Abstract
Background:
Enzalutamide, approved in China for the treatment of prostate cancer (PC), has been studied for risk of seizure, but there is limited real-world evidence on this risk in China. Cases of posterior reversible encephalopathy syndrome (PRES) among enzalutamide-treated patients have been reported.
Objectives:
To evaluate the risk of seizure and PRES in patients with PC treated with enzalutamide in China.
Design:
Observational cohort study
Methods:
Data were extracted from electronic medical records of patients with PC aged ⩾18 years treated at three clinical sites in China who started enzalutamide between March 1, 2020 and May 31, 2022. The primary objective was to estimate the proportion (%) of enzalutamide- or alternative antiandrogen-treated patients who experienced seizure. The index date was the date of the start of treatment. The follow-up period started from the index date and included both on- and off-treatment periods.
Results:
Demographic and clinical characteristics were generally similar between the enzalutamide (n = 385) and alternative antiandrogen (n = 365) groups. Risk factors for seizure were noted in 148 (38.4%) enzalutamide- and 130 (35.6%) alternative antiandrogen-treated patients. Median follow-up was 85 days for patients on enzalutamide and 196 days for those on alternative antiandrogens. During the on-treatment period, no patients on enzalutamide experienced seizure (95% confidence interval (CI): 0.00, 0.95), while one patient on an alternative antiandrogen experienced seizure (0.27%; 95% CI: 0.01, 1.48). The seizure incidence rates per 100 person-years for enzalutamide were 0.00 (95% CI: 0.00, 3.50) for the on-treatment period and 0.00 (95% CI: 0.00, 2.74) for the full follow-up period; for alternative antiandrogens, they were 0.58 (95% CI: 0.00, 2.14) during the on-treatment period and 0.43 (95% CI: 0.00, 1.61) during the full follow-up period. No cases of PRES were reported.
Conclusion:
Among patients with PC in China, the risk of seizure was low in both the enzalutamide and alternative antiandrogen groups, despite the presence of risk factors for seizure in over one-third of patients.
Plain language summary
Why was the study done? Enzalutamide is used to treat prostate cancer. Sometimes, patients with prostate cancer taking enzalutamide or other treatments have seizures. Although the risk of this happening has been studied in clinical trials, we still need to understand how often it happens in everyday life.
What did the researchers do? The researchers looked at data in China from 385 patients with prostate cancer who took enzalutamide and 365 patients who got other treatments. They aimed to find out how many patients had seizures while on enzalutamide and on other treatments.
What did the study find? During the study, none of the patients taking enzalutamide had a seizure, and one patient in the other treatment group had a seizure.
What do the findings mean? The risk of seizure was low for patients in China with prostate cancer who were treated with enzalutamide, as well as for those who got other treatments.
Introduction
Prostate cancer (PC) is the second most frequently diagnosed malignancy 1 and one of the leading causes of cancer death in men worldwide, with 1.46 million new cases and over 397,000 deaths reported in 2022. 2 According to the International Cancer Research Agency, there were around 134,000 new cases of PC reported in China in 2022, and 47,500 deaths, accounting for 2.9% of malignancy deaths in men. 3 The approval of androgen receptor pathway inhibitors, like abiraterone, apalutamide, darolutamide, and enzalutamide, expanded the array of treatments available for patients with metastatic castration-resistant PC (mCRPC), nonmetastatic CRPC (nmCRPC), and metastatic hormone-sensitive PC (mHSPC, also known as metastatic castration-sensitive PC (mCSPC)).4–6 Enzalutamide was approved in China for the treatment of patients with mCRPC, nmCRPC, and mHSPC in 2019, 2020, and 2024, respectively.7–9 The 2022 guidelines for PC in China 10 recommend apalutamide, enzalutamide, or darolutamide in combination with androgen deprivation therapy for patients with nmCRPC who are at a high risk for metastasis, and abiraterone and enzalutamide are recommended for patients with mCRPC in whom chemotherapy has been ineffective. 10
Seizure has been reported as an adverse reaction with the use of enzalutamide. In phase II,11,12 phase III,13–20 and phase IV21,22 clinical trials, treatment with enzalutamide was assessed for the risk of seizure, with a reported incidence ranging from 0.0% to 1.1%. In most of these trials, patients with risk factors for seizure at baseline were excluded.11,12,14–22 UPWARD, a global, post-marketing, single-arm study in 20 countries (excluding China) to evaluate the incidence of seizure in enzalutamide-treated patients with risk factors for seizure, reported an incidence rate of 1.1%. 21 Additionally, there have been reports of posterior reversible encephalopathy syndrome (PRES) with the use of enzalutamide in post-marketing studies, 23 but no cases have been reported in phase III clinical trials.16,19
In a similar context, the present noninterventional, post-authorization safety study evaluated the risk of seizure in patients with PC treated with enzalutamide or alternative antiandrogens in real-world clinical practice in China. The study also investigated the risk of PRES.
Methods
Study design
This was an observational cohort study using data from electronic health records from three clinical sites in China. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 24
Setting
Data from patients who started enzalutamide or alternative antiandrogens between March 1, 2020 (date of first marketing of enzalutamide in China), and May 31, 2022, and who were eligible after applying inclusion and exclusion criteria were extracted from three sites (site 1: The Second Hospital of Tianjin Medical University in central northern China; site 2: Liaoning Cancer Hospital & Institute in northern China; site 3: Sun Yat-Sen University Cancer Center in southern China).
The index date was defined as the date of the start of enzalutamide or alternative antiandrogen treatment. The baseline period was defined as the 6-month period preceding the index date. The on-treatment period was defined as the period from the index date until the end of use of enzalutamide or alternative antiandrogens. For the on-treatment period, patients were followed up from the index date until the end of enzalutamide or alternative antiandrogen treatment, transfer out of the database, or May 31, 2022, whichever was earliest. The off-treatment period was defined as the period after enzalutamide or alternative antiandrogen treatment had finished. Thus, in this study, the full follow-up period started from the index date and included both on- and off-treatment periods.
Participants
Male patients aged ⩾18 years at index, with at least one diagnosis of histologically confirmed PC, at least one follow-up visit after the index date, and a minimum follow-up period of 1 week post-index were included (Figure 1). Patients who participated in randomized controlled trials between March 1, 2020 and May 31, 2022, or who were treated with enzalutamide or alternative antiandrogens before March 1, 2020, were excluded. The initial analysis dataset contained data from patients who met the inclusion and exclusion criteria.

Study design schematic.
Patients treated with enzalutamide or alternative antiandrogens were categorized as follows:
(1) Patients treated with enzalutamide, with or without prior alternative antiandrogen treatment, were included in the enzalutamide group.
(2) Patients treated with an alternative antiandrogen without any prior use of enzalutamide were included in the alternative antiandrogen group.
(3) If patients initially included in the enzalutamide group discontinued treatment and switched to alternative antiandrogens, their data after the switch were used as a new entry in the alternative antiandrogen group (and treated as an additional independent patient record).
The dataset obtained after including the treatment switches was defined as the overall analysis dataset. Notably, patients who switched from an alternative antiandrogen to enzalutamide were considered only for the enzalutamide group.
Study objectives
The primary objective of this study was to estimate the proportion (%) of patients with PC in the study population who experienced seizure while on enzalutamide treatment, as well as the proportion of patients who experienced seizure while on alternative antiandrogens. The secondary objectives were to estimate the incidence rate of seizure and PRES (based on person-time) in the study population.
Variables
Primary variables included exposure variables (enzalutamide and alternative antiandrogens), outcome variables (seizure and PRES), and risk factors for seizure. Secondary variables included baseline demographic characteristics (age, sex, height, body weight, and smoking and drinking history) and clinical characteristics (Gleason score, clinical stage, tumor node metastasis stage, Eastern Cooperative Oncology Group classification, PC classification, metastasis, metastasis site, and prostate-specific antigen and testosterone levels).
Data sources
Two cohorts (enzalutamide and alternative antiandrogen groups) were identified using electronic medical records from hospitals in China. Basic diagnosis and treatment information was collected from linking admission and discharge records, medical history, prescription records, laboratory examination, and imaging examination databases. Treatment outcomes were captured mainly from admission and discharge records, as well as physician notes.
Statistical analyses
Projected estimates of seizure incidence during the treatment period, with a predefined level of precision, required a sample size of about 350 patients per group. A descriptive statistical analysis was performed on demographic and clinical characteristics and risk factors for seizure in the enzalutamide and alternative antiandrogen groups. Denominators for incidence rates were based on person-time of observation. We estimated seizure incidence based on both on-treatment period and total observation period (i.e., both on- and off-treatment). Continuous variables were summarized as means (with standard deviations (SDs)), medians (with interquartile ranges), as well as minimum and maximum values. Categorical variables were described using frequency and percentage (%). The proportion of missing data was shown, and a missing category was included in the reports for each categorical variable where applicable. No direct comparison was drawn between the cohorts. Subgroup analyses for seizure risk by individual risk factors and key clinical characteristics were conducted but not reported as there were few seizure cases in the overall study population. All statistical analyses were performed using SAS 9.4 software (SAS Institute, Cary, NC, USA).
No imputation was made when the patients’ baseline characteristics or outcome variables were missing, and the proportion of data missing for each variable was summarized as a “missing” category. For seizure risk factors based on medical history, such as history of trauma and history of cerebrovascular accident, a missing value was treated as absence of a risk factor.
Ethics approval
This study was conducted in compliance with the national requirements for ensuring the rights of participants in noninterventional studies in China; in accordance with the protocol, ICH guidelines, applicable regulations, and guidelines governing clinical study conduct; and in compliance with the ethical principles that have their origin in the Declaration of Helsinki.
Results
Overall, 475 patients on enzalutamide and 2977 patients on alternative antiandrogens were screened (Figure 2). A total of 750 patients with histologically confirmed PC were included in the initial analysis dataset; 385 patients received enzalutamide (with or without prior treatment with alternative antiandrogens), while 365 patients received alternative antiandrogens (without prior enzalutamide treatment). During treatment, 10 patients in the enzalutamide group switched from enzalutamide to alternative antiandrogens and were thus also included in the alternative antiandrogen group. Therefore, in the overall analysis dataset, 385 patients were included in the enzalutamide group and 375 (365 + 10), in the alternative antiandrogen group.

Patient flow for enzalutamide and alternative antiandrogen treatment groups.
Patient demographic and clinical characteristics
Demographic and clinical characteristics were generally similar between the treatment groups (Table 1). The mean (SD) age at enrollment for the enzalutamide and alternative antiandrogen groups was 72.3 (9.3) years and 72.4 (8.7) years, respectively (Table 1). In the enzalutamide group, 232 (60.3%) patients reported metastasis, of which 70 (30.2%) and 57 (24.6%) patients had bone and lymph node metastasis, respectively (Table 1). In the alternative antiandrogen group, 213 (58.4%) patients reported metastasis, of which 91 (42.7%) and 49 (23.0%) patients had bone and lymph node metastasis, respectively (Table 1). The median treatment time and median total follow-up time were 77.0 and 85.0 days, respectively, in enzalutamide group, and 115.0 and 196.0 days, respectively, in the alternative antiandrogen group (Table 1).
Demographic and clinical characteristics of participants.
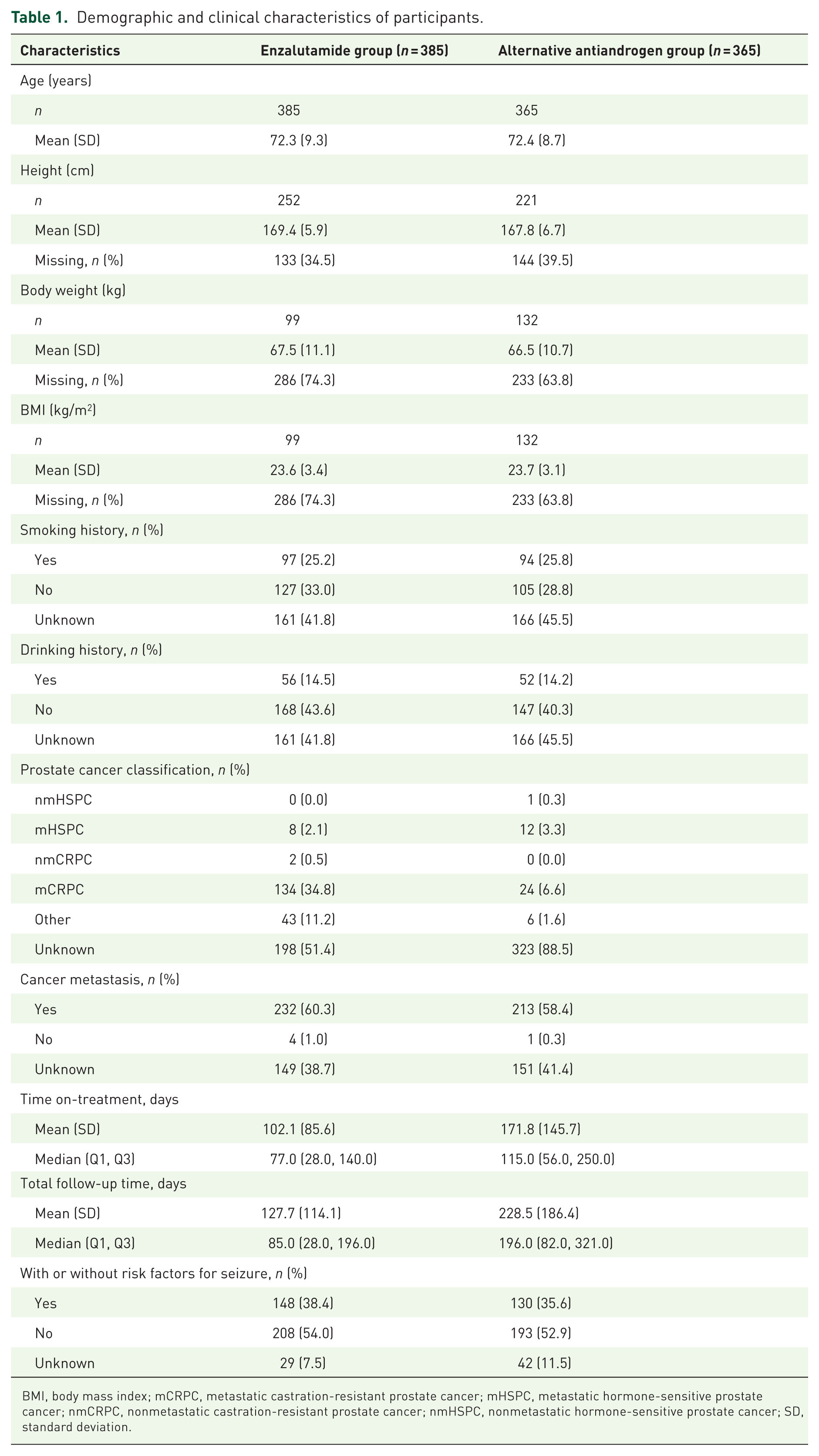
BMI, body mass index; mCRPC, metastatic castration-resistant prostate cancer; mHSPC, metastatic hormone-sensitive prostate cancer; nmCRPC, nonmetastatic castration-resistant prostate cancer; nmHSPC, nonmetastatic hormone-sensitive prostate cancer; SD, standard deviation.
The presence of risk factors for seizure was similar between the groups; 148 (38.4%) patients in the enzalutamide group and 130 (35.6%) patients in the alternative antiandrogen group had at least one risk factor for seizure (Table 1). Risk factors included taking medication that lowers the seizure threshold, history of traumatic brain injury, history of head injury with loss of consciousness, history of cerebral vascular accident, unexplained loss of consciousness in the past 12 months, and history of seizure (Table 2; Supplemental Table 1).
Risk factors for seizures in the study population.
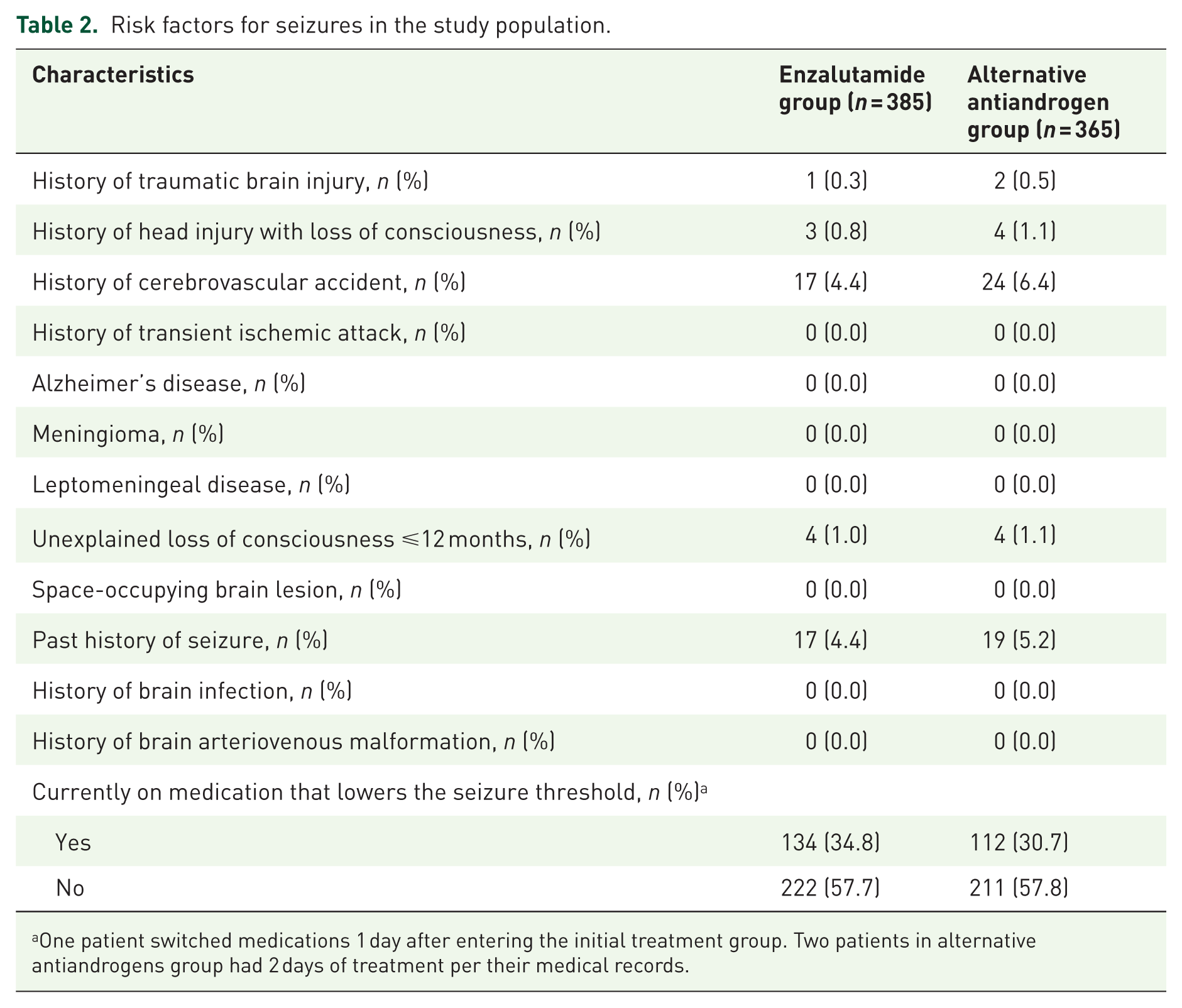
One patient switched medications 1 day after entering the initial treatment group. Two patients in alternative antiandrogens group had 2 days of treatment per their medical records.
Primary endpoint
The primary endpoint, that is, the proportion (%) of patients with PC who experienced seizure, was reported for the overall analysis dataset. During the study period, no event of seizure was reported in the enzalutamide group (95% confidence interval (CI): 0.00, 0.95; Table 3). One event of seizure was confirmed in the alternative antiandrogen group during the treatment period (0.27%, 95% CI: 0.01, 1.48), with no additional events during the off-treatment period (Table 3). The patient who experienced a seizure had no history of treatment with enzalutamide and had risk factors for seizure at baseline (Table 4), including taking medication that lowers the seizure threshold, history of traumatic brain/head injury with loss of consciousness, history of seizure, and receiving antiandrogen treatment for over 4 months.
Proportion of patients who experienced seizure during full follow-up a (overall analysis dataset).

Follow-up period starts from the index date and includes both on-treatment and off-treatment periods. The single case of seizure in the alternative antiandrogen group occurred during the on-treatment period.
CI, confidence interval.
Occurrence of seizure (%) during full follow-up a : subgroup analysis (overall analysis dataset).

Follow-up period starts from the index date and includes both on-treatment and off-treatment periods. The single case of seizure in the alternative antiandrogen group occurred during the on-treatment period.
CI, confidence interval.
Secondary endpoints
In the enzalutamide group, the seizure incidence rates per 100 person-years were 0.00 (95% CI: 0.00, 3.50) and 0.00 (95% CI: 0.00, 2.74) for the on-treatment and full follow-up periods, respectively (Table 5). For the alternative antiandrogen group, the respective incidence rates were 0.58 (95% CI: 0.00, 2.14) and 0.43 (95% CI: 0.00, 1.61; Table 5).
Incidence of seizure during full follow-up. a

Follow-up period starts from the index date and includes both on-treatment and off-treatment periods. The one case in the alternative antiandrogen group occurred during the on-treatment period.
CI, confidence interval.
No cases of PRES were reported in the enzalutamide or alternative antiandrogen groups during the on-treatment or full follow-up periods.
Discussion
This is the first real-world study investigating the risk of seizure in patients with PC treated with enzalutamide or alternative antiandrogens in China. No adverse events of seizure were observed in the enzalutamide group, despite 38.4% of patients having risk factors. Previous global phase II (STRIVE 11 and TERRAIN 12 ), phase III (PREVAIL, 14 AFFIRM, 15 ARCHES, 16 ENZAMET, 17 EMBARK, 18 PROSPER, 19 and others 20 ), and phase IV trials21,22 reported seizure in 0.0%–1.1% of patients who received enzalutamide as monotherapy or combination therapy at a dose of 160 mg/day, which is the approved clinical dose. Although these trials intended to exclude patients with risk factors for seizure, there were cases of undisclosed history of seizure.11,12,14,15
In the present study, patients treated with enzalutamide had a mean age of 72.3 years, which is similar to findings reported in previously published real-world and phase III studies in China.20,25 Further, no cases of PRES were reported among enzalutamide-treated patients, which is in line with previously published phase III trials.16,19
In the alternative antiandrogen group, one confirmed case of seizure was reported during the study period, in a patient with a medical history of seizure who had no reported exposure to enzalutamide. Considering the CIs, the estimated incidence of seizure among patients in the alternative antiandrogen group was consistent with that reported in a real-world cohort database study conducted in the United States (1.8 per 100 person-years; 95% CI: 1.5, 2.1), 26 even though the point estimates were relatively low.
Limitations
As this was a real-world study, methodological limitations such as misclassification bias, unavailability of certain data points, and underestimation of risk factors may exist. Data on seizure occurrence were extracted from the selected hospitals, which could have led to underestimation of seizure occurrence if events occurred outside the hospital setting. Additionally, the median duration of treatment with enzalutamide (77 days) was shorter than the typical treatment duration of 3–4 months, 21 and the median on-treatment and full follow-up times were shorter for the enzalutamide group (77.0 and 85.0 days, respectively) than the alternative antiandrogen group (115.0 and 196.0 days, respectively), which may have introduced further bias in the reporting of seizure. However, measures were taken to minimize the underestimation bias, specifically, data capture from physicians’ medical charts/notes, and review of suspected cases by independent neurology experts.
Conclusion
This real-world safety study in China found that the risk of seizure was low in patients with PC treated with enzalutamide, despite the presence of risk factors for seizure in over one-third of the study population. The risk of seizure was also low in patients treated with alternative antiandrogens.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251391078 – Supplemental material for Risk of seizure in patients with prostate cancer treated with enzalutamide in China: a noninterventional study
Supplemental material, sj-docx-1-taw-10.1177_20420986251391078 for Risk of seizure in patients with prostate cancer treated with enzalutamide in China: a noninterventional study by Yong Wang, Yadong Cui, Yonghong Li, Bin Hu, Xingbo Long, Qingpeng Xie, Noah Jamie Robinson, Ying Zu, Nan Sun, Xiang Qian, Mei Yang, Siyi He, Sida Wei and Yuanjie Niu in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251391078 – Supplemental material for Risk of seizure in patients with prostate cancer treated with enzalutamide in China: a noninterventional study
Supplemental material, sj-docx-2-taw-10.1177_20420986251391078 for Risk of seizure in patients with prostate cancer treated with enzalutamide in China: a noninterventional study by Yong Wang, Yadong Cui, Yonghong Li, Bin Hu, Xingbo Long, Qingpeng Xie, Noah Jamie Robinson, Ying Zu, Nan Sun, Xiang Qian, Mei Yang, Siyi He, Sida Wei and Yuanjie Niu in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
Medical writing, editorial, and graphic design support were provided by Mansi Mehta (PhD), Chrysi Petraki (PhD), Rucha Kurtkoti (MSc), and Samila Sakhabuth from IQVIA, funded by Astellas Pharma Inc.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
