Abstract
In this study, dissolving pulp from hardwood, Trema orientalis L. was used as cellulose pulp for the synthesis of cellulose acetate (CA). CA was prepared by varying time of swelling with acetic acid, duration of reaction and temperature of reaction. The degree of substitution (DS) of all prepared CA was higher than 2.5, indicating the formation of cellulose triacetate (CTA). The maximum yield of CTA was 157 wt-% after 3 hours of cellulose pulp swelling followed by a 1.5-hour acetylation reaction at 35 °C. The all prepared CA samples showed good solubility in dimethyl sulphoxide (DMSO), N,N-dimethyl formamide (DMF) and tetrahydrofuran (THF). The creation of strong absorption band at 1740 cm−1 for carbonyl (–C=O) group confirms successful acetylation reaction. The cellulose crystallinity diminishes as the acetylation progresses as observed by X-ray diffraction (XRD). The prepared CA samples were also characterised using thermogravimetric analysis (TGA), differential scanning calorimetry (DSC) and dynamic viscosity measurements. The CA/DMSO solutions exhibited shear thinning property at lower shear rate and higher operating temperature. Increased dynamic viscosity was observed with the increase in concentration of CA in the DSMO solvent. The crystallinity and thermal stability of CA samples was highly influenced by the duration of acetylation and temperature of reaction.
This is a visual representation of the abstract.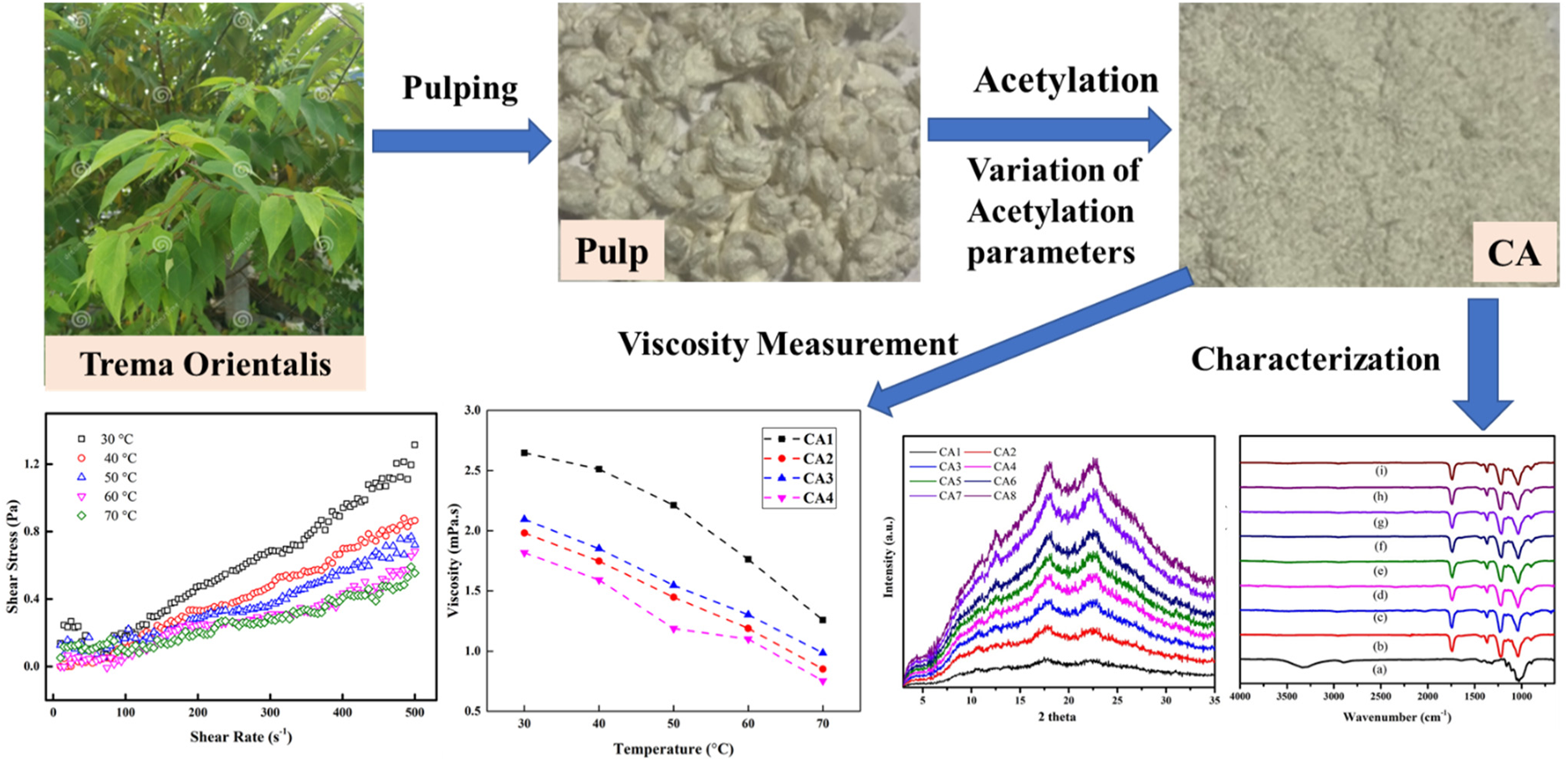
Get full access to this article
View all access options for this article.
