Abstract
Background:
Vancomycin exhibits high pharmacokinetic variability, particularly in hematopoietic stem cell transplantation (HSCT) patients, where augmented renal clearance (ARC) may compromise therapeutic exposure. This variability necessitates individualized dosing strategies to ensure optimal treatment efficacy.
Objective:
To evaluate the pharmacokinetic variability of vancomycin in pediatric and adult patients undergoing HSCT and to assess the association of ARC with serum drug concentrations.
Design:
A retrospective cohort study.
Methods:
We analyzed medical records of post-HSCT patients with febrile neutropenia who received intravenous vancomycin at a tertiary hospital in Lima, Peru. Vancomycin plasma concentrations were assessed at baseline and after dose adjustments. Pharmacokinetic parameters were estimated using Bayesian modeling via PrecisePK software. Regression models were used to evaluate associations between creatinine clearance and vancomycin area under the curve (AUC).
Results:
A total of 40 post-HSCT patients were included, 50% of whom were female. The median age was 17.0 years (interquartile range (IQR): 12.0–39.5), with 25% belonging to the pediatric group. The most frequent disease was B-cell acute lymphoblastic leukemia (47.5%). A significant change was observed in the vancomycin area under the curve (AUC0–24/MIC) before and after dose adjustment (343 mg h/L vs 523 mg h/L; p < 0.001). The median relative dose adjustment percentage of vancomycin was 45% (IQR: 20–73.3). In the multivariate linear regression model, an increase in creatinine clearance was associated with a lower vancomycin AUC (β = −0.67; 95% CI: −1.14 to −0.19). This relationship was maintained after bootstrap resampling with 3000 repetitions (β = −0.67; 95% CI: −1.11 to −0.23). Sepsis was associated with the development of ARC (aRR: 1.378; 95% CI: 1.164–1.631), and as age increased, the risk of ARC decreased (aRR: 0.991; 95% CI: 0.99–0.996).
Conclusion:
Most post-HSCT patients had subtherapeutic vancomycin levels at the beginning of treatment, which were optimized through individualized adjustments. The pharmacokinetic variability associated with ARC in young patients and those with sepsis supports the use of AUC-based therapeutic drug monitoring over trough concentration monitoring. These findings also highlight the importance of early renal function monitoring in patients treated with HSCT.
Objective: To evaluate the pharmacokinetic variability of vancomycin in pediatric and adult patients undergoing hematopoietic stem cell transplantation (HSCT), and to assess the association of augmented renal clearance (ARC) with serum drug concentrations.
Methods: A retrospective cohort study was conducted at a hospital center in Lima, Peru. Post-HSCT patients with febrile neutropenia who received vancomycin were included. Vancomycin levels were measured at the beginning of treatment and after dose adjustment. Dose adjustment parameters were estimated using the PrecisePK software.
Results: A total of 40 post-HSCT patients were included, 50% of whom were female. The median age was 17.0 years (IQR: 12.0–39.5), with 25% belonging to the pediatric group. The most frequent disease was B-cell acute lymphoblastic leukemia (47.5%). A significant change was observed in the vancomycin area under the curve (AUC) before and after dose adjustment (343 vs 523; p < 0.001). The median relative dose adjustment percentage of vancomycin was 45% (IQR: 20%–73.3%). In the multivariate linear regression model, an increase in creatinine clearance was associated with a lower vancomycin AUC (β = −0.67; 95% CI: −1.14 to −0.19). This relationship was maintained after bootstrap resampling with 3000 repetitions (β = −0.67; 95% CI: −1.11 to −0.23). Sepsis was associated with the development of ARC (aRR: 1.378; 95% CI: 1.164 to 1.631), and as age increased, the risk of ARC decreased (aRR: 0.991; 95% CI: 0.99 to 0.996).
Conclusion: Most post-HSCT patients had subtherapeutic vancomycin levels at the beginning of treatment, which were optimized through individualized adjustments. The pharmacokinetic variability associated with ARC in young patients and those with sepsis supports the use of AUC-based therapeutic drug monitoring over trough concentration monitoring. These findings also highlight the importance of early renal function monitoring in patients treated with HSCT.
Keywords
Introduction
Vancomycin is a hydrophilic glycopeptide antibiotic used to treat infections caused by resistant Gram-positive bacteria, especially methicillin-resistant Staphylococcus aureus (MRSA), and is widely used in immunocompromised patients when beta-lactams are ineffective.1–3 Its therapeutic effectiveness is closely linked to its pharmacokinetic/pharmacodynamic profile, with the AUC0–24/MIC ratio being the main predictor of clinical efficacy.4,5 The Infectious Diseases Society of America recommends achieving a vancomycin area under the curve (AUC) between 400 and 600 mg h/L to maximize antimicrobial activity and minimize the risk of toxicity.6,7 However, vancomycin shows high intra- and interindividual variability in its pharmacokinetic parameters, such as clearance (CL) and volume of distribution (Vd), especially in special populations such as pediatric and oncologic patients.8,9 This variability complicates the achievement of adequate therapeutic plasma concentrations and may lead to both therapeutic failure and nephrotoxicity. 10 In view of these limitations, the use of AUC-based therapeutic drug monitoring (TDM) has been promoted, through precision dosing strategies based on population pharmacokinetic models, to guide accurate dosing and improve clinical outcomes.11,12
Hematopoietic stem cell transplantation (HSCT) involves the infusion of hematopoietic cells with the goal of reconstituting hematopoiesis and restoring immune function in patients with both malignant and non-malignant hematological diseases. 13 The pre-transplant phase, known as conditioning therapy, consists of administering chemotherapy and/or radiotherapy to eliminate malignant cells and suppress the immune system, thereby facilitating graft acceptance. 14 Subsequently, after the infusion of hematopoietic cells, the early post-transplant period begins, characterized by prolonged immunosuppression lasting until engraftment, generally between days 10 and 30. 14 This phase is marked by profound neutropenia, disruption of mucocutaneous barriers (mucositis), and immune dysfunction, which significantly increases the risk of severe infections. 15 In post-HSCT patients, febrile neutropenia has an incidence of approximately 80% and is often associated with infections caused by both Gram-negative and Gram-positive bacteria, requiring immediate empirical antimicrobial treatment due to the high risk of morbidity and mortality. 16 The empirical use of vancomycin in this context is common since many HSCT patients present multiple risk factors for Gram-positive infections (mucositis, use of central venous catheters, skin infections, and pneumonia). However, its therapeutic management is complex due to high pharmacokinetic variability, hyperfiltration secondary to hyperhydration, and particularly the development of augmented renal clearance (ARC).
ARC is defined as a creatinine clearance (CrCl) greater than 130 mL/min/1.73 m2, and it can affect up to 65% of critically ill patients. 17 This phenomenon accelerates the elimination of vancomycin and compromises its efficacy. 18 Other factors such as hypoalbuminemia 19 and alterations in the volume of distribution further aggravate this condition, making it essential to measure vancomycin serum concentrations for the design of individualized dosing regimens. In Latin America, and particularly in Peru, there is a marked lack of studies evaluating the pharmacokinetic behavior of vancomycin in HSCT patients. Current recommendations are based on extrapolations from non-representative populations. 20 Although ARC is suspected to be a relevant factor in drug exposure variability, its specific impact in this population remains clearly undefined. Therefore, the objective of this research was to evaluate the pharmacokinetic variability of vancomycin in adult and pediatric post-HSCT patients, as well as to determine the effect of ARC on the drug’s plasma concentrations.
Methods
Study design and setting
A retrospective cohort study was conducted, collecting data from pediatric, adolescent, and adult patients who underwent autologous and allogeneic HSCT at the Edgardo Rebagliati Martins National Hospital (HNERM) in Peru between May 2023 and April 2025. HNERM is a specialized and high-complexity hospital (level III-2) belonging to the Social Health Insurance system (EsSalud). HNERM serves a population of two million insured individuals nationwide and has more than 1600 hospital beds. 21 Within this institution, the Department of Hematology has established itself as a national and international reference center in the field of HSCT, having performed over 1600 procedures, including transplants from unrelated donors, and has been recognized by the National Bone Marrow Donor Program as a globally accredited transplant center. 22 The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement guidelines. 23 The completed STROBE checklist is provided as Supplemental Material 4.
Study population
Patients were included in the cohort if they met the following criteria: (i) had received autologous or allogeneic HSCT at HNERM during the study period; (ii) had received at least three doses of intravenous vancomycin after the HSCT procedure; and (iii) had available blood biochemistry data, including serum creatinine levels, recorded within 48 h prior to the initial sample collection for TDM. Given the retrospective design of this study, all eligible patients who met these criteria during the defined period were exhaustively included, with no sampling applied. Therefore, this was a retrospective census cohort study, and no a priori sample size calculation was necessary.
Of the 267 patients who underwent HSCT between May 2023 and April 2025, 227 were excluded because they did not receive vancomycin therapy, either due to the absence of infectious complications or because they were treated solely with β-lactam antibiotics until discharge. Additionally, 15 patients who received vancomycin were excluded due to a temporary shortage of the laboratory reagent required for serum concentration measurement. Consequently, 40 patients met the eligibility criteria and were included in the analysis.
To assess the representativeness of the included sample and explore potential selection bias, a comparative analysis of baseline characteristics between included and excluded patients was performed. This information is presented in Supplemental Table 1.
The study population was divided into two age groups for analysis: a pediatric group, consisting of patients aged 2–12 years, and an adolescent-adult group, ranging from 13 to 70 years, 24 with the aim of evaluating pharmacokinetic differences between the two age groups. Given that few patients met the inclusion criteria during the study period, all eligible patients were included, and a post hoc statistical power calculation was performed. To calculate the statistical power, the “pairedmeans” command in Stata version 17.0 (StataCorp LLC, College Station, Texas, USA) was used. A paired means comparison was considered with a sample of 40 patients. Reference values were taken from the study by Sahraei et al., 25 which reported a baseline vancomycin AUC mean of 397.9 (SD: 76.02) and a post-therapeutic adjustment mean of 611.92 (SD: 148.01). Assuming a correlation of 0.5 between measurements and a significance level of 5%, the estimated statistical power was 99.9%. This indicates that the sample size was sufficient to detect a clinically meaningful difference in AUC, reinforcing the robustness of the primary pharmacokinetic findings.
During data collection and verification, no missing values were identified in clinical, biochemical, or pharmacokinetic variables; therefore, the statistical analysis was performed using the entirety of the available records.
Procedures
After ethics committee approval, access was granted to the list of patients who underwent HSCT during the study period. Data collection was carried out by W.B.-V., K.S.-L., A.W.-C, M.F.-P., L.A.-T., and A.L.-M. through a systematic review of the electronic medical records available in the EsSalud Smart Health Services system. The pharmacokinetic parameters of vancomycin were supplemented with data from the clinical pharmacy unit. The data were stored in a digital database created in Microsoft Excel with password-restricted access. After data extraction, researcher W.B.-V. conducted a first quality control check based on verifying whether the extracted data matched the medical records; if discrepancies were found, the data were corrected. The second quality control check was performed by researcher C.C.-C., who focused on identifying implausible data; if identified, the clinical records were reviewed to correct the error.
Vancomycin dosing and monitoring strategy
Initial vancomycin dosing was established according to the institutional clinical practice guideline. In adults and adolescents, doses of 1 g every 12 or 8 h were administered, based on clinical judgment. In certain cases, loading doses of 1.5 g were used, depending on individual patient assessment. In the pediatric population, vancomycin was administered at 40–60 mg/kg/day, 26 divided into four doses, without the use of loading doses, as the guideline does not specify criteria for their use, leaving the decision to the treating physician.
Therapeutic monitoring was carried out through the determination of minimum (Cmin) and maximum (Cmax) serum concentrations. For both adults and pediatric patients, Cmin and Cmax samples were obtained before the administration of the fourth dose, ensuring that steady-state conditions had been reached. Specifically, Cmin was measured within 30 min prior to the next scheduled dose, and Cmax was collected 1 h after the end of a 60-min infusion, during the post-distribution phase. Although Bayesian pharmacokinetic software can reliably estimate AUC and individual parameters even with early sampling (e.g., after the first or second dose), the study design prioritized pharmacokinetic stability by systematically waiting until steady-state was achieved. 27
Samples were analyzed at the central laboratory using the Emit® 2000 Vancomycin Assay (Siemens Healthcare Diagnostics Inc., Newark, DE, USA), a homogeneous enzyme immunoassay based on the competition between drug in the sample and drug labeled with glucose-6-phosphate dehydrogenase, performed on the AU480® Clinical Chemistry System (Beckman Coulter Inc., Brea, California, USA). The assay quantifies total vancomycin (protein-bound and unbound) concentrations in serum or plasma. The lower limit of quantification was 2.0 µg/mL. The inter-run coefficient of variation ranged from 2.5% to 3.2% for vancomycin concentrations of 7.3, 18.7, 30.2, and 39.4 µg/mL, respectively, based on internal quality control data. 28 Quality control procedures were performed in compliance with ISO 15189 accreditation standards, and results were regularly validated through participation in external proficiency testing programs. 29 The dosing data and plasma concentrations, extracted from the electronic medical record, were used to estimate individual pharmacokinetic parameters through Bayesian modeling with the PrecisePK software (PrecisePK Inc., San Diego, CA, USA). 30 PrecisePK has been part of the routine clinical practice at HNERM since 2022, implemented by the Clinical Pharmacokinetics Unit under the Department of Pharmacy. It is used as a standard tool for dose adjustment and therapeutic monitoring of vancomycin and other critical drugs, especially in high-complexity cases such as post-HSCT patients. For adults, population pharmacokinetic models by Rodvold et al., 31 Adane et al., 32 and Crass et al. 33 were used; for pediatric patients, models by Le et al. 34 and Le et al., 35 both integrated into the platform, were employed. This estimation allowed the evaluation of the adequacy of the initial regimen and the individualized adjustment of doses.
Definition of variables
Outcome variable
The primary outcome variable was the estimated AUC during the first 48 h of treatment. To determine the initial pharmacokinetics of vancomycin, Cmin and Cmax were measured within the same period. These concentrations were processed using the PrecisePK software, which estimates individual AUC and performs dose optimization simulations, using mono- and bicompartmental population pharmacokinetic models with first-order elimination kinetics.32–35 Based on the AUC0–24/MIC results, treatment adjustment was carried out, which could involve an increase or decrease in the dose and/or administration frequency, as indicated by the predictive model. Subsequently, a second measurement of Cmin and Cmax was performed 24–48 h after the adjustment, in order to reevaluate pharmacokinetic parameters and verify whether the achieved AUC0–24/MIC was within the recommended therapeutic range of 400–600 µg h/mL.
Exposure variable
We considered CrCl as the main factor involved in the pharmacokinetic variability of vancomycin. 25 CrCl was estimated in the adult population (⩾18 years) using the Cockcroft-Gault formula: CrCl = (140 − age) × weight (kg) ÷ 72 ÷ serum creatinine (mg/dL); if female, ×0.85. 36 For the pediatric population (<18 years), the glomerular filtration rate was estimated using the Schwartz formula: GFR = height (cm) × k ÷ serum creatinine (mg/dL), where k is a coefficient adjusted by age and sex.37,38 Pediatric, adolescent, and adult patients with an estimated CrCl ⩾130 mL/min were classified as having ARC. 38
Considering that Cockcroft-Gault equation may overestimate renal function in patients with low muscle mass or low serum creatinine, especially in cachectic or edematous individuals, a sensitivity analysis was conducted. In this additional analysis, renal clearance in adults was recalculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) formula, while pediatric patients continued to be assessed using the Schwartz equation. This approach aimed to evaluate the robustness of ARC classification across estimation methods. The results of this comparison are detailed in Supplemental Table 3.
In addition, to describe the population, we collected demographic data (age, sex, body mass index, weight, height), clinical data (type of hematologic disease, type of transplant, serum creatinine and albumin levels, leukocytes, neutrophils, hemoglobin, platelets), and other vancomycin pharmacological data (loading dose, initial dose, infusion time, dosing frequency, minimum and maximum serum concentrations, treatment duration, dose adjustments, half-life, volume of distribution, AUC0–24/MIC). Clinical outcomes were also analyzed, such as infection resolution, occurrence of nephrotoxicity (defined as an increase in creatinine ⩾0.3 mg/dL or ⩾50% from baseline), 39 and total duration of vancomycin therapy.
Statistical analysis
The analysis was conducted using the Stata V17 statistical software. Variables were described using absolute frequencies, percentages, measures of central tendency, and dispersion. For the bivariate analysis between the exposure variable (ARC) and categorical variables, the chi-square test (χ²) or Fisher’s exact test was used, as appropriate. For numerical variables, the Mann-Whitney U test was employed due to their non-parametric distribution. Differences in pharmacokinetic parameters (volume of distribution, clearance, half-life, AUC, daily dose, trough concentration) before and after treatment adjustment were evaluated in both pediatric and adult patients. For these analyses, the Wilcoxon signed-rank test was used.
Additionally, crude and adjusted linear regression analyses were performed to explore the relationship between estimated CrCl and the vancomycin AUC achieved. The multivariate model was adjusted for hydration, sex, age, and BMI. To further enhance the precision of our results, we adjusted the model using bootstrap resampling with 3000 repetitions. Finally, we explored factors associated with renal clearance using crude and adjusted Poisson regression with robust variance, from which we obtained the relative risk (RR) and its corresponding 95% confidence intervals (95% CI).
Ethical considerations
This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki. The protocol was reviewed and approved by the Research Ethics Committee of the Edgardo Rebagliati Martins National Hospital (code: 036-CE-GHNERM-GRPR-ESSALUD-2025). Due to its retrospective and observational nature, informed consent was not required. Confidentiality and anonymity of the data were ensured at all stages of the study, in compliance with current ethical and legal regulations. All data were extracted from the institutional electronic medical records system and stored in a secure, password-protected digital database. Patient records were anonymized by assigning unique identification codes, and no names or personally identifiable information were used during data analysis. Access to the dataset was restricted to study investigators, and two independent quality control steps were performed to ensure data integrity while maintaining patient privacy.
Results
Study population and characteristics
No missing data were found in the analyzed variables; all included patients had complete information for the variables of interest, allowing the analysis to be conducted without requiring imputation or additional exclusions.
A total of 267 patients were evaluated, and 227 were excluded for not having received vancomycin or for not having serum concentration measurements performed, due to a reagent shortage at the hospital during part of the study period (Figure 1). Of the 40 patients included, 50% were female, with a median age of 17 years (interquartile range (IQR): 12–39.5). The most common transplant type was allogeneic (80.0%), and the primary cause for transplantation was B-cell acute lymphoblastic leukemia (47.5%). ARC developed in 77.5% of the patients during hospitalization, being more frequent in the pediatric population (90%). Regarding complications, 100% of patients presented with febrile neutropenia, and the most frequent infection focus was skin and soft tissue (42.5%). Survival until hospital discharge occurred in 95% of patients, with a mean length of stay of 29.5 days (IQR: 23.0–38.5), which was longer among the pediatric population with a median of 40.0 days (IQR: 29.0–45.0; Table 1).

Flow diagram of patient selection.
Baseline characteristics of patients and differences between pediatric and adolescent/adult groups.

ALL, acute lymphoblastic leukemia; AML, acute myeloid leukemia; ARC, augmented renal clearance; B-cell, B lymphocyte; T-cell, T lymphocyte; BMI, body mass index; CRP, C-reactive protein; HSCT, hematopoietic stem cell transplantation; IQR, interquartile range.
No clinically significant differences were observed between included (n = 40) and excluded patients (n = 227) regarding age, sex, hematologic diagnosis, or survival at hospital discharge. However, a higher proportion of allogeneic transplants was noted in the included group (77.5% vs 55.1%; p = 0.012), likely reflecting a higher risk of infectious complications in these patients and, consequently, a greater clinical indication for empirical vancomycin use. Full baseline comparisons are provided in Supplemental Table 1.
Pharmacokinetic variability of vancomycin
The relative dose adjustment percentage for vancomycin was 45% (IQR: 20%–73.3%) among patients. This represents the percentage increase from the initial empirical dose to the adjusted dose needed to reach target AUC0–24/MIC values, calculated as: [(Adjusted dose − Initial dose)/Initial dose] × 100. This interindividual variability in dose requirement is illustrated in Figure 2(a). A significant improvement in pharmacokinetic exposure was observed following dose adjustment. The median vancomycin AUC0–24/MIC increased from 343 mg h/L (IQR: 295–392) to 523 mg h/L (IQR: 493–576.8), as shown in Figure 2(c) (p < 0.001), confirming that individualized dose optimization effectively improved target attainment.
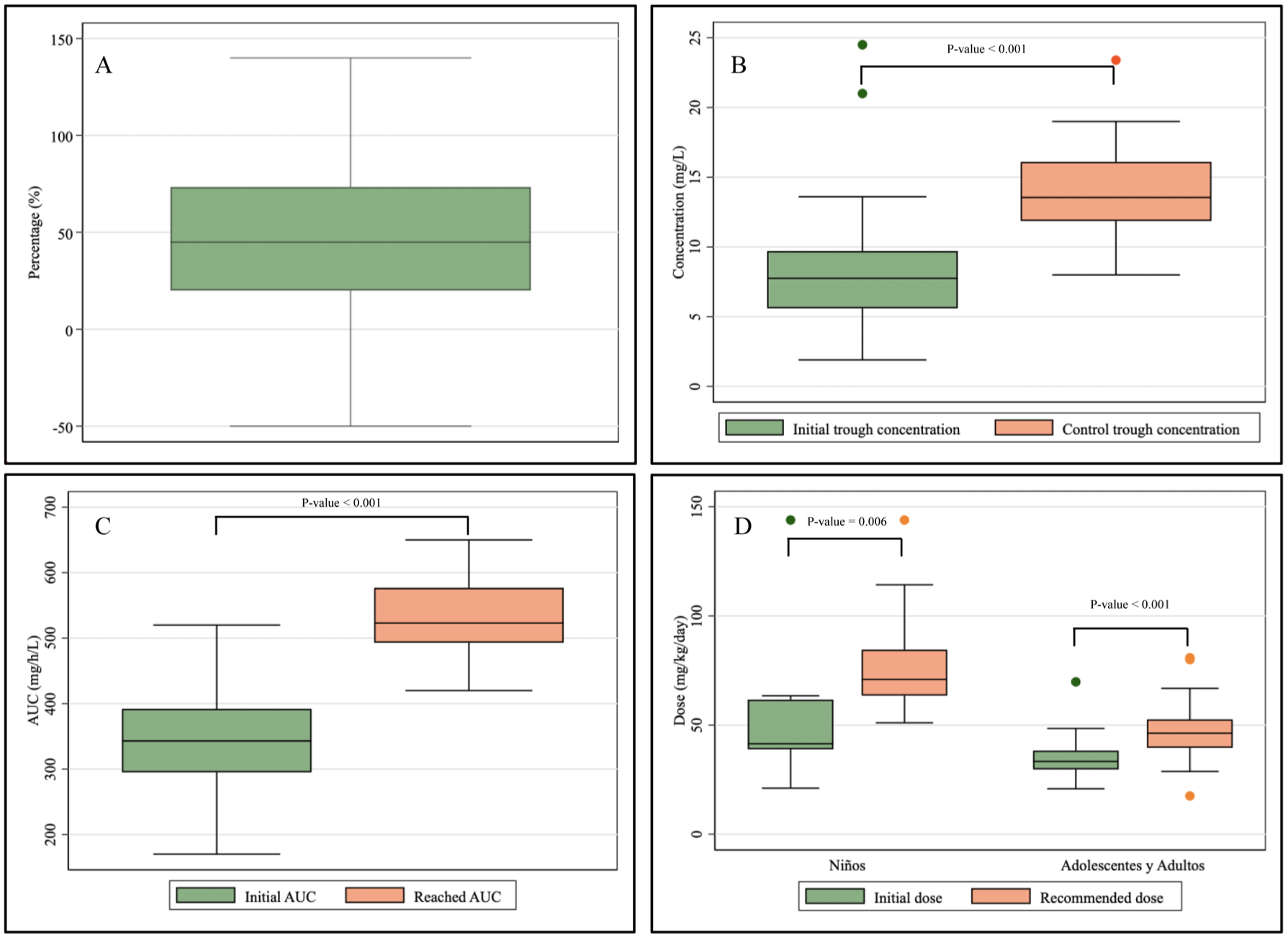
Vancomycin pharmacokinetics before and after treatment adjustment.
Similarly, a marked increase in trough serum concentrations (Cmin) was observed between the initial and follow-up evaluations (Figure 2(b)), with median values rising significantly (p < 0.001), further supporting enhanced systemic exposure after pharmacokinetic titration. In the subgroup analysis by age, Figure 2(d) shows that vancomycin dosing was significantly higher in the pediatric population (median 70.9 mg/kg/day; IQR: 63.5–84.5) compared to adolescents and adults (median 46.3 mg/kg/day; IQR: 39.6–52.6; p < 0.001), reflecting the known differences in clearance and renal function between these groups.
Regarding the pharmacokinetic characteristics of vancomycin, the median volume of distribution was 39.5 L (IQR: 24.6–48.7), clearance was 4.73 L/h/kg (IQR: 3.4–6.8), and half-life was 4.55 h (IQR: 3.3–6.34). The half-life was significantly longer in patients without ARC (7.4 h vs 4 h; p = 0.001). In the pediatric population, vancomycin clearance in L/h/kg did not show a statistically significant difference between patients with ARC and those without ARC (3.3 vs 2.8; p = 0.384; Table 2). suggesting that other age-related factors may influence variability in clearance among children.
Pharmacokinetic characteristics of vancomycin according to the presence of ARC.

ACR, augmented renal clearance; h, hours.
Factors associated with ARC
The median age was lower (median 16; IQR: 8–36) in patients who developed ARC, and ARC was more frequent in patients who underwent allogeneic transplantation (84.4%). In the multivariate Poisson regression model with robust variance, the development of sepsis was associated with the occurrence of ARC (Adjusted RR: 1.378; 95% CI: 1.164–1.631). Additionally, the risk of developing ARC was higher at younger ages, decreasing by 0.9% with each additional year of age (Adjusted RR: 0.991; 95% CI: 0.99–0.996; Table 3).
Factors associated with the development of ARC.

ARC, augmented renal clearance; BMI, body mass index; CI, confidence interval; IQR, interquartile range; Ref, Reference group; RR, relative risk.
Figure 3 shows the linear regression analysis between creatinine renal clearance and the vancomycin AUC. In the crude analysis, it was observed that the vancomycin AUC decreases as renal clearance increases (coefficient B = −0.56; 95% CI: −0.97 to −0.15). This negative association persisted after adjusting for hydration, sex, age, and BMI (B = −0.67; 95% CI: −1.14 to −0.19). Finally, after applying a bootstrap with 3000 repetitions, the relationship remained (B = −0.67; 95% CI: −1.11 to −0.23).

Linear regression between renal clearance and achieved AUC.
Discussion
In this retrospective cohort study analyzing 40 patients undergoing HSCT, it was observed that (1) there is a significant variability in the pharmacokinetic parameters of vancomycin before and after therapeutic titration. (2) The ACR is associated with a significant decrease in the vancomycin AUC ratio, suggesting that the increased renal elimination of the drug reduces its plasma concentration. (3) Age and BMI were associated with the development of ARC, with a higher incidence observed in young and underweight individuals.
A total of 77.5% of post-HSCT patients developed ARC, with the frequency rising to 90% in the pediatric population. This condition may have significantly influenced the pharmacokinetics of vancomycin, leading to reduced systemic exposure as measured by AUC. Vancomycin exhibits substantial pharmacokinetic differences between pediatric and adult populations, particularly regarding drug clearance (CL) and Vd. In children older than 1 month, weight-normalized clearance frequently ranges from 0.12 to 0.16 L/h/kg, which is approximately 2–3 times higher than typical adult values (0.05–0.08 L/h/kg), due to increased glomerular filtration per kilogram and higher renal functional reserve (RFR) in pediatric patients.40,41 Consequently, pediatric regimens commonly employ total daily doses of 60–80 mg/kg/day to reach AUC/Minimum Inhibitory Concentration ⩾ 400 mg h/L, compared to 30–40 mg/kg/day in adults. 42 In contrast, neonates particularly preterm infants demonstrate lower clearance (0.05–0.06 L/h/kg) and larger Vd (0.5–0.6 L/kg), with elimination half-lives extending up to 8–10 h, reflecting renal immaturity and higher total body water. 43
These pharmacokinetic disparities become more pronounced in the context of ARC, increasingly recognized in critically ill or immunocompromised pediatric populations, with prevalence rates up to 80%. 37 Under ARC, vancomycin clearance can nearly double, reducing half-life to <4 h, resulting in subtherapeutic exposure when standard dosing is used. Although ARC is also present in young adults with hyperdynamic circulation, the impact is particularly marked in pediatric patients due to their greater RFR. 38 These findings support precision dosing based on population pharmacokinetic models and AUC-guided TDM, especially in pediatric HSCT recipients. Clinical pharmacists are key to guiding individualized dose adjustments and ensuring timely monitoring to optimize treatment efficacy and prevent therapeutic failure.
The impact of subtherapeutic vancomycin exposure in HSCT recipients is clinically significant given their profound immunosuppression due to neutropenia, mucosal damage, and delayed immune recovery. These patients lack the physiological reserve to control infections when antibiotic exposure is insufficient. Studies have shown that an AUC/MIC ratio below 400 is associated with treatment failure, prolonged bacteremia, and increased risk of sepsis, particularly in pediatric and neutropenic adults.34,44 Moreover, ARC, a common phenomenon in this population, can markedly increase vancomycin clearance, leading to underexposure even with standard dosing. Inadequate vancomycin levels may also exacerbate systemic inflammation and potentially trigger complications such as acute graft-versus-host disease in allogeneic HSCT. 45 Therefore, achieving and maintaining therapeutic AUC is essential to ensure efficacy, minimize resistance, and improve outcomes. AUC-guided dosing is particularly valuable in this context and should be prioritized in clinical management.
Although wide variability was observed among patients, after adjusting the dosage through pharmacokinetic optimization, the vancomycin AUC increased significantly from 343 mg h/L (suboptimal range) to 523 mg h/L (optimal range). These findings are consistent with those reported by Shimamoto et al. 46 who documented the need to increase the initial vancomycin dose by 35%–65% in children with ARC post-HSCT to achieve the therapeutic AUC/MIC ⩾400 target, and with Wang et al. 37 who identified trough concentrations <5 mg/L in 45% of pediatric patients after receiving 40 mg/kg/day of vancomycin. The decrease in vancomycin concentration is attributed to renal hyperfiltration associated with age and low BMI. It is hypothesized that the development of ARC could be linked to a systemic inflammatory response state, such as that induced by sepsis, 47 a condition that in our analysis was significantly associated with the risk of ARC. This inflammatory response is characterized by the release of pro-inflammatory cytokines such as IL-6, TNF-α, and IL-1β, 48 which promote systemic vasodilation, increased cardiac output, and renal plasma flow. 49 In pediatric patients, this condition may be amplified by a greater RFR, mediated by pathways such as nitric oxide, cyclic guanosine monophosphate, and prostaglandins, which respond strongly to stimuli like fluid therapy. 50 These hemodynamic changes favor an increase in GFR, accelerating the elimination of drugs like vancomycin.
The inverse association between ARC and vancomycin AUC observed in this study aligns with findings reported by Bury et al. 51 and Bauters et al. 52 who described a significant increase in vancomycin clearance in neutropenic patients with ARC, accompanied by a reduction in half-life and suboptimal trough concentrations. In the current cohort, a decrease in half-life from 7.4 to 4.0 h was observed in the presence of ARC, as well as significantly higher clearance in adolescents and adults (6.2 L/h/kg vs 4.5 L/h/kg; p = 0.029). Similar findings were described by Izumisawa et al., 53 who indicated that ARC is associated with lower systemic exposure, shortened half-life, and a reduced probability of achieving pharmacokinetic/pharmacodynamic targets with standard dosing regimens. This phenomenon can be explained by the fact that vancomycin is a hydrophilic antibiotic with low molecular weight, whose elimination depends almost exclusively on glomerular filtration. Since it does not undergo significant hepatic metabolism or relevant tubular reabsorption, increases in glomerular filtration rate lead to accelerated drug elimination, limiting the time during which plasma concentrations remain above the MIC. This negatively impacts attainment of the pharmacokinetic/pharmacodynamic index AUC/MIC, considered the main predictor of clinical efficacy. 54
Finally, patient age was the main determinant of ARC development in this cohort, a finding consistent with studies by Shimamoto et al., 46 Wang et al., 37 , Zhao et al., 55 Bauters et al., 52 and Nelson et al., 56 who identified a higher incidence of ARC in children under 6 years of age with hematological and oncological diseases. Additionally, in our multivariate model, sepsis was independently associated with a higher risk of ARC, reinforcing the hypothesis that the systemic inflammatory state frequently observed in patients with febrile neutropenia may contribute to hyperfiltration. On the other hand, variables such as BMI, 57 hydration status, and type of transplant did not show a statistically significant association with the presence of ARC. Although these factors have been proposed as possible contributors in other populations, our data suggest that their impact may be less significant in the pediatric oncohematologic group or may be masked by other more relevant pathophysiological determinants, such as age and underlying disease.
Implications
The findings of this study highlight the importance of early and accurate assessment of renal function in patients undergoing HSCT, particularly through the estimation of GFR, in order to timely identify ARC. The presence of this condition significantly alters vancomycin pharmacokinetics, reducing its systemic exposure and compromising its clinical efficacy during critical episodes such as febrile neutropenia. Therefore, it is recommended that initial vancomycin dosing be based on precision dosing models that incorporate individual variables such as weight, height, and renal function, rather than relying on empirical methods or traditional nomograms. In this context, the role of the clinical pharmacist is crucial to ensure effective therapeutic optimization through the interpretation of pharmacokinetic parameters, the use of pharmacokinetic tools, and the recommendation of individualized adjustments. Their integration into multidisciplinary teams strengthens a personalized and evidence-based approach, especially in immunocompromised patients and in resource-limited settings.
Limitations
This is the first study to analyze vancomycin pharmacokinetics in HSCT patients in Latin America and specifically in Peru, providing valuable local evidence in a clinically underrepresented context. The application of Bayesian modeling to estimate individual parameters, the use of real-world clinical practice data, and the inclusion of both pediatric and adult populations with age stratification are relevant methodological strengths.
Among the main limitations of the study is its observational and retrospective design, which prevents the establishment of causal relationships and complicates the control of confounding factors. The small sample size and the inclusion of both pediatric and adult patients introduce clinical heterogeneity, limiting the generalizability of the findings. Additionally, the absence of key clinical variables, such as long-term mortality or disease progression, restricts the comprehensive assessment of therapeutic impact. Since the cohort comes from a single high-complexity hospital with access to advanced tools, extrapolation of the results to other settings should be done with caution. Prospective and multicenter studies are needed.
Furthermore, the heterogeneity in clinical conditions across pediatric and adult patients may have introduced variability in pharmacokinetic responses and ARC prevalence, which should be interpreted cautiously despite the stratified analysis performed.
Another important consideration is the method used to estimate renal function. While the Cockcroft-Gault formula is widely used in clinical pharmacokinetics, it may overestimate CrCl in patients with low muscle mass or low serum creatinine, such as those with cachexia or edema frequent scenarios in post-HSCT patients. To address this concern, we conducted a sensitivity analysis using the CKD-EPI equation in adults, which confirmed the robustness of our ARC classification. Although direct 24-h urinary CrCl is the most accurate method for assessing glomerular filtration, it was not routinely performed during the study period as it is not standard practice in our institution.
However, our findings have prompted a quality improvement initiative aimed at incorporating routine 24-h urinary clearance assessments and pharmacokinetic-guided dosing into standard care protocols, aligning with international recommendations for TDM in high-risk populations.
Conclusion
This study demonstrates marked variability in vancomycin pharmacokinetic parameters before and after individualized adjustment, highlighting the need for personalized dosing strategies in patients undergoing HSCT. It was shown that ARC negatively affects systemic drug exposure, significantly reducing AUC and thus compromising the achievement of pharmacodynamic targets. Furthermore, age and sepsis were identified as the main factors associated with the development of ARC, with pediatric patients being the most affected. This underscores the importance of considering age as a critical variable when estimating renal function and determining the initial dose, prioritizing AUC-based therapeutic monitoring to ensure safe and effective therapy.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251384239 – Supplemental material for Pharmacokinetic variability of vancomycin in patients undergoing hematopoietic stem cell transplantation: a retrospective cohort study in Peru
Supplemental material, sj-docx-1-taw-10.1177_20420986251384239 for Pharmacokinetic variability of vancomycin in patients undergoing hematopoietic stem cell transplantation: a retrospective cohort study in Peru by Wilder Bolaños-Vargas, Cesar Copaja-Corzo, Alfredo Wong-Chang, Karel Santamaria-Leandro, Liz Aliaga-Tabraj, Manuel Fernandez-Puycan and Arturo Yoshimar Luque-Mamani in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251384239 – Supplemental material for Pharmacokinetic variability of vancomycin in patients undergoing hematopoietic stem cell transplantation: a retrospective cohort study in Peru
Supplemental material, sj-docx-2-taw-10.1177_20420986251384239 for Pharmacokinetic variability of vancomycin in patients undergoing hematopoietic stem cell transplantation: a retrospective cohort study in Peru by Wilder Bolaños-Vargas, Cesar Copaja-Corzo, Alfredo Wong-Chang, Karel Santamaria-Leandro, Liz Aliaga-Tabraj, Manuel Fernandez-Puycan and Arturo Yoshimar Luque-Mamani in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors thank the Clinical Pharmacy Unit and the Department of Hematology at Edgardo Rebagliati Martins National Hospital for their support in accessing clinical data and facilitating the implementation of pharmacokinetic monitoring systems.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
