Abstract
Cardiovascular disease (CVD) is one of the leading causes of death in patients with severe mental illness (SMI), being ~3.3 times higher than in the general population. The adverse cardiovascular effects of psychiatric medications have short- and long-term consequences that contribute to higher rates of cardiovascular death in patients experiencing SMI. By understanding these adverse effects, clinicians can better address cardiovascular health inequalities in patients with SMI from a pharmacological perspective. This review highlights both the short- and long-term adverse cardiovascular effects of psychiatric medications. These adverse effects include QTc prolongation and torsades de pointes (TdP), which are phenomena associated with certain selective serotonin reuptake inhibitors (SSRIs), tricyclic antidepressants (TCAs) and antipsychotics. Increasing QT dispersion and induction of Brugada phenotype, both associated with serious cardiac arrhythmias and sudden death, can occur with certain antipsychotics (e.g. trifluoperazine), certain TCAs (e.g. amitriptyline), certain SSRIs (e.g. fluoxetine), methylphenidate and particular mood stabilisers (e.g. lithium). Antipsychotics themselves are associated with increased risk of myocarditis, cardiomyopathy, tachycardia, cardiometabolic derangement, hypertension, orthostatic hypotension and bradycardia. Attention deficit Hyperactivity disorder (ADHD) medications contribute to CVD and tachycardia. Acetylcholinesterase inhibitors (AChEIs) contribute towards bradycardia. Hypertension risk is elevated with serotonin-norepinephrine reuptake inhibitors (SNRIs), norepinephrine-dopamine reuptake inhibitors (NDRIs), monoamine oxidase inhibitors (MAOIs), reversible inhibitors of monoamine oxidase A (RIMA) and psychostimulants. Conversely, orthostatic hypotension is associated with certain psychiatric medications, namely TCAs and serotonin antagonist and reuptake inhibitors (SARIs). It is important to note that pharmacokinetic drug-drug interactions, such as inhibition of the cytochrome P450 (CYP450) system, can affect the pharmacodynamic profile of psychiatric medications, thereby increasing the risk of their associated adverse cardiovascular effects. By understanding the main adverse cardiovascular effects of psychiatric medications and prescribing appropriately, clinicians can reduce potential harm in patients with SMI.
Plain language summary
Patients with severe mental illness are around three times more likely to die from cardiovascular disease than those without mental illness. Some psychiatric medications for severe mental illness are associated with potentially harmful effects on the heart and blood vessels. By better understanding these effects, we can learn to improve drug choice and dosing to help reduce the risk of death caused by heart disease in patients with severe mental illness.
This review explains how certain psychiatric medications for severe mental illness can affect the heart and blood vessels in these patients. Some medications can cause problems with a patient’s heart rate and rhythm, while others may lead to long-term issues like weight gain and a higher risk of diabetes. These changes can increase the chances of having a heart attack or other serious health problems. These issues can be made worse when patients are given certain combinations of medications.
By understanding the problems that can be caused by individual or a combination of psychiatric medications for severe mental illness, healthcare professionals can work towards improved drug therapies to help reduce the unusually high risk of heart disease amongst this vulnerable patient group.
Keywords
Introduction
Severe mental illness (SMI) is defined by the National Institute of Mental Health as ‘. . .a mental, behavioural, or emotional disorder resulting in serious functional impairment, which substantially interferes with or limits one or more major life activities’. 1 This definition can include any psychiatric diagnosis meeting this criterion, but SMI is usually a concept applied to schizophrenia, schizoaffective disorder and bipolar disorder. 2
The Health Improvement Network (THIN) database shows the prevalence of SMI is ~1% in England. THIN further describes health inequalities between the general population and SMI patient demographics, with a far higher prevalence of obesity, hypertension, diabetes, coronary heart disease (CHD) and asthma in patients with SMI. 2 This partly contributes to a dramatically shortened life expectancy in some SMI patients of 15–20 years.2,3 This premature mortality is reflected in a fourfold greater death rate in SMI patients under the age of 75, with around 70% of SMI patient deaths attributable to avoidable physical illness. The predominant cause of death in SMI patients after liver and respiratory disease is cardiovascular disease (CVD), which is ~3.3 times more prevalent than in the general population. 2 The reasons underlying poor cardiovascular (CV) health in SMI patients are multifactorial, with barriers and challenges at the following levels4,5:
Patient level: Low socioeconomic status, poor treatment adherence, lifestyles that worsen CV risks, reduced health literacy and inadequate control of their mental health.
Healthcare professional level: Lack of inter-speciality collaboration, diagnostic overshadowing, unfamiliarity with psychiatric medications and adverse effects of psychiatric medications.
Healthcare system level: Discrepancies in health equity, insufficient infrastructure and lack of multidisciplinary care.
The adverse CV effects of psychiatric medications are one facet contributing to poor CV health in SMI patients.4,6 Psychiatric medications are known to cause various adverse CV effects such as QTc prolongation leading to torsades de pointes (TdP); drug-induced Brugada phenotype (BrP); increasing QT dispersion (QTd); myocarditis; cardiomyopathies; bradycardia and orthostatic hypotension (OH). Other important adverse effects are metabolic derangements, hypertension and tachycardia, which are significant long-term, modifiable CVD risk factors.7–10 Psychiatric medications are occasionally prescribed from a young age and potentially for lengthy periods of treatment, which poses a significantly higher risk for poorer CV health. Therefore, this requires clinicians to be familiar with the adverse CV effects of psychiatric medications.11,12
Although not meeting the SMI definition, depression, attention deficit hyperactivity disorder (ADHD) and dementia medications are included in this review due to their similar adverse CV effect profile and co-morbidity with SMI diagnoses.13–15 Some non-SMI diagnoses (e.g. depression) are often episodic with patients achieving full recovery. 16 This suggests that the long-term risks associated with the treatment for these non-SMI diagnoses may be of lesser severity but are still clinically relevant. Additionally, pharmacokinetic effects can have pharmacodynamic implications, such as CYP450 inhibition, causing increased plasma concentrations of psychiatric medications, which potentially worsens the associated adverse CV effects.17,18 By including treatment for non-SMI diagnoses and considering pharmacokinetic drug-drug interactions leading to pharmacodynamic effects, the review provides a broader perspective on the CV risks and adverse effects linked to psychiatric medications. While the information presented in this review is largely informed by UK-prescribing practices, psychiatric medications licensed and prescribed internationally are included to ensure the content presented here is clinically relevant in a broader global context. This review focuses on the key adverse CV effects of psychiatric medications by collating the latest information relevant to clinical practice.
Methods
A range of databases and search engines were consulted, including PubMED, PubMED Central, Cochrane, MEDLINE, EMBASE, APA PsychInfo, Google Scholar and Google using a variety of key search terms. These search terms captured a broad scope of literature pertaining to the cardiovascular side effects of psychiatric medications. Studies used to evidence cardiovascular side effects of psychiatric medications in this review were chosen based on a hierarchy of evidence, prioritising those with greater statistical power, methodological quality and recency when other factors were equal. This search was conducted from March 2024 to May 2025. To maintain relevance in a global context, this review includes psychiatric medications licensed both within the United Kingdom and internationally. Universally acknowledged and authoritative clinical guidelines addressing the cardiovascular side effects of psychiatric medications were included. Inclusion and exclusion criteria are shown in Table 1.
Exclusion and inclusion criteria.

The exclusion and inclusion criteria used to select the literature from PubMED, PubMED Central, Cochrane, MEDLINE, EMBASE, APA PsychInfo, Google Scholar and Google.
This method was applied to the construction of tables in this review, where authoritative guidelines, meta-analysis and systematic reviews were prioritised over lower hierarchies of evidence. In addition, Medicines and Healthcare products Regulatory Agency (MHRA) was used to help assess the risk of adverse CV effects of certain psychiatric medications when constructing the tables in this review. 19 Comparisons between some studies were limited by differences in study population demographics and varying criteria used for adverse cardiovascular effects (e.g. QTc prolongation). However, the information in tables has been presented as close to the literature consensus as possible to the authors’ knowledge.
QTc prolongation and TdP
Certain psychiatric medications (e.g. antipsychotics) and risk factors (see Table 2) can prolong the QTc interval. This can predispose to serious arrhythmias such as TdP, re-entry arrhythmias and sudden cardiac death.20,21 Data from The Maudsley prescribing guidelines in psychiatry, two systematic reviews, two reviews and a repository of QTc-prolonging medications (www.crediblemeds.org/druglist) are presented in Table 3, which lists QTc-prolonging and TdP predisposing psychiatric medications.22–27 Information from The Maudsley prescribing guidelines in psychiatry was prioritised where possible when creating Table 3. 22 When information from The Maudsley prescribing guidelines in psychiatry was insufficient to comment on QTc-prolonging effect for certain psychiatric medications, data from two systematic reviews and a review were used.23–25 Discrepancies in data were reconciled by prioritising data based on strength and number of study participants. Comparisons between these studies were limited due to different study populations and QTc boundary definitions, but the information in Table 3 represents the general consensus in the literature.22–24 Opioid use disorder is highly prevalent in patients with SMI; therefore, methadone and buprenorphine were included as these medications have significant QTc-prolonging and TdP risks. 28 Note that QTc prolongation does not directly correlate with TdP risk as multiple factors contribute to the precipitation of TdP, see Table 2. 23
Risk factors for QTc prolongation.

Information to make this table was taken from Wenzel-Seifert et al. 24 and Niemeijer et al. 42 Females are twice as likely as men to have TDP and have a higher QTc baseline. Common electrolyte disturbances that predispose to QTc prolongation are hypokalaemia and hypomagnesaemia. High drug concentration (e.g. high therapeutic dose, overdose, rapid infusion or impaired clearance) can predispose to QTc prolongation.
QTc, corrected QT interval; TdP, torsades de pointes.
QTc-prolonging psychiatric medications.
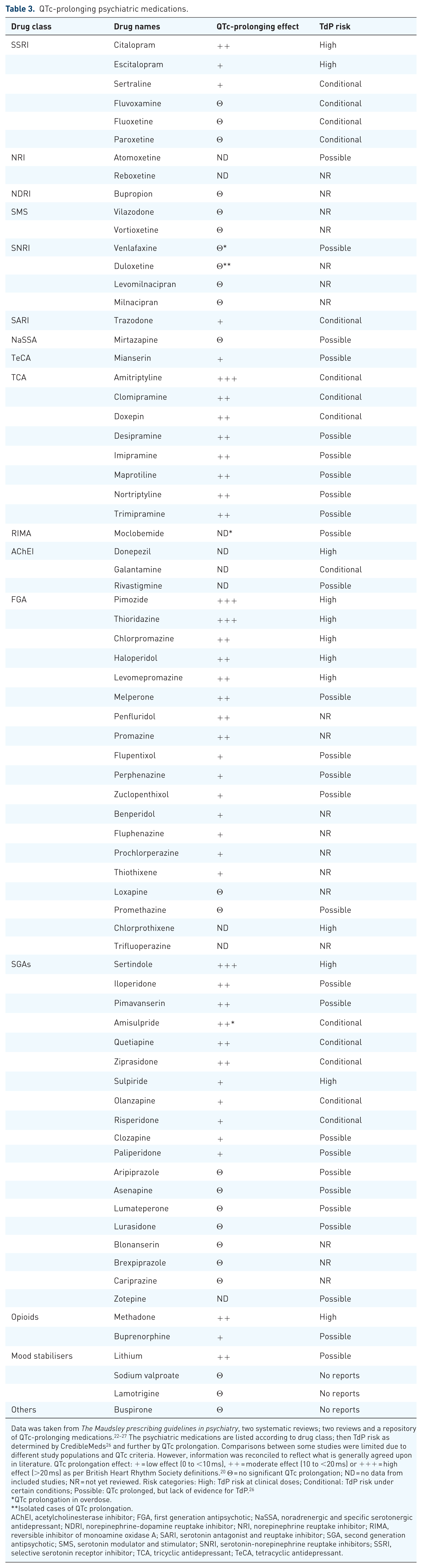
Data was taken from The Maudsley prescribing guidelines in psychiatry, two systematic reviews; two reviews and a repository of QTc-prolonging medications.22–27 The psychiatric medications are listed according to drug class; then TdP risk as determined by CredibleMeds 26 and further by QTc prolongation. Comparisons between some studies were limited due to different study populations and QTc criteria. However, information was reconciled to reflect what is generally agreed upon in literature. QTc prolongation effect: + = low effect (0 to <10 ms), ++ = moderate effect (10 to <20 ms) or +++ = high effect (>20 ms) as per British Heart Rhythm Society definitions. 20 Θ = no significant QTc prolongation; ND = no data from included studies; NR = not yet reviewed. Risk categories: High: TdP risk at clinical doses; Conditional: TdP risk under certain conditions; Possible: QTc prolonged, but lack of evidence for TdP. 26
QTc prolongation in overdose.
Isolated cases of QTc prolongation.
AChEI, acetylcholinesterase inhibitor; FGA, first generation antipsychotic; NaSSA, noradrenergic and specific serotonergic antidepressant; NDRI, norepinephrine-dopamine reuptake inhibitor; NRI, norepinephrine reuptake inhibitor; RIMA, reversible inhibitor of monoamine oxidase A; SARI, serotonin antagonist and reuptake inhibitor; SGA, second generation antipsychotic; SMS, serotonin modulator and stimulator; SNRI, serotonin-norepinephrine reuptake inhibitors; SSRI, selective serotonin receptor inhibitor; TCA, tricyclic antidepressant; TeCA, tetracyclic antidepressant.
Common psychiatric medications causing QTc prolongation and TdP include tricyclic antidepressants (TCAs), citalopram, escitalopram, pimozide, thioridazine, chlorpromazine, haloperidol, levomepromazine, melperone, penfluridol, promazine, sertindole, iloperidone, pimavanserin, amisulpride, quetiapine, ziprasidone, sulpiride and clozapine. Importantly, thioridazine is associated with significant QT prolongation and sudden death.27,29 This led to its withdrawal from global markets.30–32 A generic version of thioridazine is still licensed by Food and Drug Administration in America, 33 but its use is highly restricted. 34 Similarly, sertindole was significantly associated with sudden death, leading to a temporary market suspension. This was lifted after a retrospective, multi-country European study determined that all-cause mortality was lower than previously reported. 35 Low risk has been associated with mianserin and clomipramine. 25 Prolonged QTc and TdP risk is also associated with ADHD and dementia medications.26,36–39 Non-stimulant ADHD medications, atomoxetine (a selective norepinephrine reuptake inhibitor, NRI), may prolong QTc and cause TdP.26,36,37 Sufficient reports have been made to the pharmacovigilance database of the World Health Organization (WHO) to suggest that the psychostimulant ADHD medications, amphetamine, dexamphetamine, lisdexamfetamine and methylphenidate, may also prolong QTc. 38 However, correct reporting to the database cannot be verified, and therefore, QTc risk cannot be accurately assessed. The acetylcholinesterase inhibitor (AChEI) donepezil has known QTc-prolonging and TdP risk. Rivastigmine and galantamine, other AChEIs, prolong QTc and may conditionally cause TdP, respectively.26,39 Table 3 lists the discussed psychiatric medications and their QTc-prolonging effects. The severity of QTc prolongation increases with each additional QTc-prolonging psychiatric or non-psychiatric medication.20,40 Psychiatric medications for which QTc prolongation and TdP risk are not known were excluded from Table 3 (e.g. ADHD medications).
The ‘Tisdale Risk Score’ estimates the risk of QTc-prolonging medications for prolonging QTc beyond 500 ms. 40 However, this score was created using a coronary care unit population and may not be applicable to the general population. 41 If the QTc becomes prolonged, QTc-prolonging medications will need to be reviewed and stopped if >500 ms, along with addressing modifiable risk factors.20,41 These risk factors are listed in Table 2.24,42
QT dispersion
QT dispersion (QTd) is defined as the difference in the longest and shortest QT interval on a 12-lead ECG and is a sign of regional variation in ventricular repolarisation. 43 It is associated with ventricular arrhythmias in healthy individuals as well as patients with ischaemic cardiac tissue, heart failure, diabetes mellitus, long QT syndrome and hypertrophic cardiomyopathy.43–45 A QTd >60 ms is associated with increased risk of cardiovascular death, and a value >80 ms is considered pathological.43,45 An observational cohort study showed that psychiatric medications can increase QTd and that additional psychiatric medications compound this effect. 43 However, this study did not account for the co-morbidities that can increase QTd (e.g. diabetes). 45 A more recent retrospective cohort study using a larger patient group showed that lithium, levomepromazine, quetiapine, asenapine and clozapine increase QTd and QTd ratio in a dose-dependent manner. The QTd ratio is calculated by dividing the QTd by the cycle length in milliseconds and is another ventricular arrhythmia index derived from QTd. The same study also assessed other ECG indices associated with ventricular arrhythmias (e.g. QTd ratio; dispersion transmural repolarisation time abbreviated to Tp-e; and Tp-e/QT ratio) and found these indices to also worsen with psychiatric medications. 46 A recent prospective observational study suggested that methylphenidate may also significantly increase QTd. 47 Altogether, this suggests that psychiatric medications can affect various ECG indices associated with ventricular arrhythmias (e.g. QTc, QTd, QTd ratio, Tp-e, Tp-e/QT). However, these ECG indices are not measured clinically, and further research into these ECG indices is required to determine their true relevance to clinical decision making.
Brugada phenotype and Brugada syndrome
Brugada syndrome (BrS) is caused by loss-of-function mutations in cardiac sodium or calcium channels.48,49 These mutations are associated with arrhythmias that can cause sudden death (e.g. ventricular fibrillation).48,50 In asymptomatic patients, BrS ECG changes can be diagnosed by the pathognomonic coved ST-segment elevation seen in V1 and V2, also called type 1 Brugada pattern (see Figure 1(a)). In addition to mutations, Brugada ECG changes can be acquired through pathological factors (e.g. pyrexia, adrenal insufficiency, hypokalaemia, myocardial ischaemia) and iatrogenic factors (e.g. antiarrhythmics, anaesthetic agents), including psychiatric medications. 48 These psychiatric medications (e.g. TCAs, antipsychotics and mood stabilisers) can block cardiac sodium and calcium channels, resulting in malignant arrhythmias and potentially sudden death.48,50 Removal of the causative psychiatric medications can reverse the blockade of cardiac ion channels and prevent these malignant arrhythmias from arising. This is often seen as reversal of ECG Brugada patterns.48,51 As this is a reversible Brugada pattern, it is referred to as BrP and not BrS, which is reserved for persistence of ECG Brugada patterns in the absence of any precipitating factors. It is important to appreciate the difference between BrP and BrS as both are managed differently. Interestingly, BrS is potentially more prevalent in patients with schizophrenia (~12%) than in the general population (0.05%). 48

Brugada patterns. (a) Type 1 – Coved ST-segment elevation of ⩾2 mm in one or more of V1 and V2 (diagnostic). (b) Type 2 – Saddleback ST elevations ⩾1 mm (not diagnostic, but suspicious). (c) Type 3 – Coved or saddleback, ⩽1 mm (not diagnostic, but suspicious). 40
Patients with BrS or BrP can present with cardiogenic syncope, atrial arrhythmias (especially in those <30 years of age), atrio-ventricular block, nocturnal agonal respirations and polymorphic ventricular tachycardia causing sudden collapse or cardiac arrest.48,52 General risk factors include Asian ethnicity, male sex, middle age (30–50 years old) and a family history of BrS or unexplained cardiac death. 52 A small, retrospective cohort study suggested that drug-induced BrP can present at any time after initiating the causative psychiatric medication. 50 Importantly, BrS can occur in structurally normal hearts and is not associated with prolonged QTc. 48 If BrP or BrS is clinically suspected, ECGs should be performed in high lead positions to increase sensitivity for BrS.49,52 There are three distinct ECG Brugada patterns (see Figure 1(a)–(c)). Type 1 Brugada pattern is diagnostic of BrS or BrP, whilst type 2 (see Figure 1(b)) and type 3 (see Figure 1(c)) are considered suspicious but not diagnostic of BrS or BrP. For a definitive BrS diagnosis, the Shanghai scoring system can be used, which combines ECG findings, clinical history, family history and genetic testing to determine the likelihood of BrS diagnosis. Patients with clinical suspicion of BrS, BrP, or type 2 or 3 Brugada patterns may require a provocation test with sodium channel blockers (e.g. ajmaline, flecainide).48,49 This provocation test is positive if it can precipitate Brugada type 1 pattern.48,53 If the patient has presented with symptoms consistent with BrS or BrP, an echocardiogram is indicated to rule out structural heart disease. 52
Psychiatric medications to be avoided in BrS and those that induce BrP are listed in an online repository (www.brugadadrugs.org), which is frequently updated. 54 Certain TCAs, antipsychotics and mood stabilisers have been associated with inducing BrP and should be avoided in BrP and BrS (see ‘drugs to be avoided’ column of Table 4). It is noteworthy that certain drugs of abuse (e.g. alcohol, cannabis and cocaine) hold similar risks and should also be avoided in patients with BrP and BrS. Other psychiatric medications (e.g. certain selective serotonin receptor inhibitors (SSRIs), norepinephrine-dopamine reuptake inhibitors (NDRIs), TCAs, antipsychotics, mood stabilisers and maprotiline) have a weaker association with inducing BrP. It is therefore recommended to avoid these other psychiatric medications, albeit to a lesser extent in BrP (see ‘preferentially avoided’ column of Table 4). 54 Furthermore, BrP can be seen with overdose of certain psychiatric medications, such as TCAs (e.g. amitriptyline) and phenothiazines.51,55,56
Table of psychiatric medications that potentially induce the Brugada phenotype.
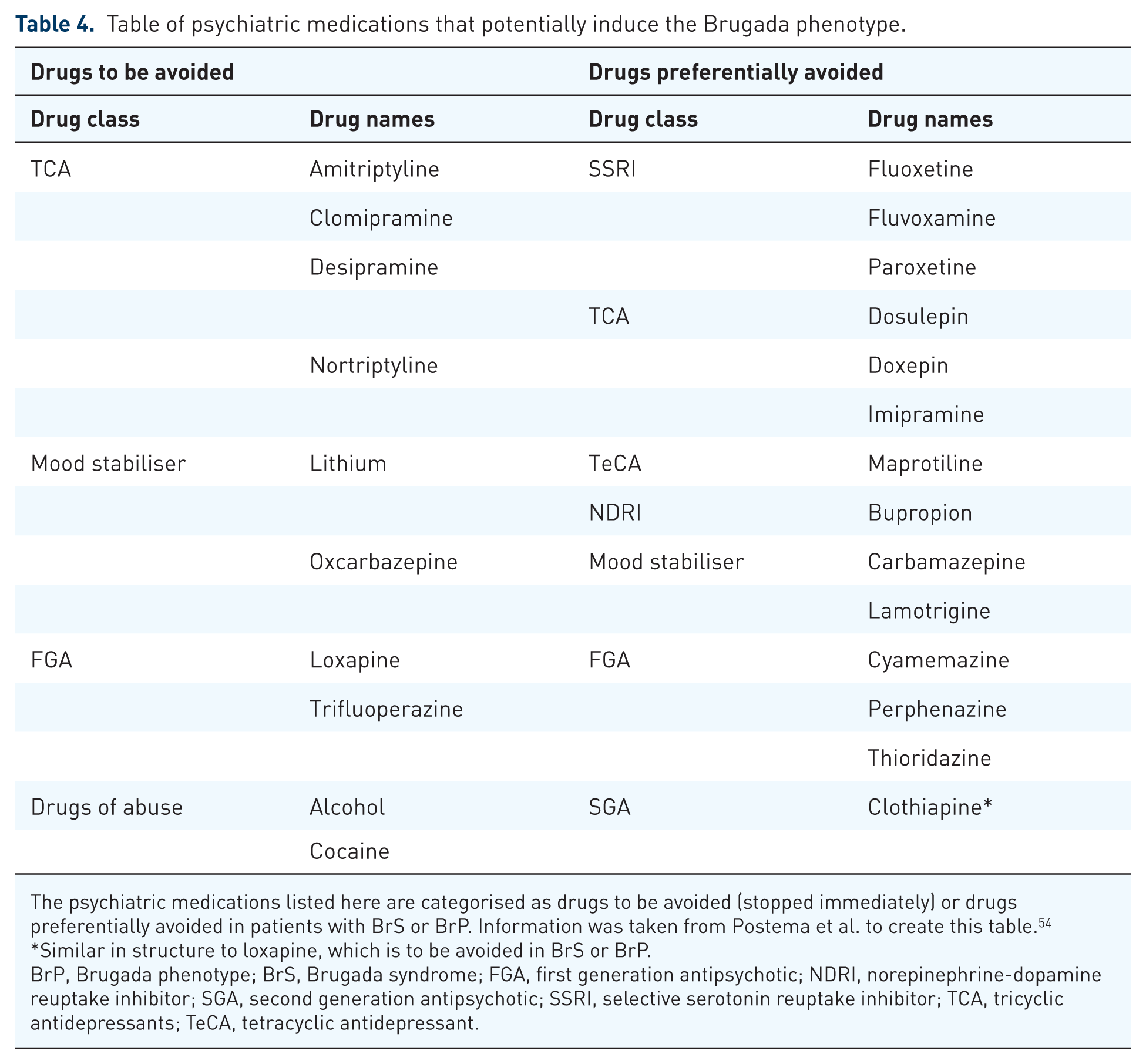
The psychiatric medications listed here are categorised as drugs to be avoided (stopped immediately) or drugs preferentially avoided in patients with BrS or BrP. Information was taken from Postema et al. to create this table. 54
Similar in structure to loxapine, which is to be avoided in BrS or BrP.
BrP, Brugada phenotype; BrS, Brugada syndrome; FGA, first generation antipsychotic; NDRI, norepinephrine-dopamine reuptake inhibitor; SGA, second generation antipsychotic; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressants; TeCA, tetracyclic antidepressant.
If drug-induced BrP is suspected with psychiatric medications, early discussion with cardiologists should take place. Conservative management is recommended initially by addressing any precipitating factors (e.g. pyrexia, metabolic abnormalities, metabolic acidosis, alcohol, cocaine) and cessation of the potential causative psychiatric medication(s).48,49 Psychiatric medication not associated with eliciting Brugada patterns should be initiated instead (see Table 4) and the patient should be advised to avoid alcohol, cannabis and cocaine. Other medications should similarly be reconciled to reduce the chance of the Brugada pattern. If the psychiatric medication cannot be stopped, then dose reduction should be considered. 48 For most cases of drug-induced BrP, conservative management is sufficient. If BrS patterns still persist, quinidine can be prescribed. If quinidine is not tolerated, it can be prescribed at lower doses in the evening to prevent significant adverse arrhythmias overnight. 49 Phosphodiesterase III inhibitors can also be considered, as well as amiodarone in BrS patients with refractory ventricular fibrillation.49,57 If multiple ventricular arrhythmias arise over a short period (termed electrical storm), isoproterenol can be used. 48 If the patient still requires ongoing pharmacological management despite initial conservative management and addressing all possible precipitating causes, BrS is the likely diagnosis. This is usually managed with an implantable cardiac defibrillator and, in some cases, radiofrequency ablation. 49 These management decisions will largely be guided by cardiologists specialising in electrophysiology.
Myocarditis and cardiomyopathy
Myocarditis is defined as inflammation of the myocardium. Myocarditis can cause cardiac dysfunction with impaired cardiac output, which is referred to as cardiomyopathy.10,58,59 Some antipsychotics can cause drug-induced myocarditis, which can progress to a dilated cardiomyopathy. 58 Cardiomyopathy has been reported as an adverse effect with the psychostimulants dexamfetamine, amphetamine and lisdexamfetamine in the WHO pharmacovigilance database, 38 but further studies will be required to determine the absolute risk of cardiomyopathy with these medications. Therefore, antipsychotic drug-induced myocarditis and cardiomyopathy will primarily be the focus here.
Both antipsychotic drug-induced myocarditis and the subsequent dilated cardiomyopathy associated with its pathophysiological progression can cause sudden cardiac death.5,10 Antipsychotic-induced myocarditis is thought to be IgE-mediated inflammation of the myocardium, but the pathological progression from myocarditis to cardiomyopathy has not yet been fully elucidated.6,60 However, the development of antipsychotic-induced cardiomyopathy is likely to be multifactorial and include10,61:
(1) Increased oxidative stress due to increased reactive oxygen species at least in in part from mitochondrial damage or dysfunction.
(2) Pro-inflammatory disposition of the cardiac extracellular matrix due to release of pro-inflammatory cytokines, myeloperoxidase and proteases (e.g. caspase 3).
(3) Increased catecholamine levels.
The drugs most associated with antipsychotic drug-induced myocarditis are clozapine, olanzapine and quetiapine. 61 The condition is less commonly induced by haloperidol, fluphenazine, chlorpromazine, lithium and risperidone.22,60,62 Clozapine is the main antipsychotic drug linked to antipsychotic-induced myocarditis, with varying incidences reported in the literature.63,64 The incidence rate reported by Electronic Medicines Compendium (EMC) in the UK places clozapine-induced myocarditis as rare, between 0.01% and 0.1% of patients prescribed clozapine. 65 Most reported cases of olanzapine- and quetiapine-induced myocarditis are complicated by overdoses and co-prescription with clozapine. 61 General risk factors for antipsychotic-induced myocarditis are established cardiomyopathy, previous pericarditis, heart failure, myocardial infarction (MI) and recent infection. 5 The following are specific risk factors for clozapine-induced myocarditis64,66,67:
(1) Rapid up-titration: Myocarditis risk increases by ~26% for every 250 mg increase in drug dose over the first 9 days.
(2) Sodium valproate co-prescription doubles the risk of clozapine-induced myocarditis.
(3) Age: There is a 30% increase in myocarditis risk with each subsequent decade of clozapine prescription.
(4) Genetics: Single nucleotide polymorphisms in GNA15.
Myocarditis and cardiomyopathy can present with non-specific symptoms (e.g. fever, lethargy, fatigue, diarrhoea, vomiting) or even asymptomatically. The classical cluster of features are raised jugular venous pressure, bibasal crackles, third heart sound, fourth heart sound, peripheral oedema, orthopnoea, paroxysmal nocturnal dyspnoea and tachycardia.63,68–70 Antipsychotic drug-induced myocarditis typically presents within the first month of commencing drug therapy, usually in the third week, but can occur years later.63,71 If myocarditis or cardiomyopathy are clinically suspected, C-reactive protein, brain natriuretic peptide, troponin, echocardiogram, electrocardiogram and chest X-ray are indicated with abrupt cessation of the suspected antipsychotic drug and immediate discussion with a cardiologist.22,63,69 Endomyocardial biopsy remains the gold standard for objectively diagnosing myocarditis but is infrequently performed due to procedure-associated risks (e.g. arrhythmias, emboli). 63 Cardiac magnetic resonance imaging is sufficient in most cases for diagnosis, prognosis and disease monitoring in conjunction with clinical history, clinical examination and aforementioned investigations. 72 Re-initiating the same antipsychotic drug is possible in rare circumstances, but this will require close discussions regarding risks and benefits between cardiology and psychiatry, along with close monitoring.22,68 Prescribing an alternative no- to low-risk antipsychotic is preferable. 22 As part of the Mental Health Strategy 2012–2015, the Scottish Government has provided monitoring advice for patients on clozapine and includes monitoring for clozapine-mediated myocarditis. 73 Although developed for a UK context, this guidance provides principles that are likely to be applicable to clozapine monitoring in other countries.
Adverse cardiometabolic effects of psychiatric medication
Antipsychotic drugs, specifically second-generation antipsychotics (SGAs), can cause metabolic syndrome. Metabolic syndrome is a cluster of at least three risk factors, including abdominal obesity, hypertension, dyslipidaemia and hyperglycaemia, which together increase the risk of CVD and type 2 diabetes mellitus (T2DM). Metabolic syndrome is prevalent in around one-third of patients with schizophrenia on long-term antipsychotic drug therapy.7,74 The incidence of T2DM and hypercholesterolaemia in patients with schizophrenia on long-term antipsychotics is 3–5 times higher compared to the general population. 7 Antipsychotic medications can cause hyperlipidaemia, which can predict poorer CVD outcomes later in life. It is apparent that antipsychotic drugs impart a significant cardiometabolic burden. To compound this further, antipsychotic medications are usually prescribed in young adults and commonly for life. This means the adverse cardiometabolic effects of antipsychotic drugs contribute significantly to CV risk, potentially throughout the life of patients with SMI.11,75,76 A recent, large, retrospective nested case-control study demonstrated an association between psychostimulant drugs and CVD. Specifically, methylphenidate and lisdexamfetamine are linked to CVD risk, especially with prolonged use. 77 A pharmacovigilance study using the WHO pharmacovigilance database has also highlighted a significant association between the psychostimulants dexamfetamine and amphetamine with CVD. 38 However, studies have largely focused on the cardiometabolic burden of antipsychotic drugs, and therefore, the quality of data is much stronger for antipsychotic drugs compared to psychostimulants. On this basis, this review will focus on the cardiometabolic burden of antipsychotics.
A systematic review and network meta-analysis assessed 18 different antipsychotic drugs and their relationship with weight change, body mass index (BMI), total, low-density lipoprotein (LDL) and high-density lipoprotein (HDL) cholesterol, triglyceride changes and fasting glucose levels. The associations are displayed in Table 5. Olanzapine, clozapine and quetiapine are significantly associated with weight gain, high BMI, elevated fasting glucose and dyslipidaemia. 7 Zotepine, sertindole, iloperidone, risperidone, paliperidone, asenapine and brexpiprazole are also significantly associated with weight gain compared to placebo, but have varying effects on BMI, fasting glucose and lipid levels. 7 This is consistent with current literature, which highlights significant associations between weight gain and olanzapine, clozapine, quetiapine, iloperidone, paliperidone, risperidone, asenapine and brexpiprazole. 22 Small increases in these metabolic parameters can significantly impact on CVD risk. For example, a 1 mmol/l increase in triglycerides can increase CVD risk by 76%. 78 Interestingly, cariprazine and lurasidone conferred beneficial effects for fasting glucose and LDL, respectively, compared to placebo. 7
Antipsychotics and their association with specific metabolic parameters.

Antipsychotics (18 in total) are listed along with their association with weight gain, high BMI, high fasting glucose and dyslipidaemia compared to placebo. Antipsychotics are numbered in each column from the most significant association (1) to the least (highest number). + = antipsychotic that has a beneficial effect compared to placebo; Θ = antipsychotic with no significant association for the metabolic parameter compared to placebo; – = association not determined. Information was taken from Pillinger et al. to create this table. 7
BMI, body mass index; LDL, low-density lipoprotein.
Estimating CVD risk from antipsychotic medications is multifactorial. The British Association of Psychopharmacology (BAP) recommends assessing glycaemic control, hypertension, BMI, features of metabolic syndrome, lipid profile, family history of CVD, BMI and smoking habits when estimating CVD risk. 79 Many models estimating CVD risk exist, with the National Institute for Health and Care Excellence (NICE) and BAP recommending QRISK3, which now includes SMI as a risk factor for CVD.79–81 Another model estimating CVD risk, the PRIMROSE risk model, was developed which specifically estimates CVD risk in SMI patients to help optimise their statin therapy in primary care. The PRIMROSE risk model requires a blood test for lipids for estimating CVD risk. 82 If obtaining blood is not possible, the PRIMROSE BMI risk model does not require a blood test and can be used to estimate CVD risk. 83 A recent systematic review assessing the adverse cardiometabolic effects of antidepressant and antipsychotic medications has resulted in the construction of an online digital tool (www.psymatik.com).84,85 This online tool can evaluate both the cardiometabolic and cardiovascular risks (e.g. QTc prolongation, hypotension, etc.) associated with antidepressant and antipsychotic drugs, providing a valuable tool to aid prescribing decisions.
The Maudsley prescribing guidelines in psychiatry and the BAP have advice and guidelines addressing the cardiometabolic side effects (e.g. weight gain, hypertension, dyslipidaemia, hyperglycaemia and T2DM) of psychiatric medications for SMI.22,79 However, little has been covered in the guidelines regarding the use, efficacy and safety of glucagon-like peptide-1 (GLP-1) agonists for antipsychotic-induced weight gain. Exenatide and liraglutide (GLP-1 agonists) demonstrate weight loss in individuals with antipsychotic-induced weight gain with no worsening of psychiatric diagnosis. 86 Trials are ongoing with semaglutide, 86 but none to the authors’ knowledge with tirzepatide which is both a GLP-1 and gastric inhibitory peptide (GIP) agonist. Tirzepatide has significant efficacy regarding weight loss in obese individuals compared with semaglutide. 87 Trials using tirzepatide to address antipsychotic-induced weight gain would be an exciting avenue for future research. In general, the use of GLP-1 and GIP agonists for weight loss in patients on weight-gaining psychiatric medications is a rapidly expanding area of research.
Hypertension
Hypertension is the leading global cause of premature death and is the most significant risk factor for CVD mortality and morbidity. 88 Psychiatric medications are known to induce hypertension initially rapidly through mechanisms not fully understood and progressively slowly over time due to weight gain. 22 Given that most SMI diagnoses are made in early adulthood, it is important to understand the hypertensive risks posed by these psychiatric medications when assessing CV health.2,8
There is no hypertension risk associated with SSRIs, tetracyclic antidepressants (TeCAs), noradrenergic and specific serotonergic antidepressants (NaSSA) and vortioxetine (a serotonin modulator and stimulator, SMS).8,22 TCAs have a low risk, with a dose-dependent effect from imipramine. 89 NDRIs and serotonin-norepinephrine reuptake inhibitors (SNRIs) are associated with moderate risk, especially venlafaxine at doses of 225 mg or higher. 8 This is supported by a dose-dependent increase in blood pressure with venlafaxine. 89 There is a significant hypertension risk with moclobemide, a reversible inhibitor of monoamine oxidase A (RIMA) and monoamine oxidase inhibitors (MAOIs), but not selegiline patch. 8 Clozapine, olanzapine and risperidone cause hypertension in ~27%, ~6% and ~3% of patients, respectively, with olanzapine and risperidone causing hypertension within the first few days of initiation of treatment.9,90 The Summary of Product Characteristics (as listed in Electronic Medicines Compendium) reports a moderate to high hypertension risk (1%–10%) with cariprazine, and a low to moderate hypertension risk (0.1%–1%) with amisulpride and the prolonged-release formulation of aripiprazole. 65 Mood stabilisers (e.g. lithium, sodium valproate) predispose to hypertension mainly through metabolic dysregulation but do not directly cause hypertension. 91 There is no association between lithium, lamotrigine and carbamazepine with hypertension.22,65 There is an association between ADHD medications (methylphenidate, amphetamine and atomoxetine) and hypertension in all age groups.77,92 Psychiatric medications and their associated hypertension risk are shown in Table 6. The tyramine reaction and serotonin syndrome are other notable causes of hypertension, both of which may require urgent clinical management.93,94 Co-prescribing SSRIs, SNRIs or TCAs (particularly clomipramine) with MAOIs is contraindicated due to the elevated risk of serotonin syndrome and hypertensive crisis. Consequently, these medications should be withheld for at least 2–3 weeks before initiating or after discontinuing an MAOI.22,65
Table of psychiatric medications and their associated hypertension risk.

Psychiatric medications are ordered by drug class and then by their hypertension risk from high to low/no risk. Information to create this was taken from Calvi et al., 8 Taylor et al., 22 Datapharm Ltd 65 and Henderson et al. 90 +++ = high HTN risk; ++ = moderate HTN risk; + = low HTN risk; θ = no hypertension risk.
Avoid in HTN.
FGA, first-generation antipsychotic; HTN, hypertension; MAOI, monoamine oxidase inhibitor; NaSSA, noradrenergic and specific serotonergic antidepressant; NDRI, norepinephrine-dopamine reuptake inhibitor; NRI, norepinephrine reuptake inhibitor; RIMA, reversible inhibitor of monoamine oxidase A; SARI, serotonin antagonist and reuptake inhibitor; SGA, second generation antipsychotic; SMS, serotonin modulator and stimulator; SNRI, serotonin-norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitors; TCA, tricyclic antidepressant; TeCA, tetracyclic antidepressant.
Orthostatic hypotension
Orthostatic Hypotension (OH) is a reduction in systolic blood pressure of ⩾20 mmHg or diastolic blood pressure of ⩾10 mmHg within 3 min of standing, with larger reductions in blood pressure required to meet the definition in patients with pre-existing hypertension. 95 OH is a risk factor for syncope, syncope-related injuries, coronary events, stroke and mortality, making it a notable adverse cardiovascular effect of psychiatric medications. 96 The aetiology and pathophysiology of OH is discussed extensively elsewhere. 95
The highest risk of OH is seen with TCAs, SGAs (clozapine, paliperidone, sertindole, quetiapine and risperidone) and low potency first generation antipsychotics (FGAs; e.g. chlorpromazine, levomepromazine and loxapine) in a dose-dependent manner.8,22,65,97 A meta-analysis and systematic review determined that TCAs and SGAs are ~6.3 and ~2.4 times respectively more likely than placebo to induce OH. 98 There is low to moderate risk of OH with SNRIs, SARIs (serotonin antagonist and reuptake inhibitors) and MAOIs, with a notable moderate risk with trazadone.8,96,98 Benzodiazepines have a moderate risk of inducing OH, especially in elderly, frail patients.96,99 A recent meta-analysis, systematic review and narrative review report no significant association of SSRIs with OH.8,98 However, OH has been reported as an uncommon adverse effect (0.1%–1% incidence) for some SSRIs (e.g. fluoxetine, paroxetine and fluvoxamine) by EMC. 65 This discrepancy in OH association with SSRIs and SNRIs may be due to CYP450 inhibition when co-prescribed with certain β-blockers. For example, fluoxetine, paroxetine and duloxetine are known as CYP2D6 inhibitors. Certain β-blockers are eliminated through metabolism by CYP2D6. The serum levels of β-blockers may rise when co-prescribed with fluoxetine, paroxetine or duloxetine due to CYP2D6 inhibition leading to OH as an adverse cardiovascular effect. This, in part, may explain the discrepancies in OH risk associated with SSRIs and SNRIs in literature. 96 Table 7 lists psychiatric medications causing OH and their OH risk.
List of psychiatric medications that induce orthostatic hypotension.
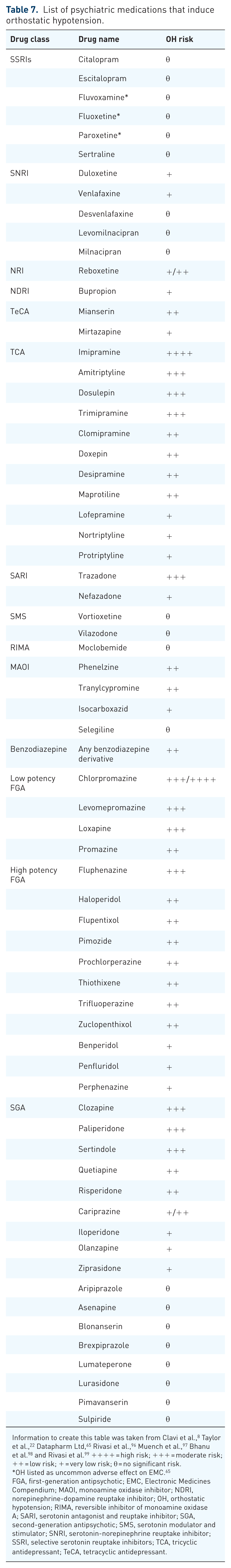
Information to create this table was taken from Clavi et al., 8 Taylor et al., 22 Datapharm Ltd, 65 Rivasi et al., 96 Muench et al., 97 Bhanu et al. 98 and Rivasi et al. 99 ++++ = high risk; +++ = moderate risk; ++ = low risk; + = very low risk; θ = no significant risk.
OH listed as uncommon adverse effect on EMC. 65
FGA, first-generation antipsychotic; EMC, Electronic Medicines Compendium; MAOI, monoamine oxidase inhibitor; NDRI, norepinephrine-dopamine reuptake inhibitor; OH, orthostatic hypotension; RIMA, reversible inhibitor of monoamine oxidase A; SARI, serotonin antagonist and reuptake inhibitor; SGA, second-generation antipsychotic; SMS, serotonin modulator and stimulator; SNRI, serotonin-norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitors; TCA, tricyclic antidepressant; TeCA, tetracyclic antidepressant.
Initial management of OH is non-pharmacological and involves ensuring adequate hydration, avoidance of precipitating events (e.g. getting up slowly), using compression stockings and medical reconciliation. This involves taking note of non-psychiatric medications that can compound OH (e.g. diuretics, α-blockers, nitrates, β-blockers, clonidine, calcium channel blockers, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, levodopa, opioids) and drug interactions.8,95,96 In clinical practice, the causative psychiatric medication (e.g. clozapine) is usually switched to an alternative or up-titrated more slowly. 22 More in-depth advice regarding OH is given elsewhere.95,96,100 There is little evidence to suggest pharmacological approaches are helpful with OH, but a randomised control trial has shown that midodrine significantly alleviates OH symptoms and is licensed for OH in America.95,96,101,102 No pharmaceuticals are licensed for OH in the United Kingdom, but some pharmaceuticals, like fludrocortisone, are prescribed for OH in an off-license manner. 101 Symptomatic relief rather than specific blood pressure targets should be targeted when addressing OH. 96
Tachycardia and tachycardia-induced cardiomyopathy
Tachycardia, defined as heart rate of >100 beats per minute (bpm), is a widely recognised adverse effect of psychiatric medications, especially antipsychotics.10,103 Tachycardia is thought to be mediated through antagonistic effects on muscarinic (M1) receptors leading to reduced parasympathetic drive along with adrenergic (α1) receptor antagonism causing reflex tachycardia. 104 This underlying sympathetic drive directly causes tachycardia but can also induce tachycardia through elevated insulin, insulin resistance, dyslipidaemia and high BMI. 88 Therefore, this underlying sympathetic drive not only causes tachycardia but worsens these important CV risk factors. If tachycardia persists for a prolonged period, it has the potential to lead to tachycardia-induced cardiomyopathy. 105 Tachycardia, along with hypertension, also promotes atherosclerosis, which reduces coronary flow reserve, vascular compliance and can cause atherosclerotic plaque instability leading to myocardial ischaemia and heart failure. 88 Indeed, increases in heart rate have been linked with CV, coronary and all-cause mortality rates, with even a modest increase in heart rate (~10 bpm) being associated with heart failure, sudden death, MI, stroke and all-cause mortality in patients with high-risk hypertension. 106 A higher resting heart rate (81–90 bpm) has an increased risk of all-cause mortality. 107 As SMI patients have a high mortality rate from CV death and antipsychotic medications are known to commonly elevate heart rate, it is important that tachycardia is well managed and discussions should take place with cardiology if the patient is symptomatic (e.g. palpitations, dyspnoea, chest pain) or has abnormal clinical findings. Lastly, tachycardia in patients on antipsychotics could be a sign of myocarditis, cardiomyopathy or neuroleptic malignant syndrome. 104 Tachycardia in these patients will require further clinical work-up for diagnosis.
Clozapine is the most recognised antipsychotic that induces tachycardia (seen in up to 33% of patients), with a significantly higher prevalence compared to other long-acting antipsychotics. 103 Clozapine is associated with an increase of ~20 bpm compared to the general population, but this may be complicated by other confounding factors (e.g. co-prescription of tachycardic-inducing medications or white coat syndrome). 108 Clozapine-mediated tachycardia is dose-dependent and is commonly seen upon initiation of treatment. It may resolve after 4 weeks, but is likely to persist during treatment. 22 High norclozapine levels and elevated clozapine:norclozapine ratios have been linked to elevated heart rate. 108 In patients on clozapine, β-blockers can provide symptomatic relief of tachycardia. 103 However, β-blockers in these patients may not significantly lower heart rates and therefore long-term sequelae of tachycardia, such as tachycardia-induced cardiomyopathy, are still possible. 108 The risk of clozapine-induced tachycardia can be reduced with initial, slow up-titration. 22 Risperidone, haloperidol, quetiapine, lurasidone and olanzapine also induce tachycardia, but not as frequently as clozapine. 104 Paliperidone, chlorpromazine and loxapine are other antipsychotics with significant tachycardia risk compared to placebo. 109 Tachycardia-inducing antipsychotics are listed in Table 8.
Tachycardia risk of psychiatric medications.

Psychiatric medications are grouped according to drug class, then by tachycardia risk and then alphabetically. + = <2% tachycardia incidence; ++ = ⩾2% to ⩽5% tachycardia incidence; +++ = >5% to tachycardia incidence; ++++ = >10% tachycardia incidence.
Specific incidence or risk is not known; therefore, the risk is stated as the general tachycardia risk of drug class. Information to create this table was taken from Taylor et al., 22 Datapharm Ltd 65 and Sweeney et al. 104
Ventricular tachycardia is listed as a rare adverse effect on EMC. 65
FGA, first-generation antipsychotic; NDRI, norepinephrine-dopamine reuptake inhibitor; NRI, norepinephrine reuptake inhibitor; SGA, second generation antipsychotic; TCA, tricyclic antidepressants.
A recent systematic review and meta-analysis has shown statistically significant associations between elevated heart rate and ADHD medications atomoxetine, lisdexamfetamine and methylphenidate when compared to placebo for adults, adolescents and children. Similar associations were found between amphetamine and bupropion with elevated heart rates, but these were only statistically significant in adults. 110 TCAs commonly cause tachycardia through anticholinergic activity and inhibition of noradrenaline uptake. 111 A recent review and meta-analysis found that TCAs increased sinus tachycardia risk threefold compared to placebo. This is largely consistent across all TCAs. 112 The tachycardia risks associated with TCAs and ADHD medications, as reported in the EMC, are largely consistent with those described here and were therefore used in constructing Table 8. 65
It is evident that antipsychotics, ADHD medications and TCAs can cause tachycardia. Persistent tachycardia can cause reversible, dilated cardiomyopathy anywhere from weeks to years after tachycardia onset. Antipsychotics causing tachycardia-induced cardiomyopathy should be considered if there is no other identifiable cause for tachycardia in individuals prescribed antipsychotics. Cessation of the antipsychotic causing tachycardia-induced cardiomyopathy would greatly improve clinical outcomes, with reversibility of the cardiomyopathy usually happening within 3 months. 105 However, this clinical decision would require discussion between cardiologists and psychiatrists.
Bradycardia
Bradycardia, defined as heart rate <60 bpm, has been reported with certain SGAs (risperidone, quetiapine, amisulpride, olanzapine, clozapine), SSRIs, MAOIs, RIMA and AChEIs.10,113 Bradycardia can clinically present as presyncope, syncope, heart failure-like symptoms and confusion (secondary to cerebral hypoperfusion). These could be signs of OH, cardiomyopathy, heart failure or concerning bradyarrhythmia (e.g. high degree heart block). 113 For these reasons, it is important to further assess bradycardia in patients on psychiatric medications.
Systematic reviews have determined little to no association between antipsychotics and bradycardia. Bradycardia is reported in 0.4% of patients on antipsychotics. A recent systematic review and meta-analysis determined that risperidone, quetiapine, amisulpride, lurasidone, haloperidol and clozapine do not significantly increase the chance of bradycardia compared to placebo or to one another. 109 However, the EMC lists bradycardia as an uncommon adverse cardiovascular effect for all SGAs apart from clozapine. Additionally, caution is advised when prescribing FGAs in patients with bradycardia, as both increase the risk of ventricular arrhythmias. 65 Interestingly, there have been a few reports of severe hypothermia with bradycardia upon initiation of risperidone and olanzapine.114–117 Neuroleptic-induced hypothermia has been suggested as the cause for this, a diagnosis that may be 10 times more prevalent than current literature suggests. 117 As this is an adverse CNS effect, it will not be discussed in this review and has been reviewed elsewhere. 118 Altogether, this suggests that bradycardia is potentially associated with SGAs (e.g. quetiapine, amisulpride, olanzapine, clozapine, haloperidol and lurasidone) but is an uncommon adverse cardiovascular effect of antipsychotics. Clinicians will need to remain aware that these antipsychotics can cause profound bradycardia in rare cases.
For decades, mild bradycardia has been associated with SSRIs and has also been associated with MAOIs and RIMAs.22,119 Importantly, to the authors’ knowledge, bradycardia is not associated with SNRI, SARI, TeCA, NRI, NDRI, SMS and NaSSA antidepressants when prescribed at normal doses.22,120 At normal doses, TCAs do not typically cause bradycardia but can in overdose due to blockade of fast sodium channels.22,121 As reported in the EMC, bradycardia is generally a rare adverse cardiovascular effect of SSRIs, with a slightly higher incidence with citalopram. 65 Long-term prescription of benzodiazepines has been associated with very rare or rare instances of bradycardia.22,65
AChEIs, in general, are associated with bradycardia. 122 Their specific use has also been linked to increased hospital admissions, syncope, permanent pacemaker insertion, heart block and fall-related hip fractures.39,123 It has been known for some time that AChEIs cause bradycardia, 115 but specific AChEI associations with bradycardia have only recently been described. A recent large retrospective, observational cohort study found that there was a significant association between donepezil and bradycardia after adjusting hazard ratios for confounding variables (e.g. age, ethnicity, beta blockers, calcium-channel blockers, antipsychotics). The same study showed that rivastigmine and galantamine do not exhibit a significant association with bradycardia. 124 However, bradycardia has been reported as a rare adverse cardiovascular effect for both medications in the EMC, and therefore, caution is advised when prescribing any AChEIs in relation to bradycardia.22,65 Interestingly, a recent systematic review and meta-analysis has shown a statistically significant association between lower heart rate and guanfacine, an α-adrenergic agonist used for ADHD.22,110 The association of bradycardia with guanfacine is reported as common in the EMC. 57 Further research is required to elucidate the bradycardia risk of guanfacine in combination with other heart rate-lowering medications, on the background of bradyarrhythmias and CYP3A4 inhibitors (e.g. eucalyptus oil). 125
In summary, certain psychiatric medications can cause bradycardia, which may be concerning in the background of conduction blocks and IHD. SGAs, SSRIs, MAOIs, RIMAs and benzodiazepines are psychiatric medications that are rarely or uncommonly associated with bradycardia. Bradycardia is more significantly associated with the ADHD medication guanfacine and the AChEI galantamine, according to EMC. 65 A summary of the bradycardic risk for each psychiatric medication is summarised in Table 9.
Bradycardia risk of psychiatric medications.

Information to make this table was taken from Taylor et al., 22 Datapharm Ltd, 65 Yekehatz et al. 120 and Khalid and Waseem. 121 Psychiatric medications are listed by drug class and then by individual psychiatric medication when bradycardic risks are different amongst medications within the same drug class. Θ = No significant association with bradycardia; + = very rare or rare adverse effect (<0.01%–0.1% incidence); ++ = uncommon adverse effect (0.1%–1% incidence); +++ = common adverse effect (>1% incidence); not reported = bradycardia may be significantly associated, but the risk has not been quantified in the source materials used.
Bradycardia risk in overdose.
AChEI, acetylcholinesterase inhibitor; BZD, benzodiazepines; FGA, first generation antipsychotic; MAOI, monoamine oxidase inhibitor; NaSSA, noradrenergic and specific serotonergic antidepressant; NDRI, norepinephrine-dopamine reuptake inhibitor; NRI, norepinephrine reuptake inhibitor; RIMA, reversible inhibitor of monoamine oxidase A; SARI, serotonin antagonist and reuptake inhibitor; SGA, second generation antipsychotic; SMS, serotonin modulator and stimulator; SNRI, serotonin-norepinephrine reuptake inhibitors; SSRI, selective serotonin receptor inhibitors; TCA, tricyclic antidepressant; TeCA, tetracyclic antidepressant; α-2a agonist, alpha-2a adrenergic receptor agonist.
Adverse cardiovascular effects from pharmacokinetic and pharmacodynamic factors
The therapeutic and adverse effects of psychiatric medications are affected by drug pharmacokinetics and pharmacodynamics. Pharmacokinetics refers to the absorption, metabolism, distribution and excretion of medications (i.e. how the body affects the drug). Pharmacodynamics refers to the physiological response to the drug (i.e. how the drug affects the body), which occurs at the drug receptor level.17,18 This section will focus on the pharmacokinetic drug-drug interactions of psychiatric medications metabolised by CYP450, as these are common in clinical practice and have potentially significant adverse cardiovascular effects. Other pharmacokinetic and pharmacodynamic interactions are covered elsewhere in the literature.17,18
Most psychiatric medications are commonly metabolised by the CYP450 (cytochrome P450) system, predominantly by CYP450 isoenzymes CYP2D6 and CYP3A4/5, although other isoenzymes are involved.42,126–128 Certain medications inhibit CYP450 enzymes, thereby increasing the concentration level of psychiatric medications and worsening associated adverse cardiovascular effects such as QTc prolongation. For example, quetiapine increases QTc in a dose-dependent manner and is metabolised by CYP3A4. There have been reports of TdP when a CYP3A4 inhibitor has been co-prescribed with quetiapine. 129 Most likely, the inhibition of CYP3A4 led to increased quetiapine serum levels, which prolonged QTc significantly enough to cause TdP. Another mechanism by which adverse cardiovascular effects of psychiatric medications can manifest is when multiple psychiatric medications (i.e. substrates) with differing affinities for the same CYP450 isoenzyme are co-prescribed. For example, risperidone is a moderate affinity substrate for CYP2D6, whilst fluoxetine is a strong affinity substrate for CYP2D6. If fluoxetine and risperidone are co-prescribed, CYP2D6 will preferentially metabolise fluoxetine over risperidone due to their differing affinities as substrates for CYP2D6. This leads to higher concentrations of risperidone, which has implications for risperidone’s therapeutic and adverse cardiovascular effects. 129 In addition to drug-drug interactions, behaviours such as smoking can impact the CYP450 system and drug metabolism. A well-established example relates to clozapine and olanzapine. Clozapine and olanzapine are predominantly metabolised by CYP1A2 isoenzyme. The polyaromatic hydrocarbons from smoking induce the CYP1A2 isoenzyme. The induction of CYP1A2 would increase the metabolism of olanzapine and clozapine, lowering their respective serum concentrations and therefore therapeutic effect. A meta-analysis clearly demonstrates that patients with schizophrenia who smoke require significantly higher doses of olanzapine and clozapine to achieve the same therapeutic levels as non-smoking patients with schizophrenia. 130 This has implications for the adverse cardiovascular effects associated with olanzapine and clozapine if the smoking habits of patients prescribed olanzapine or clozapine are variable.
It is recommended that healthcare professionals prescribing psychiatric medications aim to minimise CYP450 interactions and, where possible, preferentially prescribe psychiatric medications with multiple elimination pathways and a wide therapeutic window. 18 Furthermore, non-drug patient factors (e.g. age, sex, co-morbidities, caffeine, smoking) and drug factors (e.g. route, dose and formulation) will need to be considered when addressing pharmacokinetic and pharmacodynamic interactions, but this is out with the scope of this review. An extensive list of CYP450 inhibitors, inducers and substrates (i.e. psychiatric medications) metabolised by the cytochrome P450 can be found in the ‘Drugs Interaction Flockhart Table’.17,128,129 Tables describing relevant pharmacokinetic and pharmacodynamic drug-drug interactions which can precipitate the adverse cardiovascular effects of psychiatric medications are provided by Wijesinghe, 17 Crouse and Leung, 18 and Matos et al. 129
Limitations
This narrative review has several limitations. This is not a systematic review, as such, some relevant studies may not have been included. Predefined inclusion and exclusion criteria were applied, with prioritisation towards authoritative guidelines and studies of higher levels and quality of evidence to promote comparability between psychiatric medications. However, the included literature still remained heterogeneous in study design, populations, medication exposures and definitions of some cardiovascular outcomes, which limits comparability across studies. This review is subject to both publication and selection bias.
Conclusion
Psychiatric medications have an essential role in treating patients with a variety of mental health conditions to help control their symptoms and improve their quality of life. However, they have potential adverse CV effects which can have significant short- and long-term health consequences. A variety of adverse CV effects directly due to psychiatric medications or indirectly from drug-drug interactions have been discussed in this review. By understanding the main adverse CV effects of psychiatric medications and prescribing appropriately, it is hoped that clinicians can work towards addressing some of the disparities in CV health between the general population and patients with SMI.
Footnotes
Acknowledgements
This paper was written together by the co-authors with specialist input from Dr. Stephen Leslie (Consultant Cardiologist), Dr. Neil McNamara (Consultant Psychiatrist), Elizabeth Buist (Advanced Clinical Pharmacist in Psychiatry), Dr. Gordon Rushworth (Consultant Pharmacist) and Prof. Ian Megson (Professorial Research Fellow).
