Abstract
Performing eye movements during memory retrieval is considered to be important for the therapeutic effect of eye movement desensitization and reprocessing (EMDR). We conducted a meta-analysis of laboratory studies that compared the effects of eye movements and/or alternative dual tasks (e.g., counting) on the vividness and emotionality of negative autobiographical memories with recall only (control) conditions. The databases PsycINFO and Web of Science were queried. Fifteen studies that involved 942 participants were included. Eye movements and alternative dual tasks produced similar vividness and emotionality decreases, with the impact on vividness being strong than that on emotionality. However, eye movements yielded a stronger overall vividness reduction than alternative dual tasks, although the associated effect size was small (Cohen’s d = .29). Because eye movements and alternative dual tasks produced comparable effects, one might conclude that both tasks are therapeutic equivalents. However, it should be acknowledged that only a limited number of laboratory studies were included in our meta-analysis, and the degree to which both procedures tax working memory was not independently established. Although our conclusion cannot be generalized to clinical practice, it does raise questions about the mode of action of EMDR.
Patients with post-traumatic stress disorder (PTSD) suffer from memory-related symptoms such as intrusions (i.e., recurrent, involuntary, and distressing recollections of the traumatic event) and flashbacks (i.e., dissociative reactions in which the individual feels as if the traumatic event is recurring) after exposure to a trauma (American Psychiatric Association, 2013). Over the past decades, eye movement desensitization and reprocessing (EMDR; Shapiro, 1989) has become increasingly popular as a treatment method for PTSD.
During EMDR therapy, the patient is instructed to retrieve and visualize a disturbing memory of the traumatic experience and to focus on this memory and its related thoughts and feelings while simultaneously performing eye movements (Shapiro, 2002). A commonly voiced view is that eye movements make the negative emotions attached to the trauma memory less outstanding by reducing their vividness and emotionality.
Experimental studies examining the mechanisms of action underlying EMDR have mainly focused on the eye movement component of EMDR (see, for a review, Landin-Romero et al., 2018). The typical procedure in such studies is that participants are instructed to retrieve emotional autobiographical memories and to rate these memories on vividness and emotionality (pretest). Next, some participants have to retrieve the memory while simultaneously performing eye movements, whereas other participants recall the memory without performing eye movements (i.e., control condition). Afterward, participants again rate their memories on vividness and emotionality (posttest). Lee and Cuijpers (2013) conducted a meta-analysis on how eye movements affected emotional memories, notably their vividness and emotionality. Fifteen clinical trials and 11 laboratory studies that compared standard EMDR procedures with eye movements against EMDR procedures without eye movements were included. The clinical studies relied on the Subjective Units of Distress scale (SUDs) and Validity of Cognition (VoC) as outcome variables. The SUDs measures the degree of distress elicited by the trauma memory, while the VoC assesses the believability of a positive cognition in relation to that memory. The laboratory studies relied on Likert-type vividness and emotionality scales.
Overall, Lee and Cuijpers (2013) found that the SUDs declined more in participants who received EMDR with eye movements than in those who were given EMDR without eye movements. This between-treatment difference was associated with a medium effect size (ES) (Cohen’s d = .53). EMDR with eye movements also yielded greater increases in the VoC than EMDR conducted without eye movements, a difference that was associated with a large ES: Cohen’s d = .72. In a similar vein, the laboratory studies observed larger reductions in vividness and emotionality ratings of negative autobiographical memories following an EMDR procedure with eye movements as compared with a procedure without eye movements. Here, Cohen’s ds were .91 and .66, respectively, which can be interpreted as large and medium ESs (see van den Hout & Engelhard, 2012). Taken together, these findings suggest that the eye movement component of EMDR possesses a unique therapeutic potential.
Proposed working mechanisms of EMDR
Still, there is discussion about the working mechanism of EMDR (Landin-Romero et al., 2018). Initially, authors speculated that performing horizontal eye movements might be similar to the rapid eye movements during sleep and that both would promote a reorganization of traumatic memories and subsequent emotional processing. Others speculated that the communication between both hemispheres increases when horizontal eye movements are performed (Gunter & Bodner, 2008). However, the beneficial effect of EMDR (i.e., reductions in subjective vividness and emotionality) has also been documented with vertical eye movements, a finding that contradicts the interhemispheric communication hypothesis that emphasizes the unique therapeutic value of horizontal eye movements (Gunter & Bodner, 2008). More recently, de Voogd et al. (2018) found tentative evidence that eye movements may reduce amygdala activity, which could be a biological explanation for the observed reduction in emotionality.
An explanation for the mechanism underlying EMDR that has gained some empirical support is the working memory (WM) account (Gunter & Bodner, 2008; van den Hout & Engelhard, 2012). According to this account, retrieving a trauma memory while simultaneously performing eye movements can be seen as a dual task that taxes WM. The capacity of the WM is limited (Baddeley, 1998), and so the consequence of dual task performance is that it interferes with the full-blown retrieval of negative memories. Thus, according to the WM account, the combination of memory retrieval and simultaneously performing eye movements will lead to a degraded version—in terms of vividness and emotionality—of the trauma memory(i.e., imagination deflation; van den Hout & Engelhard, 2012). As a result, the traumatic memory will become less emotional and this degraded version is subsequently stored during reconsolidation.
Alternative dual tasks
Although eye movements are considered to be an essential ingredient of EMDR (Shapiro & Laliothis, 2015), alternative dual tasks (e.g., bilateral tones and tapping) are also used in current clinical practice because some patients prefer performing such activities to eye movements (van den Hout et al., 2012). To the extent that alternative dual tasks are as effective as eye movements in reducing vividness and emotionality, eye movements are not contributing in a unique way to the therapeutic effects of EMDR and this may cast doubts on the central role typically ascribed to eye movements (Shapiro, 1989; Solomon & Shapiro, 2008).
Gunter and Bodner (2008) reasoned that the WM account would predict any dual task to reduce the vividness and emotionality of memories as long as the task sufficiently taxes WM. To test this notion, van den Hout and colleagues (2010) investigated whether counting during recall taxed WM. Participants were asked to retrieve negative autobiographical memories and to rate them on vividness and emotionality. Following this, they participated in the following conditions: simple counting (subtracting 2 from 450), complex counting (subtracting 7 from 450), or a control condition (no counting). During counting, participants also performed a reaction time task (i.e., responding to presented letters) as a manipulation check to assess to what extent the alternative dual tasks taxed their WM. Afterward, participants retrieved the negative autobiographical memories once more and again rated them on vividness and emotionality. Manipulation checks indicated that these dual tasks indeed did tax WM. Both counting conditions were associated with decreased memory vividness and emotionality ratings as compared to the control condition.
Other studies have found less convincing evidence for vividness and emotionality reductions due to alternative dual tasks. For instance, van den Hout et al. (2001) asked participants to recall positive or negative autobiographical memories and then assigned them to an eye movement, finger tapping (i.e., tapping the tabletop with one’s index finger), or a control condition. Subsequently, the memory was retrieved again and memory vividness and emotionality were rated once more. Van den Hout et al. (2001) found that only in the eye movement condition, memory vividness and emotionality decreased.
The current meta-analysis
In sum, studies have yielded inconsistent results regarding the effectiveness of alternative dual tasks in reducing vividness and emotionality of aversive memories. To address this issue in a more systematic way, we conducted a meta-analysis. Our goal was twofold: (1) to provide an update of laboratory studies to the meta-analysis of Lee and Cuijpers (2013) and (2) to specifically examine the effect of alternative dual tasks on vividness and emotionality of autobiographical memories. As the current meta-analysis had different inclusion and exclusion criteria (see below), not all laboratory studies of Lee and Cuijpers (2013) were included. Our meta-analysis might be informative for clinicians, because if we would find, for example, that eye movements do not outperform alternative dual tasks in reducing vividness and emotionality, there is at least empirical justification for exploring the use of alternative dual tasks in clinical settings (e.g., when patients prefer an alternative dual task such as bilateral tones or tapping).
Method
Literature search and selection of studies
We used a systematic review method following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (Moher et al., 2009; see https://osf.io/qp67t/for the completed PRISMA checklist).
Our literature search was conducted in the last 2 weeks of January 2019. Figure 1 shows a PRISMA flowchart for the selection of eligible studies. The databases PsycINFO and Web of Science were queried using the following keywords: (EMDR OR eye movement desensitization OR eye movement desensitization therapy OR eye movement desensitization and reprocessing) AND (eye movements OR dual task OR bilateral stimulation OR dual taxation). An initial number of 2,852 records was found. After removing duplicates, 1,780 articles were screened. We excluded: (i) review articles, commentaries, books, and dissertations; (ii) studies in which physiological manipulations were carried out or embedded in the eye movement condition (e.g., EEG, ERP’s, etc.); and (iii) case reports or treatment studies (i.e., whole EMDR protocol).

PRISMA flow chart diagram of meta-analysis study selection. Asterisk (*) indicates articles may have met more than one exclusion criteria.
We only included articles that addressed negative autobiographical memories. This type of personal recollections seems to be the best proxy of traumatic memories in patients with PTSD. More specifically, we excluded the studies of Andrade, Kavanagh, & Baddeley (1997; black/white newspaper pictures), Littel, Remijn, Tinga, Engelhard, & van den Hout (2017; neutral memories), Nieuwenhuis et al. (2013; words), Leer et al. (2017; faces), Engelhard, van den Hout, and Smeets (2011; participants who watched the Queen’s Day tragedy via television), and Houben et al. (2018; participants watched a trauma film). We further excluded two studies that focused on flash-forwards (i.e., future-oriented images; Engelhard, van den Hout, Janssen, & van der Beek, 2010; Engelhard, van den Hout, Dek, et al., 2011). In addition, we excluded studies that did not rely on a visual analogue scale (VAS) to measure vividness and emotionality (Christman et al., 2003; Greenwald et al., 2013; Greenwald et al., 2015) and/or that solely relied on patient data (de Jongh et al., 2013; Lilley et al., 2009; Thomaes et al., 2016). A number of additional studies were identified by manual searches of review articles (e.g., Lee & Cuijpers, 2013, van den Hout & Engelhard, 2012). This initial search resulted in 24 potentially eligible articles. However, four of these studies were discarded because the manipulation (i.e., eye movements, alternative dual task, or control instruction) also involved some other task (e.g., presentations of screenshots of event, letter identification during eye movements, and a manipulation of the knowledge about EMDR; Cuperus et al., 2019; Kavanagh et al., 2001; Onderdonk & van den Hout, 2016; Littel, van Schie, & van den Hout, 2017). And five further studies had to be excluded because of missing data, although we did make an attempt to obtain the data by sending the first and/or second authors of these articles a request to share their data (Gunter & Bodner, 2008; Hornsveld et al., 2010; van den Hout et al., 2013; van den Hout, Engelhard, Beetsma, et al., 2011; van den Hout et al., 2012). Datasets from four studies were unavailable, the authors of the remaining study did not reply.
This resulted in a total of 15 studies that described 16 separate experiments and included 942 research participants. Thirteen of these studies (including14 experiments and 867 participants) involved a comparison between eye movement and control conditions (see Table 1). Seven studies (including 8 experiments and 353 participants) involved a comparison between alternative dual tasks and control conditions (see Table 2). These studies used a range of alternative dual tasks, including the shape sorter task, playing a Tetris game, binaural stimulation (beeps), simple counting (subtracting 2 from 450), complex counting (subtracting 7 from 450), tapping, articulatory suppression (counting from 1), and listening to counting (1–60). In all studies, the duration of the alternative dual task was similar to the duration of eye movements (e.g., 4 × 24 s; see Table 2). For direct comparisons involving alternative dual tasks and eye movements (see Table 3), we only included studies that comprised both conditions. Here, 5 studies describing 6 experiments and 278 participants were eligible.
Overview studies included comparison eye movements versus control.
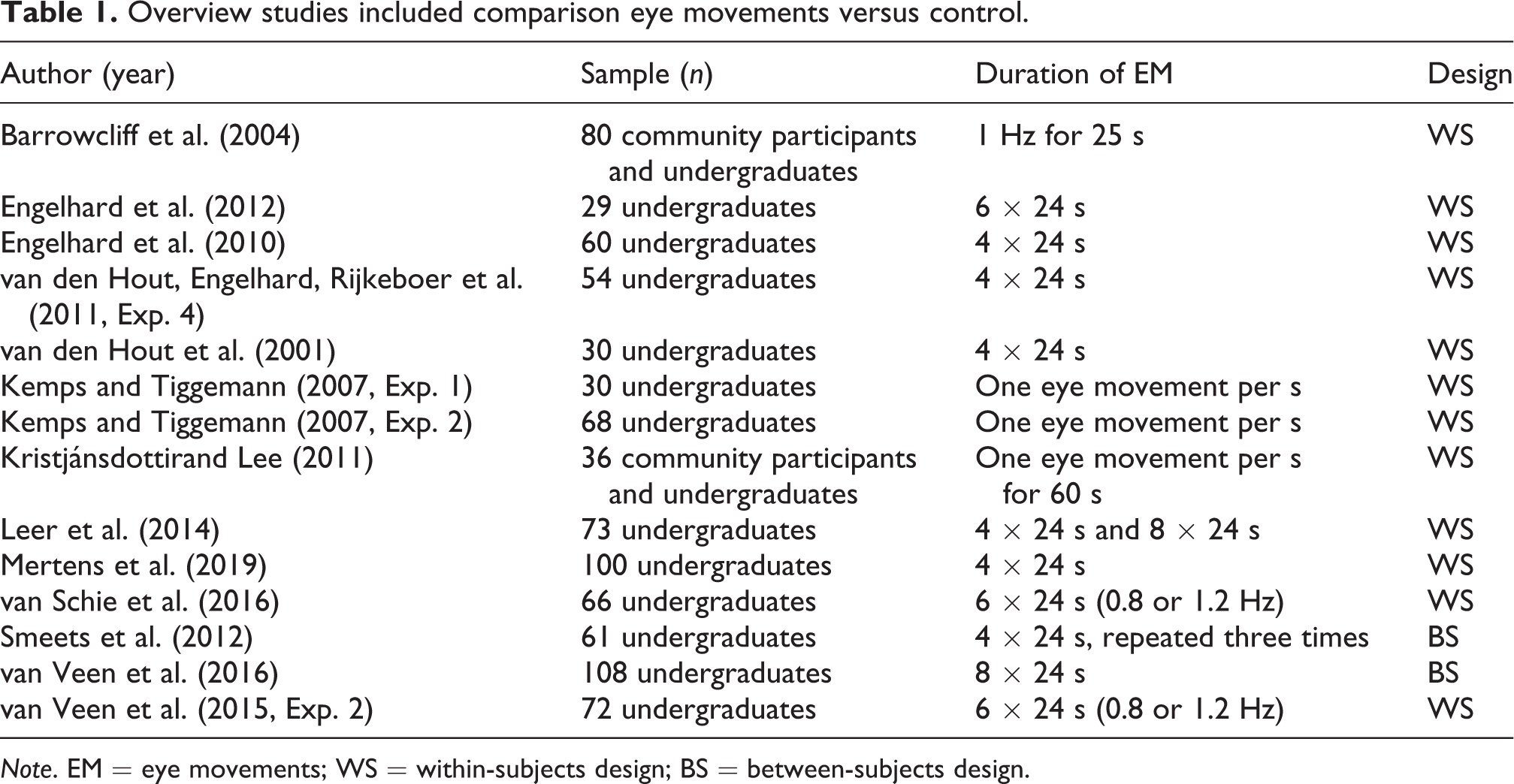
Note. EM = eye movements; WS = within-subjects design; BS = between-subjects design.
Overview studies included comparison alternative dual task versus control.

Note. BS = between subjects; WS = within subjects; n.s. = not specified.
Overview studies included comparison alternative dual tasks versus eye movements.
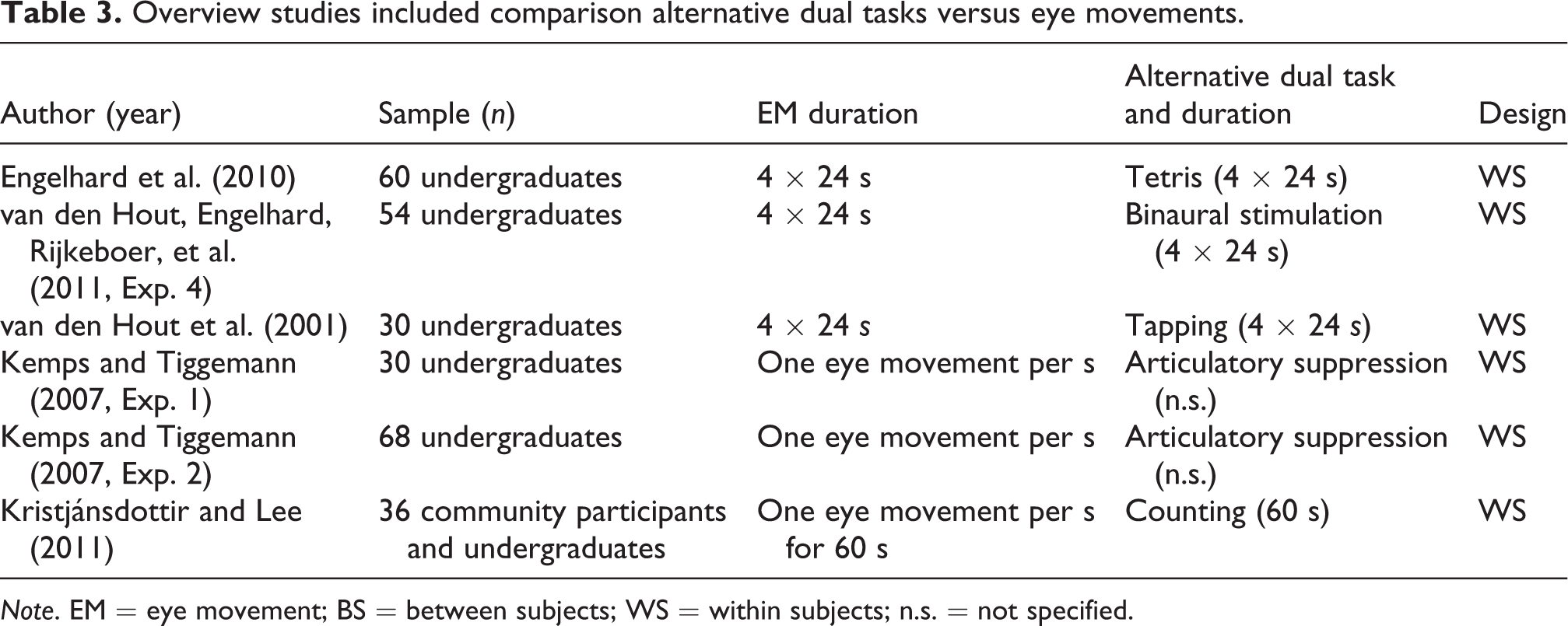
Note. EM = eye movement; BS = between subjects; WS = within subjects; n.s. = not specified.
Meta-analytic procedure
Cohen’s d was chosen as the ES indicator, because we focused on two experimental conditions (i.e., eye movements and alternative dual tasks) that each was contrasted with a control condition (memory recall only). For each study, Cohen’s d was calculated by subtracting the average posttest score of the control condition from the average posttest score of the experimental condition and dividing the result by the pooled standard deviations of post-intervention scores as reported for the control and experimental conditions. ESs were calculated for the following contrasts: (1) eye movements versus control, (2) alternative dual tasks versus control, and (3) eye movements versus alternative dual tasks. Mean Cohen’s ds were calculated for vividness and emotionality separately. ESs of 0.80 and higher were interpreted as large, ESs of 0.5–0.8 as medium, and ESs below 0.5 as small (Cohen, 1988). Several studies included multiple experimental conditions, for example, performing eye movements at different speeds (van Schie et al., 2016; van Veen et al., 2015), individual differences in WM capacity (e.g., van Schie et al., 2016), or type of memory (i.e., visual versus auditory; Kemps & Tiggemann, 2007). Hence, multiple ESs were obtained for these studies (e.g., slow eye movements vs. control and fast eye movements vs. control). Forest plots were constructed using the statistical software package JASP (JASP Team, 2019; version 0.11.1). For data file and analysis, see https://osf.io/5f6d2/.
Additionally, heterogeneity analyses were performed by calculating I 2 with JASP (JASP Team, 2019). The I 2 index can be interpreted as the percentage (range 0–100%; Higgins & Thompson, 2002) of the total variability between studies. Specifically, it is an indicator of what proportion of the observed variance would remain if sampling error variability across studies would be eliminated (Borenstein et al., 2017). A low index (<25%) means that variability in ES estimates are mainly due to sampling error, whereas high indices (>75%) imply true heterogeneity across studies (Higgins & Thompson, 2002).
Publication bias was tested by inspecting the funnel plot on the primary outcome measures (JASP Team, 2019). A funnel plot is a visual representation of study precision (1/SE) against the observed ES. In the absence of publication bias, the plot is expected to be symmetrical in the vertical axis around the meta-analytic compounded ES estimate. The points that represent the studies should lie in a confidence triangle of 95%. Points outside this interval can result from heterogeneity and/or study differences. Asymmetry of the funnel plot is regarded as evidence for publication bias (Grasman, 2017).
Results
Effects on vividness
The mean ES (Cohen’s d; see Table 4) for the difference between eye movements and the control condition was .59 (95% confidence interval (CI) [0.42, 0.77], see Figure 2(a)). For the comparison between alternative dual tasks and control, a Cohen’s d of .49 (95% CI [0.14, 0.85], see Table 4 and Figure 2(b)) was obtained. The mean ES for the difference between eye movements and alternative dual tasks was .29 and the lower part of its associated CI came close to zero (95% CI [0.03, 0.56], see Table 4 and Figure 2(c)). All comparisons were associated with substantial heterogeneity. More specifically, we found heterogeneity values (I 2%) of 75.24% (95% CI [57.99, 89.52]), 84.74% (95% CI [67.28, 95.96]), and 66.61% (95% CI [20.55, 92.35]) for eye movements versus control, alternative dual tasks versus control, and eye movements versus alternative dual tasks, respectively.
Mean Cohen’s d, 95% CI and heterogeneity.

Note. EM = eye movements; CI = confidence interval; I 2 = heterogeneity.

Forest plot for (a) eye movements versus control, (b) alternative dual tasks versus control, and (c) eye movements versus alternative dual tasks for vividness. Note. RE model = random-effects model.
Effects on emotionality
A Cohen’s d of .28 (95% CI [0.15, 0.42]) was found for the comparison between eye movements and control (see Table 4 and Figure 3(a)). For the difference between alternative dual tasks and control, a mean Cohen’s d of .32 (95% CI [0.14, 0.50], see Table 4 and Figure 3(b)) was found. A Cohen’s d of .02 (95% CI [−0.27, 0.31]) was found for the comparison between eye movements and alternative dual tasks (see Table 4 and Figure 3(c)). Moderate heterogeneity values (I 2%; 56.01% (95% CI [27.24, 83.42]) and 42.62% (95% CI [0.00, 78.72]) were found for the first two comparisons, whereas a substantial heterogeneity (72.81%, 95% CI [33.62, 94.65]) was evident for the comparison between eye movements and alternative dual tasks.

Forest plot for (a) eye movements versus control, (b) alternative dual tasks versus control, and (c) eye movements versus alternative dual tasks for emotionality. Note. RE model = random-effects model.
Publication bias
Funnel plots can be checked at https://osf.io/bwe29/. Neither visual inspection of the funnel plots nor the trim-and-fill procedure (Duval & Tweedie, 2000) identified substantial asymmetry. Most studies were projected within the confidence triangle. The few studies that fell outside the triangle tended to rely on a low sample size.
Discussion
Using a meta-analytic approach, we compared to what extent eye movements and alternative dual tasks reduce the vividness and emotionality of aversive memories. The main results can be summarized as follows. First, we found that eye movements and alternative dual tasks produced similar reductions in vividness and emotionality in comparison with control conditions. The current meta-analysis obtained a medium ES (Cohen’s d = .59) for vividness and a small ES (Cohen’s d = .28) for emotionality when eye movements were contrasted against control conditions. This is in line with the findings of Lee and Cuijpers (2013) who found a larger ES for vividness (Cohen’s d = .91) than for emotionality (Cohen’s d = .66) when they compared eye movements and control conditions of laboratory studies. Because both meta-analyses had different inclusion and exclusion criteria (e.g., randomized controlled trials and alternative dual task), the precise ES values can be expected to differ. What is remarkable is that both meta-analyses obtained an overall stronger effect of eye movements on vividness than on emotionality. In the current study, a similar pattern emerged when alternative dual tasks were compared with control conditions. We found a medium ES (Cohen’s d = .49) for vividness and a small ES (Cohen’s d = .32) for emotionality. Second, when eye movements were compared to alternative dual tasks, small ESs for both vividness (Cohen’s d = .29) and emotionality (Cohen’s d = .02) were found. Although this indicates that eye movements reduce vividness more effectively than alternative dual tasks, we do not know whether the modest ES associated with this difference is clinically relevant. Establishing such a clinically noticeable effect would require another type of metric (e.g., “minimal important difference”; Cuijpers et al., 2014). In addition, one of the main aims of EMDR is to reduce the emotional valence of the traumatic memory (Shapiro, 2002). Our results suggest that both approaches may be used to obtain this therapeutic effect.
Our meta-analytic results accord well with a main prediction that can be derived from the WM account (Gunter & Bodner, 2008), namely, that both alternative dual tasks (that presumably tax WM) and eye movements are effective in reducing the vividness and emotional load of negative memories. This raises the more fundamental question whether eye movements are an essential ingredient of EMDR. Because our analysis did not involve clinical groups, our data cannot answer this question. At the very least, they provide some empirical justification for EMDR therapists using alternative dual tasks when patients indicate that it is not feasible for them to perform eye movements.
The included studies have some constraints. For example, their sample size varied considerably: The lowest samples size was N = 29 (Engelhard et al., 2012), while the highest was N = 108 (van Veen et al., 2016). Of course, ESs generated by studies with small samples have to be interpreted with caution. Also, most of the included studies (i.e., 12 of 15) employed a within-subjects design (e.g., van Veen et al., 2015), which means that participants completed all experimental conditions. This type of design might lead to carryover effects and probably introduces some noise in the ES estimates (Quintana, 2015). In addition, although we specifically included studies that relied on VASs of vividness and emotionality, such self-report measurements can be prone to demand characteristics (Kearns & Engelhard, 2015; Kanter et al., 2004). Littel, van Schie, and van den Hout (2017; Experiment 1) found some tentative evidence that expectancy effects drive the effectiveness of EMDR. However, in their second experiment, these authors noted that providing information about EMDR did not influence the vividness and emotionality ratings of participants. Lastly, our heterogeneity analyses showed moderate to substantial heterogeneity between studies. However, I 2 estimates should be taken as provisional estimates because the current meta-analysis relied on a limited number of studies (von Hippel, 2015). Although no publication bias was observed, our test might be underpowered because of the limited number of studies (Carter et al., 2019).
The current meta-analysis has several shortcomings that limit its generalization to clinical settings. First, only a relatively small number of laboratory studies was included (k = 15). Note, however, that other meta-analyses in the EMDR domain included similar numbers of studies (see e.g. Khan et al., 2018; Moreno-Alcázar et al., 2017).
Second, all alternative dual tasks (e.g., tapping and counting) were combined for the analyses, which may mask an effect of modality. Therefore, we cannot conclude whether a specific alternative dual task might yield superior therapeutic effects. For example, Andrade et al. (1997) hypothesized that visuospatial tasks would be particularly effective as they tax the visuospatial sketch pad, which is the part of WM primarily used when retrieving memories. However, no modality-specific effect was found (see e.g., van Schie, van veen, & Hagenaars, 2019). The current meta-analysis did not evaluate the degree to which these alternative tasks tax WM. Advocates of the WM account (van den Hout & Engelhard, 2012) proposed an inverted U-shape, which means that too little or too much taxing will produce no or little effect on the vividness and emotionality of memories. However, Littel and van Schie (2019) found no evidence for the inverted U-curve. Hence, more research is required to examine modality-specific taxing and the amount of WM taxation.
Third, the search terms that we used for the meta-analysis are perhaps too specific and we may have missed potentially relevant studies. For example, studies have also focused on the effect of eye movements on positive memories (e.g., Engelhard, van den Hout, Janssen et al., 2010). Although not central to the aims of the current study, we conducted exploratory analyses on these studies and a similar pattern emerged (see https://osf.io/bf2k7/). That is, only small ESs were obtained in favor of eye movements when compared to alternative dual tasks. Unpublished data sets were requested, but either no replies were received or authors indicated that there were no unpublished data sets.
To sum up, the eye movement component of EMDR intends to diminish the emotional impact of negative memories (Shapiro, 2002). We showed that a similar effect could be obtained with alternative dual tasks in laboratory studies. Furthermore, in line with Lee and Cuijpers (2013), our current meta-analysis indicates that eye movements, or alternative dual tasks for that matter, impact vividness more strongly than the emotional load of a negative memory. This is not necessarily bad news for EMDR, because the pattern is entirely consistent with the WM account of its therapeutic potential: eye movements or alternative dual tasks interfere with WM, which reduce the vividness of negative memories, which in turn lowers their associated negative value (Gunter & Bodner, 2008; Smeets et al., 2012). The important point here is that vividness may play a more prominent role in the therapeutic effect of EMDR than initially assumed.
Although the importance of eye movements is emphasized in Dutch EMDR protocols (Beer et al., 2011), our findings do encourage EMDR therapists to explore a wider variety of alternative interventions that are perhaps equally effective in reducing the vividness and emotionality of aversive memories. EMDR theorists often assume that decreases in subjective units of distress should result in a reduction of symptomatology. However, so far no convincing link between changes in vividness and emotionality (due to eye movements or alternative dual tasks) and changes in symptomatology has been found (van Schie et al., 2019). More research on the underlying mechanisms of EMDR and on the effectiveness in reducing symptomatology is needed to draw firm conclusions about the mode of action of EMDR.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
