Abstract
Introduction
Childhood trauma is strongly associated with fear-related psychopathology, like anxiety and posttraumatic stress disorder (PTSD). Atypical fear extinction and neural responses to social threat (i.e., negative emotional faces) may serve as intermediate phenotypes preceding the emergence of fear-related psychopathology during childhood and adolescence. However, few studies have examined associations among these phenotypes in trauma-exposed youth.
Methods
29 9-year-old children with high rates of trauma exposure (Mdn = 4, min = 0, max= 14 total events) completed a fear-potentiated startle paradigm assessing fear conditioning and extinction and an emotional faces functional magnetic resonance imaging (fMRI) task assessing neural responses to fearful and neutral faces.
Results
Overall amygdala response was positively associated with anxiety (peak coordinates: x = −30, y = −6, z = −24; Z = 3.54; pFWEcorrected = 0.011; k = 24 voxels) and fear-potentiated startle during early extinction (peak coordinates x = 28, y = −6, z = −18; Z = 3.50; pFWEcorrected = 0.012; k = 19 voxels). Across the session, amygdala reactivity to fearful faces increased (F(1, 29) = 4.427, p = .044) and was positively associated with fear-potentiated startle during early extinction (r = .56, p = .002).
Conclusions
We found a positive association between increasing amygdala response to threatening faces and fear load, that is, heightened fear-potentiated startle during early extinction, in trauma-exposed children. These fear-based intermediate phenotypes may share underlying amygdala circuits, such that hyperactivity may represent an early marker of anxiety risk in trauma-exposed youth.
Introduction
During the transition from late childhood to adolescence, the prevalence of fear-related disorders (i.e., anxiety disorders, posttraumatic stress disorder [PTSD]) dramatically increases (Kessler et al., 2005), suggesting that this developmental window may be critical for identifying neurobiological mechanisms that give rise to these disorders (Stevens et al., 2016). Further, childhood trauma exposure can double the risk for developing fear-related disorders (Walsh et al., 2017). However, studies on the neurobiological sequela of trauma exposure and of fear-related disorders are largely conducted in adults. Studies identifying intermediate phenotypes of fear-related disorders during childhood and adolescence—more proximal to the traumatic exposure and the emergence of psychopathology—are needed to develop more effective prevention strategies (McLaughlin et al., 2019).
In the short term, childhood trauma may promote adaptive functional changes in neural circuits involved in the generation of threat responses, including the amygdala (van Rooij et al., 2020). Throughout development, these circuits may become dysregulated, heightening negative emotional reactivity, thereby contributing to the emergence of anxiety disorders during late adolescence and early adulthood (Teicher et al., 2016). Indeed, youth exposed to childhood trauma show amygdala hyperactivity to negative social cues, like threatening faces (Tottenham et al., 2011; White et al., 2019); a neural response characteristic of anxious adolescents (Monk et al., 2006). Studies in adults demonstrate that, relative to unexposed individuals, individuals with histories of trauma exposure show amygdala hyperactivity to threatening faces, suggesting that these neural patterns persist into adulthood (Dannlowski et al., 2013).
Amygdala function is characterized by rapid changes in reactivity with repeated stimuli presentations (Rankin et al., 2009), which has been associated with anxiety symptoms (Hare et al., 2008). Amygdala habituation refers to a typically observed decrease in amygdala blood oxygenation level dependent (BOLD) signal response occurring with repeated presentations of threatening stimuli (Pedersen et al., 2017). A lack of amygdala habituation (i.e., sustained response) or sensitization (i.e., increasing response across the session) may be related to prior trauma exposure and/or psychopathology risk. For example, to assess change in amygdala response across time during repeated presentations of threatening stimuli, Stevens and colleagues assessed amygdala BOLD signal across the early, middle, and late thirds of a negative emotional faces task in a sample of youth exposed to violence (Stevens et al., 2021). They found that youth who experienced more violence at home were more likely to demonstrate amygdala sensitization across the session, as compared to youth who experienced less violence. This finding is interesting given that patterns of sustained amygdala response across threatening face presentations has been associated with internalizing symptomology in a separate sample of trauma-exposed youth (Hein et al., 2020). Together, these findings suggest that sustained social threat-related amygdala reactivity following childhood trauma may increase risk of subsequent anxiety in trauma-exposed youth (McLaughlin et al., 2014; Thomas et al., 2001).
During Pavlovian fear conditioning, the amygdala activity supports the association of a neutral stimulus (CS+) with a naturally aversive stimulus (i.e., unconditioned stimulus, US) (Gale et al., 2004; Phillips & LeDoux, 1992). After several pairings (i.e., fear acquisition), the CS+ elicits the conditioned response alone, thereby representing the “danger cue” (Jovanovic & Ressler, 2010). Alongside the CS+ is a non-reinforced stimulus (CS−) never paired with the US, and represents a safety cue. During fear extinction, neither the safety or danger cues are paired with the US and measures one’s ability to override their learned fear response with a new memory that the CS+ is no longer paired with the aversive stimulus (Milad & Quirk, 2012). Elevated conditioned fear expression to the CS+ after fear learning, or “fear load,” has been observed in individuals with fear-related disorders (Norrholm et al., 2011). Indeed, greater symptom severity among adults with fear-related disorders was positively associated with fear load (Norrholm et al., 2015). Fear load may also serve as a potential identifier of anxiety risk in children (Waters et al., 2009), and specifically children between the ages of 8 and 10 (Jovanovic et al., 2014). A longitudinal investigation has provided further support that heightened conditioned fear expression to the CS+ is a predictor of anxiety in youth (Stenson et al., 2021). In particular, the study by Stenson et al. suggests that the overexpression of psychophysiological fear during early extinction may be a clinically informative intermediate phenotype for fear-related disorders during development.
Given that trauma exposure impacts multiple neurocognitive features of the threat response, it is plausible that these phenomena may result from dysfunction in a common threat circuitry in the brain (Javanbakht, 2019). For example, fear load was shown to be associated with attention bias to threat in individuals with PTSD, suggesting a common mechanism by which trauma influences both hypervigilance and fear learning (Fani et al., 2012). Further, a negative association was found between ventromedial prefrontal cortex (vmPFC) reactivity and fear expression during a fear-potentiated startle paradigm (Jovanovic et al., 2013). During fear conditioning utilizing social threat and safety cues, elevated fear load was associated with enhanced amygdala reactivity in adolescents (Lau et al., 2011). Amygdala hyperactivity and heightened psychophysiological arousal to threat may serve as intermediate risk phenotypes between trauma exposure and fear-related disorders during adolescence, consistent with the Research Domain Criteria (RDoC) framework (Insel et al., 2010). However, no studies have investigated the relationship between sustained amygdala response to social threat cues and fear load in trauma-exposed youth.
In the current study, we investigated fear load and amygdala reactivity to fearful and neutral faces in a sample of 9-year-old trauma-exposed youth from the Detroit metropolitan area, Michigan (USA). We assessed fear load using a fear-potentiated startle paradigm, and amygdala reactivity using an emotional faces functional magnetic resonance imaging (fMRI) task. We hypothesized that overall amygdala reactivity would be positively associated with anxiety symptoms, and that fear load would be positively associated with overall amygdala reactivity and sustained amygdala reactivity across the session (i.e., sensitization or lack of habituation).
Methods
Participants and experimental procedure
A total of 75 child participants aged 9 (M = 9.55, SD = 0.29 years) were recruited through an ongoing study of childhood trauma exposure conducted by the Detroit Trauma Project, in Detroit, Michigan. All study procedures were approved by the Wayne State University institutional review board (IRB00093684). Written informed consent was obtained from caregivers (i.e., parent or legal guardian), and verbal assent was obtained from child participants. Participants completed two visits. One visit consisted of the startle paradigm and psychological assessments detailed below. During the second visit, participants completed a magnetic resonance imaging (MRI) session, which included the emotional faces fMRI task. Visits 1 and 2 occurred approximately 4 weeks apart, (M = 29.3, SD = 26.9 days).
Participants were included if they were 9 years old at the time of enrollment, were willing to complete all study procedures, and had a consenting caregiver present at the time of enrollment. Participants were screened for the following exclusion criteria: hearing loss, neurological disorder, developmental impairment, or autism spectrum disorder. Of the 75 participants recruited, 55 completed the MRI session, 44 completed the startle session, and 37 completed both MRI and startle sessions. Of the 37 who completed both sessions, data were excluded from analysis for the following reasons: excessive motion (>1/4 of images with framewise displacement (FD) > 0.5 mm/TR, N = 5) during fMRI data collection, early termination of the MRI procedure (N = 2), or failed hearing test during administration of the fear-potentiated startle paradigm (N = 1). Therefore, the final sample was N = 29 (N = 14 Female) with complete fMRI, startle, and psychological data.
Psychological analysis and trauma exposure assessment
The Behavioral Assessment System for Children – Second Edition (BASC-2; Reynolds & Kamphaus, 2004) is a self-reported 161-item scale for children ages 6–11. Anxiety scores were measured using age-normed T-scores.
Children’s exposure to potentially traumatic events was assessed in interview format with the child. The Traumatic Events Screening Inventory (TESI) child report was used to assess the child’s experience of a variety of potentially traumatic events (Ghosh-Ippen, 2002). The total number of events reported was calculated by summing all self-reported events for a cumulative trauma score, as in our prior work in this population (Wiltshire et al., 2022). Total number of traumatic events served as the trauma exposure variable in subsequent analyses.
MRI procedures
All scans were acquired using a 3 Tesla MRI system (Siemens MAGNETOM Verio™) located at the MR Research Facility, Wayne State University. A T1-weighted, multi-echo magnetization prepared rapid gradient echo (ME-MPRAGE) sequence was acquired for co-registration with functional images using the following parameters: 176 slices, 1.0 mm slice thickness, 1.0 mm3 voxel size, 256 mm field of view (FOV), 7° flip angle (FA), repetition time (TR) = 2530 ms, echo times (TEs) = 1.79, 3.65, 5.51, 7.37 ms, total acquisition time (TA) = 6 min 55 sec. BOLD images were acquired during the same session and during the emotional faces task using a multi-echo/multi-band (ME-MB) echo planar imaging (EPI) sequence. ME-MB EPI images were collected in the axial plane using the following sequence parameters: 51 slices, 2.9 mm slice thickness, 2.9 mm3 voxel size, 186 mm FOV, k-space parallel imaging mode GRAPPA with acceleration factor 2, 2790 hz/px bandwidth, 83° FA, TR = 1500 ms, TEs = 14, 30, 45 ms, TA = 5 min 30 sec. While the imaging data were acquired using a multi-echo sequence, these data were processed as single echo images using the middle echo (TE = 30 ms), as described below, to maximize generalizability to prior studies (e.g., Stevens et al., 2021).
Image preprocessing was conducted using matlab19 and SPM12 (http://www.fil.ion.ucl.ac.uk/spm). Preprocessing steps included manual reorientation (to minimize failures of co-registration) realignment, slice-timing correction, co-registration, segmentation, normalization, and smoothing (8 mm gaussian kernel). To mitigate the effects of participant motion, we utilized the Art-Repair toolbox (https://www.nitrc.org/projects/art_repair/) to interpolate volumes which exceeded a 0.5 mm/TR movement threshold. Participant data sets with more than 25% of volumes interpolated were excluded from the study. In the final sample, the average percentage of interpolated volumes was 4.9% and the average framewise displacement was 0.115 mm.
During fMRI scanning, participants completed an emotional faces task developed by Stevens and colleagues (Stevens, van Rooij, et al., 2021). The task was a passive viewing, event-related design that involves presentation of 24 fearful and 24 neutral female faces selected from the Karolinska Directed Emotional Faces database (Goeleven et al., 2008), totaling 48 trials. Trials were presented for 500 ms each in pseudo-random order and interleaved with fixation cross presentations with variable duration (3500–9000 ms).
Fear-potentiated startle assessment
The fear-potentiated startle (FPS) protocol consisted of fear acquisition and extinction phases, separated by a 10-minute rest period, and was developed and previously used in youth populations (Jovanovic et al., 2014). The startle probe was a 106-dB [A] SPL, 40-ms burst of broadband noise delivered binaurally through headphones. Participants remained seated in a sound-attenuated booth, while visual stimuli were presented using Superlab software (Cedrus, Inc., San Pedro, CA). The trial types included a reinforced conditioned stimulus, CS+; a non-reinforced conditioned stimulus, CS−; and a noise alone (NA) trial during which no visual stimulus was presented. The US was an 80 psi, 100 ms air puff directed at the larynx. The reinforcement rate, or the percentage of CS+ presentations paired with the US, was 100%. Fear acquisition consisted of 3 blocks, in which 3 presentations of each trial type occurred, for a total of 27 trials. The intertrial intervals were randomized between 9 and 22 seconds. The CSs were colored shapes presented for 6000 ms prior to the delivery of the startle probe. The fear extinction phase consisted of 4 blocks during which the US was never paired with the CS+, for a total of 36 trials.
Psychophysiological data were collected using BIOPAC MP160 system with AcqKnowledge software for Windows (Biopac Systems, Inc., Aero Camino, CA) following our previously published methods (Norrholm et al., 2011). Eyeblink startle response data was collected at a 1000 Hz sampling rate using the electromyograph (EMG) module of the Biopac system. MindWare software (MindWare Technologies, Inc, Gahanna, OH) was used to filter and rectify the acquired EMG data, and was then exported for statistical analyses. EMG activity was recorded from two 5 mm Ag/AgCl electrodes placed over the orbicularis oculi muscle, approximately 1 cm under the pupil and 1 cm below the lateral canthus. The impedances for all participants were less than 6 kilo-ohms. The EMG signal was filtered with low- and high-frequency cutoffs at 28 and 500 Hz, respectively. The acoustic startle response was defined as the maximum peak amplitude of eyeblink muscle contraction 20–200 ms after presentation of the startle probe.
Statistical analysis
FMRI analyses were performed using SPM12 software (Wellcome Trust Centre for Neuroimaging, University College London, U.K.). To examine BOLD signal change between task conditions, first-level analyses included model fitting to subject-specific onset times for each task condition. Task conditions were created using early (first 8 trials), middle (middle 8 trials), and late (last 8 trials) onset times for fearful and neutral faces, separately. Of note, prior evidence suggests that 6–8 trials are sufficient for determining high reliability in BOLD signal across multiple regions of interest during a conventional Go/No-Go task (Steele et al., 2016). Therefore, the first-level general linear model included six task-related regressors in total (early, middle, late for fearful and neutral faces, separately) and a seventh regressor of all individual motion-related interpolated images determined with Art-Repair during preprocessing to control for variance associated with the motion-laden, interpolated images. Following Stevens and colleagues, the following first-level linear contrasts were created: (1) early fearful faces > (implicit) baseline, (2) middle fearful faces > (implicit) baseline, and (3) late fearful faces > (implicit) baseline; and (4) early neutral faces > (implicit) baseline, (5) middle neutral faces > (implicit) baseline, and (6) late neutral faces > (implicit) baseline (Stevens, van Rooij, et al., 2021). Given prior evidence that neutral facial expressions are often perceived as threatening by youth, we combined fearful and neutral faces into one contrast to measure overall response to faces, as potentially aversive social cues, (7) fearful and neutral > (implicit) baseline (Hester, 2019; Marusak et al., 2017). For completeness, we performed whole-brain analysis using the fearful and neutral > (implicit) baseline contrast, see supplementary material.
We also explored responses to fearful and neutral faces separately to assess emotional specificity in amygdala response, and change in amygdala responsivity across the session. A second level random-effects general linear model was conducted using one-sample t-tests, for each of the aforementioned contrasts across the sample. A priori beta weights were extracted for early, middle, and late fearful and neutral face presentations from the bilateral amygdala, which served as our region of interest (ROI), and was defined using a probabilistic atlas of post mortem tissue (Amunts et al., 2005). ROI extraction was completed using the SPM marsbar toolbox (https://www.nitrc.org/projects/marsbar/) and BOLD signal was averaged across all voxels in the mask. Beta weights were submitted to SPSS for further analyses.
To examine associations between amygdala reactivity and anxiety, we first conducted whole brain regression with anxiety score as the regressor of interest using the fearful and neutral faces > baseline contrast in SPM and applied small volume family-wise error correction in the bilateral amygdala (pFWEcorrected <0.05). We then conducted bivariate correlations between amygdala reactivity to fearful and neutral faces and BASC anxiety scores in SPSS. Further exploratory analysis included a hierarchical linear regression to test whether amygdala reactivity during fearful and neutral face presentation independently contributed to anxiety, with total trauma exposure in the first step and amygdala reactivity in the second step.
We then tested for change in amygdala reactivity over time using a repeated measure ANOVA with time as the within-subjects factor (3 levels; early, middle, and late). Additionally, change in threat-related amygdala reactivity was computed as a single variable, named “amygdala change,” by subtracting the bilateral amygdala beta weights in the late trials of the task from the early trials of the task, using the fearful faces > (implicit) baseline contrast. Negative change scores suggest decreasing amygdala activity, that is, habituation, over time, whereas positive change scores indicate increasing amygdala reactivity, that is, sensitization, over time.
FPS was measured using the following equation: Percent Potentiation = 100 x (startle magnitude during the CS trial – NA startle magnitude during the same session)/(NA startle magnitude during the same session), computed for both CS+ and CS− individually. To analyze fear acquisition and extinction, FPS was included in 2-way repeated measures ANOVAs with a within-subjects factor of block (3 levels for 3 acquisition blocks; and 4 levels for 4 extinction blocks), and trial type (2 levels, CS+ and CS−). Fear load was defined by the magnitude of fear-potentiated startle during early extinction, or the first and second blocks.
To examine associations between fear load and amygdala response to fearful and neutral face presentation (>baseline), whole-brain regression analyses in SPM with fear load as the regressor of interest was conducted and small volume family-wise error correction was applied to the bilateral amygdala (pFWEcorrected <0.05). To examine associations between fear load and amygdala change, bivariate correlations were conducted using amygdala change and FPS during extinction in SPSS. A Bonferroni correction for multiple comparisons was applied when comparing bilateral amygdala reactivity across the 4 blocks of extinction (alpha = 0.0125).
Results
Demographics and trauma exposure
Participant demographics, anxiety symptoms, and trauma exposure data.

Trauma and Anxiety. Association between self-reported number of potentially traumatic events reported and self-reported anxiety symptoms, as reported by the TESI and by the BASC-2, respectively (*p < 0.05).
Amygdala reactivity and anxiety
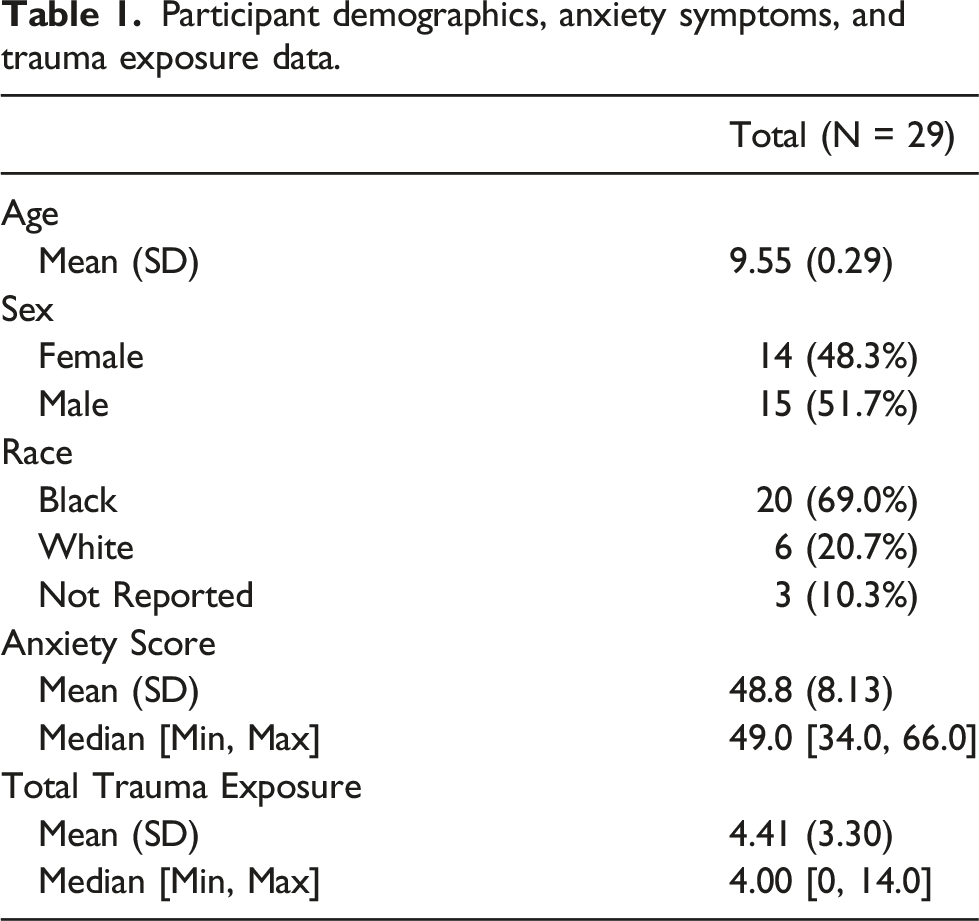
Bilateral amygdala reactivity to fearful and neutral faces > baseline was positively associated with anxiety score after controlling for trauma exposure (peak coordinates: x = −30, y = −6, z = −24; Z = 3.54; pFWEcorrected = 0.011; k = 24 voxels), Figure 2(a). Using the extracted beta weights, this association was replicated in SPSS (r = .479, p = .009), Figure 2(b). The overall hierarchical linear regression model was significant (F(2, 26) = 6.29, p = .006) and explained 34% of the variance in anxiety. Amygdala reactivity explained 15.6% of the unique variance in anxiety above total trauma exposure (R2change = .156, F(1, 24)change = 5.7, p = .025). When contrasting both fearful faces > baseline and neutral faces > baseline, amygdala reactivity was positively correlated with anxiety (r = .477, p = .01 and r = .467, p = .012, respectively). However, anxiety was not correlated with amygdala reactivity to fearful faces > neutral faces (p > 0.05). Whole brain results for the fearful and neutral faces > baseline contrast are included in the supplemental material, Supplemental Table S1. Bilateral Amygdala Reactivity and Association with Anxiety: (a) Whole brain regression with anxiety score as regressor of interest during fearful and neutral face presentation (>baseline) and controlling for trauma exposure, with small volume family-wise error correction applied to the bilateral amygdala (peak coordinates: x = −30, y = −6, z = −24; Z = 3.54; pFWEcorrected = 0.011; k = 24 voxels) (b) Using extracted beta weights averaged across the bilateral amygdala, reactivity to fearful and neutral face presentation (>baseline) is positively associated with anxiety score, replicating the small volume correction findings (**p < .01).
Amygdala change
Repeated measures ANOVA assessing change in amygdala reactivity contrasting fearful faces > baseline showed a significant main effect of time (F(1, 29) = 4.427, p = .044), Figure 3. Post hoc t-tests demonstrated that amygdala response was significantly higher during late compared to early trials (p = 0.05). There was no difference in amygdala reactivity between early and middle, and middle and late trials (ps > 0.05). This effect was not observed when contrasting neutral faces > baseline (p > 0.5), suggesting that amygdala activity increased to fearful (but not neutral) faces across the task. Amygdala change was not correlated with trauma exposure nor anxiety symptoms (ps > .05). Amygdala Change. One-way RM-ANOVA showing a main effect of time, indicating that amygdala response increased to fearful face presentation over the course of the task (*p < .05 fearful faces, only). The main effect of time was not significant for neutral faces (p < 0.05).
Fear-potentiated startle
A block x trial type (CS+, CS−) interaction (F(2, 26) = 6.689, p = .005) indicated successful discrimination between the CS+ and CS− by the last block of acquisition, Figure 4(a). Repeated measures ANOVA of extinction block x trial type showed a significant main effect of block (F(3,25) = 3.108, p = .044), indicating lower FPS across trial types during late extinction compared to early extinction and suggesting successful extinction learning, Figure 4(b). Fear load (FPS to CS+ during blocks 1 and 2 of extinction) was not correlated with trauma exposure nor anxiety symptoms (ps > 0.5). Fear Acquisition and Extinction. (a) Two-way repeated measure ANOVA showing a significant interaction between time and trial type, indicating successful discrimination between CS+ and CS− after acquisition (**p < .01 interaction effect). (b) Two-way repeated measures ANOVA showing a significant main effect of time, indicating successful reduction in fear by the end of extinction (*p < 0.05 main effect of time).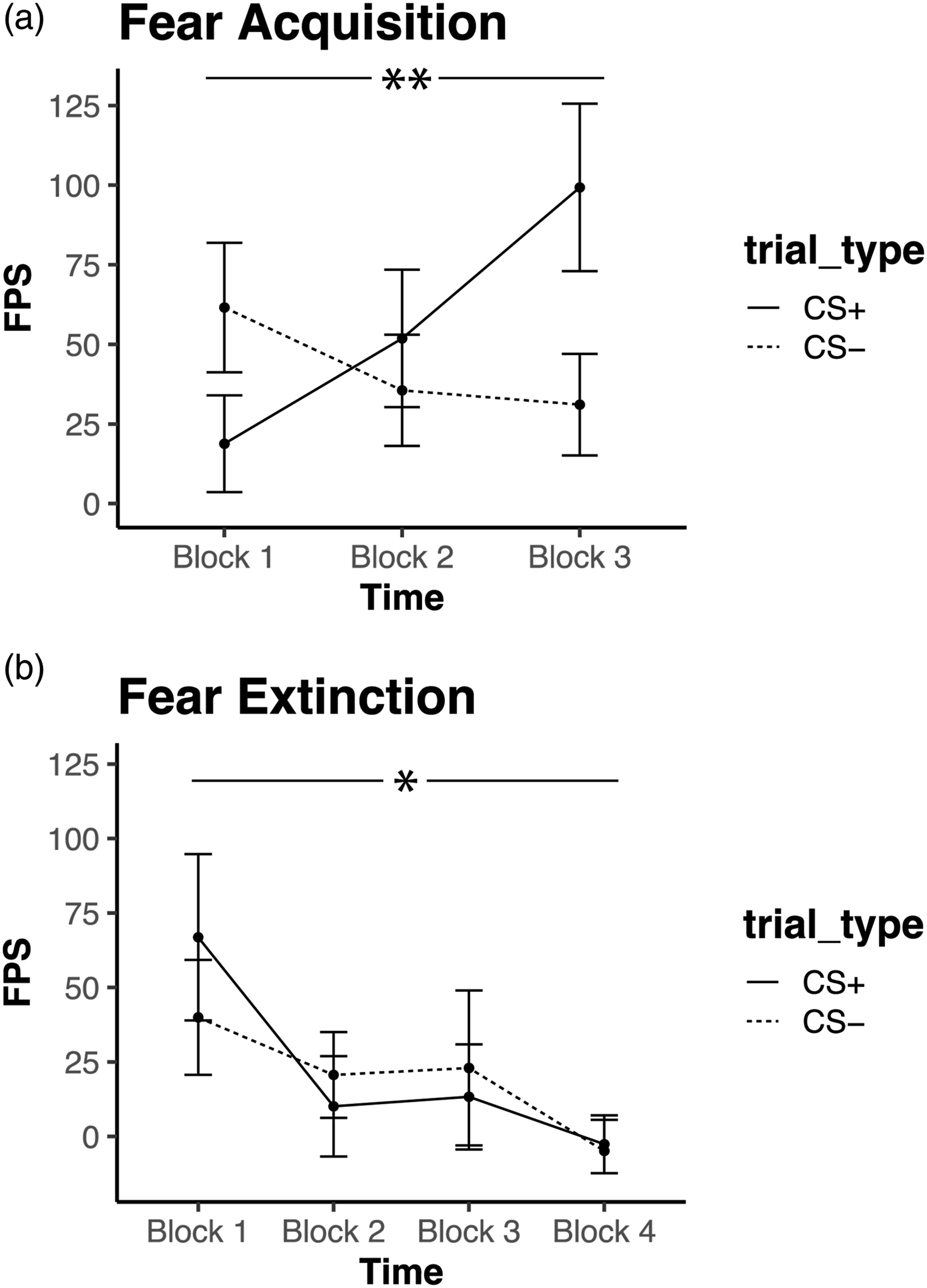
Associations between amygdala and fear load
Bilateral amygdala activity to fearful and neutral faces > baseline was positivity correlated with fear load, specifically during the second block of extinction, after small volume family-wise error correction (peak coordinates x = 28, y = −6, z = −18; Z = 3.50; pFWEcorrected = 0.012; k = 19 voxels), Figure 5(a). This finding remained significant when including trauma exposure as a covariate (peak coordinates x = 28, y = −6, z = −18; Z = 3.52; pFWEcorrected = 0.012; k = 5 voxels). Further, amygdala change to fearful faces > baseline was positively correlated with fear load, specifically during the second block of extinction (r = .56, p = .002), Figure 5(b), such that those with greater increases in amygdala reactivity across the session exhibited higher fear load. This result remained significant after correction for multiple comparisons and controlling for trauma exposure ( Amygdala and Startle Association. (a) Whole brain regression with fear load (i.e., startle response to CS+ during early extinction [2nd block]) as regressor of interest during fearful and neutral face presentation (>baseline), with small volume family-wise error correction applied to the bilateral amygdala (peak coordinates x = 28, y = −6, z = −18; Z = 3.50; pFWEcorrected = 0.012; k = 19 voxels). (b) Amygdala change to fearful face stimuli (>baseline) is positively associated with fear load during early extinction [2nd block] (***p < .005).
Discussion
This study assessed associations among anxiety symptoms, fear-potentiated startle and threat-related amygdala reactivity in a sample of 9-year-old trauma-exposed youth. We first hypothesized that amygdala reactivity to social stimuli would be positively associated with anxiety. We confirmed this hypothesis and found a positive association between amygdala reactivity to fearful and neutral face presentation and anxiety symptoms, which is consistent with prior literature in youth (Beesdo et al., 2009; Ferri et al., 2014; Herringa et al., 2013; Monk et al., 2006; Thomas et al., 2001; van den Bulk et al., 2014). This association was significant for both fearful and neutral faces, suggesting that anxiety symptoms may be associated with amygdala response to both stimuli. This is consistent with current literature suggesting that neutral faces may be perceived as threatening in youth (Marusak et al., 2017; Stevens, van Rooij, et al., 2021). Most studies investigating neural response to emotional faces in the context of trauma and psychopathology have included adolescents across a relatively wide age range, including up to age 17. The current investigation found an association between amygdala reactivity and anxiety in 9-year-old trauma-exposed children. This is important because the mean age of onset across all anxiety disorders is 11 years old (Kessler et al., 2005), and late childhood to early adolescence has been identified as a sensitive period of development for mechanisms supporting the emergence of fear-related psychopathology (Stevens et al., 2018).
Further, this investigation found that amygdala responses during fearful face presentation increased across the session, which may reflect sensitization. This is consistent with prior literature suggesting that violence exposure and internalizing symptoms are associated with sustained amygdala responses to threatening faces in adolescents (Hein et al., 2020; Stevens, van Rooij, et al., 2021). This response is in contrast to habituation where neural response decreases with repeated presentation of stimuli, which is a basic characteristic of neural activity (Bailey & Chen, 1988). Amygdala change may serve as a marker of fear circuit hyperactivity in individuals with fear-related disorders (Protopopescu et al., 2005). In adults, amygdala habituation to fearful faces has been found to partially mediate the association between childhood trauma and PTSD symptom severity (Kim et al., 2019). While studies investigating change in amygdala BOLD signal across time are scarce, these reactivity patterns can provide a reliable description of threat-related neural activity beyond average reactivity (Gee et al., 2015). In the present study, amygdala sensitization was observed during the presentation of fearful faces but not neutral faces. Amygdala sensitization may reflect an initial adaptation to trauma exposure, such that hypervigilance may require increased allocation of attentional resources to potentially threatening stimuli (Richards et al., 2014). While amygdala reactivity to both fearful and neutral faces predicted anxiety symptoms, amygdala change or sensitization did not. Further work in adolescents should examine changes in threat-related amygdala reactivity across the session as a potential predictor of risk of fear-related disorders.
Our second hypothesis was that amygdala reactivity to fearful and neutral faces would be positively associated with heightened fear during early extinction, that is, fear load. Our investigation confirmed this hypothesis. Further, we found an association between amygdala change during fearful face processing and fear load. Specifically, amygdala sensitization to fearful faces was positively correlated with FPS to the CS+ during the second block of extinction. This finding may suggest a common neurobiological mechanism supporting sustained hypervigilance and physiological arousal to threat in our sample of trauma-exposed youth. Indeed, projections from the amygdala target various hypothalamic and brainstem regions and mediate conditioned and unconditioned threat responses (Davis, 1992). Upstream interactions between the amygdala, ventromedial prefrontal cortex (vmPFC), and hippocampus are thought to modulate the threat response (Liberzon & Abelson, 2016; Milad & Quirk, 2002; Milad et al., 2007). However, the vmPFC is underdeveloped in youth (Paus, 2005) and may not yet support successful extinction learning or recall (Ganella et al., 2018; Gold et al., 2020; Marusak et al., 2021). Uncoupling of the amygdala and vmPFC during threat processing may contribute to elevated physiological arousal (Ganella et al., 2017) and dysregulation of emotional responses, a hallmark of fear-related disorders (Lee et al., 2012). As such, sustained or even increased threat-related amygdala and psychophysiological reactivity may serve as intermediate phenotypes of fear-related disorders more proximal to trauma exposure, and help identify risk for fear-related disorders (Stevens, Harnett, et al., 2021).
Strengths of this study include the use of multiple modalities, neuroimaging and fear-potentiated startle, to assess threat processing in youth, and the focus on a sample of urban trauma-exposed youth who are at elevated environmental risk of anxiety. There are also several limitations that should be noted. First, our study included a modest sample size and the paradigm included 8 trials per condition, which may introduce greater variance in the within-group effects. This investigation did not include a control group and as such, it cannot be directly interpreted that the observed patterns of amygdala change are specific to children with trauma exposure. Future studies should include direct comparisons to a control group without trauma exposure and/or compare different trauma types and frequencies of exposure. Further, this study was underpowered for mediation analysis. Further studies should aim to replicate these findings with a larger sample size, and investigate the mediating effects of social threat-related amygdala reactivity and fear load on the relationship between and childhood trauma and anxiety.
Conclusion
The present study demonstrated a positive association between physiological arousal to a previously learned threat cue during early extinction (i.e, fear load) and increasing amygdala reactivity to social threat cues (amygdala sensitization) across the session in trauma-exposed children. We also observed that threat-related amygdala reactivity was positively associated with anxiety symptoms. Together, these findings suggest a shared fear-related neurocircuitry involving the amygdala that may support hypervigilance and physiological arousal to threat in children, and increase risk of psychopathology. Further work investigating neural and physiological threat responses in youth exposed to childhood trauma will aid in identifying intermediate phenotypes of fear-related psychopathology, and promote early intervention to support healthy development into adolescence and adulthood.
Supplemental Material
Supplemental Material - Anxiety, fear extinction, and threat-related amygdala reactivity in children exposed to urban trauma
Supplemental Material for Anxiety, fear extinction, and threat-related amygdala reactivity in children exposed to urban trauma by John M. France BS, Mariam Reda BS, Hilary A. Marusak PhD, Manessa Riser BS, Charis N. Wiltshire BS, William Davie BS, Lana Ruvolo Grasser PhD, Cassandra P. Wanna BS, Anaïs F. Stenson PhD, Timothy D. Ely BS, Seth D. Norrholm, PhD, Jennifer S. Stevens PhD, and Tanja Jovanovic PhD in Journal of Experimental Psychopathology
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by support from the National Institute of Mental Health (MH111682, MH119241).
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
