Abstract
Extinction learning is considered an important underlying process of successful treatment of posttraumatic stress disorder (PTSD). However, sleep disturbances may impede this learning process: Current accounts postulate that sleep facilitates encoding by promoting neural plasticity during slow wave sleep (SWS). Based on this hypothesis, we tested whether early night sleep, with high amounts of SWS, facilitates subsequent extinction learning and recall. Sixty-three participants took part in a trauma-adapted fear conditioning experiment. One group received a three-hour sleep opportunity in the early night half, whereas the other group stayed awake. Thereafter, both groups underwent extinction training and a return-of-fear test. Retention was assessed after another sleep opportunity in both groups. Linear mixed-effects models and Bayesian inference did not support the hypothesis of strengthened fear extinction by prior early night sleep. Subsequent exploratory analyses, in contrast, point to a role of rapid eye movement sleep in promoting successful fear extinction learning. Further confirmatory research should re-investigate these effects and their implications for the treatment of PTSD.
Keywords
Introduction
Clinical practice guidelines for the treatment of posttraumatic stress disorder (PTSD) strongly recommend trauma-focused cognitive behavioral therapy (TF-CBT), comprising exposure therapy as its core element (Hamblen et al., 2019). Despite TF-CBT’s effectiveness, rates of non-responders and dropouts are high (Schottenbauer et al., 2008), indicating the need for further improvements of treatment (Michael et al., 2019). Since PTSD and sleep disturbances are highly interrelated, it has been suggested that adjunctive treatments addressing sleep may enhance the efficacy of TF-CBT (Difede et al., 2014). Therefore, the current study investigates if learning processes, which underlie successful TF-CBT, are strengthened by preceding sleep.
Sleep disturbances are assumed to be a risk factor for chronic PTSD and treatment resistance (for reviews see, e.g., Azza et al., 2020; Colvonen et al., 2019; Germain et al., 2017). This assumption is based on studies reporting a negative association between sleep disturbances and the likelihood of remission (Marcks et al., 2010) as well as PTSD symptom decline during prolonged exposure therapy (Lopez et al., 2017; Reist et al., 2017; but see Sexton et al., 2017). Consequently, it has been suggested that sleep problems impede critical recovery processes. Specifically, it is hypothesized that disturbances of sleep-related processes may impair learning, thereby compromising processes that support the dissipation of pathological fear during exposure therapy while direct evidence of this hypothesis is still missing (Colvonen et al., 2019; Davidson & Pace-Schott, 2020).
Previous research indicates that successful exposure therapy relies on processes of fear conditioning (Craske et al., 2018; but see Scheveneels et al., 2021). According to translational models of fear conditioning (Michael, 2017), traumatized individuals acquire conditioned fear to neutral stimuli that appear in contingency with threatening stimulus during trauma (i.e., unconditioned stimulus; US). As a result of conditioning, the formerly neutral stimulus (now conditioned stimulus, CS), elicits a conditioned reaction (i.e., fear and avoidance to the CS). During repeated exposure to the CS in absence of the US, conditioned reactions may decline, which is attributed to fear extinction processes. Fear extinction is assumed to rely on the formation of a new memory trace that inhibits the former CS-US trace (Bouton, 2004). The fear conditioning framework has been used to explain PTSD symptom development, especially the development of intrusive memories (Ehlers et al., 2002). Correspondingly, experimental analog studies have shown that fear acquisition of traumatic associations is related to intrusion development (Franke et al., 2021; Streb et al., 2017). Moreover, fear extinction is suggested to be the process underlying the remission of intrusive memories and thus of successful TF-CBT (Craske et al., 2018). In line with this assumption, it has been shown that successful extinction learning of fear associations reduces the probability and severity of intrusions (Franke et al., 2021). Hence, investigating effects of sleep on extinction learning could provide critical insights on how to facilitate exposure therapy during TF-CBT.
Sleep is critical for subsequent learning. That is, sleep restriction or deprivation prior to encoding negatively impacts encoding (Cousins et al., 2018) and later recall (e.g., Drummond et al., 2000; Kaida et al., 2015; Yoo et al., 2007). Moreover, findings indicate that specific processes during non-rapid eye movement (NREM) sleep, and slow wave sleep (SWS) specifically, may be critical for optimal learning during subsequent wakefulness (Kaida et al., 2015; Mander et al., 2011). SWS is characterized by slow wave activity (SWA), defined as high amplitude (>75 μV), low frequency (0.5–2 Hz) EEG activity (Berry et al., 2012). The manipulation of SWA during sleep has been shown to impact subsequent learning (Antonenko et al., 2013; Van Der Werf et al., 2009), suggesting that SWA is critical for restoring learning capabilities (but see Cousins et al., 2018, for contrasting findings). Relatedly, a prominent account hypothesizes that SWA actively functions as a homeostatic regulator of neuronal plasticity (Tononi & Cirelli, 2014). However, this assumption is debated and competing theoretical accounts propose that rapid eye movement (REM) sleep or the succession of NREM and REM sleep are more essential for restoring neuronal plasticity (Navarro-Lobato & Genzel, 2019; Niethard & Born, 2019; Poe, 2017).
Since sleep is assumed to promote subsequent learning, it may also affect learning processes involved in TF-CBT, especially extinction learning. Accordingly, a recent study showed that sleep deprivation in contrast to rested sleep was associated with alterations in brain activity during fear extinction (Seo et al., 2021). However, the interpretation of these results is limited as no psychophysiological or subjective fear conditioning indices were reported. Moreover, sleep manipulation did not directly target fear extinction since the acquisition training was performed preceding the extinction training at the same day. To the best of our knowledge, only one study to date investigated the direct impact of preceding sleep on fear extinction learning: Straus et al. (2017) examined effects of sleep on subsequent fear extinction by manipulating sleep prior to extinction training. Results show that sleep deprivation in contrast to undisturbed sleep did not lead to differences in fear expressions during subsequent extinction training. After a recovery night, however, the pre-extinction deprivation group showed enhanced fear recall during a retention test compared to the undisturbed sleep group. This was reflected in increased startle reactions—but not US expectancy or anxiety ratings—toward the aversive conditioned stimulus (CS+). These results indicate that sleep deprivation prior to fear extinction learning affects fear extinction by interfering with memory encoding and preventing successful recall. However, interpretation of these findings is limited since sleep was manipulated immediately after acquisition training (i.e., within the fear acquisition consolidation window). Thus, it is difficult to disentangle effects of sleep on the consolidation of fear acquisition and on extinction learning in this study design. Moreover, it remains unclear whether the effects are related to SWS specifically or to other sleep stages.
Therefore, we conducted a study to examine effects of early night sleep on subsequent fear extinction learning. Moreover, by using a trauma-adapted fear conditioning experiment, we sought to investigate the relationship between sleep and processes implicated in TF-CBT. Two experimental groups underwent fear acquisition training and a full night of sleep. Neutral faces served as CSs and aversive film clips as USs. On the next day, one group slept during the first 3 hours of the night while a second group remained awake. The amount of SWS and REM sleep are known to be unevenly distributed throughout night sleep. Since SWS is most prominent during early sleep cycles (Yaroush et al., 1971), this design was chosen to contrast effects of SWS-rich sleep in the early night half with wakefulness. At approximately 3 AM, both groups were subjected to fear extinction training and a return-of-fear (ROF) test. During the late night half, both groups were allowed to sleep until morning. Afterward, extinction recall was assessed during a retention test and intrusions were measured using an intrusion provocation task (IPT). We hypothesized that early night sleep, in contrast to wakefulness, enhances extinction learning and leads to a stronger decline in differential fear expressions during extinction training. Moreover, we expected lower conditioned fear expression in the ROF test and in the retention test in the sleep group compared to the wake group. We further explored whether these effects transfer to intrusion frequency. Finally, we sought to investigate whether interindividual differences in outcome measures could be predicted by preceding sleep physiology. Specifically, we hypothesized that higher amounts of SWS and numbers of slow waves are associated with strengthened fear extinction learning and recall as well as fewer intrusions in the sleep group.
Methods
Sample
Sixty-three participants took part in the experiment. Criteria for study eligibility were: age between 18 and 30 years; secondary school certificate or higher; no interpersonal trauma exposure; no clinically relevant depressive symptoms ([PHQ-9] < 10; Löwe et al., 2004) or insomnia ([RIS] < 13; Crönlein et al., 2013); no other acute mental or physical illness; no medication aside from hormonal contraceptives; no pregnancy; no heavy smoking or other drug abuse; no frequent consumption of horror or splatter movies. While participating in the experiment, subjects were instructed to go to sleep at 11 PM and to rise at 7 AM. Furthermore, they were requested to refrain from consuming alcohol and caffeine, and from napping.
Four participants were excluded from further analyses as they withdrew their participation (n = 2) or due to technical errors during the experiment (n = 2). Another eight participants did not meet the criterion for successful differential contingency learning and were therefore excluded from further analyses. Contingency learning was defined as a non-negative difference of US expectancy ratings between CS+ and CS− on the final trial of acquisition training or providing accurate responses on the contingency memory test (see the Conditioning Phases section for more details). 1 The final sample thus comprised 51 participants: wake group: n = 25, 15 females, Mage = 23.48 (SD = 3.29); sleep group: n = 26, 13 females, Mage = 24.12 (SD = 2.92). Groups did not differ in age, t W (47.77) = 0.73, p = .470, gender, X2(1) = 0.19, p = .663, nor in subjective sleep quality (PSQI; Buysse et al., 1989), tW(45.73) = 1.25, p = .217. However, the sleep group showed higher scores than the wake group for trait anxiety (STAI-T; Laux et al., 1981), tW(45.5) = 2.61, p = .012, depressive symptoms, tW(46.45) = 2.45, p = .018, and insomnia symptoms, tW(44.63) = 2.66, p = .011. Secondary analyses revealed significant positive relationships between these characteristics in our sample (rs = .31–.48; all ps < .027). To account for these unexpected pre-experimental differences in further analyses, the scales were z-standardized and summed up into an index of subclinical psychopathology. This index was introduced as a covariate in all subsequent analyses. Further information about pre-experimental group characteristics is provided in Supplementary Material B.
Study procedure
The study procedure is shown in Figure 1a. Approximately 1 week before they participated in the fear conditioning experiment, subjects filled out trait questionnaires and pre-rated a pool of potential CS stimuli (for details see the Conditioning Phases section). The fear conditioning experiment took place in a sound-proof booth on a 27” LCD monitor while participants wore headphones. At the beginning of each experimental phase (Days 1–3), psychomotor vigilance and subjective sleepiness were assessed by means of a short version of the Psychomotor Vigilance Task (PVT; Roach et al., 2006) and the Stanford Sleepiness Scale (SSS; Hoddes et al., 1973). Schematic illustration of the experimental procedure and fear acquisition procedure. Note. Figure 1(a): Experimental procedure. Approximately one week prior to the fear conditioning experiment, participants pre-rated a pool of potential conditioned stimuli (CS). At Experimental Day 1, participants filled out the Stanford Sleepiness Scale (SSS) and underwent Psychomotor Vigilance Task (PVT) and fear acquisition training. Thereafter, they went home and had a full night sleep opportunity from 11 PM to 7 AM. At Experimental Day 2, participants returned to the laboratory in the evening and were then divided into two groups. The sleep group (SG) had a sleep opportunity from 11 PM to 2 AM while the wake group (WG) remained awake during this period. After the sleep period, the SG had time to recover from sleep inertia for 30 minutes (indicated by the gray box). Both groups performed the next experimental session at approximately 2.30 AM including SSS ratings, PVT, extinction training, and return-of-fear (ROF) test. Afterward, both groups had a 3-hour sleep opportunity from 3 to 6 AM. After another 30-minute resting period, participants underwent SSS ratings, PVT, retention test and intrusion provocation task (IPT). Figure 1(b): Phase procedure during acquisition training. Participants were habituated to the CSs while subjective fear and skin conductance responses (SCRs) were recorded. Then, CSs were presented again to assess their expectation of being presented with an aversive film clip (unconditioned stimulus, US) afterward. Thereafter, ten auditory startle probes (noise alone, NA) were presented. Prior to acquisition training participants were instructed to pay attention to the following stimulus contingencies. Fear ratings, SCR and fear-potentiated startle (FPS) were recorded during acquisition training. Thereafter, US expectancy and thoughts associated with the US as well as participants’ explicit memory of the CS-US contingency were assessed. Figure 1(c): Reinforced CS+ (aversive conditioned stimulus) trial from fear acquisition training. Neutral faces, serving as CS, were presented for ten seconds. In a CS+ trial, the CS presentation was followed by 16-second aversive film clips (US). After a CS− (safety stimulus) trial, neutral film clips (control condition, CC) were presented. During the first seven seconds of CS presentation, participants were asked to rate their fear on a visual analog scale. Nine seconds after CS onset, the auditory startle probe was presented and FPS was measured afterward. SCRs were analyzed from the first to the eighth second after CS onset. After each trial, an inter-trial interval (ITI) varied between 15 and 20 seconds. Images were taken from the Chicago Face Database (Ma et al., 2015) and https://www.pexels.com.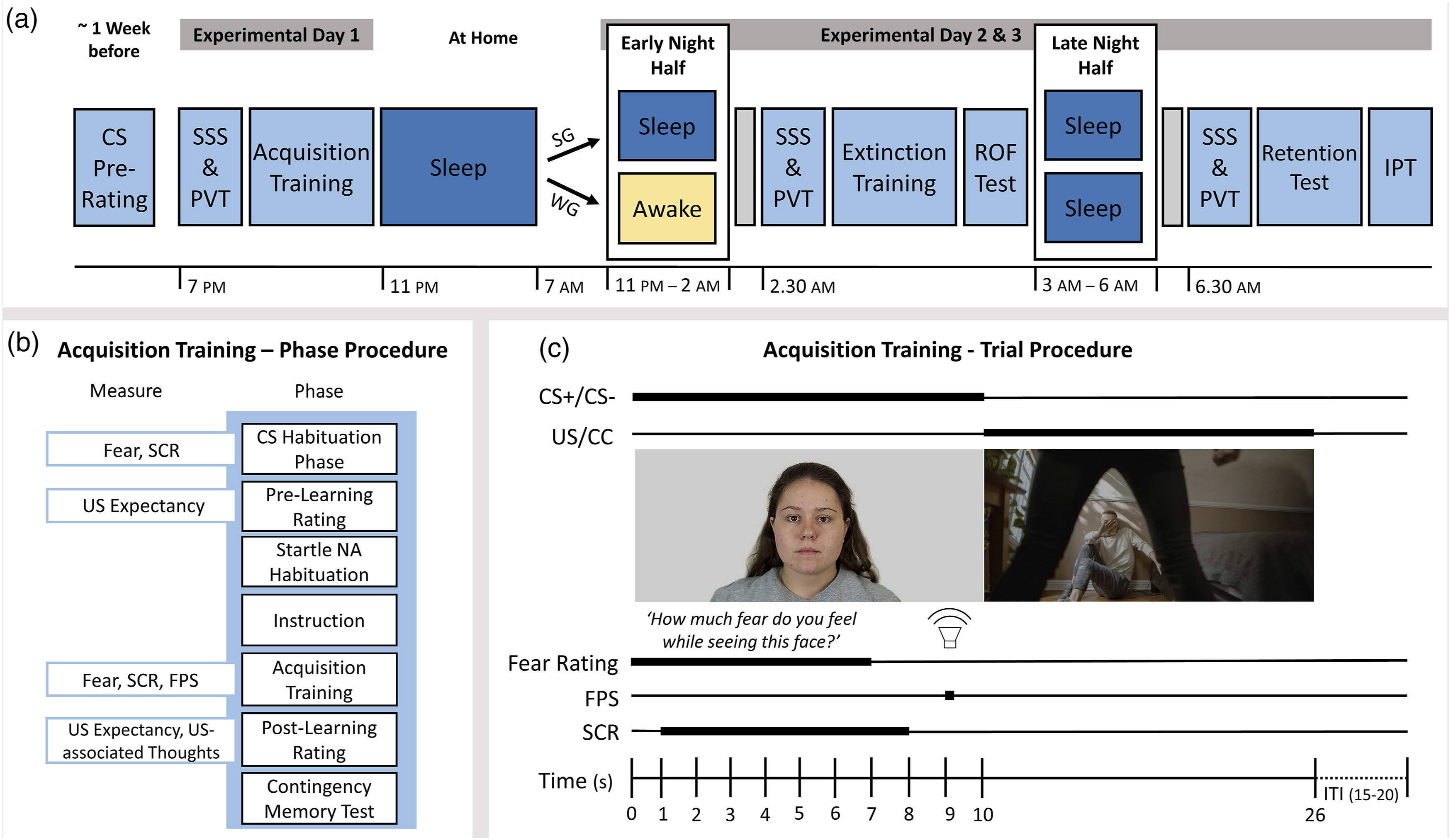
On Day 1, acquisition training took place at approximately 7 PM. Afterward, subjects went home and had a full night sleep period. On Day 2, they returned to the laboratory at 9.30 PM and were prepared for polysomnographic recordings. Thereafter, participants were pseudorandomly divided into two experimental groups. The sleep group received a sleep opportunity from 11 PM. The time of awakening was determined by the time of the first NREM2 epoch plus 3 hours. If participants had not fallen asleep after 30 minutes, the sleep opportunity was set to 3.5 hours in total. After awakening, participants had time to recover from sleep inertia for 30 minutes. The wake group remained awake during the first night half and was continuously monitored by an experimenter. Drinks without caffeine and snacks were provided, and the subjects spent their time reading, crafting, and walking through the corridors. At approximately 2.30 AM, both groups went through the second experimental session including extinction training and the ROF test. Thereafter, both groups had a sleep opportunity during the second night half. As in the early night half, the sleep period was set to 3 hours from the first NREM2 epoch while the maximum time in bed lasted 3.5 hours. After another period of 30 minutes to compensate for sleep inertia, the retention test and the intrusion provocation task (IPT) were performed during the third experimental session at approximately 6.30 AM. Subjects gave written consent according to the Declaration of Helsinki and received € 83 for their participation. The study protocol (A 15-3) was approved by the local ethics committee of the Faculty of Human and Business Sciences at Saarland University and was pre-registered (https://osf.io/fjqcm).
Fear conditioning procedure
Stimuli
Stimuli were adapted from a previous study (Brueckner et al., 2019). Pictures of White male and female persons serving as CS were taken from the Chicago Face Database (Ma et al., 2015). Based on the individual pre-rating valence scores of each participant, two equally neutral rated faces were chosen as conditioned stimuli. This procedure was applied to account for interindividual differences in face perception (Vriends et al., 2011). Nine aversive (USs) and nine neutral (control conditions, CCs) 16-second film clips were taken from commercial movies and contained scenes of interpersonal violence (i.e., physical or sexual assault) or daily activities (e.g., man brushing his teeth and people sitting in a bus), respectively. Details on film clips are provided in Supplementary Material D.
Conditioning phases
Prior to acquisition training, a habituation phase took place, in which two neutral faces (CS) were randomly presented for ten seconds six times each. Meanwhile, subjective fear toward the CS was assessed. At the end of habituation, both CSs were presented again, and participants were asked to rate their expectation of an eventually upcoming aversive film clip after the CS. Afterward, participants were habituated to the startle probe by presenting 10 bursts (50 ms) of white noise at 105 dB. During acquisition training (Figures 1b and c), CSs were each presented 12 times for ten seconds. The CS+, that is, aversive conditioned stimulus, was followed by aversive film clips (US) in nine of 12 trials (reinforcement ratio = 75%), whereas the CS−, that is, safety stimulus, was followed by neutral film clips (CC) with the same reinforcement ratio. Participants were instructed to pay close attention to the different stimuli and whether these were followed by aversive film scenes. The trial order was pseudorandomized in two blocks, assuring that each CS type was not displayed more than twice in a row. During the first 7 seconds of each trial, subjective fear was recorded. Nine seconds after CS onset, a startle probe was presented. The intertrial interval was jittered between 15 and 20 seconds. After acquisition training, the CSs were presented again to re-assess US expectancy as well as US-associated thoughts (Zenses et al., 2021). Finally, explicit memory of the contingency between the CS+ and the USs was measured by asking participants which of the three faces (CSs and a distractor picture) was repeatedly followed by aversive film clips during the task.
The same parameters used during acquisition training were also used during the following experimental sessions. Prior to extinction training and the retention test, a startle habituation phase took place. During extinction training, 12 CS+ and 12 CS− were presented without US or CC. Thereafter, the last two aversive film clips from acquisition training were re-presented to the participants without preceding CS presentation. Fear reinstatement was tested during the subsequent ROF fear test that contained six trials per CS, again without US or CC presentation. The retention test was similar to the ROF test. US expectancy and US-associated thoughts were assessed before and after extinction training, the ROF test (post-assessment only), and the retention test.
Subjective indices of fear
Subjective fear was assessed on a visual analog scale ranging from 0 to 100 (“no fear at all”–“extremely fearful”), which disappeared after responding (7 seconds maximum presentation time). Prior to each conditioning phase, participants received the following instructions to anchor the individual level of fear: Imagine the greatest fear that could occur during this experiment. Consider this your maximum on the scale ranging from “no fear at all” to “extremely fearful.” Pre-post ratings were similarly collected on a visual analog scale ranging from 0 to 100 (US expectancy: “no expectation at all”–“very high expectation”; US-associated thoughts: “not thinking about the aversive film clips”–“very strongly thinking about the aversive film clip”). Analyses of US-associated thoughts during the conditioning phases are provided in Supplementary Material A.
Psychophysiological indices of fear
We assessed fear-potentiated startle and skin conductance responses during each conditioning phase. Analyses of acquisition training did not reveal differential fear learning in fear-potentiated startle. Regarding skin conductance, successful acquisition of conditioned responses was only found in the sleep group. Therefore, no further analyses were conducted. A detailed account of analyses can be found in Supplementary Material A.
Polysomnographic recording and analyses
Nighttime sleep and wakefulness were recorded in accordance with the AASM guidelines (Berry et al., 2012), which included six EEG locations (Fz, Cz, F3, F4, C3, and C4), submental EMG, and EOG on the lower right and upper left canthi. Signals were sampled at a rate of 256 Hz using the SOMNOscreen system (SOMNOmedics GmbH, Germany). Pre-processing and sleep stage scoring was conducted using the programs EEGlab (Delorme & Makeig, 2004) and FASST.2 (Leclercq et al., 2011). Prior to sleep stage scoring, EEG signals were re-referenced to the contralateral mastoid. For wave detection, Fz and Cz were re-referenced to the average mastoid. In accordance with the AASM (Berry et al., 2012), 20-second epochs were visually scored by two independent raters as NREM1, NREM2, SWS, REM, or wakefulness. Slow waves during SWS in the early night half were automatically detected using the built-in algorithm provided by FASST.2 (Leclercq et al., 2011). Each potential slow wave was reviewed manually by a trained research assistant.
Intrusion provocation task
The procedure of the IPT was adapted from Michael et al. (2005) and James et al. (2015). Participants saw two-second blurred pictures taken from the scenes of the nine aversive film clips presented during acquisition training. After viewing all nine pictures, they were instructed to close their eyes for 2 minutes, allowing their mind to wander freely. In addition, they were asked to press the spacebar every time they experienced an intrusive memory triggered by the pictures. Intrusive memories were defined as “vivid images or sounds from the film scenes.”
Data analyses
Statistical tests were conducted in R (R Core Team, 2020) and JASP (JASP Team, 2020). Continuous changes in fear expressions over the learning periods were analyzed by means of linear mixed-effects modeling (LMM) and analysis of variance (ANOVA). LMM analyses were conducted using the R packages nlme (Pinheiro et al., 2017) and reghelper (Hughes, 2021). Plots were built with ggplot2 (Wickham, 2016).
All LMMs were built using the same sequential procedure: For each dependent variable and trials of interest, intercept-only models including a by-subject random intercept were evaluated. Subsequently, the fixed effects (CS type, Trial, and Group) and their interactions were introduced. All predictors were centered, that is, dichotomous predictors were dummy coded (0.5 = sleep group/CS+; −0.5 = wake group/CS−) and the continuous predictor Trial was mean-centered. Thereafter, by-subject random slopes of CS type and Trial were sequentially introduced to the full model if they significantly increased model fit. Grand-mean-centered covariates (sleep characteristics during the early night half, subclinical psychopathology index and SSS ratings) were added to examine their impact on model parameters. 2 Analyses of fear ratings during ROF test and retention test were confined to the first trial of the respective phase to avoid potential re-extinction confounds. Effects were considered significant at p < .05. Degrees of freedom vary across analyses due to missing data. Model parameters and coefficient tables are provided in Supplementary Material C.
The hypothesized effects of the sleep/wake manipulation on fear extinction processes were additionally tested using Bayesian inference. Reported Bayes factor BF0+ quantifies likelihood of the null hypothesis over the alternative hypothesis given the observed data and prior, that is, expected distribution (Wagenmakers et al., 2016). Tests were performed one-sided using the default JZS prior (r = 1/√2) (van Doorn et al., 2020). This was done in addition to the pre-registered analyses, to quantify evidence for the null hypotheses. In deviance to our pre-registered—and also performed—procedure of evaluating three-way interactions, setting up directional hypotheses required reductions in model complexity. The influence of Group on differential fear expressions (CS
Results
Sleep and vigilance
Objective sleep parameters
Sleep parameters in the experimental groups.

Note. NREM = non-rapid eye movement sleep; SWS = slow wave sleep; REM = rapid eye movement sleep; TST = total sleep time.
Subjective sleepiness and vigilance task performance
A mixed ANOVA including the factors Time point, Group, and SSS ratings as dependent variable revealed a significant Group*Time point interaction effect, F(1,98) = 5.67, p = .005, and main effects of Group, F(1,49) =9.94, p = .003, and Time point, F(1,98) = 70.49, p < .001. The wake group reported significantly higher sleepiness levels during the second and third experimental session compared to the sleep group, T2: tW(47.87) = −3.60, p < .001; T3: tW(42.89) = −2.58, p = .013. A mixed ANOVA of PVT reaction times and the factors Time point and Group revealed a global increase of reaction times over time, F(1.48,63.85) = 7.00, p = .004. No other effects were found. Descriptive data and Group comparisons are reported in Supplementary Material B.
Fear conditioning
Introducing the subclinical psychopathology index and SSS ratings as predictors did not significantly improve model fit and had no effect on the direction of effects that are described in the following sections. Means and standard errors of subjective fear and US expectancy ratings during the conditioning phases are shown in Figure 2. Model comparisons and coefficient tables for each conditioning phase are provided in Supplementary Material C. Fear expressions during the conditioning phases. Note. Means and standard errors of subjective fear ratings (SFR, top) and US expectancy ratings (USE, bottom) for CS+ (aversive conditioned stimulus) and CS− (safety stimulus) in the sleep (SG) and wake group (WG). Note that means and standard errors do not represent all components of the linear mixed-effects models that were built by the data; plots are shown for illustration. Brackets indicate analyses of conditioning phases and main outcomes. * < .05.
Subjective fear ratings
Habituation and acquisition training
No CS type or Group effects were found during the last habituation trial (all ps > .592), indicating no baseline differences in subjective fear after familiarization. For acquisition training, analyses revealed a significant interaction between CS type and the slope of Trial, b = 1.21, se = 0.42, 95% CI [0.91, 1.51], t(1128) = 7.84, p < .001, as well as main effects of Trial, b = 0.84, se = 0.21, 95% CI [0.43, 1.25], t(1128) = 4, p < .001, and CS type, b = 13.57, se = 2.86, 95% CI [7.97, 19.16], t(1128) = 4.74, p < .001. Post-hoc contrast revealed a significant rise in subjective fear across trials for the CS+, b = 1.44, se = 0.22, t(1128) = 6.45, p < .001, but not for the CS− (p = .292). Higher fear ratings were evident for the CS+ compared to the CS− on the first, b = 6.92, se = 2.99, t(1128) = 2.32, p = .021, and on the final trial of acquisition training, b = 20.21, se = 2.98, t(1128) = 6.78, p < .001. As CS type effects were not found prior to the acquisition training, these results indicate successful fear acquisition. Moreover, groups did not differ in response patterns during acquisition training (all ps > .659).
Extinction training
Analyses revealed a significant CS type*Trial interaction effect, b = −0.6, se = 0.09, 95% CI [−0.78, −0.43], t(1142) = −6.67, p < .001, a main effect of CS type, b = 14.03, se = 2.97, 95% CI [8.23, 19.83], t(1142) = 4.73, p < .001, and of Trial, b = −0.58, se = 0.18, 95% CI [−0.93, −0.22], t(1142) = −3.19, p = .002. Post-hoc contrasts showed a significant decline in subjective fear over extinction training for the CS+, b = −0.88, se = 0.19, t(1142) = −4.71, p < .001, while the CS− remained stable (p > .138), indicating successful fear extinction. Significant differences between CS types persist from the first, b = 17.35, se = 3.01, t(1142) = 5.77, p < .001, to the final trial of the extinction training, b = 10.71, se = 3.01, t(1142) = 3.56, p < .001, reflecting incomplete extinction. In contrast to our hypothesis, no main effect or interactions with Group were found (all ps > .493). A Bayesian analysis was conducted to examine if the sleep group showed a stronger decline in differential fear ratings compared to the wake group (alternative hypothesis). In correspondence with the LMM results, the analysis indicated that the data were almost three times (BF0+ = 2.915) more likely under the null hypothesis while evidence for this hypothesis lies between anecdotal and moderate.
Return-of-fear test
LMM analyses of the last response during extinction training and the first ROF test trial, revealed a significant interaction between CS type and the linear slope of Trial, b = 6.2, se = 2.42, 95% CI [1.50, 10.89], t(144) = 2.56, p = .012. Main effects of CS type, b = 14.16, se = 3.01, 95% CI [8.34, 19.99], t(144) = 4.71, p < .001, and of Trial, b = 8.57, se = 1.98, 95% CI [4.74, 12.39], t(144) = 4.34, p < .001, were also significant. Post-hoc analyses revealed a significant rise in subjective fear toward the CS+, b = 11.66, se = 2.31, t(144) = 5.05, p < .001, and the CS−, b = 5.47, se = 2.32, t(144) = 2.35, p = .020, reflecting successful fear reinstatement. Differential fear expression responses toward the CS+ compared to the CS− were found across all trials (last extinction training trial: b = 11.07, se = 3.24, t(144) = 3.42, p < .001; first ROF test trial: b = 17.26, se = 3.25, t(144) = 5.31, p = .001. No significant effects of Group were found (all ps > .285), not supporting our hypothesis. Bayesian analyses revealed that differential change between extinction and ROF test is over three times more likely under the null hypothesis (BF0+ = 3.557), that is, there is moderate evidence for the hypothesis of same or more elevation in fear ratings in the sleep group compared to the wake group.
Retention test
A significant main effect of CS type was found for the first trial of the retention test, b = 14.92, se = 3.35, 95% CI [8.32, 21.43], t(48) = 4.45, p < .001, reflecting higher fear ratings for the CS+ compared to the CS−. Contrary to our hypothesis, there were no effects of Group (all ps > .386). Bayesian analyses of group differences on the first trial of the retention test showed that the data are almost five times more likely under the null hypothesis (BF0+ = 4.995, i.e., there is moderate evidence for H0), stating same or higher differential subjective fear in the sleep group compared to the wake group.
US expectancy ratings
Acquisition training
A mixed ANOVA of pre- and post-acquisition US expectancy ratings revealed a significant CS type*Time point interaction effect, F(1,49) = 44.38, p < .001, a main effect of CS type, F(1,49) = 112.49, p < .001, and Time point, F(1,49) = 6.11, p = .017. All other effects were non-significant (all ps > .087). Post-hoc comparisons showed that CS types did not differ significantly prior to acquisition training (p = .377). After acquisition training, however, US expectancy was higher for the CS+ compared to the CS−, b = 64.72, se = 5.44, t(94.6) = 11.89, p < .001. These results reflect successful fear acquisition.
Extinction training
A mixed ANOVA of pre- and post-extinction US expectancy ratings revealed a significant CS type*Time point interaction effect, F(1,48) = 9.37, p = .004, and main effects of CS type, F(1, 48) = 52.90, p < .001, and Time point, F(1,48) = 16.29, p < .001. Post-hoc analyses showed that US expectancy for the CS+ significantly declined from pre- to post-extinction training, b = −18.57, se = 3.71, t(95.8) = −4.99, p < .001, but not for the CS− (p = .943). However, ratings for the CS+ were significantly higher compared to the CS− at both levels of time (ps < .001), indicating a successful though incomplete extinction. In contrast to our hypothesis, the analysis did not show effects of Group (all ps > .365). Bayesian analyses were conducted to examine if the sleep group showed a stronger decline in differential US expectancy ratings compared to the wake group (alternative hypothesis). In line with the ANOVA, results indicated moderate evidence against our hypothesis since these findings are three times (BF0+ = 3.596) more likely under the null than under the alternative hypothesis.
Return-of-fear test
A mixed ANOVA of post-extinction to post-ROF test ratings of US expectancy ratings revealed a main effect of CS type, F(1,49) = 40.97, p < .001, with higher US expectancy for the CS+ compared to the CS−. No other effects were significant, indicating no reinstatement-induced ROF and no effects of sleep manipulation as was hypothesized (all ps > .061). Correspondingly, Bayesian analyses revealed that differential change between extinction and ROF test is almost five times more likely under the null hypothesis (BF0+ = 4.816), thus providing moderate evidence for the hypothesis of same or more elevation in fear ratings in the sleep group compared to the wake group.
Retention test
A mixed ANOVA revealed a significant main effect of CS type, F(1,48) = 50.82, p < .001, with higher US expectancy ratings prior to retention test toward the CS+ compared to the CS− across both groups. Against our hypothesis, no other effects were evident (all ps > .075). In addition, Bayesian analyses of group differences prior to the retention test indicated that data is more than five times more likely under the null hypothesis (BF0+ = 5.448, i.e., there is moderate evidence for H0), stating same or higher differential US expectancy in the sleep group compared to the wake group.
Intrusion frequency
During the IPT, the groups (sleep group: M = 7.62, SD = 6.66; wake group: M = 6.04, SD = 4.87) did not, against our hypothesis, significantly differ in their reported intrusion frequency, tW(45.84) = 0.97, p = .339. Bayesian analyses were conducted to examine if the sleep group showed fewer intrusions during the IPT compared to the wake group (alternative hypothesis). The results indicated moderate evidence against this hypothesis since data is more than six times more likely under the null hypothesis (BF0+ = 6.282). Examining effects of different sleep stages during the early night half on intrusion frequency in the sleep group revealed no significant effects (all ps > .087).
Exploratory analyses on associations between early night sleep characteristics and subsequent fear extinction learning
To examine differences in subjective fear ratings during extinction training associated with preceding sleep during the early night half (minutes of NREM1, NREM2, SWS, REM, and number of slow waves), exploratory subgroup analyses were conducted in the sleep group.
3
Goodness-of-fit tests revealed significant improvements in model fit by including pre-extinction NREM2 (X2 = 13.49, p = .009), SWS (X2 = 16.18, p = .003), and REM (X2 = 38.50, p < .001) as centered predictors. Introducing NREM1 or number of slow waves did not significantly improve model prediction (ps > .820). To examine which of the sleep stages accounted for unique variance in trajectories of fear extinction learning, stepwise model comparisons including Trial, CS type, and different sleep parameters were conducted (all comparisons are listed in Supplementary Material C). Goodness-of-fit tests revealed that REM accounted for incremental variance in models including NREM2 or SWS, respectively (all ps < .001). NREM2 and SWS, however, did not significantly improve model fit when REM was included in the model (all ps > .087). Our final model thus included the factors Trial, CS type and REM. The analysis revealed a significant CS type*Trial*REM interaction, b = −0.02, se = 0.004, 95% CI [−0.03, −0.02], t(579) = −5.75, p < .001, Trial*REM interaction, b = −0.02, se = 0.01, 95% CI [−0.02, −0.01], t(579) = −2.47, p = .014, Trial*CS type interaction, b = −0.66, se = 0.12, 95% CI [−0.96, −0.38], t(579) = −5.52, p < .001, and main effects of CS type, b = 14.13, se = 4.62, 95% CI [5.14, 23.08], t(579) = 3.06, p = .002, and Trial, b = −0.64, se = 0.23, 95% CI [−0.78, −0.49], t(579) = −2.82, p = .005. All other effects were non-significant (all ps > .613). Post-hoc contrasts showed that after high amounts of REM (i.e., M + 1 SD), subjective fear decreased for the CS+, b = −1.88, se = 0.33, t(579) = −5.63, p < .001, but not for the CS− (p = .107; see Figure 3). By contrast, after low amounts of REM (i.e., M − 1 SD), no significant decline in fear ratings was found for the CS+ (p = .844) or for the CS− (p = .780). These results suggest that high amounts of REM sleep during the early night half are associated with improved fear extinction learning, indicated by differential decline in fear ratings across extinction training. We further examined whether early night REM sleep may have influenced fear responses from last acquisition trial to first extinction trial. Analysis did not support the assumption of REM amount being a significant predictor for the change in fear across sleep (p = .269). Moreover, examining effects of early night sleep on other conditioning phases or on US expectancy ratings did not reveal any significant effects. Subjective fear ratings during extinction training divided according to amounts of REM sleep during the early night half. Note. Means and standard errors of subjective fear ratings in the sleep group during extinction training divided through median splits in high and low subgroups of REM sleep (mean-centered rapid eye movement sleep in minutes). Note that subgroups and parameters do not represent all components of the linear mixed-effects models that were built by the data; plots are shown for illustration. Linear mixed model analysis revealed a significant three-way interaction between CS type, Trial and REM sleep amount (indicated by brackets) in the sleep group. Post-hoc tests showed that fear ratings to the CS+ (aversive conditioned stimulus) decreased only after high amounts of early night REM (significant slope indicated by asterisk). * < .05, n.s. >= .05.
Discussion
The current study aimed to investigate whether early night sleep compared to wakefulness facilitates subsequent fear extinction learning and recall. By using a trauma-adapted fear conditioning experiment, we further sought to explore if effects on fear extinction result in fewer intrusions on the next day. In addition, we conducted exploratory regression analyses in the sleep group with the aim of linking specific sleep stages to successful extinction learning and recall. Our analyses did not reveal a stronger decline in fear expressions during extinction training after early night sleep compared to wakefulness. Furthermore, no differences emerged between experimental groups in the subsequent ROF test and the retention test on the next day. The absence of expected group differences was confirmed by Bayesian inference. In contrast to the hypothesis that SWS is critical for learning, successful fear extinction learning was associated with early night REM sleep.
Effects of early night sleep on subsequent extinction learning
Research has frequently shown that sleep is beneficial for encoding (e.g., Cousins et al., 2018; Kaida et al., 2015). Therefore, it has been suggested that the effect of sleep is also evident in extinction learning (Davidson & Pace-Schott, 2020). Contrary to this assumption, the present study did not reveal effects of sleep on subjective ratings during extinction training, the ROF test, or the retention test. Hence, the attempt to enhance fear extinction by means of early night sleep was not successful. The absence of expected group differences was confirmed by Bayesian inference that revealed moderate evidence in favor of the null hypotheses in all analyses except for fear ratings during extinction training, where evidence was between anecdotal and moderate (van Doorn et al., 2020). Furthermore, no effect of sleep on intrusion frequency was observed. It is important to note that only one previous study investigated effects of sleep directly preceding extinction learning, yielding similar results: In line with our findings, Straus et al. (2017) did not find an effect of sleep on extinction training. With regard to extinction recall, both studies showed no differences between experimental groups in US expectancy and fear/anxiety ratings. However, Straus and colleagues did find increased startle reactions toward the CS+ in the pre-extinction sleep deprivation group during fear recall. Since we were not able to conduct analyses of psychophysiological responses during extinction training and recall, future studies are required to further address these findings. Prospective studies should investigate the robustness of extinction memory by, for instance, including a ROF test.
Effects of specific sleep stages on extinction learning
Although we did not find direct evidence for sleep-dependent fear extinction learning and recall, our results indicate that fear extinction training was affected by interindividual differences in early night sleep physiology: NREM2, SWS, and REM during early night sleep predicted trajectories of subjective fear during extinction training. Further analyses, however, suggested that only REM sleep accounted for incremental variance in predicting fear extinction performance. Slow waves during early night SWS failed to predict fear expressions during extinction training. Therefore, our results do not support an effect of preceding SWS on extinction learning. These findings are in line with results from a recent meta-analysis that show no correlation between conditioned responses during extinction training and preceding NREM sleep, including SWS, in healthy individuals (Schenker et al., 2021). Note, however, that pre-extinction SWS percentage was associated with less psychophysiological reactivity to both CS+ and CS− during extinction in patients with insomnia and PTSD.
In contrast to our hypothesis, our exploratory findings suggest a role of REM sleep on subsequent fear extinction. A significant three-way interaction revealed higher fear toward the CS+ compared to the CS− diminished across extinction training after high but not after low amounts of REM sleep. This finding indicates that REM sleep prior to extinction learning may predict successful fear extinction. Please note that these effects were also evident when including the wake group’s extinction performance. Previous investigations on the relationship between REM sleep and subsequent fear extinction have reported mixed findings (Lerner et al., 2017; Spoormaker et al., 2014) and no overall effect was found in a meta-analysis (Schenker et al., 2021). Furthermore, caution is warranted in interpreting these findings: First, although we found a robust effect of early night REM sleep on extinction training, group effects were not evident. Thus, it has yet to be proven that REM sleep contributes substantially to subsequent fear extinction. Second, early night sleep did not affect fear expressions in the ROF test and the retention test. Moreover, REM sleep amounts did not predict intrusion frequency. This lack of consistent effects might be related to the experimental design: Since we manipulated the early night half, the amount of REM sleep in the sleep group was reduced compared to full night sleep. Thus, future studies should examine whether effects of REM sleep on fear extinction may emerge in the ROF test and the retention test after a full night of sleep. Even if effects are not found to persist from extinction training to long-term fear retention, they may have implications for the improvement of TF-CBT: Diminishing the level of distress that patients experience during exposure sessions may be an important target to improve patient’s treatment adherence. As a result, patients may be less reluctant to continue treatment, which could reduce the substantial dropout rate of TF-CBT (i.e., 18 %; Lewis et al., 2020).
Potential mechanisms underlying REM sleep-dependent fear extinction learning
While the assumption that sleep plays a role in promoting optimal neuronal plasticity is widely accepted, the proposed mechanisms are still debated (e.g., Puentes-Mestril & Aton, 2017; Seibt & Frank, 2019): Previous research provided evidence for the assumption that SWA acts as a homeostatic regulator of synaptic plasticity (Huber et al., 2007; Vyazovskiy et al., 2008). However, there are likewise contradicting findings (Chauvette et al., 2012), some of which indicate a more important role of REM sleep in promoting plasticity (Grosmark et al., 2012; Watson et al., 2016). The proposed mechanism by which REM sleep is assumed to promote synaptic plasticity involves the locus coeruleus-norepinephrine (LC-NE) system, which blocks de-potentiation but is nearly inactive during REM sleep (Aston-Jones & Bloom, 1981; Poe, 2017). Several authors aim to reconcile these contradicting accounts and propose that both NREM and REM sleep contribute to neuronal plasticity, albeit in different ways (Navarro-Lobato & Genzel, 2019; Niethard & Born, 2019). Our finding that fear extinction learning was affected by early night REM sleep might therefore reflect a more dominant role of REM sleep in promoting optimal learning conditions. On the other hand, since both SWS and REM sleep are proposed to contribute to synaptic down-selection, the succession of NREM and REM sleep could have led to additive effects in synaptic homeostasis. That is, the effect of SWS may only emerge if followed by a sufficient amount of REM sleep. This could explain why we were able to find associations between REM sleep and extinction learning despite the overall small amount of REM sleep during the early night half. Moreover, this account could explain the fact that we found higher extinction rates in the high REM sleep group than the wake group (as indicated by our follow-up analyses; see footnote 3), yet no significant extinction in the low REM sleep group. This finding may suggest that the disruption of processing during successive SWS and REM sleep may negatively impact extinction learning and that beneficial effects of sleep only surface after a sufficient length of REM sleep. This hypothesis should be investigated by future research.
Beyond the framework of sleep-dependent synaptic homeostasis, REM sleep is further proposed to act as a modulator of emotional memory and reactivity (Goldstein & Walker, 2014; Tempesta et al., 2018). Similar to the proposed mechanisms of REM-dependent synaptic renormalization, the assumed modulation of emotional processing involves the LC-NE system that innervates critical brain regions associated with fear acquisition and extinction (Giustino & Maren, 2018). Based on this framework, REM sleep has been proposed to weaken affective components of memories (Goldstein & Walker, 2014). Whereas some studies have provided evidence in favor of this assumption (e.g., Van Der Helm et al., 2011; Wassing et al., 2019), others have not (e.g., Baran et al., 2012; Lara-Carrasco et al., 2009). Another important assumption arising from this framework is that REM sleep regulates emotion processing during wakefulness. In the context of fear processing, it is suggested that silencing of the LC-NE system during sound REM sleep is critical for preserving balance in fear expression and inhibition since high levels of NE disturb prefrontal control of fear expression in the amygdala (Giustino & Maren, 2018; Goldstein & Walker, 2014). However, evidence for this hypothesis is mixed so far (e.g., Franzen et al., 2009; Wagner et al., 2002). Based on these considerations, our finding that REM sleep predicted fear extinction may correspond to successful inhibition of fear associated with more REM sleep. To address this possibility, we examined whether changes in fear expressions in the sleep group from acquisition to extinction training were affected by REM sleep. These analyses did not yield any significant results, thus not providing any direct evidence of REM sleep effects on emotional reactivity.
Limitations
Our interpretation is limited by several drawbacks of the current study design. First of all, our design does not allow drawing causal inferences about the underlying mechanism by which sleep affects fear extinction. This specifically concerns REM sleep, which our study was not designed to examine, and analyses were made on an exploratory basis. Furthermore, due to the complex design, the number of analyses was relatively large, which increases the risk of false positives. Another limitation is that several confounding influences cannot be disentangled from our sleep manipulation. These include alterations in attention and working memory due to potential sleep deprivation in the wake group (Krause et al., 2017), which were reflected in increased SSS ratings. Although secondary analyses did not reveal any effect of sleepiness on subjective ratings, such effects cannot be ruled out entirely and may have influenced fear extinction learning. Furthermore, the sleep group exhibited a relatively high amount of NREM1 and NREM2 while time spent in SWS and REM was relatively low thorough the course of night. This may have influenced our effects and is likely due to the unfamiliar setting during sleeping as participants were not adapted to the sleep laboratory, that is, first night effect (Agnew et al., 1966). Another potential confound concerns the group differences in sleep physiology prior to the retention test, which also complicates measuring effects on intrusions. Deprivation of SWS induces sleep pressure, resulting in rebound effects during recovery sleep (Borbely et al., 2016). As described in Table 1, these effects were also evident in the wake group compared to the rested sleep group during the second night half. Research suggests that sleep, in particular REM sleep, promotes the consolidation of fear extinction memory while strong evidence in favor of this assumption is currently missing (Davidson & Pace-Schott, 2020). Hence, it is likely that different sleep patterns between groups may have influenced fear expressions during the retention test, obscuring effects of our initial sleep manipulation. This reduces the comparability of our study and the study by Straus et al. (2017) that tested retention after a whole recovery night. In addition, comparability with their findings is restricted as we did not find any indication of successful fear acquisition across both groups in the psychophysiological data and thus refrained from conducting further analyses. The failure of successful fear acquisition in psychophysiological measures may have resulted from gradual habituation. Moreover, non-differential skin conductance responses during acquisition training may be related to the presentation of startle probes since these have been shown to impact fear learning (de Haan et al., 2018; Sjouwerman et al., 2016). On the other hand, analyses of skin conductance revealed differences between groups in fear acquisition, which may be related to baseline differences in indices of subclinical psychopathology. Although introducing this covariate into analyses did not change the direction of effects, we cannot fully rule out potential pre-manipulation effects.
Conclusion
The current study investigated whether early night sleep compared to wakefulness facilitates subsequent fear extinction learning and recall. Our results did not confirm that preceding sleep promotes fear extinction. Moreover, sleep did not affect intrusions of the US. Not supporting our hypothesis, exploratory analyses suggest that early night REM sleep—rather than SWS— predicts successful fear extinction learning. This finding may indicate that REM sleep promotes optimal conditions for subsequent fear extinction learning. Future studies are required to confirm these findings. Moreover, clinical trials should evaluate the usefulness of boosting REM sleep and preserving the cycling alteration of SWS and REM sleep by preventing awakenings during the nighttime. Promising approaches could be, for instance, applying cognitive behavioral therapy for insomnia prior to exposure session to improve TF-CBT efficacy.
Supplemental Material
Supplemental Material - Investigation of early night sleep effects on subsequent fear extinction learning and recall
Supplemental Material for Investigation of early night sleep effects on subsequent fear extinction learning and recall by E Friesen, MR Sopp, AH Brueckner, D Ferreira de Sá, and T Michael in Journal of Experimental Psychopathology
Footnotes
Acknowledgments
Gratitude is expressed to D. Reis for her advice on statistical modeling. Furthermore, we gratefully acknowledge A. Hennes, J. Weber, R. Braun, R. Giesler, Y. Neumann, C. Daab, H. Layes, J. Groß, P. Heubl, V. Friesen, M. Hündersen, and L. Müller-Kirchen for assisting with data collection. Finally, we thank all our participants.
Author contributions
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the German Research Foundation (SO 1716/1-1).
Data availability
Materials and data are available on request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
