Abstract
Background:
There is a rising incidence of neurological adverse events (AEs), such as seizures, associated with novel anticancer agents, warranting investigation. Large-scale studies assessing seizure risk across diverse anticancer drug classes, particularly in breast cancer (BC), remain limited.
Objective:
This study aimed to systematically evaluate the association between seizures and 14 novel anticancer agents used in BC treatment, compared with traditional chemotherapy, utilizing international pharmacovigilance databases.
Design:
A large-scale, real-world pharmacovigilance study using data from the US FDA Adverse Event Reporting System (FAERS) and the Canada Vigilance Database (from Q1 2004 to Q1 2025).
Methods:
Disproportionality analysis was employed to calculate reporting odds ratios (RORs) for identifying significant seizure AE signals. Signals were assessed at both the Standardised MedDRA Query and Preferred Term levels. Pan-cancer transcriptomic data from The Cancer Genome Atlas were integrated to explore biological pathways correlated with drug-induced seizures.
Results:
Significant and consistent seizure signals were identified for five agents—Lapatinib, Tucatinib, Trastuzumab, Trastuzumab Emtansine (T-DM1), and Atezolizumab—across both databases. In FAERS, over 50% of seizures occurred after 100 days of treatment (median: 68 days); however, fatal cases exhibited a significantly shorter median onset time. Novel agents demonstrated disproportionately higher seizure reporting signals compared to traditional chemotherapy. Pan-cancer analysis revealed negative correlations between seizure RORs and pathways, including asthma and the pentose phosphate pathway.
Conclusion:
This dual-database pharmacovigilance study identifies potential associations between seizures and five novel BC therapies, underscoring the need for vigilant monitoring during their clinical use.
Plain language summary
This study investigates seizure risks linked to 14 novel anticancer drugs, a topic previously underexplored across diverse drug classes. By combining data from the FAERS and Canada Vigilance Database, researchers enhanced reliability through larger samples and diverse demographics, focusing on breast cancer (BC), a key population for these agents. Analysis revealed significant seizure signals for five drugs (lapatinib, tucatinib, trastuzumab, T-DM1, atezolizumab) consistent across both databases. Trastuzumab had high reporting volumes (193 in FAERS, 39 in Canada). Notably, novel agents showed higher seizure reports than traditional chemotherapy. Seizures typically occurred after 100 days of treatment (median 68 days), but fatal cases had a significantly shorter onset, possibly linked to BC brain metastasis or poor baseline health. Using TCGA transcriptomic data, the study explored underlying mechanisms. Pan-cancer seizure risk negatively correlated with pathways like pentose phosphate (involving glucose-6-phosphate dehydrogenase, known to mitigate seizures) and asthma (linked to mTOR, a key seizure pathway). Limitations include database biases (underreporting, duplication), residual confounding, unmeasured variables (e.g., comorbidities), and undetermined dose effects. Findings require validation via basic research and prospective studies. In summary, five BC therapies showed potential seizure associations, with fatal cases needing early vigilance. Novel anticancer drugs pose higher seizure risks than traditional chemotherapy, supported by mechanistic links to metabolic and mTOR pathways.
Keywords
Introduction
In recent years, a growing number of antitumor drugs have shown remarkable efficacy in treating breast cancer (BC).1–5 However, extensive use of these drugs has raised concerns about associated adverse drug reactions (ADRs). 6 Among these ADRs, seizures, which result from excessive and abnormal neural activity in the brain, can disrupt treatment, reduce patients’ quality of life, and even lead to death. The clinical manifestation of seizures is highly variable, including sudden loss of consciousness and limb convulsions in cases of generalized seizures, as well as cognitive decline and psycho-behavioral abnormalities caused by complex focal seizures. 7 Due to the increasing number of reports on drug-related seizures in recent years, they have become among the most closely monitored adverse reactions.8,9
Regarding BC treatment, the incidence of seizures associated with traditional chemotherapeutic drugs is relatively low and has not received sufficient attention. Currently used novel antitumor agents exhibit a stable rate of seizure occurrence. 10 However, in clinical practice, some types of seizures are difficult to detect early due to their covert nature. In certain countries and regions, standardized protocols for identifying and managing antitumor drug-induced seizures have yet to be established. According to pharmacovigilance analyses and case reports documenting seizure incidents, this gap may directly contribute to serious clinical events or even fatalities among patients with cancer. 7
Previous studies have mainly focused on individual drugs or single drug classes, with limited comparisons between different types of antitumor agents.11,12 Using two large adverse reaction databases—the Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS, the largest post-marketing safety surveillance database globally) and Canada Vigilance Database—we performed a comprehensive evaluation of seizure cases across multiple drug categories in patients with BC.13,14 This large-scale, real-world pharmacovigilance study provides new insights into the relative safety of these drugs and overcomes the limitations of relying solely on single datasets without external validation of seizure-related reports. We also analyzed the timing of seizure-related adverse events (AEs) and fatal outcomes to better elucidate the temporal patterns and mortality risks associated with drug-induced seizures.
Methods
Data sources
In this study, ADR signal mining was conducted based on the US FAERS and Canada Vigilance Database. The FAERS, a global pharmacovigilance database regulated by the FDA, compiles AE reports voluntarily submitted by healthcare institutions, pharmaceutical companies, and patients, comprising core datasets such as patient demographics, drug information, AEs, and clinical outcomes. 15 The Canada Vigilance Database, a regional drug safety monitoring system, has an architecture similar to that of FAERS, but with a relatively smaller data scale. All AEs in this study were standardized using the Medical Dictionary for Regulatory Activities (MedDRA) terminology (latest version), with a focus on the analysis of adverse reaction signals associated with BC therapeutic drugs. Signal detection was performed by calculating quantitative indicators such as the reporting odds ratio (ROR). Since both databases were fully de-identified, this study met the requirements for ethical exemption. This study utilized exclusively de-identified, publicly available data from the FAERS and Canada Vigilance Databases. As no private or identifiable patient information was involved, this analysis was exempt from the requirement for institutional review board approval and patient-informed consent in accordance with applicable guidelines and institutional policy.
Data availability
For each case included in the study, the following medication- and disease-related variables were available and used to support disproportionality analysis:
Medication-related fields: Drug name (both generic and brand), active ingredient, and drug role code (Primary Suspect, Secondary, or Concomitant). Only cases where the anticancer drug of interest was coded as Primary Suspect were included. Treatment indication was used to retain cases associated with BC.
Event-related fields: AEs were coded using MedDRA preferred terms (PTs). For this study, seizure-related reports were identified using the Standardised MedDRA Query (SMQ) for convulsions/seizures, restricted to PTs directly reflecting seizure activity.
Patient-related fields: Age and sex were available for most cases and used in descriptive analyses and deduplication. Body weight was frequently missing (approximately 67% missing in FAERS and 70% missing in Canada Vigilance) and therefore used only for descriptive purposes.
Outcome-related fields: Outcome information (e.g., death, hospitalization, life-threatening event, and disability) was used to describe clinical consequences of seizure events.
Metadata fields: Reporting country, report date, and event date were included. These fields were used for deduplication, temporal analysis, and to assess potential reporting overlap between databases.
Causality assessment: Systematic ADR causality scales (e.g., Naranjo and WHO-UMC) were not routinely captured in either database. Only a negligible proportion of reports contained any causality annotations, which were insufficient for stratified analysis. This limitation is addressed in the Discussion.
A detailed summary of field availability and missingness is provided in Supplemental Table 1.
Procedures
The selection of the 14 novel targeted and immunotherapeutic agents included in this study was based on the following criteria: FDA approval for BC indications, representation of key novel mechanistic classes (e.g., human epidermal growth factor receptor 2 (HER2)-targeted, cyclin-dependent kinase 4/6 (CDK4/6) inhibition, poly (ADP-ribose) polymerase (PARP) inhibition, immune checkpoint inhibition), and widespread clinical use during the study period to ensure sufficient report volume for signal detection in the databases. Traditional chemotherapeutic agents selected for comparison were those commonly used as cytotoxic drugs in standard BC regimens. We collected data on 14 FDA-approved novel BC therapeutic agents from the FAERS (January 1, 2004 to March 31, 2025) and the Canada Vigilance Database (January 1, 2004 to March 31, 2025). These agents included: the mammalian Target Of Rapamycin (mTOR) inhibitor (everolimus); phosphoinositide 3-kinase (PI3K) inhibitor (alpelisib); PARP inhibitors (olaparib and talazoparib); CDK4/6 inhibitors (ribociclib and palbociclib); programmed cell death protein 1 (PD-1) inhibitor (pembrolizumab); programmed death-ligand 1 (PD-L1) inhibitor (atezolizumab); antibody-drug conjugates T-DM1 (trastuzumab emtansine) and T-DXd (trastuzumab deruxtecan); and HER2-targeted therapies (trastuzumab, pertuzumab, tucatinib, and lapatinib). The generic and brand names of all medications were included.
Following FDA recommendations, duplicate data were removed prior to statistical analysis using the following criteria: for identical Case Report IDs (CASEID), the most recent submission date
Definition of adverse reaction signals
The identification of a “seizure adverse reaction signal” in this study was based on two criteria. First, at the case selection level, we only included reports where the target antineoplastic agent was explicitly designated as the “Primary Suspect” drug for the seizure event, ensuring that the analyzed signals represented the strongest perceived clinical association. Second, at the statistical analysis level, a positive signal was defined by an ROR with a lower limit of the 95% confidence interval (CI) greater than 1, supported by a minimum of three or four relevant case reports (as detailed in Section “Statistical analysis”).
Pan-cancer analysis was performed using The Cancer Genome Atlas database
Transcriptome data in FPKM (Fragments Per Kilobase of transcript per Million mapped reads) format for 23 tumor types were acquired from The Cancer Genome Atlas (TCGA) database and subsequently converted to Transcripts Per Million format. 17 Kyoto Encyclopedia of Genes and Genomes (KEGG) and Reactome pathway gene sets were obtained from the Molecular Signature Database. All tumor transcriptome data were analyzed using single-sample Gene Set Enrichment Analysis with the GSVA package in R. To determine the biological mechanism underlying seizure induction by novel antitumor drugs, we analyzed the association between the novel antitumor drug-induced target Preferred Term Reporting Odds Ratio (PTROR) and biological pathways at the pan-cancer level.
Statistical analysis
This study employed the ROR for disproportionality analysis to evaluate the association between 14 novel BC therapeutic agents and epileptic seizures. 18 Strict adherence to international pharmacovigilance research standards was maintained, requiring the lower bound of the 95% CI for the ROR to be >1, with a minimum of three seizure case reports. The formulas for calculating the ROR and its 95% CI are presented in Table 1.
Formulas for the reporting odds ratio and its 95% confidence interval.

CI, confidence interval; ROR, reporting odds ratio.
For all ROR calculations in this study, the aforementioned formulas were employed. A positive signal was defined as ⩾4 reports with the lower bound of the 95% CI for ROR exceeding 1. Lethal time distribution curves were analyzed using the Wilcoxon test, mortality proportions were compared using the chi-square test, and correlations were assessed using Spearman’s rank correlation. To compare seizure signals between novel antineoplastic agents and chemotherapeutic drugs, Bonferroni-corrected

Flow chart of the study.
Results
Descriptive analysis
As shown in Table 2, from the FAERS database, 663 seizure-related reports were analyzed. Patients aged 18–65 years accounted for the largest proportion (50.2%, n = 333); the vast majority of patients were female (97.7%, n = 648). The top five countries with the most reports were the United States, Canada, Argentina, Japan, and India. Clinical outcome analysis showed that 40.3% (n = 267) of the patients required hospitalization and 16.4% (n = 109) died. In 101 reports from the Canada Vigilance Database, female individuals still had a high proportion (84.2%, n = 85), with 80.2% (n = 81) aged 18–65 years. The mortality rate (14.8%, n = 15) was similar to that in the FAERS data. Notably, 37.6% (n = 38) of patients in the Canada Vigilance Database did not fully recover after seizures, suggesting potential long-term neurological effects.
Characteristics of seizure AE reports related to novel antitumor drugs from FAERS and Canada Vigilance Database (Q1, 2004–Q1, 2025).
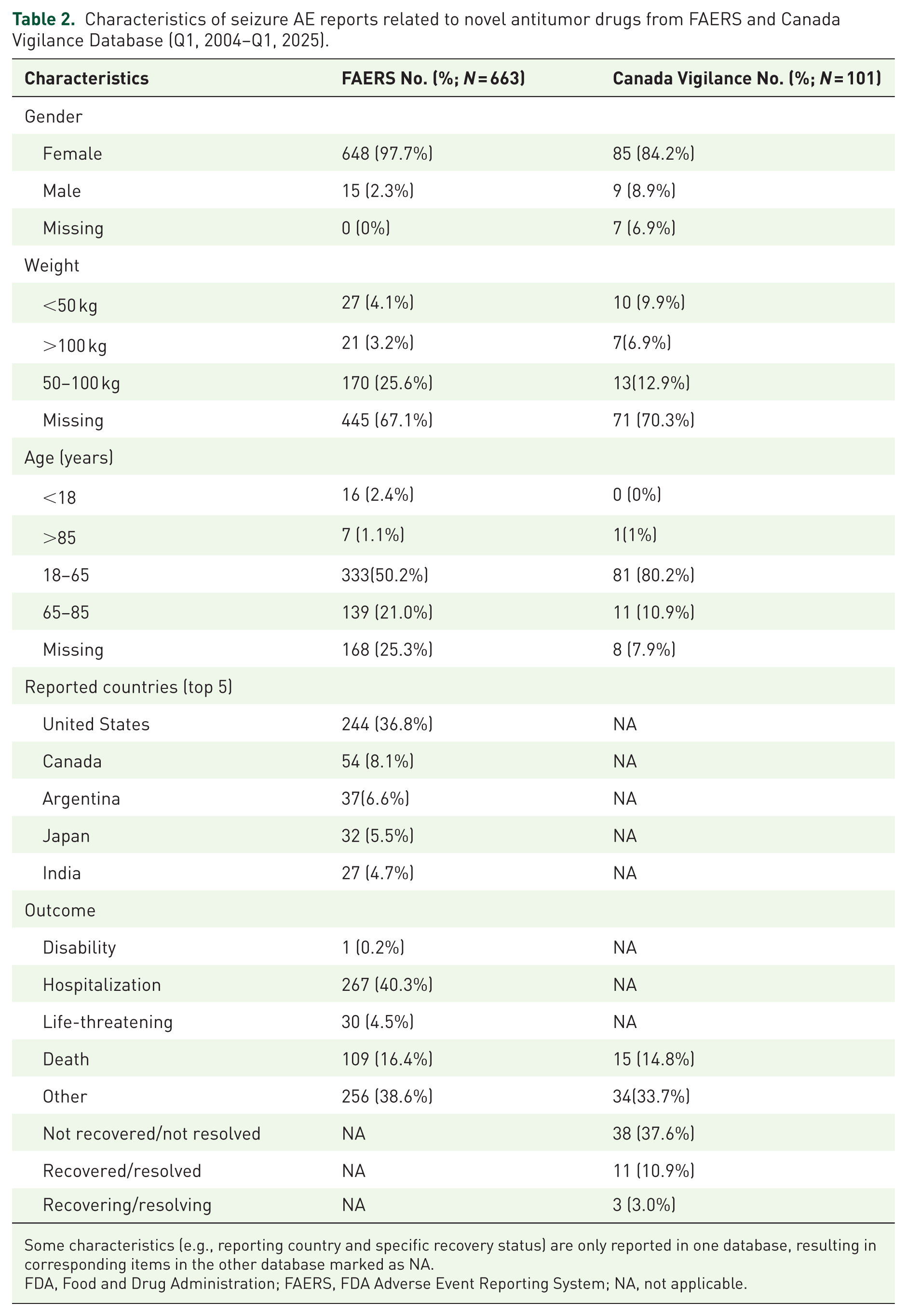
Some characteristics (e.g., reporting country and specific recovery status) are only reported in one database, resulting in corresponding items in the other database marked as NA.
FDA, Food and Drug Administration; FAERS, FDA Adverse Event Reporting System; NA, not applicable.
Disproportionality analysis
From the FAERS, eight novel antitumor drugs were identified as disproportionate seizure signals (Figure 2(a)), including: Lapatinib (ROR, 2.44; 95% CI: 1.89–3.15), tucatinib (ROR, 3.07; 95% CI: 2.21–4.25), pertuzumab (ROR, 1.7; 95% CI: 1.24–2.33), trastuzumab (ROR, 1.91; 95% CI: 1.64–2.23), T-DM1 (ROR, 3.31; 95% CI: 2.43–4.50), T-DXd (ROR, 1.83; 95% CI: 1.26–2.64), atezolizumab (ROR, 2.74; 95% CI: 1.55–4.84), and alpelisib (ROR, 1.67; 95% CI: 1.12–2.48). From the Canada Vigilance Database, five drugs were identified as disproportionate seizure signals (Figure 2(b)), specifically: Lapatinib (ROR, 3.43; 95% CI: 2.06–5.71), tucatinib (ROR, 11.45; 95% CI: 4.17–31.46); trastuzumab (ROR, 1.77; 95% CI: 1.21–2.58); T-DM1 (ROR, 2.97; 95% CI: 1.66–5.32); and atezolizumab (ROR, 7.57; 95% CI: 2.76–20.75). Notably, pertuzumab, T-DXd, and alpelisib showed seizure signals in the FAERS database, but were negative in the Canada Vigilance Database. Overall, lapatinib, tucatinib, trastuzumab, T-DM1, and atezolizumab were more likely to induce seizures when validated using dual-database analysis. In both the FAERS and Canada Vigilance Database, we analyzed the 15 most frequently reported seizure-related pharmacokinetic parameters (PT) at standardized MedDRA query levels (Figure 2(c) and (d)). Multiple PT parameters showed positive signals (10 in the FAERS and 6 in the Canada Vigilance Database), demonstrating that various novel antitumor drugs significantly impact the nervous system—a finding warranting further investigation. At the PT level, the FAERS identified 60 positive signals across the different medications (Figure 2(e)). Tucatinib and palbociclib showed the highest number of signals in nine cases. Seven patients received ribociclib and trastuzumab, respectively. Pertuzumab produced six signals, whereas T-DM1 and alpelisib showed five each. Atezolizumab produced three signals, whereas olaparib, pembrolizumab, T-DXd, and lapatinib produced two signals each. Everolimus reported one signal. In the Canada Vigilance Database (Figure 2(f)), T-DM1, trastuzumab, and pertuzumab showed four signals at their highest frequencies. Everolimus, atezolizumab, and lapatinib each showed three signals. Pembrolizumab produced two signals, whereas ribociclib, palbociclib, and tucatinib produced one signal each.

Signals of seizure associated with novel antineoplastic agents in BC treatment. (a) Forest plot of ROR for seizure associated with different novel antineoplastic agents in the FAERS database. (b) Forest plot of ROR for seizure associated with different novel antineoplastic agents in the Canada Vigilance Database. (c) ROR values of the top 15 most frequently reported PTs under seizure SMQ level in the FAERS database. (d) ROR values of the top 15 most frequently reported PTs under seizure SMQ level in the Canada Vigilance Database. (e) Heatmap showing disproportional reporting signals at the PT level among different novel antineoplastic agents in the FAERS database. (f) Heatmap showing disproportional reporting signals at the PT level among different novel antineoplastic agents in the Canada Vigilance Database. Red indicates that the lower limit of the 95% CI of the ROR > 1 and at least three seizure cases were reported. Blue indicates that the lower limit of the 95% CI of the ROR < 1, or fewer than three seizure cases were reported. Gray indicates no seizure-related reports.
Analysis of seizure onset time and death cases
In the FAERS, the median time to onset of seizures associated with all novel antineoplastic agents was 103 days (interquartile range: 20–367.5), compared with 68 days (interquartile range: 24–160.5) for fatal cases, without a significant difference

Cumulative distribution curve of seizure onset time following administration of novel antineoplastic agents and analysis of seizure-related fatal cases. (a) Time to seizure onset after antineoplastic treatment in all cases and fatal cases from the FAERS database. Panel (b) and (c) show the fatality ratio and number of different novel antineoplastic agents in the FAERS and Canada Vigilance Databases, respectively. Panel (d) and (e) compare the mortality risk between patients with seizures and non-seizure (Other PT) in the FAERS and Canada Vigilance Databases, respectively.
Comparison with chemotherapy
Compared with traditional chemotherapeutic drugs, novel antitumor agents showed significantly more reported signals (Figure 4). Using the volcano plot analysis, the FAERS database included eight novel antitumor drugs, whereas the Canada Vigilance Database comprised seven drugs with five novel candidates. The ROR forest plot results for traditional chemotherapy drugs were consistent with the volcano plot findings: no positive results were found in the FAERS database, whereas only docetaxel (ROR, 2.02; 95% CI: 1.22–3.36) and epirubicin (ROR 3.05; 95% CI: 1.45–6.41) showed positive results in the Canada Vigilance Database (Figure S1).

Volcano plots comparing seizure signals between novel antineoplastic agents and chemotherapeutic drugs. Panel (a) and (b) display comparisons within the FAERS and Canada Vigilance Databases, respectively. The x-axis represents the logarithm of the ROR, and the y-axis represents the negative base-10 logarithm of the adjusted p value, calculated via Fisher’s exact test with Bonferroni correction. The color intensity of each dot indicates the number of reports, with redder hues corresponding to higher report counts. Drugs in the upper right quadrant exhibit elevated seizure signals with statistical significance.
Analysis of potential biological mechanisms
Using pan-cancer analysis, the top five cancers with seizure levels of ROR induced by novel antitumor drugs were endometrial cancer (ROR, 4.93), sarcoma (ROR, 4.91), low-grade glioma (ROR, 2.88), hepatocellular carcinoma (ROR, 2.40), and prostate adenocarcinoma (ROR, 2.05). Cutaneous melanoma showed the lowest ROR (ROR, 0.22), whereas BC ranked eighth (ROR, 1.78), as shown in Figure S2. Biological mechanism mining and correlation analysis with the TCGA database revealed that novel antitumor drugs could induce seizure-related ROR levels, negatively correlating with six pathways. The top three correlations were the glycosphingolipid biosynthesis lacto and neolacto-series (R: −0.54; false discovery rate (FDR) < 0.01), pentose phosphate pathway (R: −0.487; FDR < 0.05), and fructose mannose metabolism (R: −0.463; FDR < 0.05), as illustrated in Figure S3.
Discussion
To the best of our knowledge, this is the first large-scale real-world study (from 2004-Q1 to 2025-Q1) to jointly utilize the FAERS and Canada Vigilance Database to investigate seizure-related AEs associated with novel anticancer agents in patients with BC. Our analysis identified potential associations between seizure occurrence and lapatinib, tucatinib, trastuzumab, T-DM1, and atezolizumab use during treatment.
Given the increasing incidence of neurological and psychiatric AEs, seizure-related ADRs have attracted increasing attention.19,20 Previous studies using the FAERS have assessed the seizure safety of lacosamide and perampanel 21 or characterized the seizure AE profiles for individual drugs.22,23 However, no previous studies have analyzed seizure signals across diverse anticancer drug classes. Our study enhanced the reliability by incorporating the Canada Vigilance Database alongside the FAERS, offering a larger sample size, diverse demographics, and an extended observation period. BC, the most common malignancy in women, represents a key population of these agents. 24
Among the 14 novel anticancer agents examined, three agents—tucatinib, talazoparib, and pembrolizumab—have been explicitly reported in the literature to be associated with a risk of seizures, warranting vigilance during treatment.25–27 Everolimus (an mTOR inhibitor), used in combination with tuberous sclerosis-associated seizures, was not directly linked to seizure induction. 28 Nonetheless, the disproportionality analysis revealed significant seizure signals for several agents. Lapatinib, tucatinib, trastuzumab, T-DM1, and atezolizumab showed consistent signals in both databases. Notably, trastuzumab had high reporting volumes (the FAERS, n = 193; the Canada Vigilance Database, n = 39). Seizure-standardized MedDRA query analysis identified four signals validated in both databases (the FAERS: n = 10; the Canada Vigilance Database: n = 6). A heatmap of the top 15 PTs revealed ten anticancer drugs with positive PT-level signals from both sources, strengthening the evidence for potential associations. In the FAERS, >50% of seizures occurred after 100 days of treatment (median time, 68 days), indicating a predominance in the later stages. However, fatal cases showed a significantly shorter median onset time, suggesting rapid progression, possibly due to factors like BC brain metastasis or poorer baseline health.10,29 Novel agents have been disproportionately reported for seizures compared with traditional chemotherapy.
Given the limited research on novel anticancer drug-associated seizure mechanisms, we integrated TCGA pan-cancer transcriptomic data.17,30 Among the 23 cancers with target PTs, BC ranked eighth in ROR. Pan-cancer ROR for drug-induced seizures negatively correlated with multiple pathways. Hu et al. described the role of glucose-6-phosphate dehydrogenase (part of the pentose phosphate pathway) in seizure mitigation, 31 which is consistent with our findings. Biagioni et al. and Karagiannis et al. linked mTOR to asthma severity, as mTOR is a key seizure pathway,32,33 which supports our observation of an asthma pathway-seizure correlation.
Furthermore, the findings of this study carry important clinical implications. They serve to alert clinicians to the potential risk of seizures in patients receiving lapatinib, tucatinib, trastuzumab, T-DM1, or atezolizumab, particularly in those with risk factors such as brain metastases. Given the shorter median time to onset in fatal cases, close monitoring for neurological symptoms and patient education during the initial treatment phase are especially crucial. This real-world evidence supports risk-benefit evaluation in clinical practice and suggests that vigilance for seizure risk should be integrated into patient management protocols for these agents.
Limitations
The study has some limitations. 34 Data from the FAERS and Canada Vigilance Database carry the risks of underreporting, duplication, and inaccuracy; selective reporting and incomplete information id inherent. Residual bias is possible despite deduplication efforts. Demographic limitations (sex, age, race, and region) also exist. Unmeasured confounders such as seizure-predisposing comorbidities or concomitant medications cannot be excluded. The dose-dependent effects of ADRs remain undetermined. These findings require validation through basic research and prospective cohort studies to further elucidate the associations.
Conclusion
This study is the first to systematically identify five novel anti-BC agents associated with a significant seizure risk through a large-scale, dual-database analysis. The findings suggest that clinical vigilance is warranted when using these drugs, emphasizing the need for enhanced monitoring of neurological symptoms, particularly during the early phase of treatment. The earlier onset in fatal cases further underscores the importance of prompt management of acute seizures. Our study provides critical evidence for the safety profile evaluation of these vital anticancer agents, calling for future prospective research to develop specific risk mitigation strategies.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251405091 – Supplemental material for Evaluating seizures associated with novel antineoplastic agents during breast cancer treatment using the Food and Drug Administration Adverse Event Reporting System and Canada Vigilance Adverse Reaction Online Database
Supplemental material, sj-docx-1-taw-10.1177_20420986251405091 for Evaluating seizures associated with novel antineoplastic agents during breast cancer treatment using the Food and Drug Administration Adverse Event Reporting System and Canada Vigilance Adverse Reaction Online Database by Yupeng Sha, Quan Yuan, Yao Qian, Xiaoming Li, Ming Niu, Yi Du, Xiaoshuan Liang, Shanshan Sun, Yige Lu and Jiguang Han in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251405091 – Supplemental material for Evaluating seizures associated with novel antineoplastic agents during breast cancer treatment using the Food and Drug Administration Adverse Event Reporting System and Canada Vigilance Adverse Reaction Online Database
Supplemental material, sj-docx-2-taw-10.1177_20420986251405091 for Evaluating seizures associated with novel antineoplastic agents during breast cancer treatment using the Food and Drug Administration Adverse Event Reporting System and Canada Vigilance Adverse Reaction Online Database by Yupeng Sha, Quan Yuan, Yao Qian, Xiaoming Li, Ming Niu, Yi Du, Xiaoshuan Liang, Shanshan Sun, Yige Lu and Jiguang Han in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986251405091 – Supplemental material for Evaluating seizures associated with novel antineoplastic agents during breast cancer treatment using the Food and Drug Administration Adverse Event Reporting System and Canada Vigilance Adverse Reaction Online Database
Supplemental material, sj-docx-3-taw-10.1177_20420986251405091 for Evaluating seizures associated with novel antineoplastic agents during breast cancer treatment using the Food and Drug Administration Adverse Event Reporting System and Canada Vigilance Adverse Reaction Online Database by Yupeng Sha, Quan Yuan, Yao Qian, Xiaoming Li, Ming Niu, Yi Du, Xiaoshuan Liang, Shanshan Sun, Yige Lu and Jiguang Han in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
