values than people with ASPD-nonPPD (r = .20) (Table 2).
During the resting task (Figure 2), the mean phasic driver values had a median of .08 for people with ASPD-nonPPD, .14 for people with ASPD-PPD, and .15 for FCs. During the SDM task, the mean phasic driver values had a median of .11 for people with ASPD-nonPPD, .11 for people with ASPD-PPD and .20 for FCs. Mean phasic driver responses during the resting and SDM tasks.
The mean phasic driver responses of FCs were significantly higher than those of the people with ASPD-PPD (r = .56) (Table 2).
Massed delta
Regarding the difference between the SDM task and the resting task (Massed delta; Table 2), people with ASPD-PPD had significantly higher difference for RMSSD (r = .60) than people with ASPD-nonPPD and higher difference for HF-nu (r = .41) and RMSSD (r = .75) than FCs. Conversely, the mean phasic driver value of FCs was significantly higher than that of people with ASPD-PPD (r = .73).
Regarding the correlation analysis, the PCL-R total score was negatively correlated with the Massed delta of the mean phasic driver (r = −.40; p = .034) and the Resting RMSSD (r = .38; p = .048). More specifically, Factor 1 was negatively correlated with the Massed delta mean phasic driver (r = −.47; p = .011) and the SDM-task mean phasic driver (r = −.44; p = .009) and positively correlated with the resting RMSSD (r = .38; p = .044). Regarding the partial correlation analysis on the facets, we found that Facet 1 was negatively correlated with the SDM-task mean phasic driver (r = −.40; p = .049). Facet 2 was positively correlated with the Resting HF-log (r = .50; p = .011) and the Resting RMSSD (r = .56; p = .003). Facet 3 was negatively correlated with the Massed delta HF-log (r = −.52; p = .010), the Massed delta RMSSD (r = −.62; p = .001), the Resting HF-log (r = −.54; p = .006) and the Resting RMSSD (r = −.50; p = .013).
Discussion
In this study, we examined the emotional responses of people with ASPD-nonPPD and people with ASPD-PPD using objective neurovegetative measures of sympathetic and parasympathetic nervous system activity elicited during the recall of autobiographical memories.
First, we hypothesized that during the resting task, people with ASPD-nonPPD and people with ASPD-PPD would both exhibit the same levels of emotion regulation (reflected by similar HRV). Our results showed that people with ASPD-PPD had significantly higher HF-log and RMSSD values than people with ASPD-nonPPD. During the resting task, people with ASPD-PPD exhibited better emotion regulation abilities than people with ASPD-nonPPD. These HRV results suggest that people with ASPD-PPD are more flexible and can more readily adapt to life events (Porges, 2009), exhibiting greater emotion regulation than people with ASPD-nonPPD. Furthermore, comparing our results with those obtained in the community samples (Dantas et al., 2017; Niveatha et al., 2022), we observed that people with ASPD-PPD had higher HF-log and RMSSD values, reflecting greater self-regulation and flexibility. Indeed, a growing body of empirical evidence suggests that people with higher resting vagal-mediated heart rate variability (VmHRV) may adaptively regulate negative emotions and behavioural emotional responses (Melzig et al., 2009; Thayer et al., 2012). Williams et al. (2015) have demonstrated a negative relationship between resting VmHRV and scores on the Difficulties in Emotion Regulation Scale (DERS) such that lower resting VmHRV was associated with greater difficulties in emotion regulation, particularly a lack of emotional clarity and impulse control.
During the SDM task, we hypothesized that people with ASPD-PPD would exhibit lower emotion regulation abilities, whereas people with ASPD-nonPPD would exhibit higher emotion regulation abilities. Regarding the three HRV indicators, we found that people with ASPD-PPD did not differ from FCs and had significantly higher HRV than people with ASPD-nonPPD. These results during an autobiographical task are consistent with those of clinical studies (Taylor & Workman, 2023), suggesting emotion dysregulation among people with ASPD-nonPPD.
Regarding the HRV difference between the SDM task and the resting state (massed delta), people with ASPD-PPD had a significantly higher HRV difference than people with ASPD-nonPPD and FCs. These results are explained by the resting-state results among people with ASPD-PPD, which were characterized by higher emotion regulation abilities (high HRV indicators). These data are consistent with those of some studies suggesting the absence of global emotional deficits among people with ASPD-PPD (Glass & Newman, 2006; Hastings et al., 2008; Marsh & Blair, 2008; Nentjes et al., 2022; Pham et al., 2010, 2021; Pham & Philippot, 2010). This finding is also congruent with the idea that ASPD-PPD might be an effective mode of functioning in emotional contexts (Harris & Rice, 2006).
Similarly, these results confirm the importance of using objective measures of emotional adjustment and coping given their incongruence with self-report measures. Indeed, dysfunctional emotion regulation abilities, particularly for the emotion of anger, are often associated with people with ASPD-PPD (Garofalo et al., 2020; Harenski & Kiehl, 2010; Kosson et al., 2020). However, this self-evaluation is problematic due to the psychological characteristics of these populations, especially given the frequency with which they pathologically lie. This difference again illustrates the variation in results depending on the methodology chosen (Brook & Kosson, 2013; Faith et al., 2022; Hicks & Drislane, 2018; Mokros et al., 2015; Newman et al., 2010). Thus, combining multiple indicators of emotional processes by measuring both self-reported subjective indicators and physiological measures is essential for studying emotional processes in these populations (Brook & Kosson, 2013).
Second, regarding the mean phasic driver responses, we hypothesized that during the resting task, people with ASPD-nonPPD and people with ASPD-PPD would have the same activation, and lower activation levels than FCs. Our results showed no significant differences between the ASPD-PPD, ASPD-nonPPD, and FC groups in the resting state. These observations are not congruent with those of studies showing lower EDA in the ASPD-PPD and ASPD-nonPPD groups (Lorber, 2004). This lack of a difference in EDA from the FC group may be related to the higher prevalence of anxiety disorders and Cluster C personality disorders among our FC group. Indeed, people with anxiety disorders have a lower electrodermal response (Birket-Smith et al., 1993; Naveteur & Freixa I Baque, 1987).
The mean phasic driver results of the ASPD groups and CP were surprising. Compared to the results of Imtiaz et al. (2020) from community people, we found that people with ASPD-nonPPD and ASPD-PPD had higher mean phasic driver responses. This indicates that their activation levels are high even in the resting state and may be partly explained by this specific characteristic of our sample (from a forensic setting and with a high prevalence of comorbidities).
We hypothesized that people with ASPD-nonPPD and people with ASPD-PPD would have similar levels of emotional activation (EDA values) during the SDM task. Our results showed that the mean phasic driver responses of people with ASPD-PPD and ASPD-nonPPD were significantly lower than those of FCs, with no significant differences observed between the ASPD-PPD and ASPD-nonPPD groups. A similar pattern of results was obtained in comparisons of values in the SDM task and resting task (massed delta). These results confirm that people with ASPD-nonPPD and ASPD-PPD exhibit low levels of EDA during a task (Broom, 2012; Farrell, 2001; Lorber, 2004). Our results highlight differences in arousal between the resting state (higher arousal) and recall task (lower arousal). Thus, experimental paradigms modulating cognitive and emotional loads may be needed to further elucidate the neurophysiological processes in people with ASPD-PPD and ASPD-nonPPD.
Concerning the results of the correlation analysis between PCL-R scores (Hare, 2003) and neurophysiological indicators during the SDM task, the interpersonal factor (Factor 1, corresponding to Facet 1, which is predominantly characterized by loquaciousness/superficial charm, grandiose sense of self, and pathological lying, and the affective facet) and Facet 2 (including lack of remorse or guilt, shallow affect, callousness/lack of empathy, and failure to accept responsibility for own actions) were negatively associated with mean phasic driver responses and positively associated with HF-log and RMSSD responses. Our results align with those of Hansen et al. (2007) on the cognitive task. On the other hand, Facet 3 (including impulsivity, need for stimulation and lack of realistic long-term goals) was associated with low HF-log and RMSSD responses. While low autonomic nervous system activity is a biomarker of aggressive and antisocial behaviour (Baker et al., 2013), the aetiologies of reactive and proactive aggression differ. At rest, the electrodermal activity is positively associated with proactive aggression, whereas the electrodermal activity in response to a minor stressor is positively associated with reactive aggression and negatively associated with proactive aggression (Amstrong et al., 2019). The correlation between HRV values and scores on Facet 3 suggest that this impulsive component is associated with lack of regulation and potentially reactive violence. Factor 1 of the PCL-R is associated with instrumental violence (Walsh et al., 2009), and it is possible that low EDA and high HRV values are the signature of ‘manipulative’ people with PPD. This ‘manipulative’ profile is characterized by high manipulative tendencies and a deep emotional deficit. They are less likely to act impulsively and antisocially (Hare, 2016). They do not have feelings for others but understand their mental states and use this information to further their personal interests (Book et al., 2007). Therefore, their callous empathy may be a key factor in their interpersonal success as cheaters (Mealey, 1995) and social predators (Hare, 2003). These traits characterize ‘successful’ psychopaths (Wallace et al., 2022). Therefore, we could hypothesize that this link between EDA and PCL-R Facet 1 would be even stronger for successful psychopaths.
This study has limitations and perspectives, particularly with regard to psychiatric comorbidity among the ASPD-nonPPD, ASPD-PPD, and FC groups. The influences of anxiety symptoms among FCs, psychiatric treatment on neurophysiological measures, and comorbid substance use disorders in the three groups on our results cannot be excluded. Substance use disorder is a common comorbidity among people with ASPD-nonPPD or ASPD-PPD in forensic settings (Pham & Saloppé, 2010). Therefore, it would be useful to include a nonclinical group without psychiatric disorders because, although we were able to compare clinical and community groups in the resting task, we were unable to compare these groups in the experimental task. This raises the question of whether people with ASPD-nonPPD and ASPD-PPD have a specific neurophysiological signature not in relation to the FC group but in relation to the community population. Similarly, including an ASPD-PPD group comprised of incarcerate individuals and an ASPD-PPD group comprised of individuals in the CP would allow us to compare their neurophysiological signatures.
Second, in this study, we measured neurophysiological responses during the recall of SDMs without specifying their possible variations across SDM characteristics (Blagov et al., 2022). It would be relevant to measure these indicators as a function of the characteristics of autobiographical recall, such as specificity, valence, integration, and life events, across the three clinical groups (Lavallée, et al., 2019).
Third, it would be useful to assess neurophysiological responses during a recovery period to test the hypothesis that people with ASPD-PPD would recover more rapidly than people with ASPD-nonPPD. Thus, a period of 5 minutes could be added after the task (Berna et al., 2014; Hansen, et al., 2003). Finally, it would be interesting to perform multilevel emotion analyses by comparing neurophysiological responses to facial expressions (Lavallée et al., 2022) and semantic, prosodic, and subjective features. Indeed, these indicators account, in the component process model of emotion (Sander et al., 2005), for the complexity of the emotional process, which includes appraisal processes, autonomic physiology, motor expression, and subjective feelings.
Conclusion
This study provides further insight into the psychological and physiological processes associated with SDMs, particularly the emotion regulation processes. Our findings highlight the adaptive capacity of people with ASPD-PPD. Our results indicate that people with ASPD-PPD exhibit high cognitive flexibility and emotion regulation abilities (characterized by high HRV). Moreover, the EDA of this group suggested lower activation levels during the SDM task than in the resting state. Taken together, these results suggest that people with ASPD-PPD can respond more flexibly to stimuli at lower levels of activation. Furthermore, PCL-R Factor 1 scores were negatively correlated with the difference in neurophysiological responses between the SDM task and the resting tasks. Therefore, people with ASPD-PPD that exhibit more loquaciousness/superficial charm, a more grandiose sense of self, and more pathological lying tend to exhibit lower levels of activation. These results are consistent with the idea that ASPD-PPD involves both functionality and subtle impairments. Furthermore, these findings support the idea that ASPD-PPD is characterized by patterns of neurophysiological functioning specific to each multi-dimensional profile.
Footnotes
Acknowledgements
This project has been carried out in collaboration with the Walloon region in Belgium. The authors wish to thank the teams of the Secure Psychiatric Hospital of the Regional Psychiatric Centre “Les Marronniers” in Tournai (Belgium). They also wish to thank Lucie Delecroix and Céline Poppe for their valuable contributions to this project. The manuscript was edited for proper English language, grammar, punctuation, spelling, and overall style by one or more of the highly qualified native English-speaking editors at AJE. A certificate was issued on July 22, 2023, and may be verified on the AJE website using the verification code DA46-3B7B-687B-9121-8C8P.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by FEDER SCV-IrDIVE under European funding through the program FEDER SCV-IrD.
