Abstract
Background:
Dexmedetomidine may expedite recovery from acute kidney injury (AKI) in critically ill adults.
Methods:
This study utilizes data from the fourth edition of the Medical Information Mart for Intensive Care (MIMIC-IV). Adult patients admitted to Beth Israel Deaconess Medical Center in Boston, Massachusetts, between 2008 and 2019 with AKI and who have at least two serum creatinine values recorded in the MIMIC-IV database meet study inclusion criteria. The primary outcome measure is the time (days) from diagnosis of AKI to recovery. Secondary outcome measures are hospital and ICU length of stay (LOS) as well as in-hospital mortality.
Results:
A total of 1893 patients are included in this study. While 293 patients received dexmedetomidine, 1600 patients did not receive dexmedetomidine. Treatment with dexmedetomidine is associated with a 53.7% (95% CI: 46.8%–59.8%) decrease in the risk of recovery from AKI, on average, and this value is statistically significant (
Conclusion:
Dexmedetomidine may be associated with slower recovery from AKI in critically ill adults. The prolonged hospital and ICU LOS associated with dexmedetomidine may be related to reduced mortality, but these results require additional investigation. These exploratory results warrant further investigation to better understand the clinical implications of dexmedetomidine exposure in the setting of AKI.
Plain language summary
Patients who receive dexmedetomidine may recover from Acute Kidney Injury (AKI) more slowly than patients who do not receive dexmedetomidine.
Introduction
Acute kidney injury (AKI) can occur for a variety of reasons: inflammation, ischemia, infection, medications, toxins, and other systemic stresses like surgery or critical illness. 1 Several classification systems have been proposed to diagnose AKI. 2 The Kidney Disease: Improving Global Outcomes (KDIGO) classification system considers urine output and serum creatinine values, and is the preferred definition for clinical and research settings. 3 With respect to changes in serum creatinine within the KDIGO classification system, stage 1 refers to an increase in serum creatinine by 1.5–1.9 times in 7 days or ⩾0.3 mg/dL in 48 h; stage 2 refers to an increase in serum creatinine by 2–2.9 times in 7 days; and stage 3 refers to an increase in serum creatinine by 2–2.9 times in 7 days or ⩾4 mg/dL or initiation of renal replacement therapy. 3
Dexmedetomidine binds centrally to alpha-2 receptors to provide sedation and attenuate the sympathetic nervous system. 4 Animal models for lipopolysaccharide-induced AKI suggest that dexmedetomidine exhibits anti-inflammatory effects. 5 Suppression of inflammatory markers such as tumor necrosis factor-alpha and interleukin-6 may halt the progression of AKI to kidney failure. 6 These studies have provided biochemical support to administer dexmedetomidine to patients at risk for AKI.
The impact of dexmedetomidine on postoperative AKI has been studied. In a meta-analysis of randomized controlled trials, dexmedetomidine decreased the odds of AKI in patients following cardiac surgery, but mortality, duration of mechanical ventilation, hospital length of stay (LOS), and ICU LOS did not demonstrate statistically significant benefits from dexmedetomidine. 7 A meta-analysis of patients following all surgeries demonstrated a decreased risk of AKI as well as reductions in ICU LOS and in-hospital mortality with dexmedetomidine. 8
There is a lack of medical literature that quantifies the effect of dexmedetomidine on AKI recovery. 9 AKI recovery is an important outcome to study because, left untreated, AKI can progress to chronic kidney disease (CKD), which has associated cardiovascular morbidity and mortality 10 . We sought to answer the following questions in this study: In adult patients admitted to the ICU with AKI, do patients who receive dexmedetomidine recover from AKI more quickly compared to patients who do not receive dexmedetomidine; and do patients who receive dexmedetomidine experience decreased LOS in the hospital and ICU or decreased in-hospital mortality compared to patients who do not receive dexmedetomidine?
Methods
Population and dataset
Medical Information Mart for Intensive Care (MIMIC-IV) includes data from 2008 to 2019 and described by the following statement11–13: MIMIC-IV is the result of a collaboration between Beth Israel Deaconess Medical Center (BIDMC) and Massachusetts Institute of Technology (MIT). Data collected at BIDMC as part of routine clinical care is deidentified, transformed, and made available to researchers who have completed training in human research and signed a data use agreement. The Institutional Review Board at the BIDMC granted a waiver of informed consent and approved the sharing of the research resource.
11
All elements of the “Data Conditions of Use” were considered prior to beginning this study and satisfied at the time of submission of this manuscript.
Covariates
Presence or absence of comorbid conditions were determined by querying the dataset for ICD-10 codes. The pulmonary comorbidity included the following diagnoses: bronchitis, emphysema, bronchiectasis, and chronic obstructive pulmonary disease (COPD). The cardiovascular comorbidity included the following diagnoses: hypertension, hypertensive disease, secondary hypertension, ischemic heart disease, cardiomyopathy, cardiomyopathy (other), and congestive heart failure. The renal comorbidity included the following diagnoses: hypertensive renal disease, acute renal failure, CKD, chronic renal failure, hypertensive heart, and renal disease. If not present in the dataset, then the subject was assumed to not have the condition. Other covariates included the following variables: age, body mass index (BMI), gender, race, baseline creatinine, total time of vasoactive infusions (epinephrine, vasopressin, phenylephrine, and norepinephrine), average rate of each vasoactive infusion, dexmedetomidine administration (binary), and average rate of dexmedetomidine infusion.
Primary and secondary outcomes
The primary outcome for this study is the effect of dexmedetomidine (binary) on time (days) to recovery from AKI. This study has three secondary outcomes: the effect of dexmedetomidine (binary) on hospital LOS; the effect of dexmedetomidine (binary) on ICU LOS; and the effect of dexmedetomidine (binary) on in-hospital mortality.
Definitions
AKI was defined as an increase in serum creatinine value by ⩾1.5 times the baseline value. The baseline serum creatinine value was defined as the first recorded serum creatinine value for that hospital admission. There are several different definitions for diagnosing AKI that include both creatinine and urine output. Although using urine output to diagnose AKI was considered, urine output is inconsistently recorded for patients who do not have Foley catheters and may over-diagnose patients with AKI, thus increasing the type I error rate. This is, nonetheless, a limitation of the current study. Timing of dexmedetomidine was defined as administration following the diagnosis of AKI.
Inclusion and exclusion criteria
Adult patients (age 18 years or older) with two or more serum creatinine values recorded while admitted to the hospital were eligible for study inclusion. Pediatric patients (age younger than 18 years) or patients with less than two serum creatinine values recorded while admitted to the hospital were ineligible for study inclusion.
Statistical analysis plan
All statistical analyses were conducted with RStudio or STATA.14,15 Survival analysis investigated the effect of dexmedetomidine administration (binary) on time (number of days) to recovery from AKI. A change in time to recovery from AKI equal to or greater than 1 day was considered a clinically meaningful result when interpreting the primary outcome. Linear and logistic regression models analyzed the effect of dexmedetomidine (binary) on hospital or ICU LOS (number of days) and in-hospital mortality, respectively. A change in hospital or ICU LOS equal to or greater than 1 day and a change in the odds of in-hospital mortality equal to or greater than 10% were considered clinically meaningful results when interpreting the secondary outcomes. Sensitivity analyses investigated dexmedetomidine as a continuous (as opposed to a binary) variable.
We used Cox proportional hazards regression to model time-to-event outcomes. Covariates were selected a priori based on clinical relevance and included age, BMI, baseline cardiovascular disease, renal disease, and duration of vasoactive medication exposure.
Results
Baseline characteristics
Of the total cohort, patients who did not receive dexmedetomidine were, on average, older (mean age 65.3 years vs 63.0 years), had lower BMI (mean 29.0 kg/m2 vs 30.2 kg/m2), and a higher prevalence of baseline cardiovascular (48% vs 40%) and renal comorbidities (42% vs 33%). They also had a shorter median duration of exposure to vasoactive medications (541.0 h vs 1064.7 h) compared to the dexmedetomidine group. Complete baseline characteristics of the study population are shown in Table 1. These imbalances informed covariate selection in subsequent regression modeling.
Baseline characteristics.

Data are presented as mean (SD) for continuous variables (age, BMI, and total vasoactive medication infusion time), and percentage for categorical and binary variables.
dL, deciliter; kg, kilograms; m, meters; mg, milligram; N, number.
Time to AKI recovery
Treatment with dexmedetomidine is associated with a 53.7% (95% CI: 46.8–59.8) decrease in the risk of recovery from AKI and this value is statistically significant (
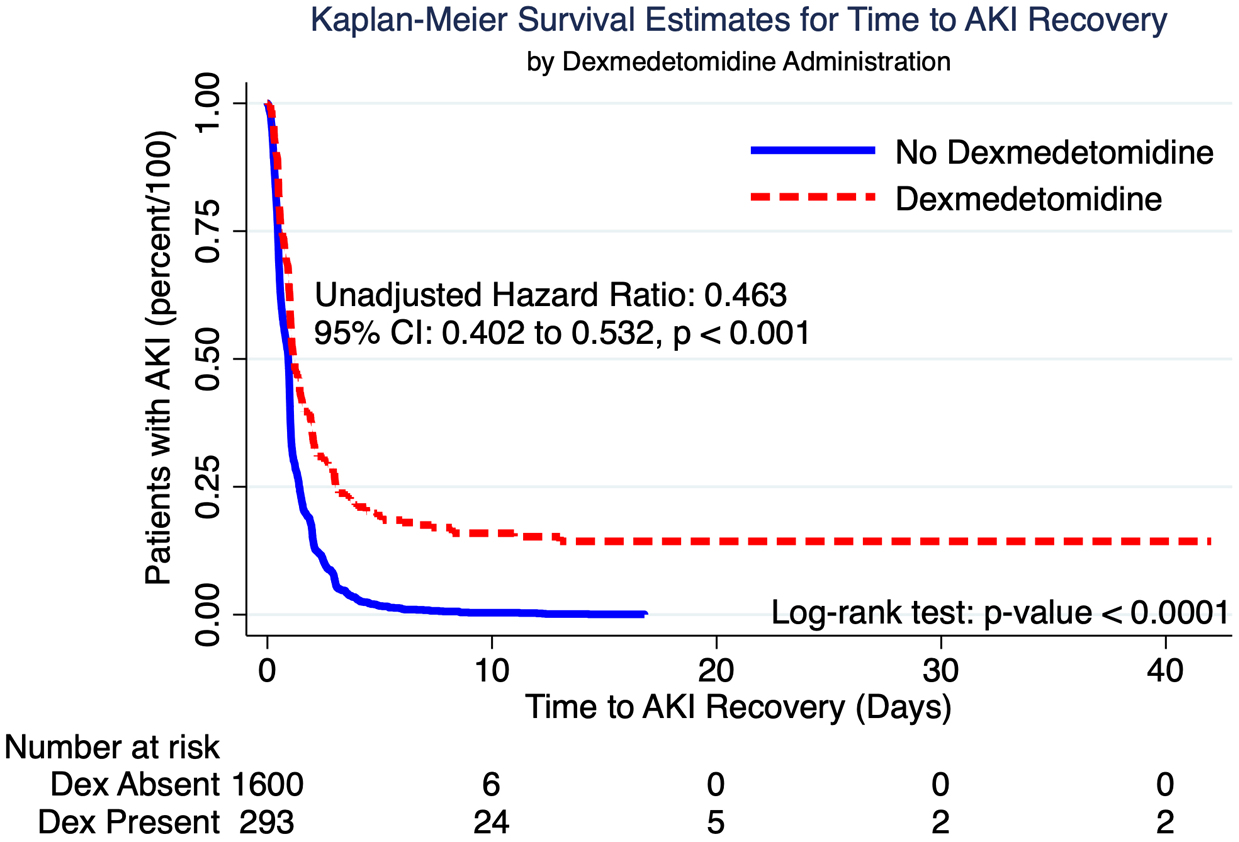
Kaplan-Meier survival estimates for time to AKI recovery by dexmedetomidine administration.
The association of dexmedetomidine on AKI recovery time is demonstrated in the Cox regression models shown in Table 2. After adjusting for all covariates, dexmedetomidine is associated with a 52.1% decreased hazard of AKI recovery that is statistically significant (
Effect of dexmedetomidine on time to AKI recovery.

Bold text indicates statistical significance.
Race: compared to white.
Baseline creatinine: compared to creatinine <1 mg/dL.
AHR, adjusted hazard ratio; CI, confidence interval; h, hour; kg, kilogram; ln, natural log; N, number of observations; u, units; μg, microgram.
Secondary outcomes
Fully adjusted models for all secondary outcomes are shown in Table 3 and discussed in the following paragraphs.
Effect of dexmedetomidine administration on natural logarithm of hospital LOS, natural logarithm of ICU LOS, and in-hospital mortality.

Bold indicates statistical significance.
Years: 10-year increments for hospital LOS and 25-year increments for ICU LOS and in-hospital mortality.
Race: compared to white.
Baseline creatinine: compared to creatinine <1 mg/dL.
CI, confidence interval; kg, kilogram; h, hour; ln, natural log; N, observations; u, units; μg, microgram.
Hospital LOS
The predicted hospital LOS in a patient who receives dexmedetomidine is, on average, 1.1898 times the hospital LOS, or is 18.98% greater, for a patient who has the same gender, age, BMI, race, baseline creatinine, comorbidities, and exposure to vasoactive infusions. The mean hospital LOS in this dataset for patients who do not receive dexmedetomidine is 19.36 days. Because 18.98% of 19.36 days is 3.67 days, the association of dexmedetomidine on hospital LOS is also clinically significant. There is no evidence to suggest that gender, BMI, and race are associated with statistically significant changes in hospital LOS in the fully adjusted linear regression model.
The predicted hospital LOS for someone with a baseline creatinine value greater than or equal to 3 mg/dL is 1.1982 times the hospital LOS (19.82% higher) in someone who has the same gender, age, BMI, race, comorbidities, and exposure to vasoactive infusions and dexmedetomidine but with a creatinine less than 1 mg/dL, which is statistically significant (
Patients who do not receive epinephrine infusions accumulate a hospital LOS, on average, of 19.85 days. Because 1.9% of 19.85 days is 0.3772 days, or 9.05 h, epinephrine rate does not demonstrate a clinically meaningful association with hospital LOS. Similarly, for every doubling of the vasoactive infusion time, the predicted hospital LOS is multiplied by 1.023 (2.3% higher). Patients who do not receive vasoactive infusions accumulate a hospital LOS, on average, of 18.58 days. Because 2.3% of 18.58 days is 0.427 days, or 10.26 h, vasoactive infusion time does not demonstrate a clinically meaningful association with hospital LOS.
ICU LOS
The predicted ICU LOS in a patient who receives dexmedetomidine is, on average, 1.3256 times the ICU LOS, or is 32.56% greater, for a patient who has the same gender, age, BMI, race, baseline creatinine, comorbidities, and exposure to vasoactive infusions. The mean ICU LOS in this dataset for patients who do not receive dexmedetomidine is 9.73 days. Because 32.56% of 19.36 days is 3.17 days, the association of dexmedetomidine on ICU LOS is also clinically significant. There is no evidence to suggest that gender, BMI, and race are associated with statistically significant changes in ICU LOS.
The predicted ICU LOS for someone with a baseline creatinine value 1–2 mg/dL is 1.1219 times the hospital LOS (12.19% higher) in someone who has the same gender, age, BMI, race, comorbidities, and exposure to vasoactive infusions and dexmedetomidine but with a creatinine less than 1 mg/dL, which is statistically significant (
Patients who do not receive epinephrine infusions accumulate an ICU LOS, on average, of 7.47 days. Because 1.9% of 7.47 days is 0.1419 days, or 3.41 h, epinephrine rate does not demonstrate a clinically meaningful association with ICU LOS. Similarly, for every doubling of the vasoactive infusion time, the predicted hospital LOS is multiplied by 1.049 (4.9% higher). Patients who do not receive vasoactive infusions accumulate an ICU LOS, on average, of 6.19 days. Because 4.9% of 6.19 days is 0.303 days, or 7.28 h, vasoactive infusion time does not demonstrate a clinically meaningful association with ICU LOS.
In-hospital mortality
Patients who receive dexmedetomidine have 0.6 times the odds of in-hospital mortality compared to patients who do not receive dexmedetomidine, which is clinically and statistically significant (
All elevated baseline serum creatinine values are associated with statistically significant increases in the odds of in-hospital mortality. An elevated baseline creatinine value of 1–2 mg/dL, 2–3 mg/dL, or greater than 3 mg/dL multiplies the odds of in-hospital mortality by 1.56, 2.03, and 2, respectively. The associations between elevated baseline serum creatinine value and in-hospital mortality are clinically and statistically significant (
Every doubling of the norepinephrine infusion rate is associated with a change in the odds of in-hospital mortality by a factor of 0.6273, which is statistically significant (
Sensitivity analyses
Sensitivity analysis utilizing Cox regression of dexmedetomidine rate on time to AKI recovery demonstrated an adjusted hazard ratio (AHR) of 1.42 (95% CI: 1.24–1.63,
Effect of dexmedetomidine as continuous variable on hospital LOS, ICU LOS, and in-hospital mortality.

Bold indicates statistical significance.
Years: 10-year increments for hospital LOS and 25-year increments for ICU LOS and in-hospital mortality.
Race: compared to white.
Baseline creatinine: compared to creatinine <1 mg/dL.
CI, confidence interval; h, hour; kg, kilogram; ln, natural log; LOS, length of stay; N, observations; u, units; μg, microgram.
Discussion
The primary outcome results suggest that dexmedetomidine is significantly associated with prolonged recovery from AKI in critically ill adults. Given the relatively small number of patients who received dexmedetomidine, further stratification in Kaplan-Meier analyses—such as by timing or dosing tertiles—would have likely created highly underpowered subgroups and unstable survival estimates. Similarly, spline-based modeling of time-varying exposure or recovery trajectories were not pursued, as the limited number of dexmedetomidine-exposed events restricted the robustness of such nonparametric approaches. We also acknowledge the presence of baseline differences between the dexmedetomidine and non-dexmedetomidine cohorts, which raises the potential for confounding by indication. However, we chose not to use propensity score matching, as it would have required excluding a substantial portion of the dataset. Given the large size of the MIMIC-IV database, we reasoned that matching could paradoxically increase bias by discarding informative data and reducing generalizability. Inverse Probability of Treatment Weighting (IPTW) was similarly not performed due to the risk of model instability in the presence of extreme weights, especially with rare exposure and high comorbidity burden among patients receiving dexmedetomidine. These decisions were made a priori and based on the study’s exploratory design. Taken together, these more advanced causal inference methods—such as IPTW, stratified tertile analysis, and spline-based modeling—may be more appropriately applied in future confirmatory studies with larger exposed cohorts and richer exposure timing information.
The primary outcome results are not supported in a Cox regression model analyzing dexmedetomidine infusion rate as a continuous variable. The effect of dexmedetomidine rate on risk of AKI recovery is either non-linear or the sample size in this study is not large enough to demonstrate its true effects. Perhaps, low doses of dexmedetomidine render protective anti-inflammatory effects while higher doses impair hemodynamics and renal perfusion, but this requires additional investigation. Prolonged recovery from AKI can increase a patient’s risk for CKD; increase costs for the healthcare system and patient; and diminish healthcare resources, such as kidneys available for transplantation and dialysis equipment.
The secondary outcome results suggest that dexmedetomidine is significantly associated with prolonged hospital and ICU LOS as well as decreased in-hospital mortality in critically ill adults with AKI. These findings are not supported in regression models analyzing dexmedetomidine infusion rate as a continuous variable. Dexmedetomidine may not exhibit a linear association with hospital or ICU LOS and in-hospital mortality. Instead, two infusion rates of dexmedetomidine may produce opposite effects, and the small sample size of this study may not adequately characterize these rate-dependent effects. Additionally, a one unit increase of 1 μg/kg/h may not be clinically relevant. Because the effect of dexmedetomidine infusion rates has not been investigated within the context of AKI, it is difficult to identify clinically meaningful categories that are not simply arbitrary whole integers. Although there is a loss of statistical power with dichotomization of dexmedetomidine into present or absent, this may be more reasonable, clinically.
Prolonged hospital and ICU LOS lead to increased costs for the healthcare system and patient; diminish healthcare resources; place financial strain on patients while they are unable to work; and lead to emotional stress while being away from their homes and families. Conversely, decreased in-hospital mortality improves patient and family satisfaction with medical care and offers patients the possibility for a return to healthy life.
Since initiating this project, a study was published investigating similar outcomes, yet utilizing the third edition of MIMIC. 9 Yang et al. compared outcomes for patients with AKI who did and did not receive dexmedetomidine utilizing data from MIMIC-III. 9 In contrast to MIMIC-III, which contains data on approximately 40,000 patients, MIMIC-IV includes data for nearly 300,000 patients and encompasses a patient’s entire hospital stay (not just the ICU), such as the medication administration record.
The methods employed by Yang et al. differ considerably from this study in several key ways. First, the authors performed propensity score matching to establish 324 pairs of patients. The use of propensity scores to decrease bias from confounding by indication is an appropriate statistical strategy for modeling rare events with limited data. 16 However, the size of the MIMIC database should be sufficiently large to minimize gross imbalances in patient characteristics, which can be paradoxically worsened after pruning the data with propensity score matching. 17 Second, Yang et al. include urine volume less than 0.5 mL/kg for 6 h in their definition for AKI. Although including urine output measurements are appropriate in defining AKI for prospective trials, including this definition in analyzing retrospective clinical data may over-diagnose AKI due to poor or untimely recording of urine output in patients. Third, Yang et al. excluded patient groups that are of clinical interest, such as patients with an ICU stay less than 48 h, which may skew the results toward more critically ill patients. Last, Yang et al. do not provide evidence to support their model assumptions. Our results would have been drastically different (perhaps statistically opposite) if model assumptions were not checked, and the analyses proceeded without log transformations.
Although the results of our study agree with the results from Yang et al. with respect to in-hospital mortality, there is disagreement regarding the association of dexmedetomidine with hospital and ICU LOS as well as AKI recovery. Yang et al. reported a HR of 1.199 (0.851–1.688,
Time to AKI recovery
Our results suggest that dexmedetomidine is significantly associated with a prolonged recovery time from AKI in critically ill adult patients admitted to the ICU. Although time to recovery is an unconventional outcome measure, we feel that it is valid to be used within this context given the significant morbidity and mortality associated with untreated AKI that progresses to CKD. Significant associations are also noted with respect to the included covariates. The increased risk of AKI recovery noted in patients with pre-existing cardiovascular disease is initially surprising but is clinically feasible when considering the medications these patients are often prescribed. Patients with hypertension or congestive heart failure are often prescribed loop diuretics to reduce hypervolemia and vasodilate blood vessels. 18 Loop diuretics may hasten AKI recovery.19,20 The possible impact of home medications on AKI recovery within the MIMIC-IV dataset requires additional investigation, such as the inclusion of loop diuretics as a covariate.
Our results also suggest that vasoactive medications are associated with decreases in the hazard of AKI recovery, yet the clinical significance of these effects are unclear. Under normal physiologic conditions in healthy volunteers, norepinephrine has been shown to reduce renal blood flow. 21 However, animal studies have shown renal blood flow to progressively increase with higher doses of norepinephrine under anesthesia. 22 Because patients who require vasoactive medications are likely more critically ill and at risk for multi-organ failure compared to patients not receiving vasoactive medications, it is feasible that patients receiving vasoactive medications would also take longer to recover from AKI.
Hospital LOS
The prolonged hospital LOS associated with dexmedetomidine may suggest prolonged recovery from acute illness or decreased early in-hospital mortality, thus allowing for longer hospital LOS. Although older patients may be expected to accrue longer hospital LOS, the results may reflect the greater severity of illness in younger patients. The threshold for hospital admission for younger patients is likely greater than that for older patients. Therefore, at the time of admission, younger patients may have more severe acute disease compared to older patients, which may manifest as longer hospital LOS in this study. Additionally, older patients might die before lengthy hospital LOS can be amassed. This hypothesis requires further investigation, such as the addition of covariates that indicate severity of illness or likelihood of mortality, such as the APACHE score. 23
Patients with pre-existing renal disease or baseline creatinine greater than 3 mg/dL experience significantly longer hospital LOS by 12.41% and 19.82%, respectively. These results are consistent with clinical expectations and highlight the importance of including these covariates in this study.
Although longer hospital LOS increases the opportunity for longer vasoactive infusion times, the total time of vasoactive medication infusion may also serve as a marker for the degree of critical illness: patients requiring vasoactive infusions for a greater length of time are likely more critically ill compared to patients who require them for less total time. Therefore, vasoactive infusion time was incorporated as a covariate in the analyses.
ICU LOS
Like the results regarding hospital LOS, the prolonged ICU LOS associated with dexmedetomidine may suggest prolonged recovery from acute illness or decreased early in-hospital mortality, thus allowing for longer ICU LOS. Although older patients may be expected to accrue longer ICU LOS, the results may reflect the greater severity of illness in younger patients. Younger patients who warrant ICU admission are likely more acutely ill compared to older patients. Conversely, older patients might be more likely to die and not reach lengthy ICU stays when compared to younger patients. This argument may also explain the significant association between cardiovascular comorbidity and decreased ICU LOS. Again, further investigation with validated clinical prediction tools for severity of illness, such as the APACHE score, is required to test these hypotheses. 23
In-hospital mortality
The results suggest a clinically significant mortality benefit with dexmedetomidine. The robustness of these results is supported by sensitivity analysis using Cox regression for the effect of dexmedetomidine on in-hospital mortality. The adjusted HR of 0.611 (95% CI: 0.443–0.843,
All elevated baseline serum creatinine values are associated with a statistically significant increase in the risk for in-hospital mortality, thus highlighting the importance of research on kidney disease. Unexpectedly, having a cardiovascular comorbidity demonstrated a decreased risk for in-hospital mortality by 51.2% (
Limitations
Several limitations should be noted when interpreting the results of this study. First, this retrospective review is limited to data already collected for clinical purposes. Data were not collected to answer the research questions presented in this study. Therefore, this study can only evaluate association (not causation). Although patients who did not receive dexmedetomidine served as the comparator group in this project, these patients are likely not representative of all patients admitted to the ICU. Furthermore, the baseline characteristics for patients who did and did not receive dexmedetomidine are significantly different, such as the amount of time patients received vasoactive medication infusions (mean (SD) 1064.7 (2828.6) h vs 541 (1284.2) h for patients who did and did not receive dexmedetomidine, respectively,
The definitions used in this study may also limit the generalizability of our results. Only serum creatinine values were used to diagnose AKI, thus excluding urine output from the criteria. Because serum creatinine values are objective laboratory values that are not subject to patient or equipment factors (Foley catheter or other urine collection system), these values allow for more accurate diagnoses of AKI. However, this likely underestimates the prevalence of AKI in the population. Furthermore, inconsistent measurement of serum creatinine introduces bias to the analyses.
Conclusion
Dexmedetomidine may be associated with slower recovery from AKI in critically ill adults. The prolonged LOS in the hospital and ICU associated with dexmedetomidine may be related to reduced mortality, but these results require additional investigation. These exploratory results warrant further investigation to better understand the clinical implications of dexmedetomidine exposure in the setting of AKI.
