Abstract
Background:
There are still some points of controversy regarding the adverse events associated with celecoxib use, particularly in terms of thrombosis.
Objectives:
To explore the relationship between celecoxib and thrombosis in the real world and to investigate the causality that exists.
Design:
We conducted pharmacovigilance analysis on spontaneously reported adverse events to evaluate the association between celecoxib and thrombotic events. In addition, Mendelian randomization studies of drug targets were used to explore the causal relationship between them.
Methods:
This study used the data from the United States Food and Drug Administration Adverse Event Reporting System (FAERS), the Japanese Adverse Drug Event Report database, and the Canada Vigilance Adverse Reaction (CVAR) for pharmacovigilance analysis. Among these, the Report Odds Ratio, Proportional Reporting Ratio, Information Component, and Empirical Bayesian Geometric Mean were used to determine the strength of adverse event signals. In addition, the Weibull shape parameter test was used in this study to investigate the trend of adverse events. Mendelian randomization was used to explore the causal link between celecoxib and deep vein thrombosis.
Results:
Pharmacovigilance signals indicated that celecoxib was associated with an increased risk of thrombosis, with deep vein thrombosis demonstrating positive signals in all three populations. In addition, Mendelian randomization analyses provided evidence to support a causal relationship between celecoxib and deep vein thrombosis and clarified that carbonic anhydrase 2, a target protein of celecoxib, is causally linked to deep vein thrombosis.
Conclusion:
The use of celecoxib leads to an increased risk of thrombosis and suggests a causal relationship.
Plain language summary
Background:
Celecoxib (trade name Celebrex) is a widely used anti-inflammatory and analgesic drug worldwide, and there has been controversy over whether it increases the risk of thrombosis. We investigated its relationship with thrombosis and explored the potential mechanisms involved.
Methods:
The United States Food and Drug Administration Adverse Event Reporting System (FAERS), the Japanese Adverse Drug Event Report database (JADER), and the Canada Vigilance Adverse Reaction (CVAR) play a key role in identifying adverse events of drugs. We retrieved adverse event reports related to celecoxib and thrombosis from three databases between 2004 and 2024 and conducted a series of statistical analyses. In addition, we also verified the causal relationship between them.
Results:
Reports from the United States, Japan, and Canada all indicate a certain correlation between celecoxib and thrombotic events. It is worth noting that there are more reports of thrombus in women than in men. Furthermore, further analysis suggests that celecoxib promotes thrombus formation by inhibiting carbonic anhydrase 2, and there is a causal relationship between them.
Conclusion:
Celecoxib increases the risk of thrombosis, which may be achieved by inhibiting carbonic anhydrase 2. This suggests that patients who take celecoxib for a long time should pay attention to the risk of thrombosis. In addition, doctors should evaluate whether patients have high risk factors for thrombosis before medication.
Keywords
Introduction
Non-steroidal anti-inflammatory drugs (NSAIDs) are a group of chemically heterogeneous drugs with anti-inflammatory, analgesic, and antipyretic properties, and are one of the most widely used prescription drugs worldwide. 1 They are being considered for short-term treatment of common pain conditions as well as long-term improvement of chronic inflammatory diseases such as osteoarthritis (OA). NSAIDs primarily act by inhibiting the cyclooxygenase (COX) pathway to block the synthesis of prostaglandins. 2 According to the different subtypes of COX they block, NSAIDS can be divided into nonselective COX inhibitors, COX-1 inhibitors, and COX-2 inhibitors. Among these, COX-2 inhibitors are widely used due to their strong antipyretic and analgesic effects and fewer gastrointestinal adverse events (AEs). 3 There are currently various types of COX-2 inhibitors in the world, such as celecoxib, meloxicam, and nimesulide. Celecoxib is the only coxib-class drug commercially available in most countries and regions, holding a considerable market share. 4 In addition, the selectivity of celecoxib for COX-2 is 30 times that of COX-1, far higher than other COX-2 inhibitors. 5 Therefore, it is widely used for anti-inflammatory and analgesic treatment of various inflammatory diseases.
Although celecoxib is currently the preferred choice for treating chronic inflammatory diseases such as OA, its safety has been controversial due to its ability to cause cardiovascular damage. The most critical point of controversy is whether the use of celecoxib leads to thrombosis. Some studies have shown that celecoxib does not cause the formation of thrombus. In the Celecoxib Long-term Arthritis Safety Study (CLASS), no significant increase in cardiovascular events was observed with celecoxib compared to nonselective NSAIDs such as diclofenac and naproxen. 6 Another experimental study demonstrated that celecoxib 200 mg/day can significantly improve the diastolic function of vascular endothelial cells, attenuate chronic inflammation and oxidative stress, and reduce the formation of thrombus after atherosclerosis. 7 In addition, a follow-up study over 10 years related to hip and knee OA did not find any association between celecoxib and thrombosis. 8 However, other studies have questioned this view, suggesting that patients treated with celecoxib have an increased risk of thrombosis. For example, a systemic sclerosis patient admitted to Carolina Medical University developed a pulmonary embolism after receiving treatment with celecoxib for 2 days. 9 This correlation has also been demonstrated in some experimental studies. For example, Mukherjee et al. pointed out that the inhibition of COX-2 by celecoxib may promote thrombosis in the coronary arteries of experimental dogs with vascular injury by reducing endogenous prostacyclin production. 6
Thrombotic events significantly elevate risks of disability, mortality, and healthcare costs, profoundly compromising patient prognosis. For example, joint replacement surgery is recommended for patients with advanced OA, but the incidence of postoperative venous thromboembolism (VTE) can reach 42.5%, and severe cases can develop into pulmonary embolism, leading to severe disability or even death.10,11 Therefore, it is very important to prevent the formation of thrombosis in perioperative patients. 12 Celecoxib, the most common medication used for pain relief in these patients, may increase thrombosis risk, which can greatly affect the success of the procedure and the quality of life of the patient, yet current clinical guidelines lack specific recommendations for thromboprophylaxis in this setting, and its potential risks remain under-recognized. Therefore, it is necessary to clarify the occurrence and causal relationship of thrombotic events in the real world with celecoxib.
The drug spontaneous reporting system is derived from spontaneously reported adverse drug reactions in the real world, used to clarify and alert potential associations between drugs and AEs that occur in clinical practice. These spontaneous reports contain data from patients who were excluded from clinical trials, making them more likely to reflect clinical practice. Based on these data, it can be determined whether the drug is related to AEs. As the database is constantly updated, it can more accurately capture the dynamics of real-world research. At present, large-scale spontaneous reporting systems mainly include the United States Food and Drug Administration Adverse Event Reporting System (FAERS), the Japanese Adverse Drug Event Report database (JADER), and the Canada Vigilance Adverse Reaction (CVAR). This study used four pharmacovigilance analysis methods, Report Odds Ratio (ROR), Proportional Reporting Ratio (PRR), Information Component (IC), and Empirical Bayesian Geometric Mean (EBGM) for signal detection from three databases.
Meanwhile, Mendelian randomization (MR) is a genetic epidemiological approach that can infer causal relationships between exposure and outcomes by using genetic variations closely related to exposure as instrumental variables (IVs). Genetic variations associated with drug target gene expression, also known as expression quantitative trait loci (eQTLs), can serve as IVs for MR to provide robust causal evidence for the relationship between drugs and adverse outcomes.
Therefore, we conducted a multinational pharmacovigilance study integrating databases from the United States, Japan, and Canada to investigate the correlation between celecoxib and thrombosis in the real world. In addition, MR of drug targets has been used to clarify the causal relationship between celecoxib and thrombosis, as well as potential mechanisms. This study aims to enhance understanding of the safety of NSAIDs in clinical practice.
Materials and methods
Pharmacovigilance data analysis
Thromboembolism adverse event data source
We identified the generic and brand names of celecoxib using a Medical Subject Headings (MeSH) terminology browser and extracted data from FAERS, JADER, and CVAR, respectively. AEs were submitted to these databases, and data were updated by healthcare professionals, consumers, manufacturers, and others in the form of spontaneous reporting systems. In the data preprocessing stage, case reports with the same gender, age, country, event date, adverse reactions, medication, and indications are identified as duplicate reports and removed. 13 Subsequently, we standardized the collected adverse event reports related to celecoxib using preferred terms (PTs) and systemic organ categories (SOCs) from the Medical Dictionary of Regulatory Activities (MedDRA) 26.1 edition International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). To improve the accuracy of the research, we limited the scope of the examination in the DRUG file to drugs whose role_cod was designated as “primary suspect (PS).” 14 In addition, we restricted PTs to “thrombosis”-related AEs in the REAC document.
In this study, we extracted only United States population-related data from the FAERS database from the first quarter of 2004 through the second quarter of 2024. The extracted data consisted primarily of patient demographics (DEMO), drug detail information (DRUG), description of adverse events (REAC), and outcomes received by the reporting source (OUTC). A relationship was established in the FAERS database that allows for the linking of each data file by a unique identification number. 15 After data preprocessing, we obtained a total of 12,103,489 DEMO data, 37,894,197 DRUG data, and 31,971,140 REAC data. Next, by limiting drug role codes and PTs, we obtained a total of 30,025 AEs related to celecoxib and 740 AEs related to thrombosis.
In the JADER database, we extracted AE data recorded from the first quarter of April 2004 through May 2024, available on the Pharmaceutical and Medical Devices Agency website (http://www.pmda.go.jp/). It comprises four datasets: DEMO, DRUG, REAC, and primary disease (HIST). We linked the DEMO tables to these tables according to the identified conditions. 16 After deduplication, we obtained 909,122 DEMO records, 4,575,592 DRUG entries, and 1,505,095 REAC entries. We obtained a total of 4571 AEs and 48 thrombotic events related to celecoxib by limiting the AEs those associated with the drug named “PS.”
AEs data recorded from January 1, 2004 to May 31, 2024 in the CVAR database were also included in this study. These data can be accessed from the official Government of Canada website (http://www.canada.ca/). Data included patient characteristics (e.g., age and sex), drug information, disease indication, AEs, and outcomes. We used “Report_ID” as a unique identifier to combine different data files. 17 We obtained a total of 940,097 DEMO data, 4,290,474 DRUG data, and 3,892,095 REAC data. Consistent with the previous process, by qualifying the drug role code and PT fields, we ended up with 1184 reports of AEs associated with celecoxib and 26 associated thrombotic events.
Time-to-onset analysis
Time-to-onset (TTO) was calculated as the time from when a patient first received a prescription to the occurrence of an AE associated with anthropometric changes. Evaluation of the TTO data was based on the median, mean, and the Weibull shape parameter (WSP) test, which is characterized by the scale parameter α and the shape parameter β, describing the risk of an increase or decrease in AEs over time. Depending on the value of the shape parameter β, the upper limit of the 95% confidence interval (CI) for β < 1 is considered to decrease the initial increase (early failure type) over time. In contrast, the lower limit of the 95% CI for β > 1 indicates an increase in risk over time (wear-and-tear failure type), whereas a 95% CI including β of 1 indicates a constant risk of AEs occurring throughout the exposure period (random failure type).
Statistical analyses
Our study used ROR, PRR, IC, and EBGM to detect safety signals. Among these, ROR is a crucial analytical method used to identify and discover potential associations between reported drugs and AEs. PRR focuses more on the combination of drugs and AEs. 18 Compared to ROR and PRR, IC reflects the strength of the correlation between drugs and AEs, with lower requirements for the quality of drug adverse event reporting data, while the correlation between detected AEs signals is relatively strong, and the EBGM method has been proven to be the most conservative among the four methods, indicating its suitability for epidemiological studies.19,20 The potential association between medication population characteristics, such as gender, and adverse drug reactions is often explored. 20 To comprehensively screen for potential drug safety risks, according to established pharmacovigilance information guidelines, any report that meets any of the above four criteria and reaches the positive signal threshold is considered a positive adverse event report. 21 Specific formulas can be found in Table S1. Data were collated and statistically analyzed using R (version 4.4.1) and the corresponding RStudio version. The R package ggplot2 was used to plot the statistical tables. The R package “TwoSampleMR” was created by Ross Ihaka and Robert Gentleman using R (version 4.4.1) and is maintained by the R core team. RStudio was developed by Posit PBC, and the R package ggplot2 was developed by Hadley Wickham and others and is maintained by Thomas Lin Pedersen.
MR analysis
Data sources
Our study used STROBE-MR guidance to assess the integrity of the entire process (Table S2). We used the DrugBank database (https://go.drugbank.com/) to obtain the active ingredient of celecoxib and identify its target gene. To identify common single nucleotide polymorphisms (SNPs) associated with the expression of celecoxib drug target genes in whole blood, eQTL analyses were performed to identify loci capable of regulating blood gene expression. eQTLs data were obtained from the eQTLGen Consortium (https://eqtlgen.org/). The eQTLGen dataset contains 16,987 genes and 31,684 cis-eQTLs in blood samples from mostly healthy European individuals. 21
Deep vein thrombosis (DVT) is the most common form of thrombosis. For example, DVT occurs in up to 600,000 people per year in the United States alone. Its annual incidence is close to 1.6/1000 inhabitants. 22 Therefore, we used DVT as an outcome indicator. Data on DVT were obtained from the UK Biobank, an outcome data set comprising 462,933 European-ancestry individuals (cases = 9241; controls = 453,692). In addition, to ensure the robustness of the data, we used data from DVT in the GWAS Catalog and FinnGen Release 12 databases as outcomes for validation. The DVT dataset in the GWAS Catalog database (ebi-a-GCST90038607), including the dataset containing 484,598 samples (case = 12,240; control = 472,358), was used as statistical associations for outcomes. 23 The FinnGen study, an extensive genomics program analyzing more than 500,000 Finnish Biobank samples, combines genetic variation with health data to elucidate disease mechanisms and susceptibility. 24 The DVT data from the FinnGen R12 database (finngen_R12_I9_VTE) consisted of 500,348 samples (case = 26,333; control = 474,015). We used data from the three datasets to validate each other, ensuring the generalizability and robustness of the findings.
Selection of IVs
To ensure the accuracy and robustness of the causal relationship between the celecoxib drug target and thrombus, the following steps were used to select IVs. In principle, genetic instruments used in MR analyses must satisfy three assumptions: (1) IVs are directly related to exposure; (2) IVs are independent of any confounding variables; (3) IVs only affect the results via exposure. First, we use the required p values <5 × 10−8 and r2 < 0.3 to screen for IVs that are strongly correlated with exposure. The strength of IVs was further quantified by calculating the individual and cumulative F-statistic for each SNP by F = R2(N − k − 1)/k(1 − R2), including only SNPs with F-statistics >10 and excluding SNPs that could lead to unwanted bias and multiple significance. Second, IVs that are not directly related to the outcome will be screened through p > 1 × 10−5. Finally, considering the potential impact of genetic instruments on potential confounding factors, we excluded pleiotropy SNPs associated with other potential confounding factors using PhenoScanner (http://www.phenosanner.medschl/).
MR analysis
To initially assess the causal relationship between exposure (celecoxib drug target) and outcome (DVT), this study performed a two-sample MR analysis with genetic variation as IVs. We used a range of MR methods for the study. The primary analytical method was random-effects inverse variance weighted (IVW), and the secondary analytical methods were weighted median and MR-Egger regression. The central tenet of IVW is that all genetic variants are legitimate IVs with strong causality detection, making IVW the most important method for assessing causality between exposure and outcome. In addition, the weighted median consistently estimates causal effects when up to 50% of the genetic variation in the SNPs is from null IVs.
Sensitivity analyses
We conducted sensitivity analyses to verify the reliability of the analyses. First, we used Cochran’s Q test, MR-Egger regression, and IVW methods to assess IVs heterogeneity. In addition, we used the MR-Egger intercept test to assess the horizontal multicollinearity between IVW and the results, where the closer the intercept value is to 0, the closer the MR regression model aspect is to IVW. Finally, the leave-one-out analysis was used to exclude single SNPs that had a large impact on the results, to avoid single SNPs that had a large impact on the results. SNPs to avoid level collapse caused by a single SNP. We used funnel plots to assess heterogeneity among SNPs and considered asymmetry as an indicator of horizontal pleiotropy.
Statistical analysis
All data were analyzed using the R software package “TwoSampleMR” (version 4.4.1) MRCIEU. In TwoSampleMR, p < 0.05 indicated that the difference between results was statistically significant. In the multivariate test, IV was considered to be horizontally multivariate with horizontal diversity when the MR-Egger regression intercept was nonzero and statistically significant (p < 0.05). In the heterogeneity test, p > 0.1 indicated that there was no heterogeneity between groups.
Results
Descriptive characteristics
Figure 1 shows the research process of this study. During the period of testing from the first quarter of 2004 and the second quarter of 2024, a total of 30,025 reports of AEs related to celecoxib were recorded in the FAERS database, of which 740 reports were related to thrombotic events. Among the cases related to celecoxib, the number of females (n = 18,390, 61.2%) is twice that of males (n = 9294, 31.0%). Specifically, when it comes to thrombotic events, the number of female patients (n = 439, 59.3%) is also higher than that of male patients (n = 276, 37.3%). In addition, Other Serious (Important Medical Events; n = 7969, 26.5%) accounted for a significant proportion of AE outcomes in celecoxib. Patients were reported to have a higher proportion of hospitalization-initial or prolonged (456, 61.6%) and Death (86, 11.6%) among all outcomes.

Flowchart of pharmacovigilance analysis and drug targets Mendelian randomization.
The JADER database recorded a total of 4571 case reports of celecoxib from the first quarter of April 2004 to May 2024, of which only 48 were related to thrombosis. The proportion of female patients (n = 2,824, 61.8%) is higher than that of males. Similarly, the number of thrombotic events in female patients (n = 37, 77.1%) was higher than that in male patients (n = 10, 20.8%). In this population, patients who experienced AEs after treatment with celecoxib had a higher proportion of final recovery (n = 2,567, 38.2%) and fewer outcomes leading to death (n = 273, 4.1%). However, the mortality rate of patients with concurrent thrombosis is relatively higher, accounting for 14.6% of all outcomes.
From January 2004 to May 2024, the CVAR database recorded a total of 1184 case reports of celecoxib, of which only 26 thrombotic events were reported. Similar to the previous two databases, the number of female patients (n = 831, 70.2%) is much higher than that of male patients (n = 339, 28.6%). It is worth noting that all 26 reported thrombotic events were in female patients. From the age distribution perspective, AE is mainly more common in the population aged 18–64.9 (n = 684, 57.8%). Furthermore, the outcomes of death and disability caused by thrombotic events accounted for 11.5%. Patient information is shown in Table 1.
Basic information on thrombus and celecoxib.

CA, congenital anomaly; CVAR, The Canada Vigilance Adverse Reaction; DE, death; DS, disability; FAERS, The Food and Drug Administration Adverse Event Reporting System; HO, hospitalization-initial or prolonged; JADER, The Japanese Adverse Drug Event Report database; LT, life-threatening; OT, Other Serious (Important Medical Event); RIPPI, required intervention to prevent permanent impairment/damage.
Signal mining of thrombotic AEs of celecoxib in three populations
At the SOC level, AEs in the three populations were divided into 27 organ systems (Figure S1). A total of six positive SOCs were detected in the American population. Among them, Cardiac disorders are the system with the highest number of reports and the strongest signals (n = 9578, ROR = 7.34, 95% CI: 7.18–7.5). There are a total of four positive signals in the Japanese population (n = 11,532), among which skin and subcutaneous tissue disorders have the highest number of SOC and the highest signal value (n = 1866, ROR = 6.02, 95% CI: 5.70–6.35). In addition, there were five positive SOC signals in the Canadian population. Systemic diseases and various reactions at the administration site are the most common SOC (n = 2,794).
From the perspective of the PT level, there are 11 thrombosis events in the American population (Figure 2). Among these, thrombosis (n = 394) and DVT (n = 331) are the most common adverse reactions, with a number even ten times that of the total of other thrombotic events. When evaluating signal strength, arteriovenous graft thrombosis is the strongest PT signal (ROR = 27.36, 95% CI: 3.64–205.61). It is worth noting that the AE of arteriovenous graft thrombosis only occurred in the American population and was not found in other populations. In the Japanese population, only three positive PT cases are associated with thrombosis. Among these, the number of DVT is the highest among Japanese people, accounting for 85% of all thrombotic events (n = 23). However, its signal strength (ROR = 1.55, 95% CI: 1.03–2.34) is 1/2 of that of the American population (ROR = 4.03, 95% CI: 3.62–4.50). Cerebral thrombosis is the strongest thrombotic event in the Japanese population (ROR = 5.62, 95% CI: 1.79–17.68), which is also higher than its signal intensity in the American population (ROR = 3.85, 95% CI: 2.45–6.04). Interestingly, the event of shunt thrombosis has only been detected with positive signals in the Japanese population, ranking second in signal value among the Japanese population (ROR = 4.74, 95% CI: 0.65–34.38, PRR = 4.74). Canadian population only reported postoperative DVT (n = 25, ROR = 74.94, 95% CI: 48.39–116.05). The results of four algorithms for positive PTs in three populations are shown in Table S3.

Signal mining of three groups at the PT level and the forest plots were used to show the ROR and corresponding 95% CIs.
We further analyzed the overall signaling patterns of adverse thrombotic events caused by celecoxib in three populations. The results showed that when considering the overall drug adverse reaction signals for thrombosis, it was positive in all three populations, providing a broader link between celecoxib and thrombosis. Specifically, the strongest positive signal among the three groups is the American population (ROR = 3.97). The Canadian population ranks second in signal value (ROR = 2.02), higher than the Japanese population (ROR = 1.48). Coagulation has always been considered a key pathway affecting thrombus formation, which is mediated by tissue factors (TFs). Research has shown that the proliferation of fibrin in blood clots is dominated by hematogenous TFs. 25 Therefore, we detected coagulation-related PT signals in three populations. Interestingly, it was found that the relevant signals were negative.
In addition, to further investigate individual characteristics, we conducted a subgroup analysis based on gender (Table 2). The results showed that there were 12 positive PTs associated with thrombosis in the American female population and 6 PTs in males. The number of thrombosis and DVT is the highest in both populations. In terms of signal strength, the strongest signal in the female population is the adverse event of postoperative DVT (ROR = 10.46, 95% CI: 1.44–76.12). The highest signal value in the male population is arterial and venous graft thrombosis, with much higher signal intensity than other thrombotic events (ROR = 72.55, 95% CI: 8.93–589.72). Except for this adverse event, the other five PTs detected in males were present in the female population. It is worth noting that there is no common thrombotic event between male and female groups in Japan. Three types of thrombotic events were identified in the female population, among which shunt thrombosis had the highest signal value (ROR = 10.76, 95% CI: 1.43–81.17). In addition, no thrombotic events with positive signals were detected in Japanese males. In the Canadian population, all thrombotic events are present in the female population.
Subgroup analysis based on gender of populations in American and Japan.

Only the PT values of positive signals detailed in both male and female populations.
EBGM, empirical Bayesian geometric mean; IC, information component; PRR, proportional reporting ratio; PT, preferred term; ROR, report odds ratio.
TTO analysis
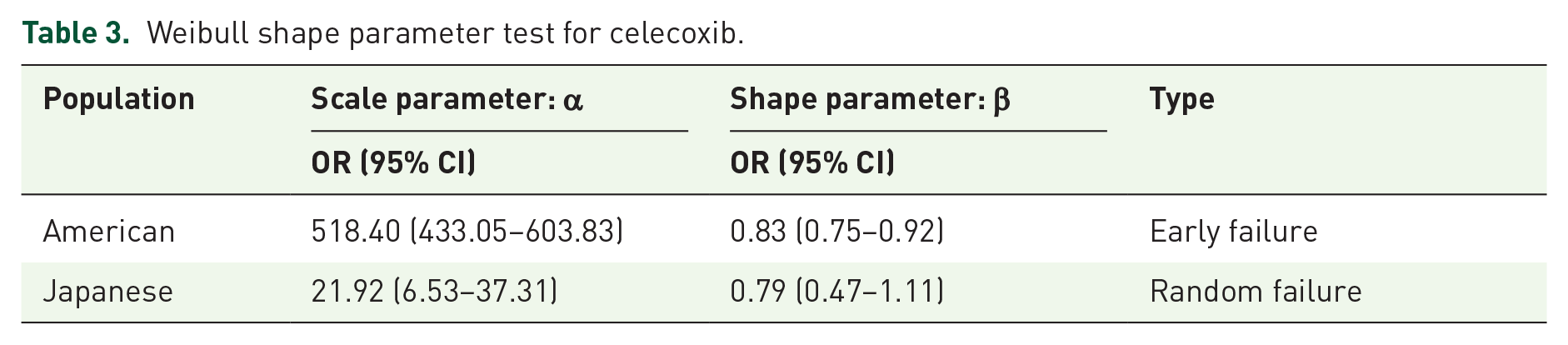
In the evaluation of the occurrence time of DVT events after treatment with celecoxib, the average induction time in the American population was about 688 days, and the median induction time was 426 days. In Japan, the average time to induction was 129 days, and the median time to induction was 12 days. Unfortunately, the CVAR database did not record the duration of any AEs. In addition, a significant statistical difference was found in the induction time of DVT between the two populations (p < 0.005). The time of AE induction in the American population was significantly later than that in the Japanese population. In addition, according to the survival curve of AEs, the incidence of DVT increases with time, and there is a statistically significant difference between the two populations (p < 0.0001; Figure 3). WSP test can be used to describe the treatment effect through the relative increase or decrease of survival time and risk ratio. The shape parameter β of the American population is less than 1, and the upper limit of the 95% CI is less than 1, indicating that the incidence of DVT in the American population is considered to decline over time. The beta value of the Japanese population is close to 1, with a 95% CI, including 1, which is considered a random failure curve. Its incidence is constantly occurring over time (Table 3).

Survival curves of adverse events induced by celecoxib in the United States and Japan.
Weibull shape parameter test for celecoxib.
Causality between celecoxib and DVT
Our research found that DVT is the most common thrombotic event among three populations. Therefore, we identified four drug targets of celecoxib through the DrugBank database and eQTLGen dataset, namely carbonic anhydrase 2 (CAII), signal transducer and activator of transcription 3 (STAT3), neuraminidase 1 (NEU1), and prostaglandin-endoperoxide synthase 2 (PTGS2). Among these, PTGS2 is a common target of NSAIDs. Subsequently, MR analysis was used to analyze the causal relationship between drug targets and DVT (Table 4). The IVW results showed that only CAII was positive and was a protective factor for thrombus formation (IVW OR = 0.9995, p = 0.0469). This suggests that celecoxib increases the risk of DVT. The other three are sites, all negative. In addition, as a supplement to IVW, weighted median and MR-Egger are also used to evaluate the correlation between the two (Figure 4). In addition, we further validated the positive results using data from the GWAS Catalog and FinnGen database. The results all showed that CAII is a protective factor for DVT (ebi-a-GCST90038607: IVW OR = 0.9995, p = 0.0256; finngen_R12_I9_VTE: IVW OR = 0.9866, p = 0.338; Table 5 and Figure S2).
MR estimates for the association between celecoxib and thrombus.

DVT, deep vein thrombosis; IVW, inverse variance weighting; MR, Mendelian randomization; MR-Egger, MR-Egger regression; SNP, single nucleotide polymorphism; Weighted median, weighted median method.

Scatter plot of MR analysis of celecoxib targets and deep vein thrombosis. (a) The results of carbonic anhydrase 2; (b) the results of the Signal transducer and activator of transcription 3; (c) the results of neuraminidase 1; and (d) the results of prostaglandin-endoperoxide synthase 2.
MR analysis of the relationship between carbonic anhydrase 2 and thrombosis in GWAS Catalog and FinnGen database.

CAII, carbonic anhydrase 2; DVT, deep vein thrombosis; IVW, inverse variance weighting; MR, Mendelian randomization; MR-Egger, MR-Egger regression; SNP, single nucleotide polymorphism; Weighted median, weighted median method.
Further sensitivity analysis results showed heterogeneity in the MR analysis of CAII and DVT (MR Egger Q_pval = 0.000000867, IVW Q_pval = 0.00000115). PTGS2, STAT3, and NEU1 show no heterogeneity (Figure S3). In addition, the intercept test of MR-Egger regression analysis was used to evaluate the horizontal pleiotropy between IV and the results, and the results showed insufficient evidence of horizontal pleiotropy (p = 0.2244). The leave-one-out indicates that after removing individual SNP analysis, there is no strong causal relationship between drug targets and DVT that is strongly influenced by a single SNP, indicating that the current MR analysis results are robust (Figure S4). There is also heterogeneity existed between CAII and DVT in the GWAS Catalog and FinnGen data (ebi-a-GCST90038607: MR Egger Q_pval = 0.000000838, IVW Q_pval = 0.000000417; finngen_R12_I9_VTE: MR Egger Q_pval = 0.00000458, IVW Q_pval = 0.00000272; Figure S5). In addition, horizontal pleiotropy was not detected in either database, (ebi-a-GCST90038607: p = 0.0788, finngen_R12_I9_VTE: p = 0.1082) and the results of the leave-one-out method supported the robustness of the causal relationship (Figure S6).
Discussion
This study aims to clarify the specific risk and potential causal relationship of celecoxib to thrombotic events in the real world through pharmacovigilance analysis and MR of AEs in three populations. Our research findings indicate that positive signals can be retrieved both at the overall level and at the PT level. In addition, further MR analysis revealed that CAII has a protective effect on thrombosis. This suggests that celecoxib may increase thrombosis risk via CAII inhibition. This provides new insights into the medication strategy of celecoxib and the improvement of patient prognosis in clinical practice.
There is currently controversy over the effect of celecoxib on thrombosis. Unlike the results of the CLASS trial mentioned above, other studies suggest that celecoxib may increase the risk of thrombosis. 6 For example, a randomized, double-blind clinical trial of selective COX-2 inhibitors, which showed a significantly higher probability of severe thrombotic events than placebo. 26 A meta-analysis further confirms this viewpoint, observing a significantly increased risk of myocardial infarction when using celecoxib to treat OA and rheumatoid arthritis, suggesting an increased risk of thrombosis. 4 Based on the above controversy, we evaluated the overall signal of thrombotic events through pharmacovigilance analysis and found that all three populations included in the study exhibited positive signals. This result provides further evidence to support the risk of thrombotic events associated with celecoxib.
When it comes to specific PT signals, our results indicate that all three groups in the United States, Japan, and Canada have specific positive thrombotic events. Specifically, DVT occurred in the highest number among these three groups, with significant positive signal intensity detected. This may be because DVT is one of the most frequent thrombotic events in the body. Elche University General Hospital reported a patient with gout who had no thrombosis and no relevant family history of thrombosis developed DVT after 5 days of celecoxib 200 mg/day. 27 This diagnosis ruled out causes other than celecoxib. In another animal study, COX-2 inhibitors were found to induce less arterial thrombosis in obese mice and significantly increase the risk of venous thrombosis in lean mice. 28 This further suggests an important role for celecoxib in venous thrombosis. It is worth noting that Canada places greater emphasis on postoperative DVT. This may be due to the fact that after its launch in Canada, celecoxib is more commonly used for the management of acute pain, including postoperative analgesia. 29 In addition, interestingly, we have found unique thrombotic events in both the American and Japanese populations. Arteriovenous graft thrombosis was only found in the American population, and the signal value was the highest among all PTs. This may be related to the large population of end-stage renal disease patients in the United States, who require long-term dialysis treatment, resulting in frequent use of arteriovenous grafts and greatly increasing the risk of thrombosis.30,31 This may be due to the fact that shunt thrombotic events frequently occur after transjugular intrahepatic portosystemic shunt (TIPS), whereas the Japanese population has a higher burden of liver disease and a higher number of patients undergoing TIPS.32,33 Based on this multicenter study, the geographic distribution of celecoxib-associated thrombotic events was shown to be heterogeneous. This heterogeneity likely reflects interethnic genetic variations and lifestyle factors.
Furthermore, to investigate whether there is heterogeneity in this risk among different characteristic populations, we conducted a subgroup analysis based on gender. The subgroup analysis results indicate that women reported more AEs of thrombosis, compared to men. This may be due to women’s higher estrogen levels, smaller blood vessel diameters, and the expression of some sex-specific genes that place women at a higher risk of thrombosis compared to men.34–36 When focusing on specific PT signals, we found that pulmonary thrombosis was only detected in the female population. The reason for this may be related to the differences in pulmonary vessel diameter between genders. A study showed that women have smaller pulmonary vessel diameters and narrower lumens than men, which can lead to slower blood flow and increase the risk of pulmonary thrombosis. 37 In addition, it is worth noting that among Japanese women, there is a relatively high number of cerebral thrombosis and the signal value is also relatively high. Research shows that the cerebral blood flow of women is significantly higher than that of men, and the incidence rate of migraine is higher. 38 Migraine has been increasingly shown to be associated with the onset of ischemic stroke in more and more studies, and cerebral thrombosis is a common cause of ischemic stroke. 39 Therefore, this may be the reason for the higher number of cerebral thrombosis in women.
Interestingly, although coagulation function is often considered a common factor in triggering thrombosis in the study of thrombosis mechanisms. However, our research results showed that PT signals related to coagulation were negative, indicating that celecoxib may affect the occurrence of thrombotic events through pathways other than coagulation. At present, research has delved into the underlying mechanism, and celecoxib may promote thrombosis by increasing fibrinolysis inhibitors, leading to a decrease in fibrinolysis. 40 In addition, celecoxib may cause vasoconstriction by affecting endothelial vasodilation, thereby increasing the risk of thrombosis.
In drug safety evaluations, it is critical to assess the interval between drug administration and the onset of AEs. This assessment can elucidate the underlying mechanisms of AEs, identify specific time windows of risk during treatment, and help in the early prevention or diagnosis of adverse events. 14 TTO analyses showed that the mean induction time for DVT in the United States population was approximately 688 days, with a median induction time of 426 days. In Japan, the mean induction time was 129 days and the median induction time was 12 days. The possible reason for the shorter latency period for adverse thrombotic events in the Japanese population is related to its relatively poor metabolism of celecoxib. Celecoxib is primarily metabolized by CYP2C9, and studies have shown that CYP2C9 activity is lower in Asians than in Europeans.41,42 This increases the risk of drug-related AEs. We observed two different pathogenic models for the drugs in the TTO analysis. The US population showed an early failure type curve and the Japanese population showed a randomized failure model. This suggests that no single mechanism (tolerability effect/accumulation effect) can explain the temporal association between the onset of celecoxib and thrombosis. These findings suggest that physicians need to monitor patients closely during the early stages of treatment to detect thrombotic tendencies promptly. In addition, regular assessment of thrombosis-related markers is crucial during long-term treatment. In conclusion, our findings emphasize the need for continuous vigilance in clinical practice.
Our study suggests that CAII, a drug target of celecoxib, is protective against thrombosis. When celecoxib inhibits CAII, it significantly increases the risk of DVT. The current study confirms that the protective effect of CAII on DVT is primarily achieved by affecting changes in pH. CAII has been found to maintain intracellular and extracellular pH homeostasis by acidifying the microenvironment through catalyzing the hydration of carbon dioxide to bicarbonate and protons. 43 Significantly delays clot formation, prolongs clotting time, and reduces platelet aggregation under acidic conditions.44,45 Meanwhile, a study addressing the mechanism of endothelial dysfunction generation demonstrated that endothelial cells are able to catalyze the formation of arachidonic acid to form five COX-driven end-products, which act as endothelium-derived diastolic and endothelium-derived constrictor factors to regulate vascular function and further reduce thrombotic events. CAII has been demonstrated to maintain endothelial cell viability under acidic stress, and when it is inhibited, endothelial cell homeostasis is disrupted, causing vascular dysfunction and an increased risk of DVT.46,47 In addition, when endothelial function is impaired, endothelial NO synthase (eNOS) is inhibited, resulting in a decrease in NO release and an increase in blood viscosity, which affects hemodynamic stability and promotes thrombosis. 48
Interestingly, our results found no causal relationship between PTGS2 and thrombosis. This is despite previous studies suggesting that PTGS2 plays a role in celecoxib’s promotion of thrombosis. For example, PTGS2 deficiency was found to alter Annexin A2 distribution in monocytes/macrophages, which led to an increase in TF expression and activity, and ultimately the formation of stable thrombi. 49 However, our results suggest that there is no causal link between the two. Therefore, it is inappropriate to simply view PTGS2 as a bridge between COX-2 inhibitors and DVT. Furthermore, our study suggests that the key target of celecoxib for the development of DVT is its unique inhibition of CAII. Therefore, when patients are treated with COX-2 inhibitors, they should be evaluated first, and if high-risk factors for thrombosis are present, celecoxib should be avoided and replaced with other COX-2 inhibitors.
Our study has several strengths. Firstly, our findings combine real-world data studies with MR analyses to provide not only real-world evidence but also strong genetic evidence for a causal relationship between celecoxib and DVT with high reliability, bridging pharmacoepidemiologic evidence with genetic causality to inform clinical guidelines. In addition, this study, which addresses the controversial point of celecoxib, clarifies the effect of celecoxib on thrombosis during treatment, allowing healthcare professionals to make the best choice for their patients in clinical decision-making, to take precautions ahead of time, and to use medication appropriately.
Limitations
It must be acknowledged that this study has some limitations, first the FAERS, JADER, and CVAR databases rely heavily on spontaneous reporting, which leads to some reporting bias, for example, consumers have different definitions of an AE, their lack objective public knowledge and their reporting may not be as reliable and comprehensive as that of healthcare professionals; there may be sampling bias and not all AEs may be collected in countries and regions with a high number of reports. On the other hand, controlling for confounding factors such as the dose of medication used, duration of treatment, drug interactions, and specific disease types poses a challenge. Therefore, MR analyses can only provide a preliminary judgment of causality, and their specific biological mechanisms should be further investigated in depth.
Conclusion
The widespread use of celecoxib in clinical practice has raised concerns about its safety, and there is some controversy over whether it can cause thrombotic events. This study reflects the specific risk of thrombotic events associated with celecoxib in the real world by jointly analyzing three different populations. In addition, a possible mechanism was proposed through MR analysis. This promotes its better use in clinical practice.
Supplemental Material
sj-docx-7-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-docx-7-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-1-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-tif-1-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-2-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-tif-2-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-3-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-tif-3-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-4-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-tif-4-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-5-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-tif-5-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-6-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-tif-6-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-10-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-xlsx-10-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-8-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-xlsx-8-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-9-taw-10.1177_20420986251347354 – Supplemental material for Beyond pain relief: the thrombosis threat of celecoxib
Supplemental material, sj-xlsx-9-taw-10.1177_20420986251347354 for Beyond pain relief: the thrombosis threat of celecoxib by Jingkai Di, Yujia Xi, Likun Qi, Yicong Zhao, Zijian Guo, Nan Yang and Chuan Xiang in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
