Abstract
Background:
Current guidelines recommend the use of only on a limited basis in patients with normal or minimal structural heart disease. The CAST study, the only randomized controlled trials, showed increased mortality from long-term flecainide use in post-myocardial infarction (MI) patients with reduced left ventricular ejection fraction (LVEF). However, many later studies have revealed its safety when used in other structural heart diseases.
Objectives:
This study investigates the incidence of ventricular tachycardia (VT) or ventricular fibrillation (VF) VT/VF in patients with structural heart disease compared to those with a normal heart when using flecainide.
Methods:
We retrospectively recruited patients who had received at least one dose of flecainide in the past 5 years. Baseline characteristics, indications for flecainide use, and echocardiography results were reviewed. After 1 year, we evaluated the incidence of ventricular arrhythmias and all-cause mortality.
Results:
After screening, 447 patients had received at least one dose of flecainide, and 336 patients were included in the study. Forty-seven patients (14%) had structural heart disease as defined by our protocols. Left ventricular hypertrophy (LVH) and impaired LVEF accounted for 28% and 25% of cases, respectively. There were five patients with coronary artery disease (CAD). After 1 year, ventricular arrhythmias occurred in two patients (4.7%) in the structural heart group; these patients had also experienced arrhythmias before receiving flecainide. In the non-structural heart group, ventricular arrhythmias were detected in three patients (1.1%). In multivariate analysis, structural heart disease was not associated with an increased incidence of ventricular arrhythmias (OR = 4.8 (0.6–38.44), p = 0.139).
Conclusion:
Our study showed that no patients died due to ventricular arrhythmia, and the incidence of VT/VF was not increased in patients with structural heart disease. A prospective study is needed to further evaluate the safety of flecainide in patients with structural heart disease other than ischemic heart disease.
Plain language summary
Background:
Flecainide is a medication usually recommended for people with normal or mild heart conditions to treat irregular heartbeats. The CAST study, the only large clinical trial on flecainide, showed that it can increase the risk of death in patients with heart damage after a heart attack. However, more recent studies have suggested that flecainide may be safe for people with other types of heart problems.
Objectives:
This study examined whether using flecainide leads to more heart problems (such as dangerous arrhythmias) in people with structural heart disease, compared to those with healthy hearts.
Methods:
We reviewed the medical records of patients who received at least one dose of flecainide in the past 5 years. We looked at their heart health before taking the medication and checked for any heart problems or deaths after one year of taking the drug.
Results:
A total of 447 patients had taken flecainide, and 336 were included in the study. Forty-seven patients (14%) had structural heart disease. The most common problems were thickening of the heart muscle (28%) and reduced heart function (25%). Five patients had coronary artery disease. After one year, only two patients with structural heart disease had dangerous arrhythmias, and they had arrhythmias before starting flecainide. In the group without structural heart disease, three patients had arrhythmias. Overall, having structural heart disease was not linked to a higher risk of arrhythmias.
Conclusion:
Our study found that no patients died from heart arrhythmias, and the use of flecainide did not increase the risk of dangerous arrhythmias in people with structural heart disease. Further studies are needed to better understand the safety of flecainide in people with heart problems other than those caused by a heart attack.
Keywords
Introduction
Flecainide is a well-known antiarrhythmic medication with efficacy against various types of arrhythmias. However, its use is limited due to contraindications in patients with structural heart disease. According to current guidelines,1,2 conditions such as heart failure, valvular heart disease, abnormal LVH (left ventricular wall thickness ⩾ 14 mm), coronary artery disease, and left ventricular systolic dysfunction are listed as contraindications. The CAST study is the only one that demonstrated harm from flecainide use. In the CAST study, 3 flecainide was used in post-myocardial infarction (MI) patients to reduce the burden of PVCs. After 10 months of follow-up, the number of arrhythmic deaths in the flecainide group was 3.6 times higher than in the placebo group.
In developed countries, there are many alternative medications, but in many developing countries, options are limited, with flecainide being one of the few available. Its efficacy is superior to amiodarone for atrial fibrillation in patients with normal structural hearts. Several studies4–10 have shown the safety of flecainide in various conditions, such as hypertrophic cardiomyopathy (HCM), arrhythmogenic right ventricular cardiomyopathy, congenital heart disease, tachycardia-induced cardiomyopathy, rheumatic mitral stenosis, post mitral valve replacement, ischemic heart disease, and heart failure. We conducted a retrospective cohort study to evaluate the outcomes of patients with structural heart disease who received flecainide at King Chulalongkorn Memorial Hospital (KCMH) over the past 5 years.
Methods
Trial design
Retrospective cohort study
Setting
This study was conducted at King Chulalongkorn Memorial Hospital (KCMH), Thai Red Cross Society, Bangkok, Thailand. Data were collected from January 1, 2016, to November 30, 2020.
Participants
Inclusion
Patients who had received at least one dose of flecainide.
Exclusion
Patients younger than 15 years old.
Patients who did not have an echocardiogram in their medical records.
Patients who were lost to follow-up before 1 year.
Data sources
The reporting of this study conforms to the STROBE statement. 11 Electronic medical records were reviewed for baseline characteristics, underlying diseases, and indications for flecainide use. Echocardiography was assessed for structural heart disease as defined by our protocols. Patients were followed up for the incidence of ventricular tachyarrhythmias and all-cause mortality 1 year after the first dose of flecainide.
Variables and definitions
Ventricular arrhythmias: Ventricular tachycardia (VT) or ventricular fibrillation (VF) lasting more than 30 seconds, or if symptoms occur, lasting less than 30 seconds.
Arrhythmic death: Death resulting from ventricular arrhythmias recorded during resuscitation. If deaths occurred without ECG recordings, they were presumed to be due to arrhythmia if they had the following characteristics (as defined by the CAST study):
Witnessed and instantaneous death without preceding symptoms.
Witnessed and preceded by symptoms of myocardial ischemia in the absence of cardiogenic shock.
Witnessed and preceded by symptoms of cardiac arrhythmias, such as syncope.
Unwitnessed, but without another explainable cause.
Structural heart disease: Defined by the following features:
Left ventricular ejection fraction <50%
Significant severe valvular abnormalities (both stenosis and regurgitation)
Significant left ventricular hypertrophy (Any segment of LV wall thickness ⩾ 14 mm measured by echocardiography or cardiac MRI).
Chronic coronary syndrome with or without myocardial infarction, documented myocardial ischemia with an exercise stress test, stress echocardiography, MIBI, stress cardiac MRI, or coronary angiography showing more than 50% stenosis.
Congenital heart disease with or without surgical correction
History of cardiac surgery, such as CABG, valve surgery, etc.
HFpEF diagnosed by a cardiologist
Bias
Due to the retrospective nature of the study, potential biases in detecting ventricular arrhythmias may exist. We excluded some patients who received flecainide without a baseline echocardiogram. To reduce the risk of bias, we also analyzed data comparing echo and no-echo groups.
Sample size
We included all patients based on the inclusion and exclusion criteria during the period from January 1, 2016, to November 30, 2020.
Statistical analysis
Continuous variables are reported as means or medians, and categorical variables as numbers and percentages. Baseline characteristics between the two groups were compared using the Chi-square test. Categorical predictor variables were screened for their association with death and VT using contingency tables and Fisher’s exact test. Logistic regression was used to compare unadjusted and adjusted odds ratios (95% CI) of factors associated with VF and death. For continuous variables (e.g., age), the linearity of the association against the log odds ratio was assessed.
Results
After screening 447 patients who were prescribed at least one dose of flecainide, 4 patients were excluded due to being <15 years old, 49 patients were excluded due to loss to follow-up before 1 year, and 58 patients were excluded due to lack of echocardiography data. Of the 336 patients with complete echocardiographic data, 47 patients (13.9%) had structural heart disease as defined in our protocols, and 289 patients were in the non-structural heart group. The flowchart of patient recruitment is shown in Figure 1. The baseline characteristics of the two groups are shown in Table 1.

Consort diagram.
Baseline characteristics of patients in structural and nonstructural heart disease.

CKD, Chronic kidney disease; DM, Diabetes mellitus; DLP, Dyslipidemia; HTN, Hypertension; PACs, Premature Atrial Contrcations; PVCs, Premature Ventricular Contractions; SVT, Supraventricular tachycardia; VT/VF, ventricular tachycardia/ventricular fibrillation.
Among the 47 patients in the structural heart group, 13 had increased LV wall thickness (⩾14 mm); 12 had impaired LV systolic function (all patients had LVEF ⩾ 35%); 7 had a history of cardiac surgery; 5 had chronic coronary syndrome; 4 had severe valvular heart disease; 3 had heart failure with preserved EF; 1 had impaired RV systolic function, and 1 had congenital heart disease (shown in Table 2). Detailed information about the five patients with chronic coronary syndrome (CCS) who received flecainide is provided in Supplemental Table 1. No patient in the CCS group experienced VT/VF after receiving flecainide.
Types of structural heart disease.

In the structural heart disease cohort, VT/VF was detected in two patients during follow-up, but these patients had VT/VF before receiving flecainide. One patient died during the 1-year follow-up due to metastatic renal cell carcinoma. In the non-structural heart disease cohort, VT/VF was detected in three patients during follow-up, and two patients died during the 1-year follow-up, both from non-cardiac conditions. Detailed data about patients in whom VT/VF occurred are provided in Supplemental Table 2.
We also analyzed data from patients without baseline echocardiography. Among the 58 patients with complete 1-year follow-up data, there was no difference in baseline characteristics between the echocardiography group (echo group) and the no echocardiography group (no echo group), as shown in Table 3.
Baseline characteristics of patients in echo group and no echo group.
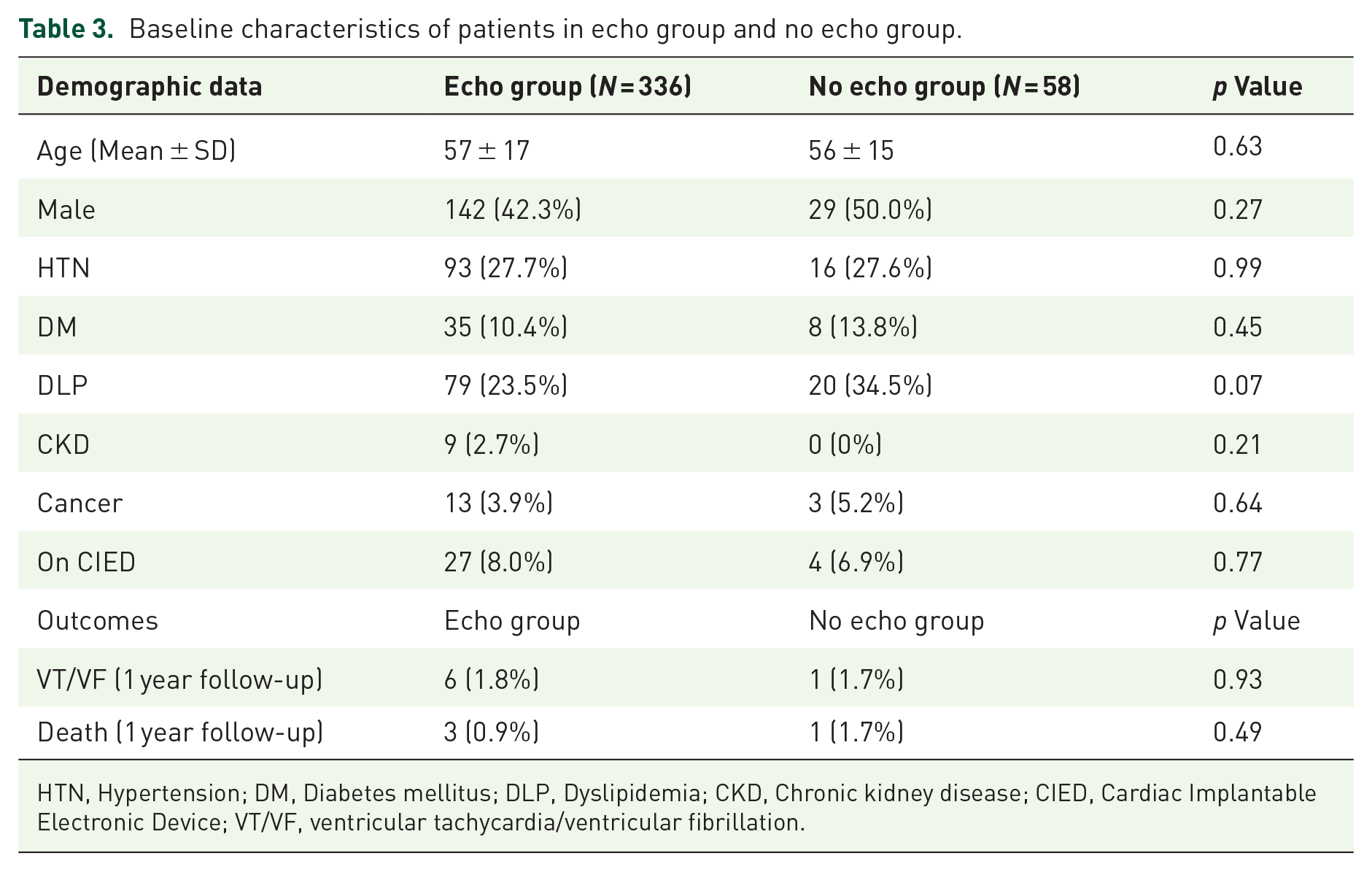
HTN, Hypertension; DM, Diabetes mellitus; DLP, Dyslipidemia; CKD, Chronic kidney disease; CIED, Cardiac Implantable Electronic Device; VT/VF, ventricular tachycardia/ventricular fibrillation.
Categorical predictor variables for death and VT were screened using contingency tables and Fisher’s exact test. Logistic regression was then used to compare unadjusted and adjusted odds ratios (95% CI) for factors associated with VF and death. For continuous variables (e.g., age), the linearity of the association was assessed. Due to non-linearity, age was modeled in quartiles, with the upper three quartiles collapsed together because the OR and 95% CI were similar. Factors associated with VT at p < 0.015 in univariate models included age ⩽46 years, structural heart disease versus no structural heart disease, and CIED.
After adjustment in a multivariable model, both age ⩽ 46 and CIED were independently associated with an increased risk of VT. Structural heart disease did not show an independent association (p > 0.05), but the OR and 95% CI suggest that structural heart disease may be associated with an elevated risk of VT after adjustment for age and CIED, although the magnitude of the effect is less than that observed with age and CIED (Table 4). A plot of the odds ratios and 95% CI from the adjusted model is shown in Figure 2.
Predictor variables for their association with VT/VF.

VT/VF, ventricular tachycardia/ventricular fibrillation.

Odds ratio (OR) from adjusted analysis of factors associated with VT.
Cancer was the only risk factor associated with death by Fisher’s exact test, and because this and other deaths were unrelated to heart disease, we did not create logistic models for this endpoint.
Discussion
In current practice, we avoid using flecainide in patients with “structural heart disease,” as defined by standard guidelines, which encompass a broad spectrum of heart diseases such as valvular heart disease, heart failure, LVH, and others. Our study provides data on flecainide prescriptions in patients with structural heart disease. The structural heart disease definitions in our protocols were broad, but most were consistent with contraindications listed in guidelines for flecainide use. Our results showed that 13.7% of patients who were prescribed flecainide had structural heart diseases. Most patients in our study had non-ischemic heart disease and mildly abnormal structural heart conditions. The maximum LV wall thickness was 16 mm, and the minimum LVEF was 35%. This study also supports previous evidence regarding the safety of flecainide use in patients with LVH and non-ischemic cardiomyopathy.
A retrospective cohort study 4 conducted by Haruki in 2015 found that flecainide was used in 15 patients with hypertrophic obstructive cardiomyopathy (mean LV wall thickness 20.4 mm), and after 8.9 years of follow-up, no malignant arrhythmias were detected. Another retrospective study 7 in 2018 found that among 13 patients with PVC-induced cardiomyopathy treated with flecainide, with a mean LVEF of 37%, no sustained VT or deaths occurred after 3.8 years of follow-up. In our study, no VT was reported in the LVH group. One patient in the impaired LV systolic function group experienced VT during follow-up, but this patient had recurrent VT before taking a 25 mg single dose of flecainide, making it unlikely that the VT was due to the flecainide.
For valvular heart disease, our study included only four patients diagnosed with severe valvular heart disease, and no VT/VF occurred in this group after receiving flecainide. Our results are consistent with those of two studies8,9 that included patients diagnosed with mild to moderate mitral stenosis and patients with post-mitral valve replacement, both of which showed no arrhythmic complications following flecainide administration. Our study also reported the outcomes of five patients diagnosed with CCS. Three patients in the CCS group received flecainide before PCI with no adverse events, and two patients received flecainide after PCI safely. These patients differed from those in the CAST study in several respects. In the CAST study, patients had post-myocardial infarction within 2 years, with impaired LV systolic function, and were given flecainide to treat PVCs. However, in our study, most CCS patients had good LV systolic function and no history of myocardial infarction, which could explain the discrepancy between our findings and those of the CAST study. Our results are also consistent with a systematic review 10 that included 20 randomized controlled trials in which Class IC antiarrhythmic agents were used to convert atrial fibrillation, compared with placebo. A significant portion of patients (about 20%) in this review had ischemic heart disease or mild heart failure (mostly excluding NYHA III-IV). The incidence of VT was very low (0.1%), with no reported deaths.
These findings align with previous studies4–10 that the incidence of death or ventricular arrhythmias did not increase with flecainide use in patients with structural heart disease after the exclusion of significant CAD with left ventricular dysfunction.
Limitations
There are a few limitations in our study including missing baseline echocardiographic data and the potential underreporting of documented VT/VF. A prospective study or randomized controlled trial is needed to provide more safety data on the use of flecainide in patients with structural heart disease other than ischemic heart disease.
Conclusion
The results show that a substantial proportion of structural heart disease patients received flecainide. Several subgroups of structural heart disease in our study were defined as contraindications according to current guidelines, such as impaired LV systolic function and LV wall thickness ⩾14 mm. Our retrospective cohort review indicated that structural heart disease was not associated with an increased incidence of VT/VF in patients who received flecainide.
Supplemental Material
sj-docx-1-taw-10.1177_20420986251316462 – Supplemental material for Flecainide use in arrhythmic patients who have structural heart disease
Supplemental material, sj-docx-1-taw-10.1177_20420986251316462 for Flecainide use in arrhythmic patients who have structural heart disease by Naruepat Sangpornsuk, Voravut Rungpradubvong, Tachawut Tiensantisuk, Pattranee Leelapattana, Ronpichai Chokesuwattanakul and Somchai Prechawat in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986251316462 – Supplemental material for Flecainide use in arrhythmic patients who have structural heart disease
Supplemental material, sj-docx-2-taw-10.1177_20420986251316462 for Flecainide use in arrhythmic patients who have structural heart disease by Naruepat Sangpornsuk, Voravut Rungpradubvong, Tachawut Tiensantisuk, Pattranee Leelapattana, Ronpichai Chokesuwattanakul and Somchai Prechawat in Therapeutic Advances in Drug Safety
Footnotes
Appendix
Acknowledgements
We would like to thank all of the other supporting personnel who assisted us by supplying the data that was essential and vital, without which we would not have been able to perform efficiently on this project.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
