Abstract
Background:
Baricitinib is widely used for immune-mediated diseases, yet real-world safety in underrepresented age groups and the temporal dynamics of adverse drug reactions (ADRs) remain insufficiently characterized.
Objective:
To identify age-stratified ADR signals of baricitinib and to examine potential causal roles of Janus kinase (JAK) 1/2 inhibition in key ADRs.
Design:
A retrospective pharmacovigilance study integrating disproportionality analysis, Mendelian randomization (MR), and time-to-onset (TTO) assessment.
Methods:
Baricitinib-associated ADRs reported to the FDA Adverse Event Reporting System (FAERS; Q3-2018 to Q1-2024) were analyzed using Reporting Odds Ratio and Bayesian Confidence Propagation Neural Network, stratified by age (18–65 vs ≥66 years). TTO was modeled to characterize temporal patterns. Two-sample MR using eQTL-based instruments of JAK1/2 expression evaluated causal links with thrombosis and atrial fibrillation (AF).
Results:
Among 5354 reports, infections were most frequent (28.3%). Thrombotic events (deep vein thrombosis, pulmonary embolism) were more prominent in the elderly (≥66 years), whereas hepatic enzyme elevation and malignancies were more frequent in adults aged 18–65 years. MR suggested that higher JAK2 expression was protective against thrombosis (OR = 0.998, p = 0.028), whereas higher JAK1 expression conferred increased risk of AF (OR = 1.255, p = 0.043). TTO analysis showed that thrombotic ADRs tended to occur early after baricitinib initiation, whereas certain malignancies emerged later.
Conclusion:
This study highlights distinct age-dependent vulnerabilities to baricitinib-associated ADRs, with genetic evidence suggesting target-specific mechanisms underlying cardiovascular risk. These findings underscore the importance of age-tailored monitoring strategies and proactive pharmacovigilance in clinical practice.
Plain language summary
Baricitinib is a medicine used to treat diseases caused by an overactive immune system, such as arthritis. Like many strong medicines, it can cause side effects. We wanted to know whether younger and older patients face different risks when taking this drug, and whether genes might help explain these risks.
To study this, we looked at more than 5000 reports of side effects linked to baricitinib that were collected by the U.S. Food and Drug Administration between 2018 and 2024. We compared patients aged 18–65 with patients aged 66 and older. We also used genetic data to see whether natural differences in certain genes could explain why some patients experience heart- or blood-related problems.
We found that the most common side effects were infections. Older patients were more likely to develop blood clots, such as deep vein thrombosis or pulmonary embolism. Younger patients were more likely to show signs of liver problems and, in some cases, breast cancer. Genetic analyses suggested that activity in two genes, JAK1 and JAK2, may partly explain differences in risks: higher JAK2 activity seemed to protect against blood clots, while higher JAK1 activity appeared to increase the risk of irregular heartbeats.
Most side effects happened soon after starting the medication. Overall, our study shows that younger and older patients face different risks when using baricitinib. Doctors should consider age and genetic factors when monitoring patients to make treatment safer and more personalized.
Keywords
Introduction
Baricitinib, a Janus kinase (JAK) inhibitor, has emerged as a crucial therapeutic option for the treatment of rheumatoid arthritis (RA), COVID-19, and other immune-mediated disorders due to its remarkable efficacy.1,2 By inhibiting the JAK-STAT signaling pathway, baricitinib reduces the release of pro-inflammatory cytokines, thereby controlling inflammation and mitigating the pathological effects of immune hyperactivation. 3 As an immunomodulatory agent, baricitinib has significantly improved the quality of life for many patients and has shown unique therapeutic potential in specific populations. However, with its expanding clinical use, concerns about its safety—particularly the threat of adverse drug reactions (ADRs)—have become more prominent. 4 This issue is especially critical in real-world patient populations, where responses to baricitinib may vary significantly across different age groups, presenting additional challenges for its clinical application.
Despite its efficacy, safety concerns are compounded by patient heterogeneity, particularly age-related differences. 5 Elderly patients, due to factors such as immunosenescence, reduced organ function, and the presence of multiple comorbidities, may be more vulnerable to the ADRs of immunomodulators. These patients are at an elevated risk of severe infections, thrombotic events, and malignancies. 6 In addition, the decline in renal function that often accompanies aging can result in reduced clearance of baricitinib, increasing the hazard of drug accumulation and toxicity. By contrast, while younger patients generally have better metabolic capacity and immune function, long-term use of JAK inhibitors in this population may raise concerns about immunosuppression, with potential safety concerns such as opportunistic infections or cancer. Variations in drug metabolism, immune status, and comorbidity profiles across age groups underscore the heterogeneity in patient responses to baricitinib. 7
Although the safety profile of baricitinib has been preliminarily established through numerous randomized controlled trials, these trials typically involve more homogeneous patient populations and often exclude the elderly and other age extremes. 8 As a result, real-world patient populations, which exhibit a broader age range and more complex comorbidities, may experience different safety and efficacy outcomes. Systematic research addressing the distribution and threat profiles of ADRs across various age groups has remained limited, creating a knowledge gap that hinders the rational use of baricitinib in diverse patient populations.
The distribution and clinical significance of baricitinib-associated ADRs likely vary across age groups. Some studies indicate that elderly patients face significantly higher hazards of infections and thrombotic events, particularly when baricitinib is co-administered with other immunosuppressants or when underlying chronic conditions are present. 9 However, data on ADR types and long-term safety concerns in younger patients, especially those using baricitinib over extended periods, remain scarce. Furthermore, many existing studies tend to focus on overall ADR incidence rather than conducting age-stratified analyses, potentially overlooking high-threat subpopulations and limiting the ability to optimize treatment protocols and clinical decision-making.
Real-world data offer a valuable opportunity to fill these gaps. 10 The FDA Adverse Event Reporting System (FAERS) provides a large-scale, global database of drug-related ADRs, which can be leveraged to conduct age-stratified analyses. Such analyses can uncover patterns in ADRs distribution, identify high-threat subgroups, and provide evidence for optimizing therapeutic strategies. 11 Compared to traditional clinical trials, FAERS data offer the advantages of extensive coverage, large sample sizes, and diverse patient representation, making it particularly well-suited to addressing age-related drug safety concerns. Although existing studies have explored the overall safety of baricitinib, research on the distribution of ADRs and threat signals across different age groups is still insufficient.
To further validate these findings and eliminate confounding factors, this study applied Mendelian randomization (MR) analysis. 12 MR uses genetic variations closely related to exposure as instrumental variables (IVs) to infer causal relationships between exposure and outcomes. Expression quantitative trait loci (eQTLs) regulating drug target gene expression are widely used in MR analysis to assess the causal relationship between drug exposure and disease risk. This method has been successfully applied in several studies, such as evaluating the role of the sodium-glucose cotransporter 2 (SGLT2) target in reducing gout risk and the association between GLP-1 receptor agonists (GLP1RAs) and the risk of chronic obstructive pulmonary disease (COPD) based on drug target MR evidence.13,14 Based on these findings, this study will use MR analysis to verify the causal relationship between JAK1/JAK2 targets and drug-related adverse reactions, providing genetic evidence for the safety of JAK inhibitors.
This study aimed to analyze baricitinib-related ADR signals across age groups using the FAERS database and apply MR to verify the causal link between JAK1/JAK2 targets and specific ADRs. It sought to identify age-related differences in ADR patterns and provide evidence to optimize treatment strategies and personalized prescribing, ultimately enhancing baricitinib’s safety profile and improving patient management.
Methods
Data source
This study was a retrospective pharmacovigilance analysis based on publicly available data from the FAERS database evaluating the safety of baricitinib (Olumiant) FAERS, which has been operational since 2004, collects ADRs globally from healthcare professionals, patients, and drug manufacturers, covering various drugs and their associated safety issues. 11 The data are updated quarterly and publicly available. The data used in this study were downloaded and preprocessed via the OpenVigil 2.1 platform, 15 a pharmacovigilance tool developed by Kiel University in Germany, which effectively removes duplicate and incomplete data to enhance data accuracy and reliability. The Summary of Product Characteristics (SPCs) for marketed drugs provide essential information about the drug and are publicly available on the drug regulatory authority’s website. SPCs assist in guiding drug use and supplement ADRs. The FAERS database monitors drug safety in real time and, by comparing with known information from SPCs, helps identify new drug hazards, supporting safety evaluations (Figure 1). This is a retrospective secondary data analysis using publicly available FAERS reports.

Study flow.
The study period, from Q3 2018 to Q1 2024, was chosen to capture the full post-marketing experience of baricitinib, with the latest data available up to Q1 2024. The FAERS database consists of several key data tables, including patient demographic information (DEMO), drug usage (DRUG), adverse event reports (REAC), patient outcomes (OUTC), drug treatment duration (THER), indications (INDI), and report sources (RPSR). 16 FAERS query terms and constraints. Reports were retrieved from structured FAERS fields by restricting the DRUG table to entries where role_cod = “PS” and the product name matched “baricitinib” or “OLUMIANT”; For this study, ADRs reports were encoded using MedDRA (Medical Dictionary for Regulatory Activities) version 26.1, 17 which provides standardized terminology for ADR coding, with the focus on Preferred Terms (PTs) and the System Organ Classes (SOCs).
During data processing, a standardized cleaning procedure was applied. First, duplicate, invalid, and structurally incomplete reports were excluded to ensure dataset consistency. For reports with the same Case ID (CASEID), the most recent FDA report date (FDA_DT) was retained; if dates matched, the report with the highest Primary ID (PRIMARYID) was selected. Only those reports in which baricitinib was identified as the “Primary Suspect Drug” (PS) were included in the analysis. 18 Notably, “structurally incomplete reports” referred to reports lacking essential FAERS fields for valid case identification, and were distinct from “missing covariates” within otherwise complete reports, which were addressed analytically rather than excluded at the extraction stage. Confounding signals related to the drug indication or disease course were also excluded from this study. Through these data processing and filtering methods, this study ensured a comprehensive analysis of ADRs associated with baricitinib and provided a solid data foundation for further pharmacovigilance assessments.
Inclusion criteria and exclusion criteria
Inclusion criteria were as follows: (1) FAERS reports in which baricitinib was recorded as the PS; (2) unique reports after deduplication using CASEID, FDA_DT, and PRIMARYID; and (3) valid adverse events coded under MedDRA at the PT/SOC level.
Exclusion criteria were as follows: (1) reports in patients aged ≤17 years due to insufficient case volume for meaningful stratification; (2) duplicate or structurally incomplete records; and (3) entries where the reported event reflected indication/disease course or efficacy rather than safety, which were removed to minimize confounding.
Signal mining methods
Signal detection framework using multiple algorithms
We employed disproportionality analysis (DPA) to assess the potential association between baricitinib and ADRs using two signal detection algorithms: Reporting odds ratio (ROR) and Bayesian confidence propagation neural network (BCPNN). These methods, with distinct statistical properties and applicability, enhanced signal detection stability and accuracy through cross-validation.
- ROR: ROR is a traditional signal detection method that estimates relative threat and is computationally simple. Due to its high sensitivity, this study combined it with BCPNN to reduce the occurrence of false positives and false negatives, 16 and the results were presented with a 95% confidence interval (CI).
- BCPNN: Based on Bayesian inference models, BCPNN can identify more complex patterns in the data, offering a significant advantage in detecting signals for rare ADRs. 17 By using a multi-algorithm approach, these methods effectively reduced noise and improved the reliability of the signals. Signals were confirmed when the signal scores from both algorithms exceeded the pre-set threshold, indicating a potential true association between baricitinib and the adverse event. All mathematical formulas and threshold standards for signal detection are detailed in Table S1. Proportional Reporting Ratio and Empirical Bayesian Geometric Mean were not included because they are methodologically redundant with ROR and BCPNN and would not provide additional signal information.
MR analysis
Exposure variables and IVs selection
For the analysis of JAK1 and JAK2 as independent primary exposure genes, we identified common single-nucleotide polymorphisms (SNPs) significantly associated with peripheral blood expression of JAK1 or JAK2. To do this, we used cis-eQTL summary data from the eQTLGen Consortium, which integrates results from multiple cohorts predominantly of European ancestry. This dataset includes 16,987 cis-eQTLs across 31,684 individuals in blood tissues (accessed on August 14, 2025). 19
For each gene, we downloaded the corresponding summary statistics from eQTLGen, selecting SNPs that were significantly associated with gene expression (p < 5 × 10−8). SNPs were required to be independent, with linkage disequilibrium defined as r² < 0.001 and a distance of 10,000 kb between SNPs, ensuring independence of the variants. To guarantee strong instruments for causal inference, only SNPs with an F-statistic >10 were retained. This careful selection process ensured that the chosen SNPs were robust and reliable for MR analysis. 13 For valid causal inferences in MR analysis, three key assumptions must be satisfied: Relevance ensures that the genetic variants used as IVs are strongly associated with the exposure (JAK1/JAK2 inhibition). Independence requires that these variants are not linked to any confounders that could distort the exposure–outcome relationship. Lastly, Exclusion Restriction mandates that the genetic variants influence the outcome (ADRs) solely through the exposure (JAK1/JAK2 inhibition), without any direct effect on the outcome itself. 14 In this study, we only used publicly available summary-level genetic data involving European individuals, which were derived from original studies that underwent ethical review and obtained participant consent. Therefore, no additional ethical approval was required.
Outcome data source
In selecting the outcomes for ADRs from the FAERS database, we did not include all ADRs. Instead, we prioritized the most prominent PTs and further incorporated three ADRs that were not reported in the SPCs. This selective strategy was guided by the clinical relevance of the outcomes to the exposure under investigation and the availability of high-quality genetic summary data.
Consequently, we included deep vein thrombosis (DVT) and pulmonary embolism (PE), atrial fibrillation (AF), acute pancreatitis (AP), and interstitial lung disease (ILD) as the outcomes for MR analysis. The datasets were obtained as follows (all datasets were accessed on August 14, 2025). Detailed dataset characteristics are provided in Table S10.
Analytical methods
In this study, the Inverse Variance Weighted (IVW) method was employed as the primary analysis to estimate the causal effect of JAK1/JAK2 inhibition on ADR risk. 14 In situations where only a single genetic IV is available (i.e., one SNP), conventional multi-instrument approaches, such as the IVW method, are not applicable. Under such circumstances, the causal effect should be estimated using the Wald ratio method. To assess the robustness of the results, several sensitivity analyses were performed. MR-Egger regression was used to detect potential directional pleiotropy, which could indicate that the genetic variants affect the outcome through pathways other than the exposure. The Weighted Median method was employed to provide a more robust causal estimate in the presence of invalid instruments, assuming that at least 50% of the instruments are valid. A heterogeneity test using Cochran’s Q test was conducted to evaluate the variability among the instruments, with p < 0.05 indicating significant heterogeneity and potential bias in the causal estimates. The pleiotropy test was conducted by assessing the intercept from the MR-Egger regression, with p < 0.05 indicating the presence of horizontal pleiotropy. In addition, a leave-one-out sensitivity analysis was carried out by excluding each SNP one by one to assess the robustness of the results and identify any outliers that could influence the findings. 13 Because this study was hypothesis generating, no multiplicity correction was applied. Instead, concordance across ROR and BCPNN and MR sensitivity analyses was used to increase credibility.
Steiger directionality test
To ensure the direction of causality is correct, the Steiger directionality test was incorporated. This test verifies that the IVs influence the exposure (JAK1/JAK2 inhibition) and not the outcome (ADRs). 20 This helps confirm that the causal relationship flows from exposure to outcome, adhering to the correct directionality. Reporting of this study follows the STROBE-MR guidelines for MR studies (see Table S2 for the checklist). 21
Age-stratified subgroup analysis
Patients were divided into three age groups to identify age-related differences in disease progression, treatment responses, and outcomes, as well as to reveal significant differences in the incidence of ADRs across these groups. This approach allowed for the development of more tailored and effective treatment strategies for baricitinib:
(1) ≤17 years group.
(2) 18–65 years group.
(3) ≥66 years group.
We excluded individuals under 17 years of age, as their numbers were too small to provide meaningful analysis. Therefore, we focused on the 18–65 and ≥66 age groups for the final analysis. We screened the positive PTs and SOCs that met the criteria and analyzed the intersection of the 18–65 and ≥66 age groups, resulting in 55 shared PTs. At the same time, we analyzed the unique PTs for 18–65 and ≥66 age groups to identify age-specific ADRs. Chi-square tests were conducted to evaluate the statistical differences in the number of SOC reports across different disease categories between the 18–65 age group and the ≥66 age group, with a p-value of <0.05 considered statistically significant.
Analysis of time-to-onset and cumulative statistical analysis
This study included all reports containing time-to-onset (TTO) data, with the analysis focusing on positive SOC categories across all age groups. The TTO was defined as the interval between the initiation of baricitinib therapy and the onset of an ADR. We analyzed TTO data from the FAERS database using Weibull shape parameters (WSP) to evaluate the temporal distribution of ADRs associated with baricitinib. 16 Invalid dates were excluded, and missing dates were imputed with the midpoint of the month. TTO was summarized using medians and interquartile ranges (IQRs). The WSP model, characterized by the shape parameter β, confirmed the distribution type: early failure type (β < 1), random failure type (95% CI of β including 1), and wear-out type (β > 1). Kaplan–Meier analysis was applied to compare cumulative ADR incidence across baricitinib. Data analysis and visualization were conducted using R software (R Foundation for Statistical Computing, Vienna, Austria), Python (Python Software Foundation, Wilmington, DE, USA) and the BioLadder platform (BioLadder, China).
Results
ADRs report analysis of baricitinib: Demographics, outcomes, and report sources
A total of 5354 baricitinib-related ADR reports were included in the final analytical dataset, covering the period from Q3 2018 to Q1 2024 (Table 1). During the cleaning process, duplicate, invalid, and incomplete reports were excluded to ensure the accuracy and reliability of the analyzed data. Regarding gender distribution, the proportion of ADRs reported by females was higher than that reported by males, at 63.41% and 26.91%, respectively. In terms of age distribution, the 18–65 age group accounted for 44.11%, making it the largest group, while the ≤17 and ≥66 age groups represented smaller proportions, suggesting that baricitinib was more widely used among adults. The majority of reports originated from the United States, accounting for 56.14%, followed by Japan, the United Kingdom, and other countries, reflecting differences in drug usage across nations. The outcomes of the ADRs were diverse, with hospitalization being the most common clinical outcome, accounting for 28.48% of reports. Deaths and life-threatening events each accounted for 8.87%, disabilities accounted for 1.53%, and other significant medical events were stated in 30.58% of reports. In terms of report sources, consumer-submitted reports were the most prevalent, comprising 50.00% of the total, followed by reports from physicians (14.48%) and pharmacists (16.08%). The group aged ≤17 years was excluded from the analysis due to an insufficient sample size (58 reports), which could compromise the statistical power and reliability of the findings. Our findings highlighted a peak in both total numbers and percentages in 2021, followed by fluctuations through 2024 (Figure 2).
Case characteristics on ADRs related to baricitinib.

Since a case may experience different clinical outcomes during drug therapy, it is reasonable that the sum percentage of the outcome under this item may exceed 100%.
ADR, adverse drug reaction.
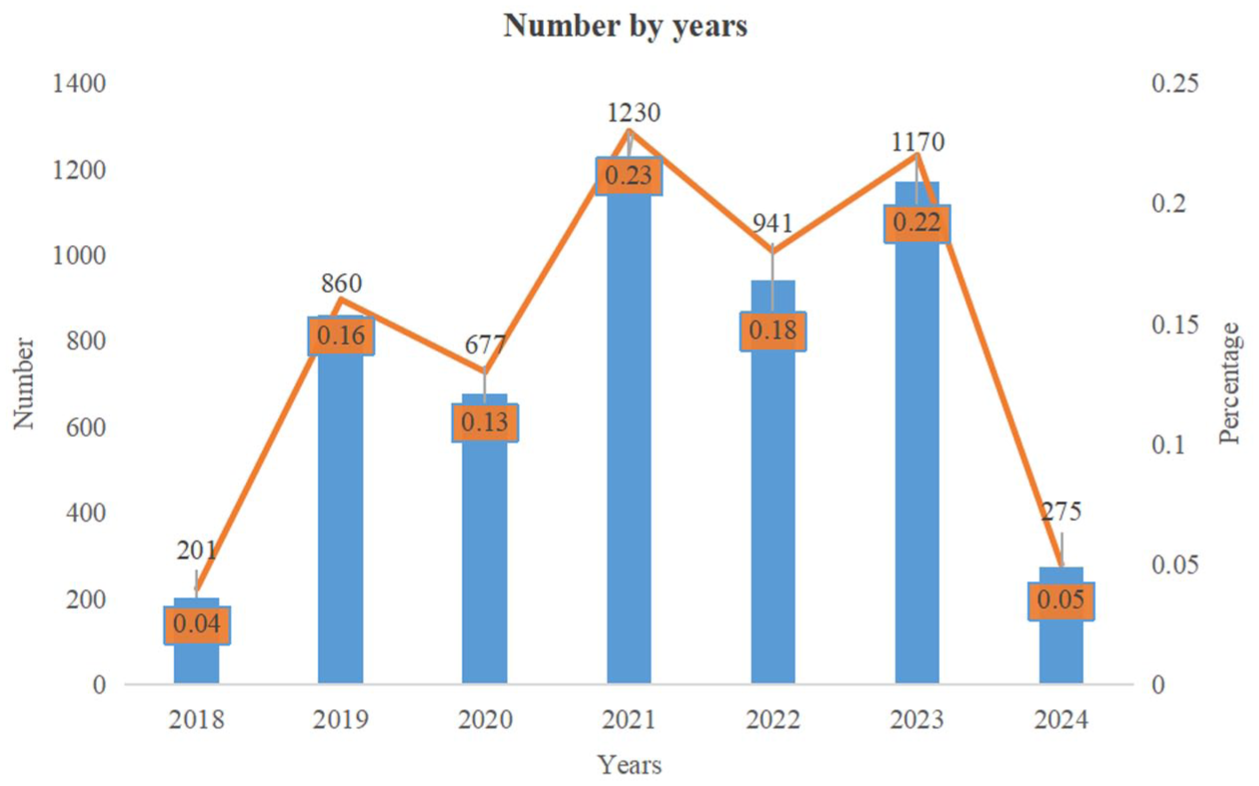
Annual trends in baricitinib FAERS data (Q3 2018 to Q1 2024): Trends in numbers and percentages by year (2018–2024). The blue bars represent the total numbers (left y-axis), while the orange line indicates the corresponding percentages (right y-axis) for each year.
Signal detection and ADRs of baricitinib across organ systems
In the signal detection analysis of baricitinib across all age groups, a total of 261significant signals were identified that met the criteria of two algorithms at the PT level. Among the signals, 127 were found in the age group of 18–65 years, while 103 signals were identified in individuals aged ≥66 years. The five most frequently stated PT signals across all age groups were PE (246 reports, ROR = 25.78, 95% CI: 2.48–4.28), DVT (170 reports, ROR = 3.68, 95% CI: 4.19–4.62), therapy interrupted (148 reports, ROR = 7.98, 95% CI: 2.67–3.14), herpes zoster (116 reports, ROR = 10.72, 95% CI: 3.04–3.57), and pneumonia (110 reports, ROR = 1.93, 95% CI: 0.62–1.16). ADRs such as cerebrovascular accident (ROR = 2.61, 95% CI: 0.92–1.67), ILD (ROR = 5.53, 95% CI: 1.92–2.73), AP (ROR = 2.89, 95% CI: 2.67–3.14), and duodenal perforation (ROR = 7.98, 95% CI: 0.33–2.13), which were not mentioned in the SPCs, suggested a potential clinical hazard associated with the use of the drug (see detail in Table S3).
In the signal analysis at the SOC level of all age groups, baricitinib was significantly associated with ADRs in multiple organ systems (Figure 3 and Table S4). The top five SOC categories by the number of reports were as follows: infections and infestations (IAIs, ROR = 3.60, 95% CI: 1.58–1.70); investigations (ROR = 1.66, 95% CI: 0.57–0.75); respiratory, thoracic, and mediastinal disorders (RTMDs, ROR = 1.89, 95% CI: 0.75–0.94); vascular disorders (VDs, ROR = 2.56, 95% CI: 1.17–1.42); and neoplasms benign, malignant, and unspecified with incl cysts and polyps (ROR = 1.25, 95% CI: 0.16–0.41). In the category of IAIs, the 18–65 age group (862 reports) had significantly more reports than the ≥66 age group (695 reports). In the VDs category, the 18–65 age group had 237 reports, while the elderly group had 180 reports. However, in the category of RTMDs, the 18–65 age group had 404 reports, while the ≥66 age group had 180 reports, representing a reduction of 55.4%. See Figure 4 for details. The chi-square test results indicated a significant statistical difference in the SOC report counts between the 18–65 age group and the ≥66 age group across different disease categories (p = 7.42e-05). Overall, the emergence of these SOC signals suggested the need for close monitoring of the associated ADRs during baricitinib treatment, and further evaluation of its safety and threat–benefit ratio in clinical practice.

Proportional distribution of SOCs for baricitinib in the FAERS database: Pie chart showing the proportional distribution of SOCs for baricitinib in the FAERS database.

Age distribution of SOCs for Baricitinib in the FAERS database: Stacked bar chart showing the distribution of SOCs for Baricitinib in the FAERS database, stratified by age groups (18–65 and ≥66 years).
MR analysis of JAK1 and JAK2 on specific ADRs
In our MR analyses, higher genetically predicted expression of JAK1 was significantly associated with an increased risk of AF (Wald ratio: OR = 1.255, 95% CI: 1.008–1.563, p = 0.043). No significant associations were observed between JAK1 expression and the risk of deep vein thrombosis and pulmonary embolism (DVT&PE), ILD, or AP (all p > 0.05). For JAK2, genetically predicted higher expression was associated with a protective effect on both DVT&PE and AF. Using the IVW method, higher JAK2 expression was associated with a reduced risk of DVT&PE (OR = 0.998, 95% CI: 0.997–0.999, pIVW = 0.028) and AF (OR = 0.937, 95% CI: 0.888–0.989, pIVW = 0.019). Results were consistent using the weighted median approach (Table S11). Sensitivity analyses, including MR-Egger regression and leave-one-out analysis, revealed no evidence of horizontal pleiotropy or heterogeneity (p > 0.05); see Tables S12 and S13 and the Supplemental Figure for further details. No causal associations were found between JAK2 expression and ILD or AP (pIVW > 0.05). Evidence for JAK1 comes from a single SNP, while JAK2 relies on multiple SNPs, providing more robust results. Clinically, JAK2 inhibition may increase thrombosis risk, consistent with genetic data. The Steiger directionality test confirmed that the causal effect runs from JAK1/JAK2 inhibition to ADRs.
Subgroup analysis in baricitinib treatment
To further explore the ADR profiles of baricitinib, we conducted analyses on both the intersecting signals and the unique PTs for the age groups 18–65 and over 66, providing a more detailed assessment of the drug’s safety across these distinct age categories.
In analyzing the distribution of treatment-related PTs across different age groups (≥66 and 18–65 years), it was found that DVT (53 reports, ROR = 17.43, 95% CI: 3.43–4.21) and PE (77 reports, ROR = 15.08, 95% CI: 3.37–4.02) were significantly more common in the ≥66 years group, whereas pneumonia staphylococcal was more prevalent in the younger group (18–65 years), with ROR = 78.97, 95% CI: 2.80–4.70. Building on the previous analysis, 55 common PT signals were identified across the 2 age groups (18–65 and ≥66 years), indicating shared ADRs among all demographics, see heatmap in Table S5 and Figure 5 for details. Common high-frequency PTs in the elderly population included pneumonia (53 reports), lymphocyte count decreased (16 reports), diverticular perforation (8 reports), and ILD (27 reports). In the 18–65 age group, the frequency of liver function test increased (25 reports), therapy was interrupted (109 reports), alanine aminotransferase increased (44 reports), and breast cancer (18 reports) were higher. The relative frequency of lung adenocarcinoma (7 reports) was also higher. Overall, the results reflect differences in disease types, diagnostic patterns, and health status across the age groups.

Heatmap showing the differential distribution of PTs associated with baricitinib, based on the intersection of 55 PTs across age groups (18–65 and ≥66 years). The color intensity represents the relative frequency of each PT, with red indicating higher frequencies and blue indicating lower frequencies.
To be precise, in the 18–65 age group, influenza (30 reports, ROR = 2.96, 95% CI: 0.91–1.95), cerebrovascular accident (29 reports, ROR = 4.94, 95% CI: 1.58–2.64), and acute myocardial infarction (AMI, 20 reports, ROR = 12.62, 95% CI: 2.54–3.81) were found exclusively, primarily representing acute respiratory and cardiovascular conditions (Tables S6 and S8). By contrast, the ≥66 age group experienced a unique prevalence of gastroenteritis (14 reports, ROR = 18.12, 95% CI: 10.69–30.72), renal impairment (19 reports, ROR = 2.33, 95% CI: 0.40–1.70), lung neoplasm malignant (13 reports, ROR = 3.43, 95% CI: 1.99–5.91), COPD (12 reports, ROR = 2.83, 95% CI: 1.61–5.00), and other diseases, indicating a greater burden of severe respiratory and thrombotic disorders in the elderly population (Tables S7 and S9). Overall, the 18–65 age group was predominantly affected by acute diseases, while the ≥66 age group faced a higher incidence of chronic and severe conditions, the top 20 unique PTs refer to Figure 6(a) and (b).
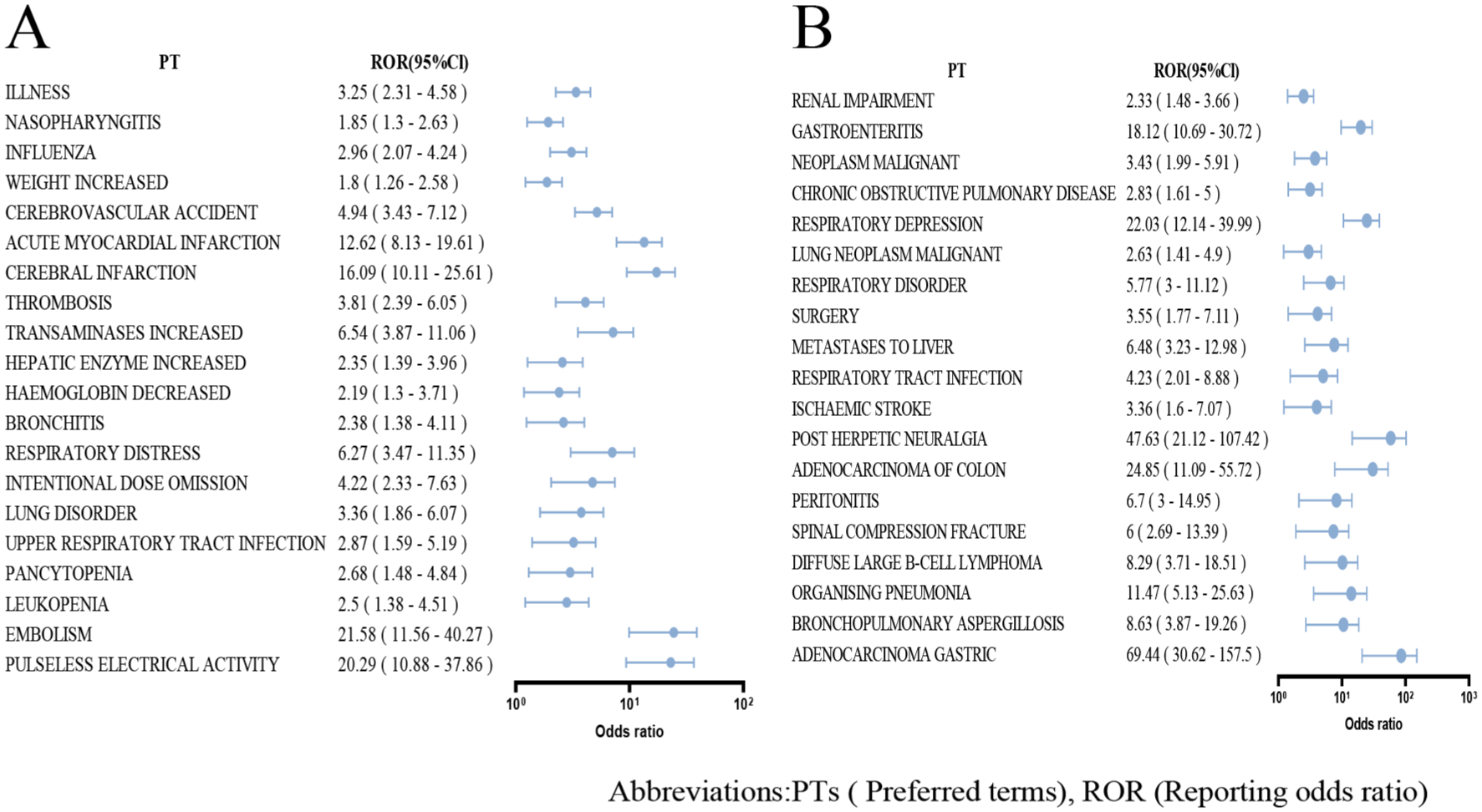
Forest plot comparing PTs for baricitinib between age groups (≥66 vs 18–65): The forest plot compares the PTs reported in the ≥66 age group (a) versus the 18–65 age group (b) for baricitinib, based on FAERS data. The plot includes point estimates and 95% CIs for each PT, with a vertical dashed line indicating the reference point. The red diamonds represent the estimated proportions for the ≥66 group, while the horizontal blue lines denote their CIs.
Cumulative risk of ADRs by SOC categories in baricitinib treatment
In this study, the TTO of SOC in the FAERS database was analyzed using the WSP model to evaluate the occurrence patterns of baricitinib-related ADRs, see details in Figure 7. The results showed that β = 0.49, indicating that the hazard of baricitinib-related ADRs decreases over time (early failure type), as detailed in Table 2.

Time-to-onset (TTO) of adverse drug reactions (ADRs) for baricitinib: X-axis represents the time (in days) from the start of baricitinib treatment to the onset of ADR. Y-axis indicates the cumulative proportion of patients experiencing ADRs over time. Values range from 0 to 1, representing the cumulative probability of ADRs occurring at a specific time point.
WSP of top 15 PTs in baricitinib treatment.

α: Scale parameter, represents the scale of the distribution function as the quantile at which 63.2% of ADEs occur. β: Shape parameter, could be used to confirm the distribution type: early failure type (β < 1), random failure type (95% CI of β includes 1), and wear-out type (β > 1).
95% CI, 95% confidence interval; IQR, interquartile range; PT, preferred term; WSP, Weibull shape parameter.
The WSP analysis for the top 15 PTs revealed significant variability in the occurrence patterns of ADRs; see Table 3 for details. For DVT (β = 0.59, 95% CI: 0.51–0.67), PE (β = 0.51, 95% CI: 0.46–0.57), ILD (β = 0.63, 95% CI: 0.42–0.83), urinary tract infection (β = 0.45, 95% CI: 0.31–0.59), cerebrovascular accident (β = 0.43, 95% CI: 0.31–0.55), pneumonia (β = 0.49, 95% CI: 0.39–0.59), anemia (β = 0.65, 95% CI: 0.45–0.85), and renal impairment (β = 0.44, 95% CI: 0.31–0.56), the results indicated early failure patterns, suggesting that the threat of these ADRs was higher shortly after drug exposure.
TTO analysis for SOC in baricitinib treatment.

α: Scale parameter, represents the scale of the distribution function as the quantile at which 63.2% of ADEs occur. β: Shape parameter, could be used to confirm the distribution type: early failure type (β < 1), random failure type (95% CI of β includes 1), and wear-out type (β > 1).
95% CI, 95% confidence interval; IQR, interquartile range; PT, preferred term; SOC, System Organ Classes; TTO, time-to-onset; WSP, Weibull shape parameter.
By contrast, retinal vein occlusion (β = 0.52, 95% CI: 0.00–1.04), herpes zoster (β = 0.87, 95% CI: 0.65–1.09), sinusitis (β = 0.93, 95% CI: 0.25–1.60), myocardial infarction (β = 0.76, 95% CI: 0.46–1.06), and breast cancer (β = 1.79, 95% CI: 0.66–2.91) exhibit random failure patterns, indicating a consistent safety concern over the treatment period.
Notably, an increase in fibrin D dimer (β = 1.91, 95% CI: 1.35–2.46) demonstrated wear-out failure patterns, suggesting a higher likelihood of ADRs occurring later during the treatment course. Median TTO and corresponding α parameters further highlighted the variability in timing and magnitude of these events, such as the median TTO of 7.5 days (IQR: 4.5–13.0 days) for DVT compared to 841.5 days (IQR: 325.5–1036.0 days) for breast cancer.
Discussion
Post-marketing reviews have highlighted emerging safety concerns as the clinical use of baricitinib continues to expand.22,23 Consistent with these observations, disproportionality analyses from real-world pharmacovigilance databases have detected non-trivial safety signals, 24 providing the rationale for an integrated real-world and mechanistic evaluation in this study.
Study objectives and innovation
To our knowledge, this study provides the first large-scale integration of age-stratified FAERS signals with MR of JAK1/2 inhibition, enabling precise characterization of real-world toxicity and a causal framework to interpret the efficacy–toxicity paradox.25,26
Demographic variations and reporting trends
Females accounted for 63.41% of ADR reports, compared to 26.91% for males, consistent with the higher prevalence of immune-mediated diseases (e.g., RA, systemic lupus erythematosus) in females. 27 Adults aged 18–65 years represented 44.11% of reports, reflecting baricitinib’s primary use in RA management within this age group. 28 Consumer-submitted reports comprised 50%, suggesting heightened patient awareness, while physicians and pharmacists contributed 14.48% and 16.08%, respectively.
Key findings and comparison with existing literature
By conducting DPA on 5354 ADR reports from Q3 2018 to Q1 2024, we identified age-related patterns consistent with existing literature, including common events such as upper respiratory tract infections, herpes zoster, and liver dysfunction. Notably, DVT and PE signals align with MR evidence showing that physiological JAK2 elevation reduces thrombotic risk (OR = 0.998, pIVW = 0.028), whereas pharmacological JAK2 inhibition by baricitinib may counteract this protective effect. Other frequently reported PTs included hepatic enzyme elevation, urinary tract infections, hyperlipidemia, acne, and sinusitis, corroborating the SPCs and clinical trial data for baricitinib, 23 and supporting the reliability of our pharmacovigilance approach. ADR reports peaked in 2021, followed by a downward trend, potentially linked to updated safety warnings from regulatory agencies (e.g., the United States and United Kingdom), which may have prompted stricter patient selection and monitoring practices. We identified 261 statistically significant ADR signals across SOCs, including infections, blood/lymphatic disorders, and VDs. The emergence of AF as a novel signal is mechanistically plausible, given MR evidence linking JAK1 (not JAK2) expression to increased AF risk (OR = 1.255, pIVW = 0.043), suggesting that baricitinib’s JAK1 inhibition may incompletely mitigate this pathway. Conversely, the absence of ILD and AP signals aligns with null MR associations for JAK2 (both pIVW > 0.05), reinforcing the disease specificity of JAK-STAT modulation. These novel signals warrant further clinical scrutiny to differentiate drug-induced effects from underlying comorbidities.
Infection-related ADRs
In our study, DPA demonstrated that IAIs constituted 28.3% of signals, consistent with prior studies. 24 Several of these ADRs had been reported in clinical trials. 29 Herpes zoster (9.3%), pulmonary infections (including bacterial pneumonia) (18.2%), and urinary tract infections (7.0%) were prominent, while lymph node tuberculosis remained rare (0.4%). Meta-analyses indicate baricitinib increases the relative risk (RR) of infections (RR = 1.34) and opportunistic infections (RR = 2.69), though severity is typically mild. The cumulative incidences of herpes zoster and pneumonia were 1.13% and 0.26%, respectively. 30 A 2-year cohort study following 4720 RA patients treated with baricitinib found the incidence of ADRs per 100 person-years to be: herpes zoster 4.68, severe infections 3.05. 31 A dose-dependent relationship with herpes zoster incidence has been reported,32,33 emphasizing the need for proactive and stringent monitoring of patients undergoing baricitinib treatment. Our study suggested that individuals aged ≥66 were more prone to chronic infections, which may have been related to the effects of baricitinib on the immune system. Baricitinib inhibited the JAK-STAT signaling pathway, weakening the immune system’s ability to produce antiviral cytokines, particularly suppressing Type I interferon signaling. In older populations, whose immune systems had already weakened with age, the immunosuppressive effects of baricitinib may have exacerbated the reactivation of latent viruses such as herpes zoster, 29 emphasizing the need for vigilant monitoring, particularly in elderly patients with age-related immune decline. This hazard is critical in managing older patients with underlying conditions or latent infections. 34
Cardiovascular and thrombotic events
Regarding VDs, including thrombotic events such as DVT and PE, our FAERS analysis confirmed the well-established risk associated with baricitinib, as highlighted in its boxed warning. In the 18–65 age group, elevated RORs were observed for embolism (ROR = 21.58), pulseless electrical activity (ROR = 20.29), cerebral infarction (ROR = 16.09), and AMI (ROR = 1.62). Mechanistically, this paradox—where JAK2 inhibition appears harmful—can be explained by MR evidence indicating that physiologically higher JAK2 expression confers vascular protection (p = 0.028), a signal that—despite its numerically small magnitude—reflects causal directionality under lifelong genetic modulation rather than a clinically sized effect. 35 JAK2 maintains endothelial homeostasis via STAT-mediated signaling, promoting nitric oxide (NO) production, limiting platelet activation, and preserving vascular barrier integrity. 36 Pharmacologic JAK2 inhibition by baricitinib may therefore reverse this protective mechanism, consistent with a “dose–response inversion” hypothesis: moderate physiological activity is protective, whereas inhibition or constitutive activation promotes thrombosis. This is further supported by the pro-thrombotic effect of constitutive JAK2 activation (e.g., V617F mutation) in myeloproliferative neoplasms 37 and aligns with meta-analyses showing that JAK inhibitors, evidence from a recent meta-analysis does not show a significant increase in venous thromboembolism (VTE) risk with JAK inhibitors compared with TNF inhibitors (RR = 0.94, 95% CI 0.33–2.69), while lower-dose tofacitinib was associated with a lower risk of VTE versus higher dose (RR = 0.51, 95% CI 0.30–0.86). 38 Patients with RA faced a higher threat of VTE due to their elevated inflammatory state, 39 particularly in patients with immune-mediated inflammatory diseases (IMIDs), who often received a combination of medications, including corticosteroids, biologics, and JAK inhibitors. Baricitinib’s inhibition of JAK1/2 may exacerbate thrombotic susceptibility via platelet regulation. 40 Interestingly, the newly observed AF signal had biological plausibility. Our genetic studies showed that higher JAK1 expression was associated with increased AF risk (Wald ratio: OR = 1.255, p = 0.043), potentially via IL-6/JAK1/STAT3-mediated atrial fibrosis. 41 While baricitinib inhibits both JAK1 and JAK2, the effect of selective JAK1 inhibition on AF risk remains unclear, suggesting complex roles for JAK1 or compensatory mechanisms in the heart. Clinically, patients treated with baricitinib showed an elevated incidence rate ratio (IRR) for VTE (IRR = 1.51) and major adverse cardiovascular events (IRR = 1.54), 42 although some cohort studies reported no significant risk in RA patients treated with baricitinib (HR = 1.1, p = 0.63), 43 underscoring the need for cardiovascular assessments in high-threat patients.
Oncologic concerns
The occurrence of malignancies, especially malignant tumors, was also an important focus of the study on the use of JAK inhibitors. In our study, the more commonly reported tumor-related PTs included 10 cases of malignant melanoma, 41 cases of solid tumors such as breast cancer, and 11 cases of lung adenocarcinoma, among others. The crude incidence rate of lymphoma in patients receiving treatment with baricitinib is 0.10. 44 The JAK/STAT pathway’s role in tumor proliferation 6 and evidence linking long-term JAK inhibitor use to non-melanoma skin cancer 45 highlight the importance of regular cancer screening. Subgroup analysis suggested that the 18–65 age group had a higher tumor reporting rate than the ≥66 age group. Notably, the TTO for breast cancer was characterized by a median of 841.5 days (325.5–1036.0) post-treatment, indicating the need for regular monitoring for early cancer detection during this period. Chronic inflammation and immunosuppression in RA patients may further elevate malignancy. 46 A comprehensive peril assessment and informed consent had to be obtained before initiating baricitinib treatment in these patients.
We identified AP as a new ADR signal. Although case numbers were low, clinical severity was notable. MR analysis did not support a causal relationship between JAK2 expression and AP risk, suggesting that the observed risk may arise from off-target mechanisms. Potential contributing factors include drug-induced hypertriglyceridemia, biliary dysfunction, and immunosuppression-related viral reactivation.47–49
Age-specific vulnerabilities
Patients ≥66 years exhibited unique ADR patterns, including adenocarcinoma gastric (ROR = 69.44), adenocarcinoma of the colon (ROR = 24.85), gastroenteritis (ROR = 18.12), and gastrointestinal perforation, particularly diverticular perforation (diverticular perforation, 8 cases, ROR = 52.76). Age-related declines in gastrointestinal barrier integrity, gut microbiota dysbiosis, and gastric mucosal oxidative stress may collectively heighten susceptibility to gastrointestinal complications in elderly populations.50–52 For patients aged ≥66 receiving baricitinib, proactive monitoring and personalized safety concern stratification are critical to prevent severe ADRs such as gastrointestinal perforation. Enhanced surveillance strategies and tailored interventions are recommended to optimize safety outcomes.
While RA frequently coexists with ILD, 53 baricitinib-associated ILD remains rare (0.2% in ≥66 years vs 0.06% in 18–65 years) and is not documented in the SPCs. 54 This age disparity may reflect cumulative vulnerabilities in elderly populations, including immunosenescence, preexisting cardiopulmonary comorbidities, diminished lung reserve, and altered drug pharmacokinetics. Although the absolute incidence is low, clinicians should maintain vigilance for ILD in older adults receiving baricitinib, particularly those with baseline respiratory compromise.
Lastly, renal impairment was uniquely observed in patients ≥66 years, with a rapid median onset of 6.5 days (IQR: 1.0–10.5). Baricitinib is primarily excreted through the kidneys. Age-related declines in renal clearance, compounded by baricitinib’s primary renal excretion, 55 heighten drug accumulation hazards. To mitigate toxicity, strict renal function monitoring and dose adjustments are essential, particularly in severe impairment.
Several studies have explored the influence of germline variation within the JAK/STAT axis on IMID and therapeutic responsiveness (e.g., rs10758669 in JAK2 associated with RA pharmacogenetics; STAT4 risk haplotypes in IMIDs).56–58 However, to date, no clinical pharmacogenomic study has directly linked JAK/STAT polymorphisms to inter-individual susceptibility to baricitinib-related ADRs. This lack of baricitinib-specific pharmacogenetic evidence highlights an important gap in the literature and underscores the need for prospective validation.
TTO analysis
Longitudinal analysis of 4283 cases revealed distinct temporal hazard profiles. The median TTO was 12.5 days (range: 5.5–122.5 days), with early-phase predominance of thrombotic events such as PE and DVT consistent with reported surges in venous thromboembolism hazard during initial treatment. 59 Through PT’s WSP analysis, we found that the safety concern of baricitinib-related ADRs, such as DVT, PE, and anemia, decreased over time. Tuttle et al. found that patients treated with a high dose (4 mg/day) of baricitinib had a higher incidence of anemia. 60 WSP modeling further delineated three dynamic patterns: (1) declining threats for anemia and thrombosis beyond 6 months, potentially reflecting immune adaptation 61 ; (2) stable chronic safety concerns for sinusitis, herpes zoster, retinal vein occlusion, myocardial infarction, and other conditions, aligned with persistent inflammatory mechanisms62,63; and (3) progressive elevation of D-dimer levels underwent a wear-out failure pattern. In patients with RA, elevated D-dimer levels may be closely associated with the occurrence of DVT, and elevated D-dimer levels could serve as an important biomarker for assessing thrombosis hazard, 64 suggesting cumulative thrombotic susceptibility warranting long-term monitoring.
Strengths
This study extends prior analyses 28 by integrating causal inference with expanded FAERS data, rigorous disproportionality assessment, and age-stratified insights. A major strength is the use of MR, which complements passive surveillance by providing genetic evidence on target-specific effects of JAK1/2 inhibition and mitigates confounding by indication through the simulated random allocation of variants linked to JAK1/2 expression.
Limitations
However, several limitations must be acknowledged. First, FAERS’s passive surveillance design relies on voluntary reporting, resulting in underreporting, incomplete demographics, and limited generalizability. Second, geographic disparity persists in our dataset, 56.14% of reports originated from the United States, followed by Japan, the United Kingdom, France, and Germany-indicating that FAERS submissions remain predominantly from North American and European regions, which limits extrapolation to Asian/African populations. Third, while MR addresses unmeasured confounding for target-mediated effects, its limitations include potential horizontal pleiotropy, the assumption that genetic proxies reflect lifelong inhibition rather than acute drug effects, and the fact that blood-derived eQTLs may not fully capture JAK-STAT signaling in disease-relevant tissues (e.g., vascular endothelium, atrium). In addition, we used a standard two-sample MR framework with population-level eQTL instruments rather than individual-level linkage to FAERS cases, and the strongest instruments were from European-ancestry cohorts; this introduces ancestry mismatch and should be interpreted cautiously. Finally, no formal multiplicity correction was applied at the discovery stage, so false-positive signals cannot be excluded, and results should be regarded as exploratory. To address FAERS constraints, we implemented dual signal detection strategies (ROR and BCPNN). 24 Nevertheless, these remain probabilistic tools for hypothesis generation. Our findings—both FAERS signals and MR associations—require validation in prospective, diverse cohorts and mechanistic studies. Notably, these limitations reflect structural challenges in post-marketing surveillance and genetic epidemiology, not study-specific deficiencies. Our integrated approach aligns with READUS-PV guidelines 65 and provides a framework for future target safety deconvolution.
Conclusion
In conclusion, this integrated pharmacovigilance and MR study delineates age-specific adverse event patterns of baricitinib and provides genetic evidence linking JAK1/2 inhibition to thrombotic and AF risks. These findings support personalized risk mitigation, including vigilant infection monitoring, proactive cardiovascular assessment, and age-tailored cancer surveillance. Future research should validate these signals in prospective cohorts and clarify the molecular mechanisms underlying target-mediated toxicities to guide safer clinical use.
Supplemental Material
sj-tiff-2-taw-10.1177_20420986251406106 – Supplemental material for Differential risk of adverse drug reactions with baricitinib across age groups: integrating real-world pharmacovigilance and genetic causal inference
Supplemental material, sj-tiff-2-taw-10.1177_20420986251406106 for Differential risk of adverse drug reactions with baricitinib across age groups: integrating real-world pharmacovigilance and genetic causal inference by Huiqiong Zeng, Wei Liu, Aidong Li, Hanjiang Liu, Xiaojuan Li, Junda Lai, Miaoqian Chen, Gaofeng Xiong and Ye Zhang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-1-taw-10.1177_20420986251406106 – Supplemental material for Differential risk of adverse drug reactions with baricitinib across age groups: integrating real-world pharmacovigilance and genetic causal inference
Supplemental material, sj-xlsx-1-taw-10.1177_20420986251406106 for Differential risk of adverse drug reactions with baricitinib across age groups: integrating real-world pharmacovigilance and genetic causal inference by Huiqiong Zeng, Wei Liu, Aidong Li, Hanjiang Liu, Xiaojuan Li, Junda Lai, Miaoqian Chen, Gaofeng Xiong and Ye Zhang in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors wish to thank the FAERS data, which was provided by the FDA for academic purposes. The authors acknowledge the FDA but are solely responsible for the analysis, interpretation, and conclusions, acknowledging any data limitations or biases.
Declarations
Ethics approval and consent to participate
This study was based on publicly available, anonymized FAERS data, which is exempt from institutional review board approval and patient consent requirements. All MR data were derived from publicly available GWAS summary statistics, with prior ethical approval and informed consent obtained in the original studies. Consent to participate: Not applicable.
Consent for publication
Not applicable.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported in part by research grants from Futian Healthcare Research Project (FTWS084).
Competing interests
The authors declare that there is no conflict of interest.
Availability of data and materials
Data are available on the FAERS database (https://www.fda.gov/drugs/fdas-adverse-event-reporting-system-faers/fda-adverse-event-reporting-system-faers-public-dashboard). Detailed sources of the MR data are provided in ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
